Abstract
Alzheimer’s disease (AD) is the most common type of dementia, and AD individuals often present significant cerebrovascular disease (CVD) symptomology. AD with significant levels of CVD is frequently labeled mixed dementia (or sometimes AD-CVD), and the differentiation of these two neuropathologies (AD, AD-CVD) from each other is challenging, especially at early stages. In this study, we compared the gray matter (GM) and white matter (WM) volumes in AD (n = 83) and AD-CVD (n = 37) individuals compared with those of cognitively healthy controls (n = 85) using voxel-based morphometry (VBM) of their MRI scans. The control individuals, matched for age and sex with our two dementia groups, were taken from the ADNI. The VBM analysis showed widespread patterns of significantly lower GM and WM volume in both dementia groups compared to the control group (
Keywords
Introduction
Dementia is a general medical condition that affects an individual's cognitive and behavioral abilities and has diverse subtypes, including Alzheimer’s disease (AD), vascular dementia (VaD), Parkinson’s dementia, frontotemporal dementia, Lewy body dementia, and so on. 1 AD and VaD are common types of dementia, which comprise about 60 and 20% of total cases, respectively, 2 and both have characterized pathology. 3 In AD pathology, the deposition of amyloid plaques and tau tangles in the brain leads to neuronal synaptic loss and subsequently deteriorates cognitive functions.4,5 In contrast, VaD refers to the cognitive decline resulting from vascular issues, including ischemic strokes, hemorrhages, and hypoperfusion. 6 Frequently, individuals diagnosed with AD present significant cerebrovascular disease (CVD) symptomology; herein, we labeled this group as AD-CVD. 3 Due to the confounded pathology of AD-CVD and its overlapping symptoms with AD, 7 the differential diagnosis of AD-CVD from AD individuals is challenging, especially at early stages and thus a focus of this study.
While there is no unified approach for the diagnosis of AD-CVD, 8 one of the common approaches is utilizing the Hachinski Ischemic Score (HIS), a clinical scoring tool comprising 13 items,9,10 to separate AD-CVD from AD. In particular, the representative HIS score for AD-CVD is >4 but <7, while for AD, it is ⩽4. 11 However, this rating scale alone offers limited accuracy 12 and lacks taking advantage of CVD-related brain imaging data.7,13 Another approach to diagnose VaD and AD-CVD is the National Institute of Neurological Disorders and Stroke—the Association Internationale pour la Recherche et l’Enseignement en Neurosciences (NINDS-AIREN) scale 14 that incorporates brain imaging outcomes.
Imaging biomarkers can play an important role in increasing the certainty of the dementia diagnosis.15,16 Depending on the modalities, imaging biomarkers trace structural, functional, and molecular changes in the brain.16,17 Magnetic resonance imaging (MRI) is a widely used tool that can assist in identifying dementia types and their underlying pathologies. 15 Dementia causes regional atrophy in gray matter (GM) and white matter (WM) of the brain, which can be detected using structural MRI techniques.18,19 Previous studies reported patterns of this structural alteration in AD and VaD.20 -23 However, to the best of our knowledge, this pattern in AD-CVD is not well characterized. For identifying dementia types, cerebrospinal fluid (CSF) markers (levels of amyloid beta 42 and tau protein) have been reported in past studies,24,25 but the CSF collection process is invasive, requiring lumbar puncture. The amyloid burden tracer called 11 C-Pittsburg compound-B positron emission tomography 26 has been utilized in recent studies27,28; however, it is expensive and has limited use in clinical practice. 29 Alternatively, analysis of structural MRI data in relation to AD and AD-CVD might provide additional information, such as any trend of atrophic patterns in GM or WM that would facilitate their differentiation.
