Abstract
Even before the advent of fMRI, the amygdala occupied a central space in the affective neurosciences. Yet this amygdala-centred view on emotion processing gained even wider acceptance after the inception of fMRI in the early 1990s, a landmark that triggered a goldrush of fMRI studies targeting the amygdala in vivo. Initially, this amygdala fMRI research was mostly confined to task-activation studies measuring the magnitude of the amygdala’s response to emotional stimuli. Later, interest began to shift more towards the study of the amygdala’s resting-state functional connectivity and task-based psychophysiological interactions. Later still, the test-retest reliability of amygdala fMRI came under closer scrutiny, while at the same time, amygdala-based real-time fMRI neurofeedback gained widespread popularity. Each of these major subdomains of amygdala fMRI research has left its marks on the field of affective neuroscience at large. The purpose of this review is to provide a critical assessment of this literature. By integrating the insights garnered by these research branches, we aim to answer the question: What part (if any) can amygdala fMRI still play within the current landscape of affective neuroscience? Our findings show that serious questions can be raised with regard to both the reliability and validity of amygdala fMRI. These conclusions force us to cast doubt on the continued viability of amygdala fMRI as a core pilar of the affective neurosciences.
Keywords
Introduction
Now more than 20 years ago, Merboldt et al. 1 published a brief commentary titled: “Functional MRI of the Human Amygdala?.” At the time, Merboldt and his colleagues wrote this commentary to draw attention to the (then often overlooked) presence of magnetic susceptibility artifacts occurring in the amygdala due to the proximity of bone and air-filled cavities (ie, sinuses). Although these artifacts are less of a concern now that we scan at (much) higher field strengths, the question itself is now relevant as it was back then—albeit for somewhat different reasons. The purpose of this review is to provide an overview of the contributions amygdala functional magnetic resonance imaging (fMRI) has made over the years to the affective neurosciences at large, in order to critically assess its enduring role as a research tool in the current scientific landscape
Academic interest in the amygdala as an emotion processing region originally arose due to observations of hypo-emotional behavior in rhesus monkeys after (bilateral) ablation (part of the “Klüver-Bucy syndrome”).2 -4 A more explicit proposal for the amygdala’s role in emotion processing would not follow, however, until the structure came to be closely associated with Pavlovian fear conditioning in animal models (eg, see Maren & Fanselow 5 for a review). From this perspective, the amygdala (particularly its basolateral subdivision) is seen as the locus at which stimulus-reward/punishment associations are forged within the brain, thus linking noxious unconditioned stimuli (eg, an electric shock) on the 1 hand, to otherwise innocuous conditioned stimuli (eg, a tone) on the other hand. The autonomic response to this conditioned fear is then mediated indirectly by the (centromedial) amygdala via descending pathways projecting to lower brain regions such as the periaqueductal gray, while the activity of the amygdala itself can be modulated by regulatory prefrontal regions such as the medial prefrontal cortex (MPFC).6,7 While not always explicitly addressed anymore in the literature, this fear conditioning framework has in fact shaped much of our way of thinking about the amygdala’s role in emotion processing.
With the advent of fMRI in the early 1990s, the study of amygdala and emotion was no longer confined to animal models, as the early work on Pavlovian fear conditioning often was, but could also be extended to human populations. Initially, this burgeoning field was mostly limited to studies that utilized some form of emotion provocation to measure the magnitude of the amygdala’s blood-oxygen-level-dependent (BOLD) response to in-scanner stimuli (see section “Amygdala Activation fMRI”; Figure 1, top-left panel). From the mid-to-late 2000s onwards, however, scientific interest began to shift more and more towards the study of the brain’s (and by extension, the amygdala’s) functional- and effective connectivity architecture (see sections “Amygdala PPI fMRI” and “Amygdala RSFC fMRI”; Figure 1, top-right and middle-left panels, respectively). In the 2010s, a general trend towards more reproducible science saw some research-groups more closely examining the test-retest reliability of amygdala fMRI (see section “Amygdala Test-Retest Reliability fMRI”; Figure 1, middle-right panel). At around the same time, advances in the field of real-time fMRI neurofeedback enabled researchers to target the human amygdala in vivo (see section “Amygdala Real-Time fMRI Neurofeedback”; Figure 1, lower panel). We end this review by integrating the main insights garnered by these subbranches of research, after which we aim to answer the question: What part (if any) can amygdala fMRI still play within the current landscape of the affective neurosciences (see section “Integration”)?
