Abstract
Smoking is associated with a lower incidence of Parkinson's disease (PD), which might be related to a neuroprotective action of nicotine. Postmortem studies have shown a decrease of cerebral nicotinic acetylcholine receptors (nAChRs) in PD. In this study, we evaluated the decrease of nAChRs in PD
Introduction
Parkinson's disease (PD) is a neurodegenerative movement disorder that is characterized by a progressive loss of the nigrostriatal dopaminergic neurons leading to a severe dopamine depletion state. In addition to the decline of the dopaminergic function, other neurotransmitter systems are involved in PD, including the nicotinic cholinergic system (Jellinger, 1991). There is now accumulating evidence that there are significant declines in nicotinic acetylcholine receptors (nAChRs) in the postmortem brains of both demented and nondemented patients with PD (Aubert et al, 1992; Rinne et al, 1991; Quik, 2004). Moreover, converging evidence suggests that nAChRs could be potential targets for PD therapy and neuroprotective strategies. For example, numerous studies have shown that the stimulation of the nicotinic receptors located on presynaptic dopaminergic neurons by nicotine increases dopamine release in the striatum (McCallum et al, 2006; Salminen et al, 2004). In parallel, smoking markedly reduces the risk of developing PD (Allam et al, 2004). Finally, nicotine protects against neuronal insults in experimental models of PD (Jeyarasasingam et al, 2002; Parain et al, 2003; Quik et al, 2006). Hence, a deeper knowledge with regard to the nAChRs located in the basal ganglia and the changes related to PD is necessary.
Neuronal nAChRs are pentameric ligand-gated ion channels composed of α-subunits (homomeric receptors) or of α- (α2 to α7) and β-subunits (β2 to β4) (heteromeric receptors). The α4β2 nAChRs are the prominent subtype in the human brain (Gotti and Clementi, 2004). Nontoxic and selective ligands of nicotinic receptors have been recently developed and validated in humans for the assessment of cerebral nAChRs with SPECT (single photon emission computed tomography) or positron emission tomography (PET) imaging. Among those, the derivatives of 3-[2(
A-85380 has also been radiolabeled with fluorine-18 and characterized for PET imaging of nicotinic receptors in the human brain (Dollé et al, 1999; Koren et al, 1998; Valette et al, 1999a, b ; Bottlaender et al, 2003). Among these compounds, 2-[18F]fluoro-A-85380 has a high affinity for the α4β2* subtype (Gao et al, 2008) and is the most studied PET radioligand.
This study aimed at measuring the
Materials and methods
Subjects
Thirteen nondemented PD patients, aged 61.2 ± 10.8 years (mean ± s.d.) were enrolled (Table 1). All subjects satisfied the United Kingdom Parkinson's Disease Society Brain Bank Diagnostic criteria for the clinical diagnosis of idiopathic PD (Hughes et al, 1992). Symptoms duration ranged from 0.75 to 19 years and the Hoehn and Yahr stage varied from I to IV, when off medication. All but one, were on anti-Parkinsonian medication. None of them had been treated with drugs interacting with the cholinergic system in the 2 months before the study. None of the PD patients had dementia according to the MMSE (Mini-Mental State Examination) score; they all scored higher than 26/30, except patient no. 4 who scored 25. However, this Polish patient had trouble with the French language, which explained most of his mistakes. Five patients had never smoked. Eight patients had quit 7 to 26 years earlier.
