Abstract
Parkinson’s Disease (PD) occurs as a result of the progressive loss of dopaminergic neurons within the substantia nigra causing motor and non-motor symptoms and has become more prevalent within the last several decades. With mitochondria being essential to cellular survival, mitochondrial dysfunction contributes to the disease progression by increasing neuron loss through (1) insufficient ATP production and (2) reactive oxygen species generation. MicroRNAs (miRNAs) are small molecules located throughout cells that regulate gene expression, particularly mitochondrial function. Through their own dysregulation, miRNAs offset the delicate balance of mitochondrial function by altering or dysregulating the expression of proteins, increasing neuroinflammation, increasing retention of toxic substances, limiting the removal of reactive oxygen species, and preventing mitophagy. Improper mitochondrial function places cells at increased risk of apoptosis, a major concern in individuals with PD due to their reduced number of dopaminergic neurons. This article has identified the 17 most promising mitochondrial associated miRNAs within PD: hsa-miR-4639-5p, miR-376a, miR-205, miR-421, miR-34b/c, miR-150, miR-7, miR-132, miR-17-5p, miR-20a, miR-93, miR-106, miR-181, miR-193b, miR-128, miR-181a, and miR-124-3p. These miRNAs alter mitochondrial function and synaptic energy by impeding normal gene expression when up or downregulated. However, there is limited research regarding mitochondria-localized miRNAs that are typically seen in other diseases. Mitochondria-localized miRNA may have a greater impact on mitochondrial dysfunction due to their proximity. Further research is needed to determine the location of these miRNAs and to better understand their regulatory capabilities on mitochondrial and synaptic function within PD.
Parkinson’s Disease
A neurodegenerative disorder that presents with reduced mobility and dementia among the elderly, Parkinson’s Disease (PD) has rapidly become the focus of many researchers. The increased average age of an individual combined with exposure to a multitude of factors (ie, pesticides, air and water pollutants, chemicals, and toxins), increases the risk for many diseases, including PD. 1 The combination of an aging population and increased exposure to environmental toxins has drastically increased the prevalence of several diseases, specifically PD. Approximately 6.1 million individuals have been diagnosed with PD in the world. 2 A neurological disease that has quickly become a major economic and social burden, PD is characterized by the loss of dopaminergic neurons in the substantia nigra. 3 The substantia nigra is essential for dopamine production and movement control, which corresponds with the typical symptoms of PD. 4 The classic presentation of PD includes both motor and non-motor symptoms: tremors at rest, depression, anxiety, cognitive impairment, reduced movement, poor balance, sleep irregularities, orthostatic hypertension, and gut imbalances.5,6 PD is typically seen in older populations, which is concerning due to the increasing age of the average individual. While PD is more common in males than females, the risk for both increases with age. 6 This may also arise from males tending to have greater exposure to probable risk factors than women, due to gender disparities within the workforce. 1 Other possible risk factors include family history, genetic factors, pesticide exposure, milk consumption, hydrocarbon solvent exposure, high iron intake, metal exposure, and industrial byproducts.1,3 The major risk factors for PD are described in Figure 1.

The most common risk factors of PD. This figure depicts the most common factors that are attributed to the development of PD, which includes: genetic factors, toxins, pesticide exposure, air and water pollutants, aging, family history, being a male, chemical exposure, and hydrocarbon solvent exposure.
Pathology of Parkinson’s Disease
There are 2 recognized forms of PD: familial and sporadic. The familial form is thought to originate from genetic mutations, while the sporadic form is thought to emerge from a combination of environmental factors. 5 Both forms of PD present similarly but distinguishing the 2 is made problematic due to the difficulties of diagnosing PD itself. Additionally, the lack of a single biomarker that can gage the extent of the disease and its slow progression makes it incredibly challenging to differentiate PD from other neurodegenerative diseases. 1
Diagnosis typically occurs after the onset of symptoms, in a patient who has fallen and is admitted to the hospital. 6 Many of the symptoms occur after 60% to 70% of dopaminergic neurons have been lost. 3 Neuronal loss is suspected to be related to abnormal alpha-synuclein aggregation which causes neurotoxicity and eventually leads to degeneration of dopaminergic neurons. 7 Alpha-synuclein is an insoluble protein that constitutes a major component of Lewy Bodies. 8 The consequences of Lewy Body formation are dependent upon the onset of dementia. Dementia that occurs 1 year after the development of motor symptoms indicates Parkinson’s Disease Dementia, while an earlier onset indicates Lewy Body Dementia. Lewy Body Dementia presents with neuropsychiatric symptoms such as hallucinations, apathy, and depression. 9
The initial cause for the accumulation of alpha-synuclein is presently a target of researchers with microRNAs (miRNAs) and mitochondrial dysfunction being heavily implicated. Other probable causes of neuronal loss include oxidative stress, inflammation caused by unregulated overactive immune cells, or endogenous neurotoxins.5,10 Understanding the pathologic blueprint would help treat PD before the development of symptoms and substantial neuronal loss. The current understanding of the development of PD is the Braak hypothesis, which consists of 6 stages, depicted in Figure 2. 5
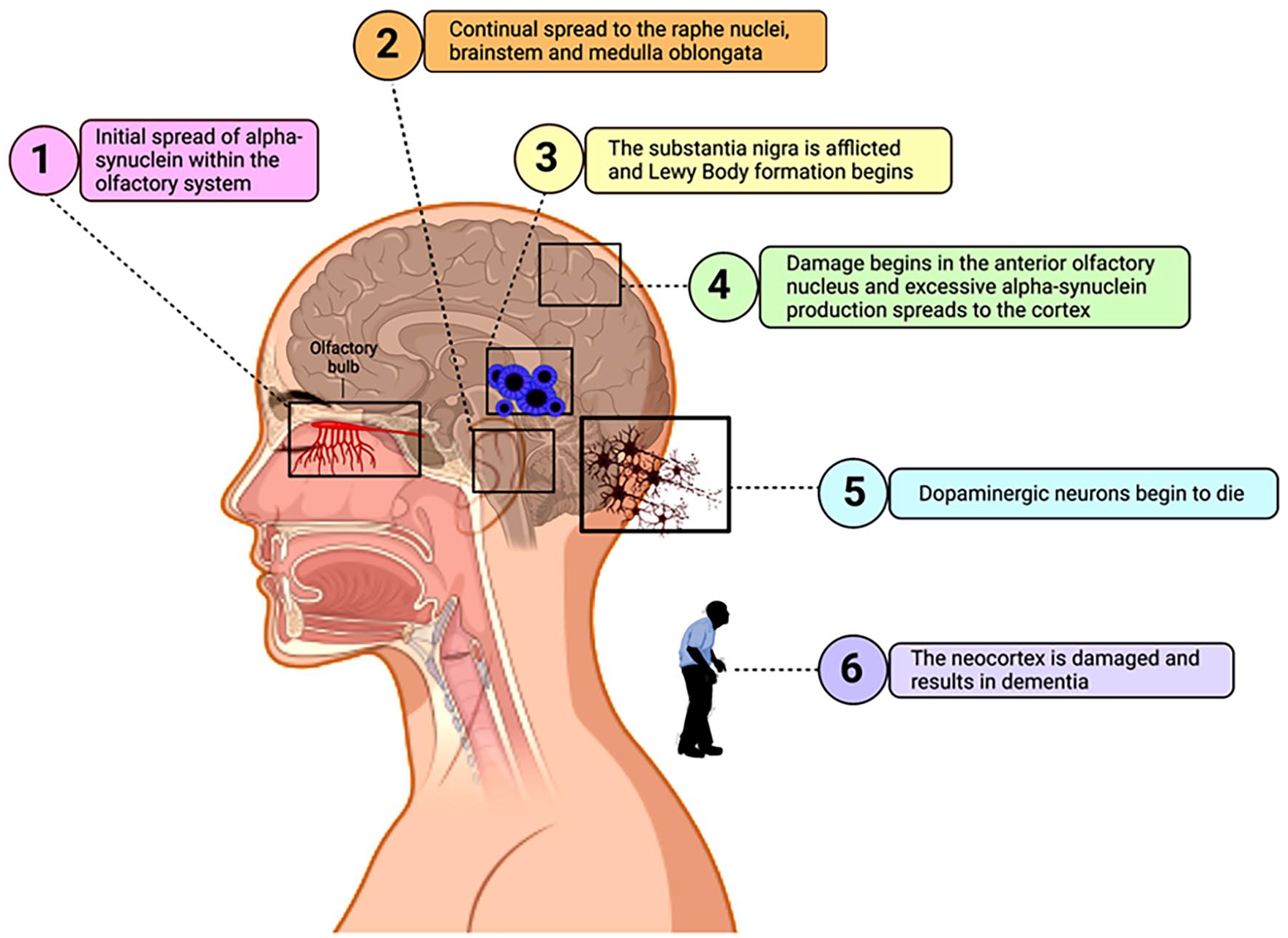
Progression stages of PD. The 6 stages of PD are numbered to match the corresponding stages of the Braak Hypothesis. Stage 1: Initial spread of alpha-synuclein within the olfactory system caused by unknown circumstances. Stage 2: Continued spread to the raphe nuclei, brainstem, and medulla oblongata. Stage 3: The substantia nigra is afflicted and Lewy Body formation begins. Stage 4: Damage begins in the anterior olfactory nucleus and excessive alpha-synuclein production continues to spread reaching the cortex. Stage 5: Dopaminergic neurons begin to die. Stage 6: The neocortex is damaged and results in dementia. 5