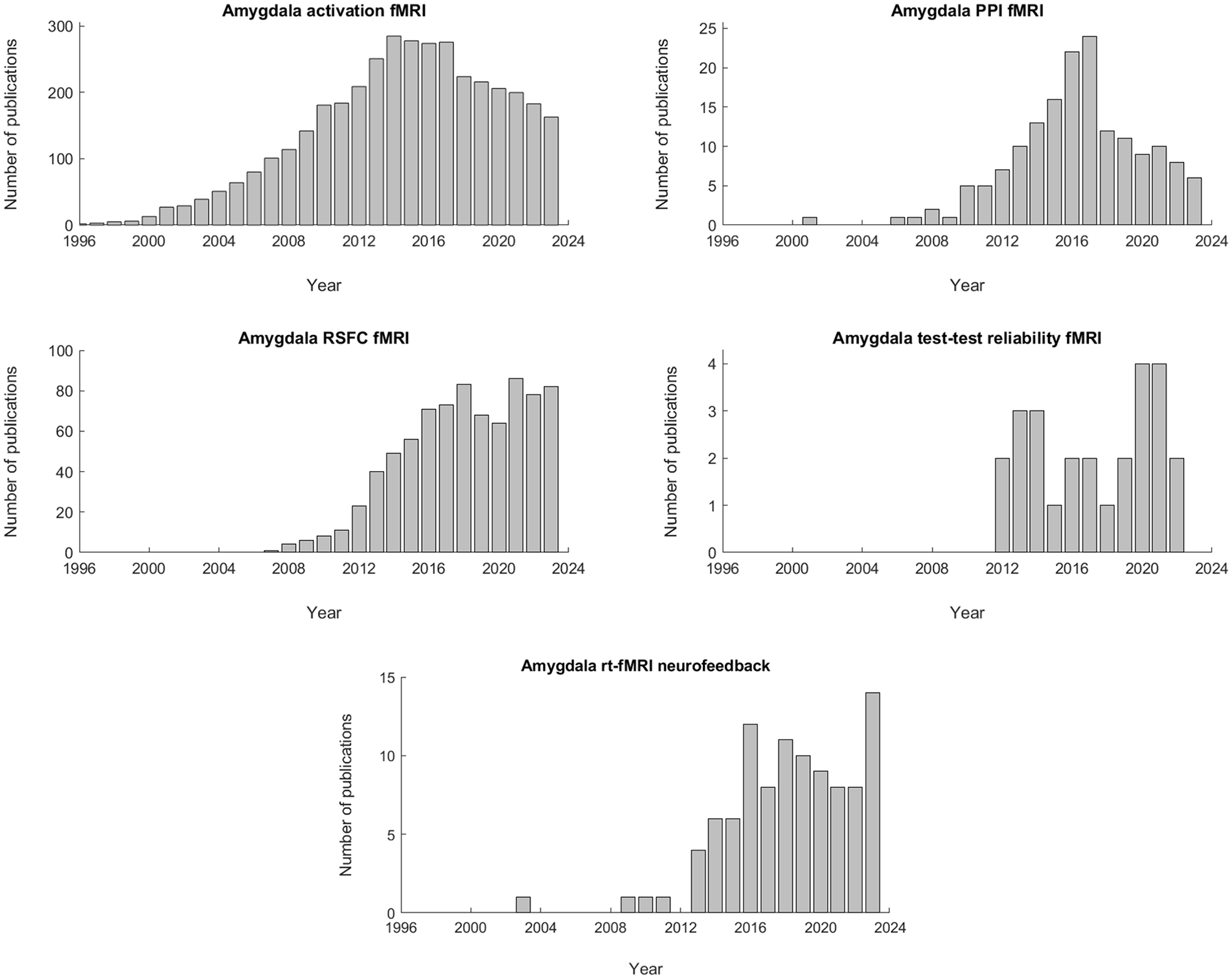
Number of publications per year for each of the amygdala fMRI subtopics discussed in the main body of the text. For the top-left panel (amygdala activation fMRI), a combination of the following search-terms was used: “amygdala” AND “fMRI” AND (“activity” OR “activation”). For the top-right panel (amygdala PPI fMRI), the following search-terms were used: “amygdala” AND “fMRI” AND (“PPI” OR “psychophysiological interaction” OR “psycho-physiological interaction”). For the middle-left panel (amygdala RSFC fMRI), the following search-terms were used: “amygdala” AND “fMRI” AND (“resting-state” OR “resting state”) AND “functional connectivity”. For the middle-right panel (amygdala test-retest reliability fMRI), the following search-terms were used: “Amygdala” AND “fMRI” AND (“test-retest reliability” OR “intraclass correlation coefficient” OR “ICC”). Finally, for the lower panel (amygdala rt-fMRI neurofeedback), the following search-terms were used: “amygdala” AND “fMRI” AND (“neurofeedback” OR “real-time fMRI”). All searches were conducted on PubMed based on keyword matches in the title/abstract field.