Characteristics of the PD patients
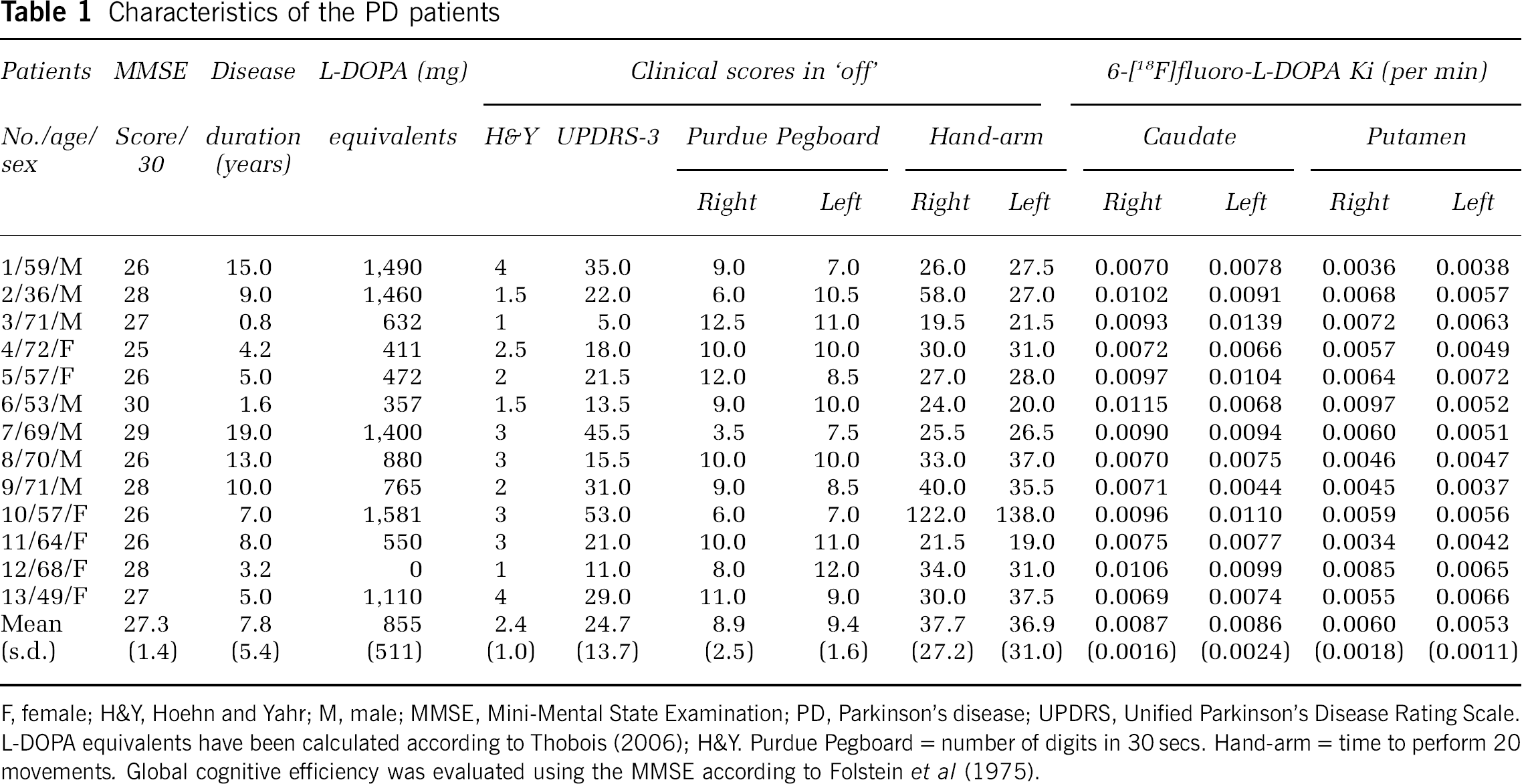
F, female; H&Y, Hoehn and Yahr; M, male; MMSE, Mini-Mental State Examination; PD, Parkinson's disease; UPDRS, Unified Parkinson's Disease Rating Scale.
L-DOPA equivalents have been calculated according to Thobois (2006); H&Y. Purdue Pegboard = number of digits in 30 secs. Hand-arm = time to perform 20 movements. Global cognitive efficiency was evaluated using the MMSE according to Folstein et al (1975).
The control group consisted of six nonsmoking healthy volunteers, aged 61.2 ± 13.9 years (range 37 to 74, three men and three women) without any psychiatric or neurologic illness or cholinergic medication. All of them had a negative plasma cotinine determination and a normal brain magnetic resonance imaging (MRI). There was no significant difference in age (
Study Design
The severity of PD was measured using the UPDRS (Unified Parkinson's Disease Rating Scale) motor score and timed motor tests (Purdue Pegboard, hand-arm tests) in the defined ‘off’ condition (i.e., at least 12 h after the last anti-Parkinsonian medications) (Defer et al, 1999). This evaluation was performed by one neurologist, an expert in movement disorders. He was blinded to the PET results.