Current treatment of PD involves a dopamine prodrug, Levodopa, to replace the lost dopamine. Unfortunately, as the disease progresses, individual responses to the medication decrease and other forms of therapy must be pursued. 6 Other alternative medications include dopamine agonists: pramipexole, bromocriptine, and apomorphine. 3
Mitochondria
The mitochondrion is an essential cellular organelle. They reproduce through cycles of fission and fusion, are highly scrutinized for quality control, contain their own DNA and help with apoptosis. 11 Both mitochondrial fission and fusion preserve mitochondrial functions by repairing damage caused by normal mitochondrial functions that generate harmful substances, including free radicals. Mitochondrial fission promotes the equal division of mitochondria into 2 daughter cells produced during cellular reproduction and removes damaged mitochondria via autophagy, preventing replication of damaged mitochondria. Mitochondrial fusion, induced by Bcl-2 and Bak, permits the rearrangement of the mitochondrial membrane to merge the inner and outer membranes of 2 individual mitochondria to facilitate repair and gene product transfer between the 2 mitochondria. This process also utilizes Mfn1, Mfn2, and Opa1 proteins to conduct gene product transfer. While fission and fusion allow the preexisting mitochondria to be divided amongst daughter cells, phagocytose damaged mitochondria and repair damaged mitochondrial membranes, replicating mitochondria requires a process called biogenesis. Mitochondrial biogenesis creates new mitochondria by replicating the mitochondrial genome, a transcript for 13 essential electron transport chain proteins, and the appropriate mitochondrial genes stored in the nucleus of the cell. 12
Mitochondria produce energy, in the form of ATP (adenosine triphosphate), through respiration and oxidative phosphorylation. ATP is synthesized by creating an electrochemical proton gradient using the 4 mitochondrial Electron Transport Chain complexes and the electrons generated from oxidative phosphorylation. During this process, reactive oxygen species are produced by complexes 1 and 3 and are used by macrophages and neutrophils to eliminate bacteria.13,14 While other enzymes also produce reactive oxygen species, their mechanisms are less well-known. 13 Additionally, normal processes of the mitochondria generate reactive oxygen species, as products or byproducts, that are released by mitochondrial permeability transition pores. 15 Mitochondria possess several beneficial functions that are essential to cell survival. These functions can also cause cell death under different circumstances. It is a delicate balance between beneficial mitochondrial function and detrimental mitochondrial dysfunction.
Mitochondrial Dysfunction in Parkinson’s Disease
Mitochondrial dysfunction can be caused by a combination of genetic and/or environmental factors and contribute to the progression of PD. Aging is often considered a mitochondrial disease as the mitochondrial genome acquires mutations over a lifetime that provoke neurodegeneration. 16 These acquired mutations eventually result in mitochondrial dysfunction, ultimately destruction and cell death. Mitochondrial dysfunction affects the high energy demanding tissues or tissues with high energy demands or requirements, the brain, and muscles. 16 In individuals with PD, these mutations can have a devastating impact. Mitochondrial dysfunction may arise from any number of causes, individually or in concert, including oxidative stress and disorganized mitophagy.
Mitochondrial dysfunction permits complexes 1 and 3 to produce excessive reactive oxygen species causing oxidative stress and damage to the mitochondrion and the cell, furthering the development of PD. 13 In addition to complexes 1 and 3, alpha-glycerophosphate dehydrogenase also produces reactive oxygen species and contributes to overproduction. 15 An abundance of reactive oxygen species is the result of disproportionate reactive oxygen species removal and reactive oxygen species production. 15 Extensive oxidative stress damages the mitochondrial DNA and dramatically reduces the mitochondria’s capability to perform quality control and leading to neuronal death. 5 Furthermore, Subramaniam and Chesselet 14 discusses deficiencies of complex 1 in the frontal cortex due to repeated exposure to oxidative stress. Frequent increases in reactive oxygen species lead to mitochondrial destruction by a reactive oxygen species burst. 15 Interestingly, alpha-synuclein accumulation increases sensitivity to oxidative stress. 14 This may be in direct relation to the mitochondrial proteins that regulate alpha-synuclein also acting as a neuroprotective barrier to oxidative stress.
Another potential mechanism for mitochondrial dysfunction is disordered mitophagy, often caused by gene mutations. PINK1/Parkin, leucine rich repeat kinase 2 (LRRK2) and alpha-synuclein help to regulate mitophagy, however, in patients with PD, these molecules are usually mutated.17,18 PINK1 and Parkin together impact on the health status of mitochondria and initiate mitochondrial degradation. 17 Reduced mitophagy impairs cellular function by permitting diseased mitochondria to wreak havoc on the cell. PINK1 and Parkin remove mitochondria with impaired mechanisms, which can be induced by alpha-synuclein accumulation. 18 Alpha-synuclein contributes to the impairment of mitochondrial import by opening mitochondrial permeability transition pores, depolarizing the mitochondrial membrane, and facilitating mitochondrial DNA damage, eventually causing apoptosis. 18 In addition to PINK1 and parkin, LRRK2 contributes to the progression of PD. Overexpression of LRRK2 results in neurotoxicity, phosphorylation of GTPases, and possibly disruption of organelle trafficking.19,20
Mitochondrial death and dysfunction contribute to PD progression as they are undeniably fatal to the cell. While researchers have achieved a level of understanding about neuronal death caused by mitochondrial death, it was unclear as to why only dopaminergic neurons were targeted. miRNA may be the answer to this question.