Amygdala Activation fMRI
Functional MRI first appeared in the early 1990s.8 -11 The first 2 studies to specifically target the amygdala via fMRI, Breiter et al. 12 successfully elicited an amygdala response by using stimuli based on human facial expressions (ie, fearful or happy), whereas Irwin et al. 13 utilized pictures taken from the International Affective Picture System (IAPS) to the same avail. Interestingly, these 2 tasks have remained the 2 gold standards for emotion provocation/amygdala fMRI to this very day.
Following in the footsteps of Breiter et al. 12 and Irwin et al., 13 multiple studies were published that similarly examined the amygdala’s role in emotion processing using task fMRI (see Figure 1, top-left panel; see Costafreda et al. 14 and Sergerie et al. 15 for meta-analyses). In most of these studies, either (a variant of) the facial expression task described by Breiter et al. 12 was used (eg, 16 -22), or an emotional pictures (ie, IAPS-based) task like the 1 detailed in Irwin et al. 13 (eg, 23 -27)—although (written) emotional words (eg, 28 -30), and tasks based on Pavlovian fear conditioning (eg, 31 -34) were also quite common. In 1 landmark study, Hariri et al. 35 directly compared the efficacy of stimuli based on facial expressions (ie, fearful or angry) or (negative) IAPS pictures, and found that while both types of stimuli were indeed able to induce a significant increase in amygdala (re)activity relative to baseline, facial expressions were significantly better at doing so than IAPS pictures were; it should be mentioned, however, that subsequent studies have been unable to replicate this finding.36,37 While the amygdala was initially hypothesized to play a distinct role in the processing of facial emotion (fear, anger, happiness, disgust, etc.; eg, 16,18,38), fMRI researchers later began to consider the amygdala more as a region involved in the detection of salience in general (eg, 39 -42). This altered viewpoint fitted well with observations of relatively high magnitudes of amygdala activation when using either scrambled pictures, a fixation cross, or a blank screen, instead of non-expressive faces, as a neutral baseline for contrast discriminability.15,40,42 When viewed in this light, the amygdala responds to all stimuli that signal some form of personal relevance or threat, including emotional stimuli, be they of the facial variety or otherwise.
Given the above, it is perhaps not surprising that the amygdala has (also) garnered much attention by researchers interested in face processing. In fact, some authors have advocated that the amygdala should be seen as a core component of a larger network involved in processing faces (eg, see Mende-Siedlecki et al. 43 ). Consequently, much effort has been spent over the years in trying to uncover the optimal task and stimulus parameters to adequately activate the amygdala using facial expressions. A number of consistent findings have emerged from this research: (1) the amygdala responds more strongly to the presentation of dynamic (ie, rather than static) expressions of affect;44 -46 (2) the amygdala response is higher when the faces are looking directly toward (versus away from) the observer; 21,47,48 and (3) paying overt attention to facial expressions seems to augment the amygdala’s response, 17,20—although non-consciously perceived faces are apparently still able to elicit a significant increase in amygdala activity relative to baseline.18,49,50 More recently, Kätsyri et al. 51 observed that the amygdala’s response to facial stimuli may also be higher when participants are exposed to real versus computer-generated faces, although Moser et al. 52 were unable to record a similar effect previously. Overall, this body of literature seems to indicate that the amygdala response is strongest when participants are paying overt attention to facial stimuli that are presented to them in as much of a naturalistic and personally relevant (ie, salient) manner as possible.
An early systematic review of the amygdala fMRI literature by Baas et al. 53 was the first to point towards a possible lateralization effect of the amygdala’s reactivity to emotion stimuli, with more of the included studies reporting activation in the left than in the right amygdala—regardless of the stimulus type, task instructions, habituation rate, or complexity of the in-scanner task that was used. Sergerie et al. 15 later replicated this finding in a quantitative meta-analysis of the emotion processing fMRI literature; however, these authors were unable to record a significant difference in the magnitude of left versus right amygdala activation. In another meta-analysis, Costafreda et al. 14 recorded a left lateralization effect for the processing of (static) emotional expressions only when the stimuli contained language elements, at the same time recording right lateralization only when the stimuli were masked to prevent consciously perceiving the facial expressions. Importantly, no other indications of lateralization were observed in that study. In a voxel-based meta-analysis of facial expression task fMRI studies, Fusar-Poli et al. 54 were similarly unable to record significant lateralization of the amygdala’s task reactivity. Finally, in a meta-analysis of the dynamic facial expression task fMRI literature, Zinchenko et al. 46 recorded significant activation within the left amygdala but not the right amygdala. Together, these findings paint a somewhat unclear picture of the possible lateralization of amygdala activation.