All subjects underwent a
Magnetic Resonance Imaging and Anatomic Volumes-of-Interest
Magnetic resonance imaging scans were performed on a 1.5 T imager (Signa, General Electric, Milwaukee, WI, USA). A
Volumes-of-interest were drawn manually on each patient's
6-[18F]fluoro-l-DOPA Positron Emission Tomography Acquisition and Analysis
All dopaminergic medications were stopped at least 12 h before the scan (Brooks et al, 2003; Ribeiro et al, 2002). All patients received 100 mg of carbidopa, a peripheral decarboxylase inhibitor, 1 h before tracer injection. Nine frames were acquired over 90 mins, after tracer intravenous injection (159.7 ± 31.2 MBq). Positron emission tomography scans were reconstructed with filtered back projection with a ramp filter producing images with a resolution of 6.8 mm full-width at half-maximum. Positron emission tomography images were corrected for scatter, γ-ray attenuation, and fluorine-18 decay. The voxel size was 2.4 times 2.4 times 2.4 mm3.
A parametric image of 6-[18F]fluoro-
2-[18F]fluoro-A-85380 Positron Emission Tomography Acquisition and Analysis
A-85380 was labeled with fluorine-18 by a no-carrier-added nucleophilic aromatic substitution (Dollé et al, 1999). 2-[18F]fluoro-A-85380 (188 ± 22 MBq; 2 to 3 nmol) was injected intravenously as a 1-min bolus. Positron emission tomography acquisition started 90 mins later and lasted for 2 h, a period in which the apparent steady state of 2-[18F]fluoro-A-85380 was reached as shown by Gallezot et al (2005) and Mitkovski et al (2005). Fifteen sequential frames were acquired with the acquisition duration ranging from 5 to 10 mins. Twenty venous blood samples were collected, initially every 5 mins during the first hour after injection, and then subsequently every 10 mins until the end of the PET emission (Picard et al, 2006). Plasma radioactivity was measured in a γ-counter (Cobra Quantum D5003, Perkin-Elmer, France). On the basis of the fact that the metabolites of 2-[18F]fluoro-A-85380 do not cross the blood—brain barrier (unpublished data), we applied a metabolite correction only for the plasma as previously described (Gallezot et al, 2005). Positron emission tomography emissions were reconstructed using the same procedure as that of the 6-[18F]fluoro-
The distribution volume (DV) of 2-[18F]fluoro-A-85380 was determined using a simplified method based on the ratio of brain tissue concentration to unchanged 2-[18F]fluoro-A-85380 plasma concentration at equilibrium as defined by Mitkovski et al (2005). However, venous plasma was used instead of arterial plasma in our series. This simplified approach has been previously validated in 10 subjects (5 PD patients and 5 controls) of our study. These subjects had dynamic PET scan acquisition, and arterial and venous blood collections started with the bolus administration of 2-[18F]fluoro-A-85380, which enabled the calculation of DV with a two-tissue compartmental model analysis as described in Gallezot et al (2005). We found that the DV values defined by the region-to-venous plasma ratio were strongly correlated with those obtained from a two-tissue compartmental model analysis (data not shown). It showed that the venous blood sampling was an acceptable choice for input function and that 2-[18F]fluoro-A-85380 DV values expressed by this ratio can be used to quantify cerebral nAChRs concentration.
Parametric images of 2-[18F]fluoro-A-85380 DV were created using the BrainVisa software. For each frame collected during the last 30 mins of the emission scan, the radioactivity in each voxel was divided by the value of individually metabolite-corrected 2-[18F]fluoro-A-85380 concentration in venous plasma measured at the same time point.
Statistical Analysis
The 2-[18F]fluoro-A-85380 DV images were analyzed by combining an individual analysis using VOIs and a voxel-based approach using the Statistical Parametric Mapping software (SPM2, Wellcome Department of Cognitive Neurology, London, UK), which enables exploratory analysis of the entire brain volume without requiring an
Distribution volume values in the VOIs were measured from the PET parametric image, after coregistration with the MRI. The DV values of the patients and controls were compared by an analysis of variance with
The SPM2 software implemented in Matlab 6.5 (MathWorks, Natick, MA, USA) was also used to localize mean group differences in 2-[18F]fluoro-A-85380 DV between PD patients and controls throughout the entire brain volume. The DV images were spatially normalized to a standard anatomic orientation (Montreal Neurological Institute space) using an appropriate gray matter template; first, individual parametric images were registered to individual
Results
2-[18F]fluoro-A-85380 Volume Of Interest Analysis
In controls, the greatest DV value was found in the thalamus. High-to-intermediate levels were observed in other regions, and the lowest DV values were found in the occipital cortex and corpus callosum (Table 2, Figure 1).