In the context of AD-CVD, it may be challenging to characterize its differential atrophic patterns due to its diverse localization of brain lesions and co-occurred pathologies. 30 Our previous pilot study 31 regarding AD and AD-CVD separation executed a region of interest (ROI)-based analysis on a few selected vulnerable areas in brain MRI, such as ventricles, hippocampus, frontal gyrus and precuneus and estimated white matter hyperintensities (WMHs), a marker of CVD. In that study 31 we did not notice a substantial distinction between AD and AD-CVD in those limited areas and WMHs, suggesting further analysis incorporating other additional brain regions in a larger sample. Herein, we conducted a whole-brain comparison in our two AD and AD-CVD groups and their age and sex-matched cognitively healthy controls. We aimed to explore whether the AD, AD-CVD, and controls differed in GM and WM while comparing them with each other and voxel-based morphometry (VBM) was applied herein. While previous studies on VBM20,21 widely focused on GM or WM volumetric atrophic patterns in AD people compared to controls, we explored these patterns in the AD-CVD group.
The novel contribution of this paper is to investigate the AD-CVD brains’ structural differences using their MRI scans for the first time in comparison to those in the AD population.
Methodology
Participants
Brain MRI data has been collected from 141 participants as part of a clinical trial studying the effects of repetitive transcranial magnetic stimulation (rTMS) on Alzheimer’s individuals in three locations (Winnipeg, Montreal, and Melbourne). 32 We excluded 21 participants with insufficient image quality and significantly different MRI scanning metrics (echo time, inversion time, and repetition time). We utilized the modified HIS similar to our team’s previous study 13 to group our remaining 120 participants as AD (n = 83) or AD-CVD (n = 37). Because of accuracy improvement in modified HIS, as reported in Molsa et al, 12 the highest cut-off was ⩽3 for AD, and the score range for AD-CVD was ⩾4 but ⩽7. The Mean ± SD of modified HIS for the AD and AD-CVD groups were 1.3 ± 1.1 and 4.5 ± 1.1, respectively. Our classified AD-CVD subjects also fulfilled the definitions of “AD with CVD” following the NINDS-AIREN criteria. 14 Additionally, we considered the clinical neuropsychiatrist or neurologist’s diagnosis utilizing patient history, imaging findings and cognitive assessments. All participants gave written consent prior to participation in the trial 33 according to the Declaration of Helsinki, which was approved by the Biomedical Research Ethics Boards of all three study sites (Winnipeg, Montreal, and Melbourne).
Since our clinical trial of rTMS did not include any control participants, we downloaded brain MRI data of 85 age- and sex-matched cognitively normal controls from the ADNI database (http://adni.loni.usc.edu/) to compare our AD and AD-CVD participants. Notably, the ADNI study included subjects with their self-defined modified HIS ⩽ 4. 34
MRI acquisition