The amygdala has frequently been linked to alterations in BOLD-reactivity in individuals suffering from some form of psychopathology. Indeed, the list of psychiatric disorders to which task fMRI studies have now been able to link the amygdala is long and includes (but is not limited to) disorders such as social anxiety disorder (SAD; eg, 55,56; see Etkin & Wager
57
for a meta-analysis), schizophrenia (eg, 58
-60; see Anticevic et al.
61
for a meta-analysis), posttraumatic stress disorder (PTSD; eg, 62
-65; see Hayes et al.
66
for a meta-analysis), borderline personality disorder (BPD; eg, 67,68; see Ruocco et al.
69
for a meta-analysis), major depressive disorder (MDD; eg, 70
-72; see Groenewold et al.
73
for a meta-analysis), bipolar disorder (eg, 74,75; see Chen et al.
76
for a meta-analysis), intermittent explosive disorder (IED)77,78—and even Turner syndrome
79
and Alzheimer’s disease
80
. (We note that the meta-analyses of Etkin & Wager,
57
Anticevic et al.,
61
and Hayes et al.
66
also include Positron Emission Tomography [PET] studies; however, a full list of the included studies is presented in each meta-analysis in table-form, along with the imaging methodology that was employed in each incorporated study [ie, fMRI or PET].) Overall, this body of research suggests that hyperreactivity of the amygdala may be present in individuals suffering from disorders such as SAD, PTSD, BPD, bipolar disorder, or IED, whereas
It should be mentioned that while the above body of literature may seem consistent in its reporting of amygdala reactivity in response to emotion provocation, there are, in fact, many examples of studies that failed to record such an effect (eg, 81 -85). What is more, there are also many examples in which no significant difference in amygdala reactivity could be recorded when patients with a psychiatric disorder were compared to those without (eg, 86 -90). This indicates that activation of the amygdala via emotion provocation fMRI may not be as robust a phenomenon as often assumed in the literature. It is also important to point out that only 3 of the 66 research articles cited in this section had a sample size of N ⩾ 30 (median N = 12; IQR = 10-15 participants). Such small sample sizes may increase the risk of false positive findings and inflate effect sizes, especially when considering that the shadow of publication bias looms large over the overarching field of functional neuroimaging. 91
Amygdala Connectivity fMRI
In the early years of fMRI, most studies focusing on the amygdala only examined the region’s magnitude of BOLD-reactivity in task-based settings. From the mid-2000s and onwards, however, fMRI researchers became increasingly interested in (also) examining the connectivity patterns of the amygdala, following a broader trend taking place in neuroscience at the time. These studies can be (roughly) subdivided into the following 2 categories: (1) psychophysiological interaction (PPI) studies that have examined task-dependent effective connectivity during active task periods, and (2) functional connectivity studies examining task-free (ie, intrinsic) fluctuations of the BOLD-signal at rest (ie, resting-state functional connectivity [RSFC]). An overview of the insights garnered by these 2 research areas is provided in the following 2 subsections.
Amygdala PPI fMRI
PPI is a measure of effective connectivity designed to ascertain instances of communication between brain regions that only take place under specific task demands, as maintaining these connections might otherwise prove costly in terms of energy consumption. In PPI analysis, linear regression is used to test for an interaction between a physiological variable (ie, the time-series of a seed region) on the 1 hand, and a psychological variable (ie, the experimental task) on the other hand. If significant, the brain region expressing the interaction is said to exhibit context-dependent effective connectivity with the seed ROI. 92
PPI analysis was developed by Karl Friston and co-workers in the mid-to-late 1990s.93,94 Although the first PPI studies focusing on the amygdala date from the early 2000s, most of the work conducted in this field was actually published in the 2010s (see Figure 1, top-right panel). As before, the majority of these studies employed either a facial expression task (eg, 43,95 -99), or an emotional pictures task based on IAPS-photographs depicting non-facial objects or scenes (eg, 100 -104). In many of these studies, amygdala PPI was examined while participants were performing some form of emotion regulation training (eg, 100,104,105; see Berboth & Morawetz 106 for a meta-analysis). Furthermore, some of these studies were conducted in patients with (versus without) a psychiatric disorder—examples of which include schizophrenia107,108 (although see Fakra et al. 109 ), MDD, 110 bipolar disorder,111,112 PTSD113,114 (although see Van Rooij et al. 88 ), IED77,78 (although see Heesink et al. 90 ), BPD, 115 and generalized anxiety disorder (GAD).116,117 Still others report on the relationship between amygdala PPI and personality constructs such as trait neuroticism, 118 aggression, 77 and psychopathy. 119 Whatever the exact research aims, however, most of these studies converge on the same target regions exhibiting significant PPI with the amygdala during task performance. These regions include the early visual cortex (ie, Brodmann areas 17-19), fusiform gyrus—including the fusiform face area (FFA)—the anterior cingulate cortex (ACC) and insula; the 2 main constituents of the salience network, 120 the inferior frontal gyrus (IFG), orbitofrontal cortex (OFC), and dorsolateral prefrontal gyrus (DLPFC)—the last of which is considered to be a main constituent of the central executive network. 121 Other regions of the brain with which PPI research has often associated the amygdala include the MPFC (both its ventral and dorsal aspects) and the ventrolateral prefrontal cortex (VLPFC), as indicated by meta-analyses of the amygdala PPI literature by Smith et al., 122 Di et al., 123 and Berboth et al. 106 . Importantly, the results of these PPI studies are (largely) consistent with the patterns of task-based effective connectivity of a landmark study using structural equation modelling by Stein et al. 124 . (Note: 1 complicating factor in reviewing this literature is that authors tend to differ in their operational definitions of anatomical or functional brain regions. For instance, [part of] what is labelled as the ACC in 1 study, may instead be labelled as the MPFC or the OFC by others. This overlap should be kept in mind when reading both the sections on amygdala PPI and RSFC.)