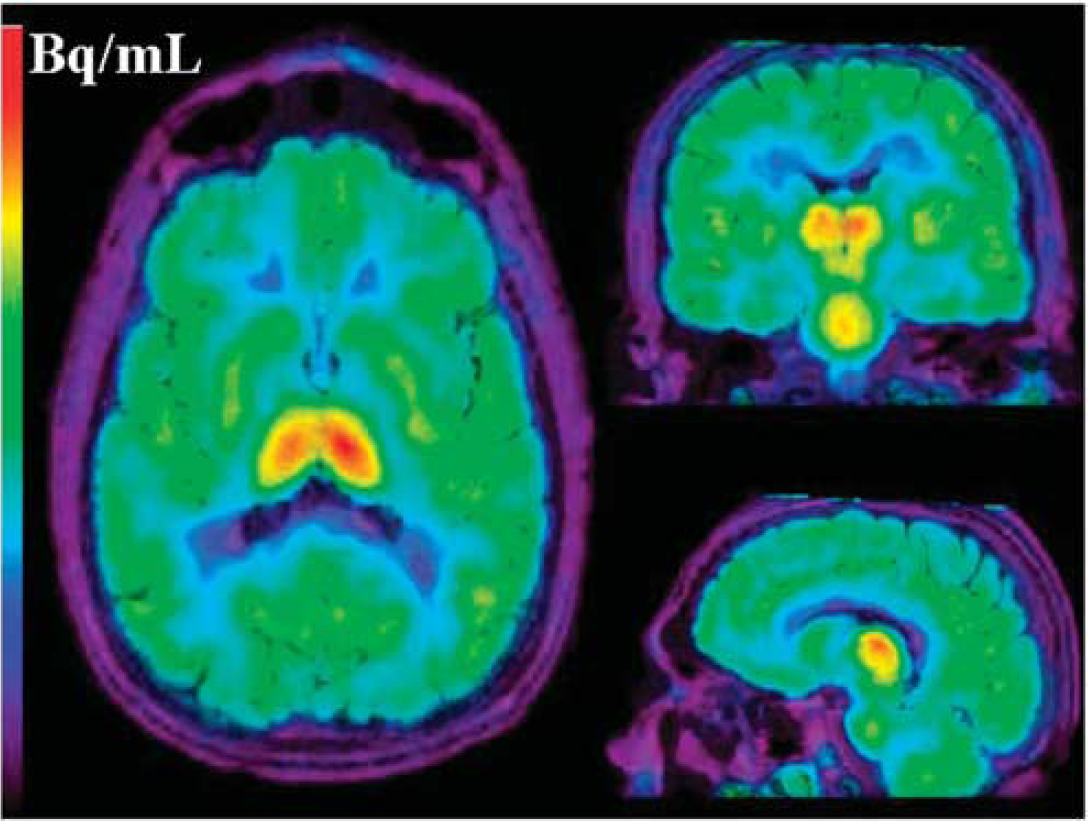
Axial, sagittal, and coronal mean PET images obtained from one control between 90 and 120 mins, after the injection of 2-[18F]fluoro-A-85380. PET images are coregistered with MR images.
2-[18F]fluoro-A-85380 DV values: mean ± s.d.