MicroRNAs
MicroRNA molecules are small non-coding RNAs that assist in the regulation of gene expression by degrading or stabilizing the target mRNA. 7 miRNAs are produced through a cellular process known as miRNA biogenesis. The genomic precursors of miRNA are found in either intergenic or coding-intronic sequences within the genome. These sequences are then transcribed by 1 of 2 nuclear enzymatic proteins: RNA polymerase 2 or RNA polymerase 3. RNA polymerase 2 primarily transcribes miRNA which oversees the process of DNA transcription into primary mRNA, while RNA polymerase 3 transcribes primary miRNA which keeps the cell cycle and cell growth in check. After transcription of the primary miRNA, the first cleavage occurs within the nucleus, producing precursor miRNA. To prevent degradation within the nucleus and transportation into the cytoplasm, the precursor miRNA forms a protein complex with the nucleocytoplasmic transporter factor Exportin-5 and RanGTP. Once the precursor miRNA is in the cytoplasm, it is cleaved, once again, into a miRNA duplex. The miRNA duplex is then cleaved a third time by Dicer into single-stranded mature miRNA molecule. This cleaving process does not require ATP; however, the release of the single-stranded miRNA molecules may require ATP. The miRNA forms a protein complex with RISC, an RNA-induced silencing complex, allowing it to regulate the target mRNA. 20 A brief miRNA biogenesis pathway is depicted in Figure 3. The use of enzymes also utilized in DNA replication allows for the regulatory factors from DNA replication to be used for miRNA biogenesis to regulate expression levels. Further regulation of miRNA biogenesis is conducted via regulatory proteins including: HnRNPA1, SMAD1, SMAD5, and Lin 28. Lin 28 binds specific miRNA and promotes its degradation. 20
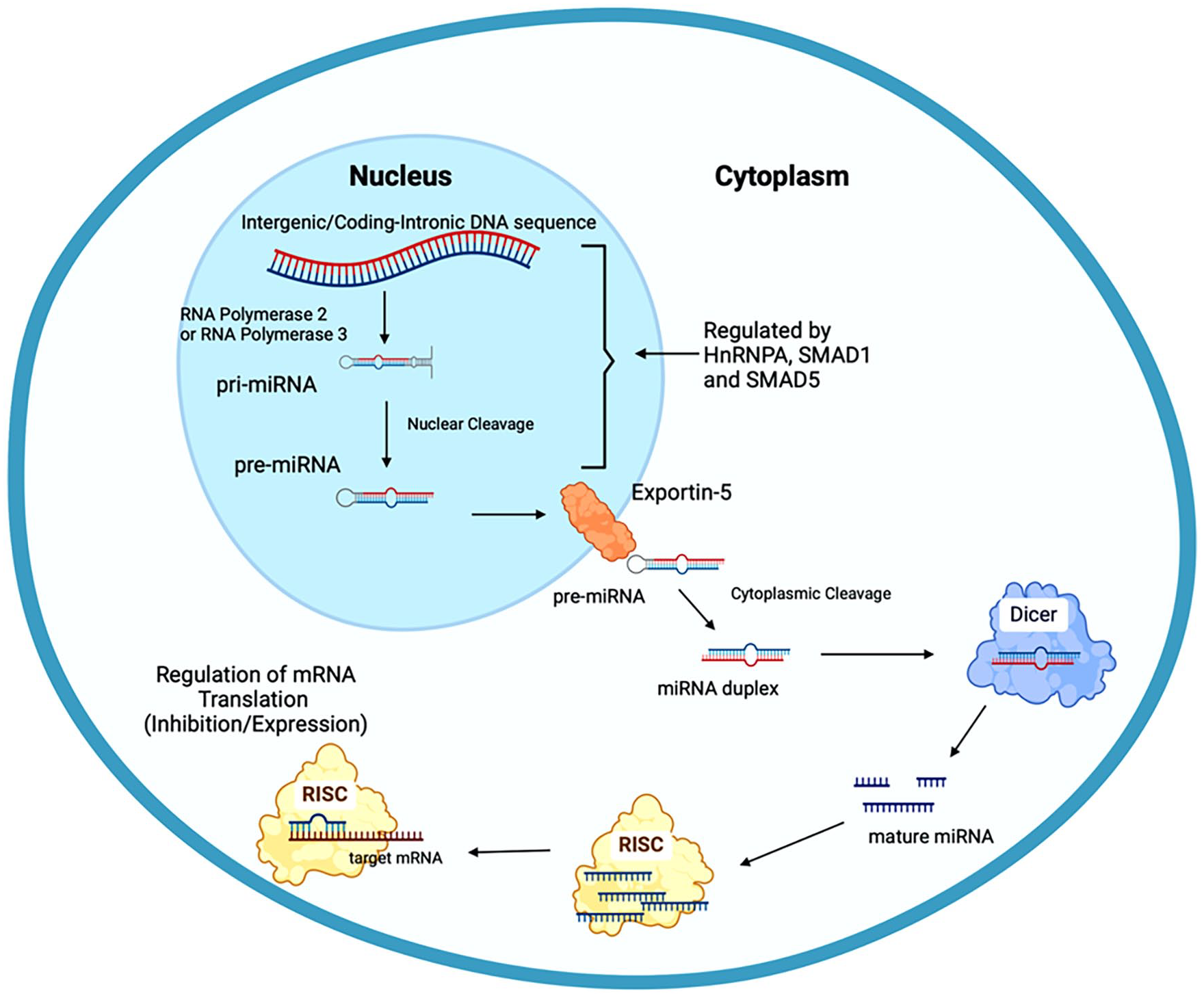
The miRNAs biogenesis pathway. miRNA biogenesis begins with the transcription of the intergenic or coding-intronic DNA sequence by RNA polymerase 2 or RNA polymerase 3 into primary miRNA. This pri-miRNA is then cleaved into precursor miRNA, within the nucleus before being exported by Exportin-5 into the cytoplasm, where it is cleaved again into a miRNA duplex. The miRNA duplex is then cleaved by Dicer into single stranded miRNA, before forming a complex with the RNA induced silencing complex (RISC), allowing it to regulate mRNA translation.
Within PD, miRNA expression is altered in the brains of individuals who have been diagnosed with PD. Aberrant miRNA expression is often the result of stressors, oxidative, physiological, pathogenic, or psychological stressors, leading to the dysregulation of genes related to PD. 5 miRNAs aggravate the symptoms by manipulating the genes to produce more or less of their target products. For instance, miR-181 is upregulated in PD and increases the alpha-synuclein levels, despite miR-181 being neuroprotective and preventing alpha-synuclein accumulation in healthy individuals. 10 Researchers are currently looking into utilizing miRNAs as a form of therapy. 7
In addition to PD, miRNAs are being explored as potential therapeutic agents in other disorders including numerous cancers, neurologic disorders, cardiovascular disease, sepsis and many more. This is related to the advantages of miRNA compared to other molecular treatment or surveillance methods. miRNAs are an ideal therapeutic agent as they are small and may potentially cross the blood brain barrier with some assistance and direction. They are highly accessible, specific and sensitive molecules. miRNA can be found in most bodily fluids and discriminates against various stages of cancer and neurodegenerative diseases. Furthermore, the existing technology is sufficient to detect miRNA and reduces the economic burden of researching and developing new techniques and technology. One major limitation of utilizing miRNA is determining accurate values for the normal and diseased states. Current research depicts a wide range of values proposed due to limited sample sizes. 21
Mitochondria-Associated miRNAs
MicroRNAs have significant impacts throughout the body. While other RNAs may have a role in the pathogenesis or the dysregulation leading to PD, this article focuses on miRNA. These particular miRNAs were chosen as they demonstrated potential usage in the diagnosis, prevention, or treatment of PD through a mitochondrial pathway. In particular, the focus is on mitochondria-associated miRNA- miRNA located within the cytoplasm and peripheral circulation that alters gene expression affecting mitochondrial function. These miRNAs have been summarized in Table 1.10,22 -33 They can be up or downregulated causing mitochondrial functions to become imbalanced and ultimately destroying the mitochondria and cells. Figure 4 shows the most potential mitochondria-associated miRNAs and their roles in mitochondrial events in relation to PD.