Taken together, this body of research seems to support the notion that the activity of the amygdala is gated by executive control regions in the prefrontal cortex. Diminished coupling between these prefrontal regions and the amygdala might lead to the development of mental health issues, particularly those marked by emotion dysregulation problems. The amygdala’s PPI with the primary nodes of the salience network (ACC and insula) further support the region’s role in salience detection. Its effective connectivity with the fusiform gyrus is likely to reflect the amygdala’s role in facial processing.
Amygdala RSFC fMRI
Almost since the inception of fMRI, researchers were aware that spontaneous low-frequency fluctuations (<0.1 Hz) occur in BOLD-weighted data.
125
While initially (dis)regarded as noise, it was not until Biswal et al.
126
observed significant correlations between the resting-state signals of the left and right sensorimotor cortices that fMRI researchers truly began to take notice of these fluctuations, and realized that they are, in fact, of neuronal origins. Even then, however,
Biswal’s seed-based approach remains a very common way to assess RSFC today. The first RSFC studies to use this method to target the amygdala were published in the late 2000s/early 2010s (see Figure 1, middle-left panel). In a study that would become a major landmark in the literature, Roy et al. 128 characterized the patterns of amygdala-based RSFC in a (large) sample of healthy volunteers (N = 65), showing that the amygdala exhibits (1) positive RSFC with the hippocampus, parahippocampal gyrus, and superior temporal gyrus, as well as with medial prefrontal regions such as the ACC and (medial) OFC, a finding that is largely consistent with the amygdala’s purported role in associative learning; (2) positive RSFC with the insula (and ACC)—which again points towards its role in salience detection; (3) negative RSFC with (dorso)lateral regions of the prefrontal cortex, including the middle and superior frontal gyri, which is in line with a top-down (executive) control model on emotion regulation; and (4) negative RSFC with the precuneus/posterior cingulate cortex (PCC)—two main components of the “task-negative”, or default mode network. Following in the footsteps of Roy et al., 128 many others subsequently sought to chart the landscape of amygdala-based RSFC in populations suffering from psychiatric disorders, such as GAD (eg, 129 ), SAD (eg, 130,131), PTSD (eg, 132 -134; see Koch et al. 135 for a systematic review), MDD (eg, 136,137; see Tang et al. 138 for a meta-analysis), bipolar disorder (eg, 139,140; see Vargas et al. 141 for a systematic review), and to a lesser extent, schizophrenia.142 -144 In our own work, we conducted amygdala-based RSFC analysis in war veterans with versus without a IED, recording group differences only when applying a rather lenient threshold of significance. 145 Intriguingly, even though (task) activation studies on BPD were quite common in the heyday of emotion provocation fMRI, relatively little research has focused on the amygdala-based RSFC of this Axis II disorder. Possibly, the high degree of psychiatric comorbidity common in this population has prevented researchers from conducting RSFC fMRI research in BPD patients (eg, see Table 1 in Shafie et al. 146 ). Nevertheless, taken as a whole, this body of literature tends to show that the strength of many of the functional connections reported by Roy et al. 128 may be disrupted in psychiatric disorders marked by emotion regulation problems.