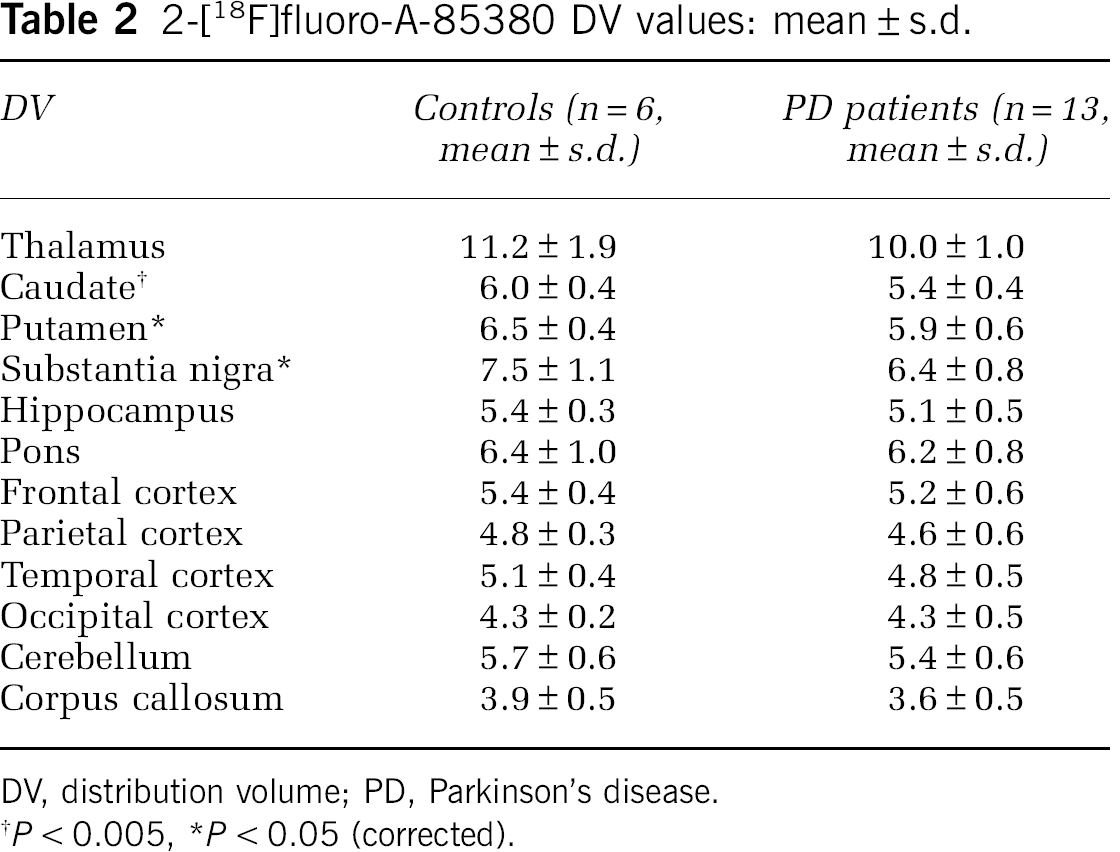
DV, distribution volume; PD, Parkinson's disease.
In PD patients, there was no right-to-left asymmetry of DV values despite the asymmetry of motor symptoms. Accordingly, statistical tests were performed using DV values averaged over both hemispheres. Compared with controls, we found a widespread decrease of DV values in the PD patients, ranging from −.4% in the occipital cortex to a −.9% loss in the substantia nigra (Table 2). After Bonferroni correction, the decrease remained significant only in the substantia nigra (−.9%,
2-[18F]fluoro-A-85380 SPM Analysis
We found a significant bilateral decrease of DV in the striatum in the PD group (Table 3). The analysis performed with a small volume correction (
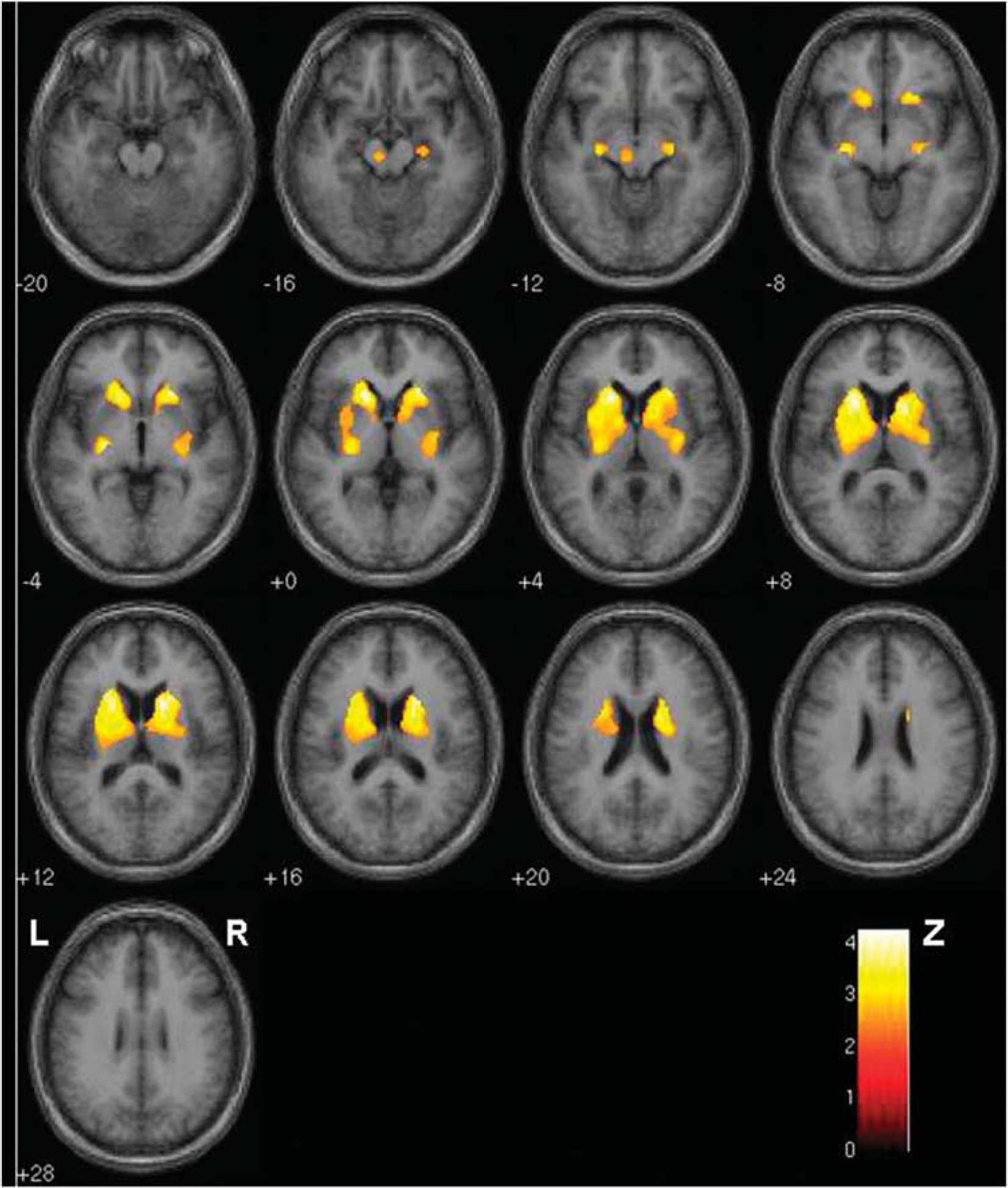
SPM analysis: Brain areas with a significant decrease of 2-[18F]fluoro-A-85380 DV in the striatum and the substantia nigra, in the PD patients compared with that in controls. Colors reflect Z-scores. R: right; L: left.
Decrease of 2-[18F]fluoro-A-85380 DV in PD patients: SPM results
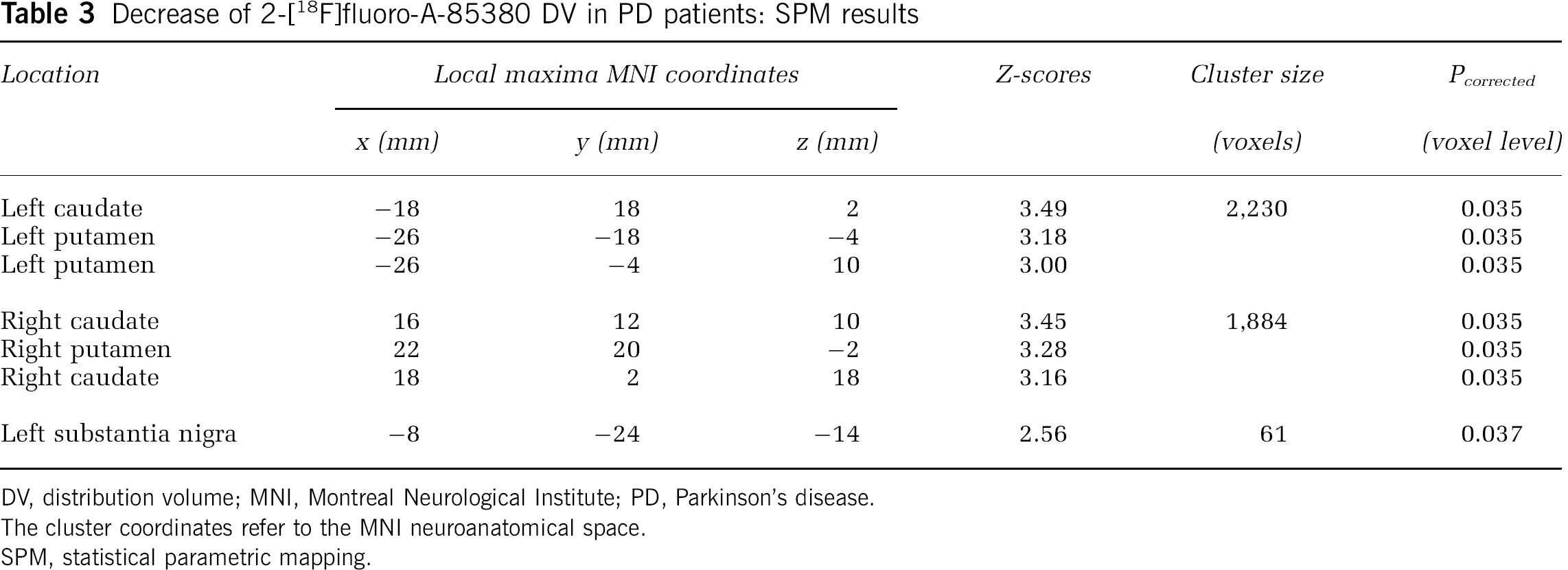
DV, distribution volume; MNI, Montreal Neurological Institute; PD, Parkinson's disease.
The cluster coordinates refer to the MNI neuroanatomical space.
SPM, statistical parametric mapping.
Correlations
No significant correlation was observed using both VOI (Spearman rank correlation test, all