Details of mitochondria associated miRNAs in Parkinson’s disease.

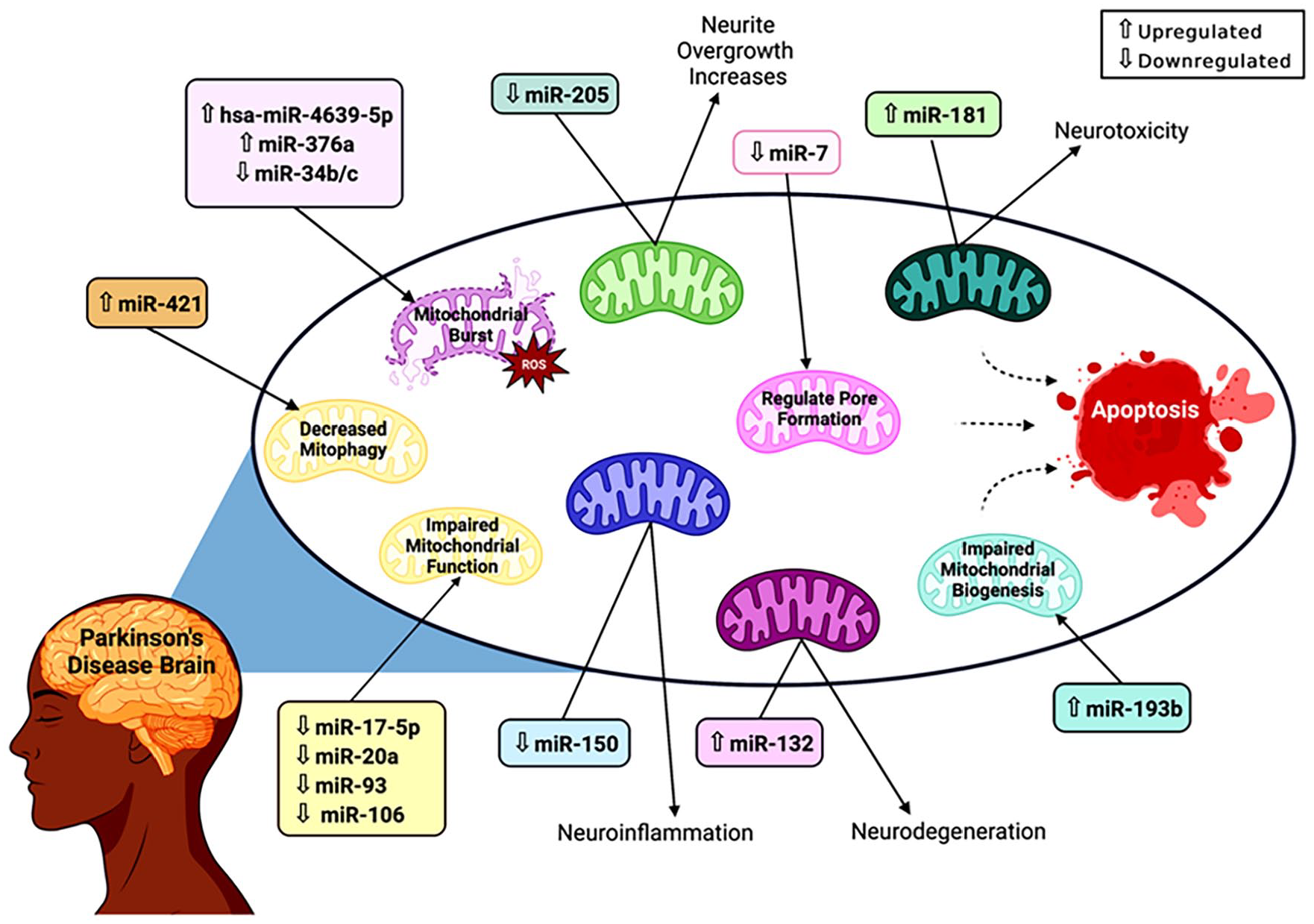
Impact of mitochondrial-associated miRNAs on mitochondrial function in PD. miRNAs impact mitochondrial function through a variety of mechanisms. Upregulated miR-421 and miR-193b decrease mitophagy and impair mitochondrial biogenesis, preventing the removal and replacement of disordered mitochondria. Some miRNAs, including hsa-miR-4639-5p, miR-376a, and miR-34b/c, increase the production of reactive oxygen species, resulting in a build-up of toxins that leads to a mitochondrial burst. miR-7 is downregulated in PD and prevents the mitochondrial permeability transition pore from opening. Other miRNAs have indirect effects that aid the progression of PD. Downregulated miR-205 increases neurite overgrowth, possibly contributing to the loss of dopaminergic neurons. miR-181 and miR-132 are both upregulated and cause neurotoxicity and neurodegeneration, respectively, by altering mitochondrial functions. miR-150 is downregulated and indirectly causes neuroinflammation in individuals with PD. However, the mechanism of some miRNAs is still unknown, with miR-17-5p, miR-20a, miR-93, and miR-106 all being downregulated and contributing to the impairment of mitochondrial function. The effects of these miRNAs individually are devastating to the mitochondria and the cell, but the culmination of their effects, as seen in Parkinson’s Disease, results in extensive mitochondrial destruction and cell death.
Mitochondria Associated miRNAs and Oxidative Stress in the Progression of Parkinson’s Disease
Normal byproducts of aerobic metabolism, Reactive Oxygen Species (ROS) are molecular compounds that are neutralized by antioxidants. 34 Under pathogenic circumstances, the balanced ROS production and elimination is disrupted leading to an accumulation of ROS within the cell. Elevated levels of ROS generate oxidative stress that manifests as cell damage and ultimately cell death. The characteristic feature of oxidative stress-induced cell death is a ROS burst. 35 While the exact mechanism of dopaminergic neuron death in PD has not been uncovered, oxidative stress may be a part of the answer. Researchers have investigated the possibility of miRNA altering the balance of ROS production and elimination to cause cell death.