We used 3D T1-weighed magnetization prepared rapid acquisition gradient-echo (MPRAGE) scans for the AD, and AD-CVD participants, and all data were acquired using 3T Siemens MRI systems—specifically, a 3T Verio (Winnipeg), Prisma (Montreal), and Skyra (Melbourne). Scanning parameters were slightly different in each of the three locations; however, slice thickness, repetition time, echo time, inversion time, and flip angle were 0.9 to 1.2 mm, 1800 to 2300 milliseconds, 2.19 to 2.98 milliseconds, 900/1100 milliseconds, and 8 to 12°, respectively. We also downloaded 3T T1-weighed MPRAGE MRI data for the cognitively normal controls from the ADNI database. The MRI scanning parameters of the controls were: slice thickness = 1.2 mm, echo time = 3 milliseconds, repetition time = 2300 milliseconds, inversion time = 900 milliseconds, and flip angle = 9°. The detailed MRI acquisition procedures following the ADNI protocol can also be found here. 34
Voxel-based morphometry (VBM) analysis
We conducted a VBM analysis on the MRI data utilizing the CAT12 toolbox (http://www.neuro.uni-jena.de/cat/) within the SPM12 software (https://www.fil.ion.ucl.ac.uk/spm/). 35 All T1-weighted MR images were passed through a spatially adaptive non-local means 36 denoising filter (strength “medium”) before an initial automatic registration and segmentation into GM, WM, and cerebrospinal fluid (CSF) tissues, utilizing the “unified segmentation” method. 37 This initial segmentation was further refined to reach the adaptive maximum a posteriori segmentation. 38 Geodesic shooting registration 39 was then applied to nonlinearly normalize the segmented tissues (GM and WM) to the Montreal Neurological Institute (MNI) template. 40 The spatially normalized GM and WM tissue segmentations were modulated to preserve their relative volumes, 41 and the modulated tissue images were then smoothed by a full width at half maximum (FWHM) Gaussian kernel [size: 8 × 8 × 8 mm3] before statistically evaluating the smoothed outputs. Finally, the automatic quality assurance metrics in each participant’s CAT12 report were manually checked to verify that the images had acceptable quality ratings. Moreover, prior studies reported the usage of a larger-sized smoothing kernel in the VBM analysis42 -44; we smoothed the images using the FWHM Gaussian kernel of size 12 × 12 × 12 mm3 and presented the VBM results in the supplementary file for your interest.
Statistical analysis
Statistical analysis of the subjects’ demographic and group characteristics (Table 1) was conducted using RStudio (ver. 1.4.1106) in the R platform.45,46 A two proportions test was considered for comparing male/female ratios between groups. Depending on the distribution of continuous data (i.e., age and other group characteristics), we ran either a parametric two-sample
Subjects’ demographic details and group characteristics.

Abbreviations: AD, Alzheimer’s disease; ADAS-Cog: Alzheimer’s disease assessment scale-cognitive subscale; CDR, clinical dementia rating; AD-CVD, AD with cerebrovascular disease; MoCA, montreal cognitive assessment; SD, standard deviation.
In three group comparisons, statistically significant difference was only considered, if
Two-proportions test.
Two-sample