Discussion
Using 2-[18F]fluoro-A-85380 and PET, we found a mild but significant reduction of the density of nAChRs in the striatum and substantia nigra of 13 nondemented PD patients. Despite the wide range of disease severity in these patients, we found no correlation between the different measures of PD severity and the reduction of nAChRs density.
The cerebral distribution of nAChRs in our group of elderly healthy subjects is in agreement with previous studies in nonsmoking healthy volunteers (Gallezot et al, 2005; Kimes et al, 2003; Mitkovski et al, 2005; Picard et al, 2006) and is consistent with data obtained
Although most studies converge to show a reduction of nAChRs density in the nigrostriatal system in PD, an emerging question is whether these changes are related to disease progression. We found no correlation between DV values and the different clinical markers of PD progression, including the amount of medication taken by the subjects, nor with the loss of 6-[18F]fluoro-
Eventually, it has been suggested that nAChRs, especially in the cerebral cortex, might be involved in the cognitive deterioration of PD patients (Rinne et al, 1991; Whitehouse et al, 1988). In our series of 13 nondemented patients, 2-[18F]fluoro-A-85380 DV was reduced by an average of 5% in the cortical regions, which did not reach statistical significance. In their study of 10 nondemented patients Fujita et al (2006), found a similar reduction (−6% on average) of cortical binding of 5-[123I]iodo-A-85380. In addition, Oishi et al (2007) reported a decrease of 5-[123I]iodo-A-85380 DV in the cerebral cortex of 10 nondemented PD patients that only reached significance in the frontal cortex. These slight discrepancies might be related to methodological reasons, such as the use of two different derivates of A-85380, the blood sampling and the plasma analysis, or differences between PET and SPECT procedures, which do not have the same resolution and accuracy. Alternatively, this might simply show individual variations in three different small groups of patients. It could be hypothesized that some individuals have a more severe reduction of cortical nAChRs, which might be predictive of future dementia. Further longitudinal studies are needed to confirm such a hypothesis. Nevertheless, considering the high affinity of 2-[18F]fluoro-A-85380 for the α4β2 nAChRs, the most widespread nAChRs population in the cortex, this ligand has a great potential for investigating the relationship between cognitive deficits and nAChRs density in Parkinsonian patients.
Footnotes
Acknowledgements
This work was supported in part by a grant from ‘Les Journées de Neurologie de Langue Française’.
The authors declare no conflict of interest.