Three independent research studies discovered that dysregulation of their respective miRNAs resulted in the dysregulation of ROS production and elimination. Baghi et al 23 collected samples from individuals grouped into 3 categories: healthy, early stage, and advanced stage based on their level of disability. Disability was defined as limiting movement by the authors of this study. The early stage was defined as minimal or no functional disability without an impairment in walking and the advanced stage was defined as mild to severe disability impaired postural reflexes and being able to stand independently. Individuals in the healthy category were age-matched to the individuals in the other categories. A total of 58 individuals participated in the study and provided a sample of venous blood. The researchers isolated peripheral blood mononuclear cells within 2 hours of sample collection, which were then further explored utilizing to MPP+, a neurotoxin. MPP+ induced toxicity within SHSY-5Y cell line. In addition to studying the expression of miR-376a, the researchers also measured the levels of intracellular ROS using fluorescent dye dichlorofluorescein-diacetate. The upregulation of miR-376a was significantly higher in the samples acquired from individuals in the advanced stage, followed by the early stage. ROS production was increased in the PD cells compared to the control group. Further research illustrated that the upregulation of miR-376a suppressed PGC1-α, TFAM, and GSK2-β. Typically, these 3 genes are protective against oxidative stress. PGC1-α regulates mitochondrial responses to increased ROS production and is neuroprotective at high levels. TFAM is increased in response to oxidative stress to stabilize and protect mitochondrial DNA and helps regulate apoptosis in neurons. The suppression of these targets prevents the protective mechanisms from reducing ROS accumulation and should be attributed to miR-376a dysregulation. These findings implicate miR-376a in ROS production and oxidative-stress-induced cell death. 23
The dysregulation of miR-34b and miR-34c were also investigated for their role in increased ROS production. Miñones-Moyano et al 26 collected amygdala and frontal cortex samples 3 to 11 hours postmortem and categorized the samples into 3 groups: control (without neuropathological lesions), incidental PD (not diagnosed with neuropathologic lesions), and PD (diagnosed with neuropathologic lesions). The investigators utilized RT-PCR to analyze miR-34b/c levels in the samples and found that both categories of PD samples contained significantly downregulated miR-34b/c. After determining the downregulation of miR-34b/c, the researchers transfected SHSY-5Y cells with miR-34b/c inhibitors and found that the cells exhibited a decreased ability to reduce ROS accumulation in comparison to the control cells. Based on predictive databases and a literature search, the authors concluded that Parkin, DJ-1 and PINK1 are potential indirect targets of miR-34b/c that alter the balance of ROS production and elimination. 26
Another study collected plasma samples from 439 individuals who were either diagnosed with PD, essential tremors, or were healthy. Using qRT-PCR, the researchers were able to determine 50 miRNAs that were significantly dysregulated in the PD samples compared to the healthy controls. Of the 50 miRNAs, hsa-miR-4639-5p was selected for its potential regulatory effect on DJ-1. DJ-1 protects against oxidative insults and ROS bursts, especially when levels are elevated. Based on these predictions, SH-SY5Y cells were transfected with either hsa-miR-4639-5p or a hsa-miR-4639-5p inhibitor to determine the impact of upregulating hsa-miR-4639-5p on DJ-1. The researchers found that upregulation of hsa-miR-4639-5p causes repression of DJ-1, thus leading to an increased likelihood of cell death by ROS burst. 22
The compilation of these studies facilitates further understanding of the etiology and progression of PD. miRNA dysregulation alters the balance of ROS production and elimination, leading to neuronal cell death via oxidative stress. Currently, PD is diagnosed based on the symptoms that occur after the majority of dopaminergic neurons are lost. This limits treatment to symptomatic relief. However, dysregulation of miRNA occurs in advance of symptom presentation, which allows for a diagnosis that is independent of symptoms. miRNA would serve as a serum biomarker that can be conducted as part of routine bloodwork. Additionally, the regulatory mechanisms of miRNA could potentiate a treatment by correcting the dysregulated miRNA, effectively preventing dopaminergic neuron loss before symptoms appear.
Mitochondria Associated miRNAs and Neurotoxicity
In addition to oxidative stress, another potential factor contributing to dopaminergic neuron loss is miRNA dysregulation which facilitates neurotoxicity. Cells regularly undergo apoptosis when normal function is impaired. This prevents the continued reproduction of dysfunctional cells. Neurotoxicity is described as increased apoptosis in neuronal cells as a result of changes in cellular function. Apoptosis is initiated by the release of cytochrome c from the mitochondria making apoptosis a mitochondrial process and dysregulation of apoptosis is typically due to mitochondrial dysfunction. miRNAs suppress and promote the production of genes or genetic products that prevent neurotoxicity in healthy individuals. In individuals with PD, miRNA dysregulation leads to disruptions in genes and genetic products. These changes permit the development of factors resulting in neurotoxicity and the characteristic increases in apoptosis of neuronal cells.
Several studies have associated neurotoxic dopaminergic neuron loss with miRNA dysregulation. Zhang et al 33 focused on the expression of miRNA-124-3p in cerebrospinal fluid and serum samples of 92 PD and 95 control patients. miR-124-3p was determined to be downregulated in the PD patient samples using qRT-PCR. 33 Normal levels of miR-124-3p suppress circRNA homeodomain interacting protein kinase 3 (circHIPK3), thus preventing neuroinflammation through the miR-124-3p/STAT3/NLRP3 signaling pathway. In addition to the downregulation of cicrHIPK3, determined by qRT-PCR, miR-124-3p also regulates signal transducer and activator of transcription 3 (STAT3), which controls microglial inflammation. The activation of STAT3 can interact with NLRP3 and increase neuronal apoptosis and dopaminergic neuron death. In a healthy individual, miR-124-3p regulates the expression of cicrHIPK3, preventing neuroinflammation. In individuals with PD, miR-124-3p is downregulated allowing cicrHIPK3 to be overexpressed and enhancing neuroinflammation. SHSY-5Y cells that were transfected with downregulated miR-124-3p expression had significantly reduced cell viability, increased apoptosis rates and an increased vulnerability to future stresses. The increased rates of apoptosis are the result of stresses building up within the mitochondria that lead to mitochondrial dysfunction and subsequently, apoptosis which is initiated by the mitochondria. This leads to the progression of PD, due to the increased loss of dopaminergic neurons. However, the addition of miR-124-3p reverses the damaging effects, indicating that miR-124-3p has the potential to be utilized as a therapeutic agent to prevent dopaminergic neuron loss. The healing capabilities of miR-124-3p may also be useful in preventing PD in susceptible individuals. 33
Neuroinflammation is also indirectly caused by miR-150 through mitochondrial activity. Li et al 27 obtained blood samples from 140 individuals with and without PD to compare the expression of miR-150. After measuring miR-150 expression levels using qRT-PCR, the researchers found that miR-150 was significantly downregulated in the samples from PD patients. 27 Additionally, the PD samples had increased procytokine levels, indicating a negative correlation between miR-150 and proinflammatory cytokine levels. Further research elucidated v-akt murine thymoma viral oncogene homolog 3 (AKT3), a mitochondrial gene as the source of elevated levels of proinflammatory cytokines, which are typically associated with neuroinflammation. The regulation of the mitochondrial gene AKT3 by miR-150 is decreased in PD and suggests that the miRNA acts either directly or indirectly to suppress AKT3 and impair mitochondrial functions. AKT3 is suppressed by miR-150 except in PD where downregulated miR-150 fails to suppress AKT3 leading to an increase in neuroinflammation. As mentioned in the above discussion, neuroinflammation leads to the progression of PD due to an increased vulnerability to neuronal apoptosis. To better understand the relationship between miR-150 and neuroinflammation, microglia BV2 cells were transfected with vectors that either upregulated or inhibited by miR-150 expression. Inhibition of miR-150 led to an increase in neuroinflammatory markers, whereas upregulation of miR-150 suppressed the release of neuroinflammatory markers. The results of this study potentiate miR-150 as a biomarker and/or a therapeutic agent. The use of blood samples to identify the downregulation of miR-150 would allow a simple blood draw to diagnose PD in the early stages. This could potentially prevent progression of PD before the symptomatic stages. Additionally, the therapeutic effects of upregulating miR-150 reducing neuroinflammatory markers could serve as a treatment to prevent further dopaminergic neuron loss. 27