Mann–Whitney
The hypotheses were: a) the AD-CVD group has substantial GM and WM differences compared to those in controls, and b) the AD and AD-CVD groups would present a volumetric difference in VBM.
Group differences in normalized, modulated, and smoothed GM and WM images were separately assessed using a general linear model in SPM12. A full factorial design with three levels (AD, AD-CVD, controls) was performed with the inclusion of age, sex, total intracranial volume (TIV), and MRI location (Winnipeg, Montreal, Melbourne, and ADNI) as covariates in the design matrix. The following t-contrasts were investigated to compare between groups: AD < controls, AD-CVD < controls, and AD < AD-CVD, representing lower volume in AD than in controls, lower volume in AD-CVD than in controls, and lower volume in AD than in AD-CVD, respectively (as well as the opposite t-contrasts: i.e., AD > controls, AD-CVD > controls, and AD > AD-CVD). The family-wise error (FWE) was corrected in the model at
Results
Group comparisons
Table 1 details the subjects’ demographic and cognitive data. From the two-proportions tests for sex and two-sample
Gray matter VBM
Compared to controls, the AD group had significantly lower GM volume in several brain regions after correcting for age, sex, TIV, and MRI location (Table 2 and Figure 1a-c). The largest of these clusters (216 810 voxels) was primarily located in the right inferior temporal gyrus and right parahippocampal gyrus but also extended to other brain areas. The second largest cluster (2921 voxels) was mostly centered in the left thalamus, and the third largest cluster (1528 voxels) was in the bilateral gyrus rectus and the orbital part of the right superior frontal gyrus. Another large cluster (1096 voxels) was observed in the dorsolateral part of the left superior frontal gyrus and left precentral gyrus. Other significant clusters were also found in the right angular gyrus and right superior occipital gyrus, right precentral gyrus, the temporal pole of the right superior temporal gyrus, orbital part of the right superior frontal gyrus, right thalamus, and left anterior cingulate and paracingulate gyri. Other clusters with a size of <100 voxels were found in brain regions, as detailed in Table 2. No substantial differences were found for the opposite
Regions of lower gray matter (GM) volume in AD compared with controls (

Abbreviations: MNI, montreal neurological institute; PFWE-corr, family-wise error (FWE) corrected

Anatomical rendering in SPM displays VBM results on gray matter (GM). The left, middle, and right columns are anterior, right, and bottom views of the 3D brain template, respectively. In the t-contrasts of AD < controls (a-c) and AD-CVD < controls (d-f), the family-wise error (FWE) was corrected at