In 2001, Raichle and colleagues 147 at Washington University proposed the existence of a network of (primarily) midline brain structures that activates when not engaged by a specific task, based on observations in PET-data. A few years later, Greicius et al. 148 used Biswal’s seed-based approach to demonstrate that the BOLD-signals of these same brain regions are highly intercorrelated during rest. It is now clear that this default mode network (or DMN)—which mainly comprises the (dorsal) MPFC and PCC/precuneus—can routinely be extracted from resting-state fMRI data by using independent component analysis (ICA).149,150 The discovery of the DMN would mark the first of many large-scale connectivity networks to be uncovered (through ICA) over the years. Other well-known examples include the central executive network (CEN), a constellation of brain regions centering around the DLPFC and dorsal posterior parietal regions, 121 and the salience network (SN), which is anchored around the anterior insula and dorsal ACC. 120 Importantly, rather than (sub)serving any 1 function in particular, the activity and/or connectivity dynamics of these large-scale networks are purported to support a broad range of psychological faculties, operating in a much more domain-general fashion than often assumed by traditional views on brain functioning, which tend to focus on functional segregation (rather than integration). 151 For instance, the DMN has often been linked to functions that vary from remembering personal memories, to moral cognition and reasoning, and imagining the future, while the SN is associated with the detection of personally-relevant stimuli, be they internally or externally generated, of the emotional, cognitive, or social variety (or otherwise).120,151 The interaction (switching) between these networks is thought to give rise to complex phenomena such as emotion and cognition. Importantly, the amygdala is often considered to be a part of the SN,120,152 which on the 1 hand, fits well with its hypothesized role in salience detection, but on the other hand, somewhat trivializes the region’s importance, as when viewed from this angle, the amygdala is only a very small and non-central component of a much larger apparatus. It cannot be denied, however, that this (network-based) perspective does far more to consider the complex nature of brain functioning than do traditional small-scale circuit models on emotion processing. It also provides a viable explanation as to why much of the brain’s energy consumption actually takes place during the resting-state. 153
Amygdala Test-Retest Reliability fMRI
In the 2010s, researchers became increasingly interested in (re)evaluating the test-retest reliability of (amygdala) fMRI (see Figure 1, middle-right panel), perhaps prompted by the reproducibility crisis that was slowly making its way over from the psychological sciences.
91
This is not to say that earlier work had not already explored the retest reliability of amygdala fMRI to some extent. For instance, Johnstone et al.
154
recorded mostly poor test-retest reliability for task-evoked amygdala responses to neutral or fearful faces measured over 3 (scan) sessions separated by several weeks (most intraclass correlation coefficients [ICC’s] <.4), indicating low replicability at the subject-level—although ICC’s tended to be somewhat higher when averaging across runs
To our knowledge, no study has ever specifically examined the test-retest reliability of amygdala-based RSFC. By and by, however, research focusing on the general test-retest reliability of RSFC tends to record rather low ICC’s of seed-based connectivity metrics, with higher ICC’s often being observed for network-based connectivity measures. For instance, in a meta-analysis of all test-retest reliability studies conducted on seed-based RSFC conducted (up until that point), Noble et al. 165 recorded a mean ICC of only 0.29. In a systematic review conducted in that same study, Noble et al. 165 found that connections within the same connectivity networks were generally stronger, particularly those within the DMN or CEN. These latter results are largely consistent with a replicability study of networks extracted via ICA by Zuo et al., 166 although Wisner et al. 167 recorded rather lower internal consistencies of large-scale connectivity networks in another (similar) study—especially at the intra-subject level. Furthermore, although Noble et al. 165 did not explicitly target the amygdala in their systematic review/meta-analysis, they did observe that the seed-based connectivity of subcortical brain regions was relatively low when compared to cortical areas. Finally, 1 study by Nord et al. 168 showed that amygdala’s PPI with the DMPFC during emotion provocation exhibited good test-retest reliability at the intra-subject level (ie, most ICC’s close to or above 0.59). However, this last finding awaits further confirmation/replication.
Amygdala Real-Time fMRI Neurofeedback
Many of the milestones of fMRI research discussed thus far can trace their roots to proof-of-concept papers already published in the 1990s. Real-time fMRI (rt-fMRI) is no exception. 169 Initially, this branch of research focused primarily on optimizing methodological aspects such as online quality assurance and motion correction/realignment. The first studies to apply rt-fMRI in a neurofeedback setting were published in the early 2000s (see Weiskopf 170 for a historical overview).
Neurofeedback is a form of biofeedback that has participants receiving “live” and ongoing information on their own brain (re)activity, so that they may learn to gain volitional control over it. Applications of neurofeedback in humans were initially based on electroencephalography (EEG) recordings. However, due to the lack of localization precision and limited coverage of EEG, many researchers were keen to discover novel ways of administering brain-based biofeedback to their study participants. As mentioned, the first studies to deliver on this promise and successfully apply rt-fMRI neurofeedback were published in the early 2000s. One of these early rt-fMRI neurofeedback studies already targeted the amygdala. In that study, Posse et al.