In addition to AKT3, microRNAs target several other genes, including leucine-rich repeat kinase 2 (LRRK2). LRRK2 dysregulation has been associated with familial PD and is considered a genetic risk factor for sporadic PD. Mitochondrial impairment is strongly associated with LRRK2 mutations, and LRRK2 localization in the mitochondria regulates mitochondrial function. 36 Utilizing software that identifies miRNA that targets LRRK2, Cho et al 24 found that miR-205 matches the criteria. Cerebral cortex samples were collected from individuals diagnosed with PD and a control group of individuals. qRT-PCR was utilized to measure miR-205 expression levels and LRRK2 levels. miR-205 was significantly downregulated and qRT-PCR analysis demonstrated a statistically significant inverse correlation between LRRK2 and miR-205 levels. Under healthy circumstances, miR-205 decreases LRRK2 expression. However, in individuals with PD, miR-205 is downregulated and consequentially liberates LRRK2, allowing LRRK2 expression to increase neurite growth in a disorderly manner. To confirm miR-205’s role in LRRK2 regulation, neuron cells were transfected with either miRNA-205 inhibitors, pre-miR-205, or co-transfected with both pre-miR-205 and miR-205 inhibitors to determine the impact on LRRK2. Cells transfected with miR-205 inhibitors had significantly elevated LRRK2 and cells that were co-transfected did not exhibit any changes in LRRK2 levels. In addition to elevated LRRK2 in the cells transfected with miR-205 inhibitor, neurites were shortened and diminished in number in comparison to the control group of cells. Additionally, Cho et al 24 investigated the impact of miR-205 overexpression on cells that expressed shortened and diminished neurites by introducing miR-205 into hippocampal neurons of mice. Both of the negative outcomes associated with miR-205 downregulation were prevented by overexpression of miR-205, serving as a rescue effect. miR-205 is a potential therapeutic agent that can regulate LRRK2, a gene associated with both types of PD, and prevent or decrease the impacts of miR-205 downregulation. 24
Another aspect of neurotoxicity within PD is neurodegeneration via alpha-synuclein accumulation. An abnormal protein that aggregates in dopaminergic neurons, alpha-synuclein majorly contributes to the formation of Lewy Bodies, which are associated with early onset dementia. In order to better understand the source of alpha-synuclein aggregation, Lungu et al 29 isolated several miRNAs from mouse brain tissue and quantified them into upregulated and downregulated miRNAs. miR-17-5p, miR-20a, miR-93, and miR-106 were detected to be significantly upregulated in PD samples using qPCR analysis. These miRNAs help regulate the cell cycle at the G1/S checkpoint. Additionally, miR-132 was detected to be significantly upregulated in the PD samples. Abnormally low levels of miR-132 significantly downregulate Nurr1 and brain-derived neurotrophic factor (BDNF) both of which maintain normal neuronal health, Nurr1 aids in the prevention of alpha-synuclein accumulation and regulates dopaminergic neuron development and differentiation. BDNF is a dopaminergic neurotrophin that regulates neuronal health. The dysregulation of miR-132, and subsequently Nurr1 and alpha-synuclein allows for an increase in dopaminergic neuron loss. The loss of dopaminergic neurons is the defining characteristic of PD and is the source of PD symptoms. The downregulation of miR-132 results in the loss of regulation of Nurr1 and BDNF’s neuroprotective barriers permitting the neurodegeneration to begin. Similarly, Stein et al 10 investigated miR-181 due to prior research that established its role in suppressing neuronal growth, neurite extension, synaptic signaling, ion transport, mitochondrial respiration, mitochondrial removal, and cell survival genes. miR-181 is upregulated in PD samples and in aging brain samples allowing it to downregulate its target genes. These genes include Gabra1, Kenj6, and Chchd10. Gabra1 encodes a subunit of the GABAA inhibitory neurotransmitters, which regulates dopamine-driven movement. Kcnj6 aids in limiting neuronal firing and dopamine release. Chchd10 supports mitochondrial cristae maintenance and respiratory function. Utilizing adeno-associated viruses, Stein et al 10 injected overexpressed miR-181 and inhibited miR-181 into mouse models. The models with overexpressed miR-181 exhibited neurotoxic effects with a loss of dopaminergic neurons. Additionally, the researchers combined the overexpressed miR-181 and an inducer of alpha-synuclein into 1 model which exacerbated the neurotoxicity and elicited increased alpha-synuclein production. The upregulation of miR-181 extends the downregulation of miR-181’s gene targets. The combination of inhibited miR-181 and the alpha-synuclein inducer protected the model against the neurotoxic effects of alpha-synuclein accumulation. This indicates that downregulation of miR-181 is neuroprotective against PD-related neurotoxicity. As with most of the previously mentioned miRNAs, preventing miR-132 or miR-181 upregulation would help minimize dopaminergic neuron loss and, by extension, the progression of PD. This could potentially also prevent the onset of PD entirely if it eliminates the accumulation of alpha-synuclein.
Neurotoxicity is an important consequence of miRNA dysregulation in PD pathology and disease progression. It is primarily caused by the accumulation of alpha-synuclein which is possibly caused by the dysregulation of miRNAs leading to decreased regulation of the miRNA’s targets. While the exact sources of alpha-synuclein accumulation and subsequent formation of Lewy Bodies are currently unknown, miRNA dysregulation contributes to the exacerbation of alpha-synuclein accumulation through the loss of regulation of the target genes and gene products. This facilitates the progression of PD through the degeneration of dopaminergic neurons. PD is a complex disease and obtaining a better understanding of how the dopaminergic neurons are lost and how alpha-synuclein accumulates can provide the roadmap to a therapeutic agent that reduces or stops the accumulation. This would prevent the progression of PD and could serve as a treatment for individuals with early PD.
Mitochondria Associated miRNAs and Dopaminergic Neurons
In 2016, Cheng et al 32 studied the damage to mitochondria, which results in the activation of both mitophagy and mitochondrial apoptosis. The elimination of dysfunctional mitochondria is necessary for mitochondrial quality maintenance and efficient energy supply. miR-181a, an endogenous inhibitor of mitophagy, is normally downregulated by mitochondrial uncouplers in human neuroblastoma SH-SY5Y cells. This accelerates the autophagic degradation of damaged mitochondria. Overexpression of miR-181a inhibits mitochondrial uncoupling agents-induced mitophagy by inhibiting the degradation of mitochondrial proteins without impacting autophagy. miR-181a directly targets Parkin E3 ubiquitin ligase and partially blocks the colocalization of mitochondria and autophagosomes or lysosomes. Administering exogenous Parkin reinstates the inhibitory effect of miR-181a on mitophagy. Furthermore, miR-181a increases the sensitivity of neuroblastoma cells to mitochondrial uncoupler-induced apoptosis, whereas miR-181a antagomir prevents cell death. As mitophagy defects are associated with a variety of human disorders, these findings indicate an important link between miRNAs and Parkin-mediated mitophagy and serve as a potential therapeutic strategy for human diseases. Additionally, the dysregulation of mitophagy and mitochondrial apoptosis furthers the loss of dopaminergic neurons as they are heavily dependent on proper mitochondrial function.
Another potential therapeutic target for the treatment of PD includes miR-128, as it also impacts the survival of dopaminergic neurons through the regulation of axis inhibition protein 1 (AXIN1). In this study, Zhou et al 31 established mouse models of PD to investigate the expression of miR-128 and the mechanism through which it affects apoptosis of dopamine neurons and the expression of excitatory amino acid transporter 4 (EAAT4) via binding to AXIN1. Gene expression microarray analysis was performed to screen differentially expressed miRNAs that are associated with PD. The targeting relationship between miR-128 and AXIN1 was verified via a bioinformatics prediction and dual-luciferase reporter gene assay. After separation, DA neurons were subjected to a series of inhibitors, activators and shRNAs to validate the mechanisms of miR-128 in controlling AXIN1 in PD. Positive protein expression of AXIN1 and EAAT4 in DA neurons was determined using immunocytochemistry. miR-128 expression and the mRNA and protein levels of AXIN1 and EAAT4 were evaluated via qRT-PCR and Western blot analysis, respectively. DA neuron apoptosis was evaluated using TUNEL staining. The author identified AXIN1 as an upregulated gene in PD based on the microarray data of GSE7621. AXIN1 was targeted and negatively mediated by miR-128. Evidence from experimental models revealed that miR-128 might reduce apoptosis of DA neurons while increasing the expression of EAAT4 which might be related to the downregulation of AXIN1. 31
Mitochondria Associated miRNAs and Impaired Mitochondrial Functions
Dopaminergic neurons are particularly susceptible to mitochondrial changes as they have high energy requirements and require an increased number of mitochondria to supply their energy requirements. As a consequence of dopaminergic neurons relying so heavily upon mitochondria, alterations in mitochondrial function and regulation have profound impacts on the survival of dopaminergic neurons. Mitochondrial processes are determined and influenced by mitochondrial, both localized and involved, RNA’s expression. Variations in mitochondrial RNA expression led to mitochondrial dysfunction which cripples mitochondrial function and eventually dopaminergic neurons. In addition to the dopaminergic neurons, alterations to the number of functioning mitochondria prevent normal cellular functions and have devastating effects on a variety of cells. As mitochondria are essential organelles to many cells, their functions are heavily regulated by a number of pathways. Several of these regulatory pathways include miRNAs and can be severely disrupted in PD development. Under healthy circumstances, miR-193b, miR-421, and miR-7 maintain normal mitochondrial function. miRNA dysregulations associated with PD can cause damage to mitochondria, potentially resulting in neuronal cell death and increasing the progression of PD.
Baghi et al 30 studied the expression levels of miR-193b and their impact on mitochondrial function in peripheral blood mononuclear cells (PBMCs) from 20 control plasma and 20 PD plasma samples. The use of PBMCs allows the researchers to exclude red blood cells (RBCs) that do not contain mitochondria while maintaining a simple collection process that could easily be replicated in clinical applications. After analysis of the samples using qRT-PCR, miR-193b was found to be significantly upregulated in PD samples. Utilizing an AUC value, Baghi et al 30 confirmed that miR-193b could be a biomarker for the diagnosis of PD. An AUC value indicates the efficiency with which a marker can differentiate 2 conditions, in this case, PD and control. Additionally, SHSY-5Y cells were transfected with miR-193b inducers to study the impact on miR-193b. The elevated levels of miR-193b demonstrated repression of peroxisome proliferator-activated receptor gamma coactivator-1 alpha (PGC1-alpha) with a significant inverse correlation between the expression of PGC1-alpha and miR-193b. This is particularly important as PGC1-alpha is part of a neuroprotective pathway that regulates mitochondrial biogenesis and respiration. Mitochondrial biogenesis is the mitochondrial replication process and is similar to bacterial replication, known as binary fission. The mitochondrion duplicates its internal components and then splits into 2 new daughter mitochondria. Dysfunctional mitochondrial biogenesis would impair the production of daughter mitochondria and fail to prevent the accumulation of damage within the mitochondria. Furthermore, PGC1-alpha also regulates mitochondrial respiration, which produces ATP. Dysregulation of mitochondrial respiration has catastrophic effects that prevent essential cellular functions from occurring. The suppression of PGC1-alpha impedes normal mitochondrial replication and mitochondrial protective mechanisms, increasing the likelihood of mitochondrial death. This is another possible etiology for the dopaminergic neuron death that results in the progression of PD symptoms. Reducing the expression of miR-193b improves mitochondrial biogenesis by increasing the expression of PGC1-alpha. 30
The toll of oxidative phosphorylation and the subsequent oxidative damage are cumulative and are seen in older mitochondria. Similar to cell senescence, mitochondria also undergo senescence and need to undergo their version of apoptosis, mitophagy. Mitophagy digests disordered mitochondria before replication to prevent the reproduction of dysfunctional mitochondria. Decreased mitophagy leads to an accumulation of improperly functioning mitochondria contributing to overall mitochondrial dysfunction. Eventually, failure to replace these mitochondria results in apoptosis of the whole cell. The regulation of mitochondrial dysfunction has piqued the interest of many researchers.
miR-421 has been implicated in mitophagy regulation in several diseases, including pancreatic cancer and PD. Dong et al 25 hypothesized that miR-421 upregulation could exacerbate the neurodegenerative processes typically observed in PD through the regulation of PINK1 and Parkin. PINK1 and Parkin are signaling molecules that flag improperly functioning mitochondria for removal via mitophagy. After transfecting SHSY-5Y cells with miR-421 inducer, PINK1 and Parkin levels were found to be suppressed when compared to the control cells. PINK1 is thought to have a binding site that allows miR-421 to prevent mitophagy, which was confirmed using western blotting. The results of this study indicate that the upregulation of miR-421 suppresses mitophagy and permits the reproduction of dysfunctional mitochondria, leading to cell apoptosis and the progression of PD. However, the possibility of utilizing a miR-421 inhibitor as a therapeutic agent could reduce dopaminergic neuron loss.
As previously mentioned, mitochondria play a major role in the regulation of cell apoptosis. Impaired mitogenesis and disordered mitophagy tell the cell to undergo apoptosis to prevent the reproduction of the dysfunctional mitochondria. Another aspect of mitochondrial control over regulation includes the production of mitochondrial permeability transition pores to release cytotoxic elements that diminish the mitochondrial membrane potential and facilitate cell death. One of the main components of these pores is voltage-dependent anion channel 1 (VDAC1), which is regulated by miR-7. miR-7 prevents cell apoptosis by downregulating the production of VDAC1 in healthy individuals. However, miR-7 is downregulated in individuals with PD and permits an increase in VDAC1 expression and increased mitochondria-facilitated cell apoptosis. Chaudhuri et al 28 transfected SH-SY5Y cells with miR-7 and found that VDAC1 levels were reduced. They applied JC-1, a dye used to distinguish healthy and disordered mitochondria, to observe the mitochondria. The cells that were transfected with miR-7 had significantly less disordered mitochondria and were able to withstand efforts to cause membrane depolarization, by inhibiting the mitochondrial permeability transition pore from opening. Moreover, miR-7 combats VDAC1 production by downregulating the gene. This preserves functional mitochondria and prevents unnecessary cell death, which may potentially be the cause of dopaminergic neuron death. In individuals with PD, miR-7 expression is downregulated and VDAC1 levels are rampantly elevated. This allows an increase in cell death and may potentially be the cause of dopaminergic neuron death. Countering the downregulation of miR-7 could stop the progression of PD and prevent VDAC1 expression from elevating.
MiRNAs play an important role both in the development and progression of PD. While the exact mechanisms of dopaminergic neuron death are not currently known, ongoing research is quickly approaching the answers. It is known that mitochondrial dysfunction plays a major role in PD progression and is related to the numerous mitochondria located in dopaminergic neurons. MiRNAs have demonstrated the ability to regulate mitochondrial function and create mitochondrial dysfunction when dysregulated. Mitochondrial dysfunction plays a major role in PD progression. While the combined impact of miRNA dysregulation has not been studied within PD, the results of the above studies indicate that more than 1 dysregulated miRNA may be the cause. Upregulation of miR-193b and miR-421 plus downregulated miR-7 have exhibited impaired mitochondrial biogenesis, disordered mitophagy, and increased mitochondria-facilitated cell death. However, miRNAs may also be the answer as biomarkers, preventative therapies, or even in the treatment of advanced PD.