171
provided their participants with real-time feedback on amygdala activation in order to (successfully) augment neutral or sad feelings in their subjects via pictures of (corresponding) facial expressions. The majority of amygdala neurofeedback studies was published roughly ten years after this initial report by Posse et al.,
171
in the 2010s (see Figure 1, lower panel). Invariably, the express goal of these later studies was to employ neurofeedback in order to reduce the amygdala’s responsiveness to stimuli that were explicitly
With the exception of 1 study by Brühl et al., 180 who described the use of a facial expression task, most other neurofeedback works cited here used either IAPS stimuli (eg, 181,183,184), autobiographical (happy) memories (eg, 173,176,179,182), or personalized (trauma) words 187 to elicit an emotional amygdala response in their study participants. This is a bit surprising, given the predominance of facial expressions in other areas of amygdala fMRI. In some of these inquiries, only healthy volunteers were included,179 -183 while in others, patients suffering from psychiatric disorders, such as MDD173,174,176,178 (see Young et al. 193 for a review), PTSD,172,175,186 -188,190 or BPD 184 (also) participated. Importantly, in all this work, at least some degree of self-regulation success was reported by the authors, with the observed (and expected) direction of effect (ie, up- or down-regulation) depending on task instructions. Insofar as clinical populations were recruited, successful BOLD regulation was frequently associated with significant reductions in symptom self-report questionnaire scores (eg, 172,174,177,178,184). One study even recorded an increase in hippocampal volume one-to-two weeks after self-regulation training of the amygdala’s BOLD activity in a sample of PTSD patients. 190 Overall, these findings tend to show that it is possible to gain volitional control over the amygdala’s activity via rt-fMRI neurofeedback, and that significant (clinical) improvements in emotion regulation may follow after (successful) amygdala neurofeedback training. 189
Finally, some of the rt-fMRI neurofeedback work discussed thus far has also examined the amygdala’s connectivity with other brain regions either during active feedback runs (ie, via PPI),181,184,185,187 or immediately thereafter, during rest.173,175,176,184 Overall, this (small) body of research tends to report an increase in RSFC of the amygdala with regions such as the VLPFC, DLPFC, ACC, PCC, and precuneus, from pre- to post-training, with more variable effects being recorded in the hippocampus and parahippocampal gyrus. With the addition of the insula and ventral- and dorsal MPFC, largely these same brain regions have been associated with an increase in amygdala PPI during active neurofeedback runs. Interestingly, 1 study based their neurofeedback on task-evoked effective connectivity between the DMPFC and amygdala, rather than the magnitude of the amygdala’s BOLD responsivity, 194 showing effects similar to the other works discussed here. In sum, these are largely the same brain regions for which amygdala-based connectivity effects were recorded previously, in the literature described in sections “Amygdala PPI fMRI” and “Amygdala RSFC fMRI.”
Integration
In this review, an overview was presented of the major developments that have occurred within the field of amygdala fMRI since its first appearance in 1996. We note that the body of literature discussed here should be considered as a general overview, and that it by no means is meant to be exhaustive; wherever possible or relevant, we have referred to other (systematic) reviews and meta-analyses for further reading. We also wish to emphasize that the conclusions drawn here are based entirely on our review of the fMRI literature, inspired by our personal experiences in that field; it does not cover preclinical (ie, animal) work on the amygdala’s role in emotion processing. For an overview of that line of research, we refer the reader to other sources (eg, Maren & Holmes 195 ). To visually complement the narrative provided here, a timeline of all the major landmark papers cited throughout this manuscript is presented in Figure 2.

Timeline depicting some of the major landmark publications in amygdala fMRI research, color-coded by theme.
To recapitulate what we have discussed here: In section “Introduction,” we saw how early work in animal models, particularly within the sphere of Pavlovian fear conditioning, has shaped much of our way of thinking about the neurobiology of emotion processing. Next, we saw that soon after the first two amygdala fMRI publications, a veritable goldrush of studies ensued that similarly explored the amygdala’s BOLD response to emotional stimuli, most of which used either facial stimuli or IAPS photographs. Several key findings stand out from this literature: First, although many studies reported an increase in amygdala activation in response to emotional stimuli, there are also quite a few examples in which no such effects could be recorded. Second, as the years went by, the general consensus gradually shifted towards a frame of reference in which the amygdala was no longer seen as a brain region devoted specifically to the processing of emotional information, but more as an area involved in the detection of salience in a much broader sense. This idea resonated well with observations of higher activation magnitudes when using non-emotional
So, what part (if any) can amygdala fMRI still play in the current landscape of the affective neurosciences? For 1 thing, if the amygdala is indeed just a small portion of a much larger network devoted to salience detection, and if the activity of the amygdala cannot be measured reliably at the single-subject level, than further targeting only the amygdala via rt-fMRI neurofeedback might be ineffective. Indeed, if the explicit purpose of such an approach is to facilitate emotion processing by influencing how the brain processes potentially salient information, than according to the literature reviewed here, some measure of clinical efficacy might only be expected if at least some of the other brain regions within the same salience network (ie, the anterior insula and/or ACC) are targeted as well. Even then, however, the poor test-retest reliability of amygdala fMRI suggests that the method may not (yet) be viable for real-time neurofeedback applications. How then should we interpret the body of amygdala rt-fMRI neurofeedback research reviewed in section “Amygdala real-time fMRI neurofeedback”? To answer this question, it is perhaps best to quote Thibault et al.,
197
who’s critical assessment of the (larger) rt-fMRI neurofeedback literature aligns well with our own observations:
Whatever the precise function of the amygdala may be, however, the reliability of its task-induced BOLD reactivity, ie, insofar as emotion provocation fMRI is concerned, is at present simply too low to warrant its reputation as a robust single-subject neuroimaging biomarker of emotion processing. Interestingly, a recent overview of the most common 3 Tesla scanning protocols used in amygdala fMRI, suggests that methodological aspects such as the type of scan sequence, the spatial resolution (voxel size), imaging plane (axial, coronal, sagittal), brain coverage, scan time, and type of radiofrequency coil used, can all significantly impact the quality of the data (Foster et al. 198 ). These observations highlight the possibility that suboptimal imaging parameters, along with other methodological details, may lie at the root (at least partly) of the reliability-related issues amygdala fMRI currently faces. We certainly do not discount this possibility. Hence, we encourage future research efforts to take (even) further steps to identify the optimal scan parameters for adequately imaging the amygdala in vivo.
As mentioned above, our review of the amygdala fMRI literature is by no means meant to be exhaustive; rather, it is intended as a broad-strokes historical overview of the major themes in the field of amygdala fMRI. There are several lines of fMRI research that we have not discussed here that, although important in their own right, have had—at least in our opinion—somewhat less of an impact on the field overall. Examples of these include the effective connectivity of the amygdala as measured via Granger causality (eg, see Liao et al. 199 ) and/or dynamic causal modeling (DCM; eg, see Sladky et al. 200 ), the dynamic functional connectivity of the amygdala (eg, Cisler 201 ), which measures changes in intrinsic connectivity that occur over shorter periods of time, and the (fractional) amplitude of low frequency fluctuation ([f]ALFF) of the amygdala (eg, Sato et al. 202 )—a measure of the magnitude of spontaneous (versus task-based) fluctuations in the BOLD-signal. In addition, we re-emphasize that the current work does not cover the developments that took place over the years in preclinical/animal research on the role of the amygdala in emotion processing. Although such work was instrumental for providing much of the foundation on which the field of amygdala fMRI was eventually able to flourish 5 —a point we already highlighted in section “Introduction”—the preclinical field has since progressed—largely independently—in its own disparate directions. We do note, however, that the overall picture emerging from the preclinical/animal literature is, by and large, mostly compatible with a domain-general role of the amygdala in salience processing as described here (eg, see Maren & Holmes, 195 McEwen et al., 203 and Zhang et al. 204 ; see also Koen et al. 205 for a more translational perspective).
We started this review by reiterating the question Merboldt et al.
1
posed in the title of their commentary: “Functional MRI of the Human Amygdala?”. In answer to this question: If within the confines of emotion processing, than barring some justified exceptions, it is perhaps time we set our sights towards a new—or at least, a broader—horizon, 1 in which the intricacies of emotion processing are understood as the complex interplay between entire constellations of interacting brain regions, rather than any single brain region acting more or less in isolation. The amygdala simply does not respond selectively—or even very reliably—to emotional content; it responds to all things that are new, exciting, threatening or otherwise relevant to an individual at any given time of day, and does so as part of a much larger network devoted to that same purpose. In our opinion, these are important conclusions to draw from roughly 30 years of fMRI research on the amygdala’s role in emotion processing. We hope that further improvements in imaging equipment and methodology will help amygdala fMRI to finally fulfill its long-standing promise to the broader field of affective neuroscience. In the meantime, however, we are forced to reassess the possible therapeutic efficacy of rt-fMRI neurofeedback training regimens that target
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Dutch Ministry of Defence.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