Mitochondria Associated miRNAs and Synaptic Energy in Parkinson’s Disease
At its core, PD is a neurodegenerative disorder that involves synapses. Healthy brain function involves abundant synaptic function and neurotransmitter release. Synapses, the communicating portion of the neuron, utilize neurotransmitters to send and receive messages quickly. Neurotransmitters are stored within synaptic vesicles within the synapse and released by action potentials that open voltage-gated calcium channels. This requires a tremendous amount of energy that is supplied by the copious amounts of mitochondria present in the synapse. The release of neurotransmitters is energetically expensive and utilizes an abundance of ATP, the energy currency of life. ATP is required for the use of Sodium-Potassium ATPase pumps, Calcium-ATPase pumps, Sodium pumps, and the Proton-ATPase pumps that help create favorable environments for the release of neurotransmitters. 37 In addition to the pumps, energy is also required for the creation, storage, and mobilization of synaptic vesicles. Once the mitochondria have completed their lifecycle, energy is also required to remove them and synthesize new ones. Cellular energy is high in demand throughout these cells and production must meet the demands. ATP is produced by mitochondria through the electron transport chain and oxidative phosphorylation. Besides the synaptic function, ATP also sustains cell function. Insufficient ATP would prevent normal synaptic function and eventually lead to neuronal death, as seen in PD. In addition to synaptic dysfunction, mitochondrial dysfunction significantly reduces the cell’s ATP supply. As previously mentioned, the downregulation of miR-34b/c prevents sufficient ATP production in PD. 26 The combination of mitochondrial and synaptic dysfunction leads to dopaminergic neuronal death and the progression of PD. These circumstances demonstrate the importance of healthy mitochondrial and synaptic function. However, there is a deficit of research in this area. Mitochondrial-mediated synaptic function in PD has largely been avoided as a factor. Additional research should be conducted regarding mitochondrial-mediated synaptic function in PD.
Therapeutic Relevance of Mitochondrial Associated miRNAs in Parkinson’s Disease
Mitochondrial-associated miRNAs have a drastic impact on the expression of essential mitochondrial genes, with some having a 7-fold increase in gene expression. 30 The majority of miRNAs were found to have an inverse relationship with their target gene, however, some presented with a negative correlation, in which both the miRNA and the target gene were reduced. Upregulation of miR-376a directly reduces PGC1-alpha, its target gene, effectively preventing mitochondrial biogenesis. 23 miR-421, when upregulated, reduces the expression of Bax, PINK1, and Parkin impeding mitophagy. 25 miR-132 is upregulated in PD and decreases Nurr1, a neuroprotective gene and protein that maintains neuronal health. An example of miRNA and a target gene that are negatively correlated are miR-150 and AKT3. This negatively correlated pair increases neuroinflammation and the expression of neuroinflammatory markers that exacerbate the effects of PD. 27 Another aspect of utilizing miRNA for the treatment of PD includes the method of delivery. The blood brain barrier is a significant barrier to drug delivery, especially targeted deliveries. MiRNAs are incredibly small molecules that are ideal for crossing the blood brain barrier, however, they require assistance to cross the blood brain barrier and reach the target. While the current miRNAs are in the early stages of research, Tryphena et al 38 have suggested the utilization of nanoparticles to cross the blood brain barrier to deliver targeted miRNA to the brain. This research is fundamental to the utilization of miRNA in the treatment of PD as it permits targeted miRNA therapy to prevent or restore mitochondrial dysfunction, as a potentially curative measure. 38 Nanotechnology has been seen in other chronic disorders to deliver therapeutic miRNA. The direct impact of these mitochondrial-associated miRNAs on mitochondrial function and the eventual pathologic effects that develop in PD are critical relationships that can help facilitate our understanding of the causes and potential treatments for PD. These miRNAs hold the key to preventing the onset and progression of PD. Based on the importance of the miRNAs and their target genes, mitochondrial-associated miRNAs in PD are a field of research that requires further investigation and understanding.
Potential Usage of Mitochondrial Associated miRNAs as Biomarkers in Parkinson’s Disease
In addition to the use of miRNAs as potential treatments for PD, they may be utilized to serve as biomarkers. The current diagnostic criterion for PD is the presence of symptoms, by which the disease has progressed and exhibits significant dopaminergic neuron loss. The applications of miRNA as a biomarker would serve as a pre-symptomatic diagnostic criterion. Many of the miRNAs discussed in this article are found in the blood and can be monitored via a routine blood draw. MiRNAs that can potentially serve as biomarkers include miR-193b, miR-150, miR-124-3p and hsa-miR-4639-5p, which can be observed using blood or plasma samples.22,27,30,33 Diagnosing PD early would provide ample time for treatment before the progression of the disease to the symptomatic stages. Diagnostic biomarkers have successfully been implemented in a variety of other diseases, including Diabetes Mellitus. Fasting blood glucose is a relatively quick blood test that diagnoses the patient as healthy, pre-diabetic, and diabetic. These diagnoses allow the patient and healthcare team to make informed decisions regarding future treatment options. miRNA can serve a similar purpose for PD. Despite the current lack of preventative measures for PD, miRNA biomarkers can facilitate early diagnosis and treatment that reduces the severity of symptoms.
Summary and Conclusion
PD has grown tremendously in prevalence, affecting individuals above the age of 60, and posing a concern to the aging global population. The lack of diagnostic markers and the slow progression impede diagnosis. In the meantime, mitochondrial dysfunction is wreaking havoc on the remaining dopaminergic neurons. As mitochondria reproduce through fission and fusion, they depend on mitophagy to remove diseased mitochondria. Mitochondria also generate energy for the cell, during which reactive oxygen species are generated and quickly disposed of. In PD, both disordered mitophagy and reactive oxygen species buildup cause the cells to be placed under severe oxidative stress and ultimately, destroy the mitochondria and the cell. Mitochondrial-associated miRNAs contribute heavily to the development of PD, by influencing genes to increase or decrease their product generation. These imbalances develop into the accumulation of alpha-synuclein and Lewy Body formation, causing the progression of PD. miRNAs are currently being explored as biomarkers and/or therapeutic agents to stop or slow the progression of PD. Biomarkers would help definitively diagnose PD and could include: miR-193b, miR-150, and hsa-miR-4639-5p. The above-mentioned miRNAs have great potential to become therapeutic targets to restore mitochondrial function and reduce Parkinson’s pathology. These miRNAs may be useful as targets or as messengers and include: miR-376a, miR-205, miR-421, miR-34b/c, miR-7, miR-132, miR-17-5p, miR-20a, miR-93, miR-124-3p and miR-106. Further research is needed to understand the molecular mechanism and roles of these miRNAs in PD pathogenesis.
Future Direction
We have investigated the roles of mitochondrial miRNAs in Alzheimer’s disease (AD).39 -43 The mitochondrial-associated miRNAs are located within the cell, but their precise location is unknown. Amongst other neurodegenerative disorders, mitochondria-localized miRNAs have been discovered and are being studied for their regulatory capabilities.42,44 Mitochondrial-localized miRNAs have been extensively researched within heart disease, diabetes cancers, liver transplants, AD, and Huntington’s Disease, however, none have been mentioned in PD. With mitochondrial dysfunction playing a major role in PD, mitochondrial-localized miRNAs should be the next step in understanding PD. In addition, the mitochondria-associated miRNAs have immense potential to be utilized as therapeutic agents within PD due to their ability to regain control of their regulatory functions.
Footnotes
Acknowledgements
The authors are exceedingly grateful to Prof. Rajkumar Lakshmanaswamy, Chair of the Department of Molecular and Translational Medicine, TTUHSC El Paso for the immense research support.
Author Contributions
Conceptualization, S.K.; Original draft preparation, G.R.A. and Y.C.; Review and editing, G.R.A. and S.K. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Institute on Aging (NIA), National Institutes of Health (NIH), grant number K99AG065645, R00AG065645, R00AG065645-04S1, SARP mini grant TTUHSC EP, EARN Grace V Chope Estate fund and TTUHSC EP MTM Startup Funds to S.K.