The AD-CVD individuals also had substantially lower GM volume in brain regions relative to the control group, as described in Table 3 and Figure 1d to f. Similar to the AD group, the largest of these clusters (183 821 voxels) was primarily positioned in the right inferior temporal gyrus and parahippocampal gyrus and covered several brain regions of AD-CVD. The second largest cluster with 2072 voxels was mainly centered in the left thalamus; the third largest cluster (1756 voxels) was in the bilateral gyrus rectus and orbital part of the right superior frontal gyrus, similar to the AD group. Large clusters of GM differences in AD-CVD compared to controls were also found in regions of the dorsolateral part of the left superior frontal gyrus and left precentral gyrus, left superior occipital gyrus, left parts of the postcentral gyrus, supramarginal gyrus, and inferior parietal gyrus, right precentral gyrus and middle frontal gyrus, right inferior parietal gyrus, the temporal pole of the right superior temporal gyrus, and lobule 2 of left cerebellum curs. Other clusters with <100 voxels were found in different brain regions, as shown in Table 3. No significant differences were noticed for the opposite t-contrast (AD-CVD > controls).
Regions of lower gray matter (GM) volume in AD-CVD compared with controls (

Abbreviations: MNI, montreal neurological institute; PFWE-corr, Family-wise error (FWE) corrected
Amongst the contrasts between the two dementia groups (AD < AD-CVD, AD > AD-CVD), a significant difference was not observed in VBM analysis using PFWE-corr < 0.05. However, using an uncorrected threshold of
Regions of gray matter (GM) and white matter (WM) difference in AD compared to AD-CVD.

Abbreviations: MNI, montreal neurological institute.
Results are presented here at a liberal threshold of uncorrected
White matter VBM
Relative to the control group, AD individuals were found to have lower WM volume in several regions after correcting for age, sex, TIV, and MRI location (Table 5 and Figure 2a-c). These were mainly positioned in the left sides of the insula, middle cingulate gyrus, and sub-gyral regions, with the largest cluster of 82 442 voxels. The second largest cluster, with a size of 28 511 voxels, was primarily in the left middle temporal gyrus and left hippocampus; the third cluster (2281 voxels) was mainly noticed in the parahippocampal gyrus and lenticular nucleus of the left side of the brain. The right lobule (6) of the vermis and right cerebellar lobules (4, 5, 6), right hippocampus, left cerebellar lobules (4, 5, 6), and right inferior temporal gyrus presented a substantially lower WM in AD than in controls. Small clusters of <100 voxels were also found, as shown in Table 5 and Figure 2a to c. No substantial differences were found for the opposite
Regions of lower white matter (WM) volume in AD compared with controls (
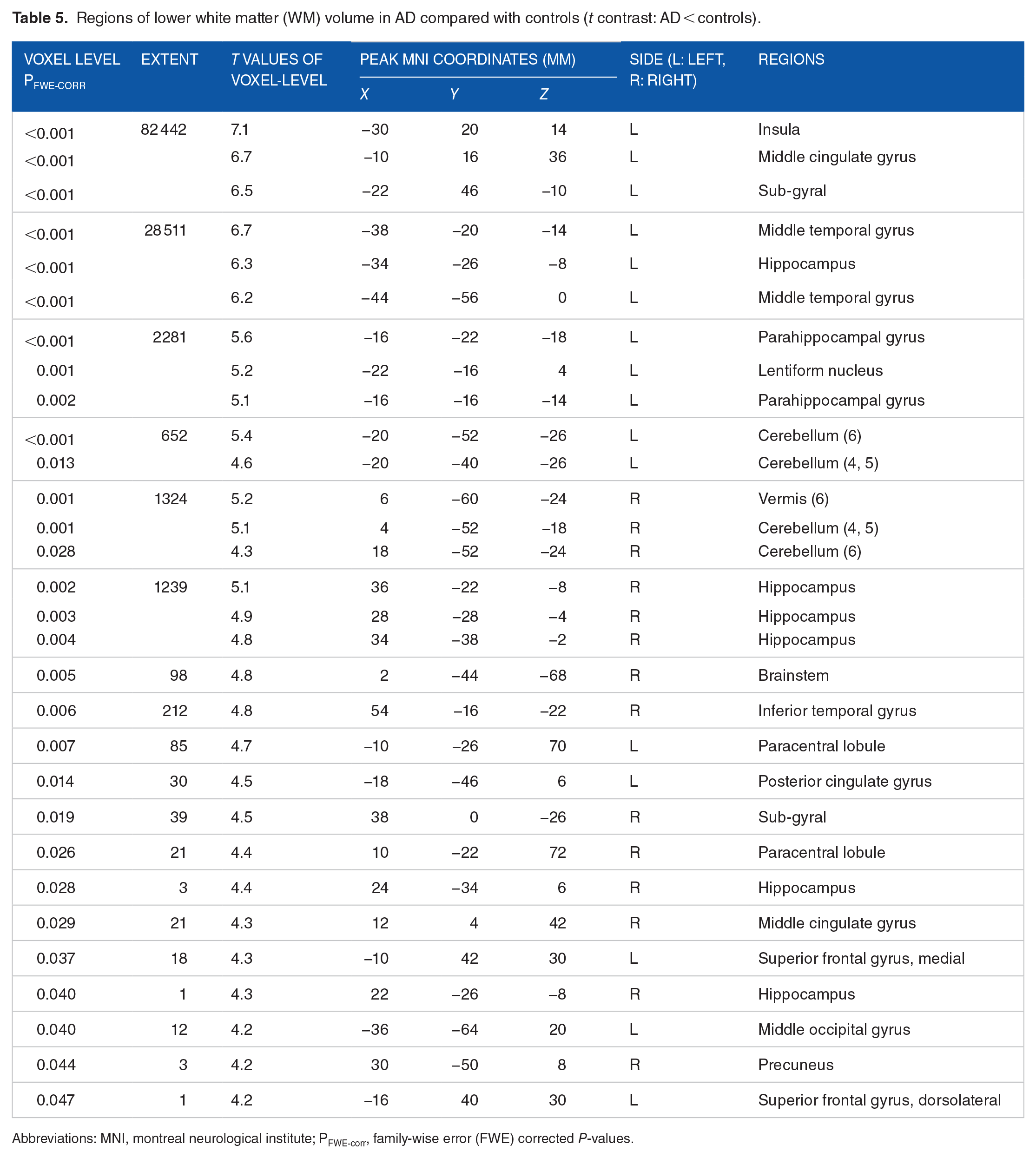
Abbreviations: MNI, montreal neurological institute; PFWE-corr, family-wise error (FWE) corrected

Anatomical rendering in SPM displays VBM results on white matter (WM). The left, middle, and right columns are anterior, right, and bottom views of the 3D brain template, respectively. In the t-contrasts of AD < controls (a-c) and AD-CVD < controls (d-f), the family-wise error (FWE) was corrected at
Comparing the AD-CVD and control groups (Table 6 and Figure 2d-f) revealed that the AD-CVD group showed substantially lower WM volumes in several regions, particularly in the left superior frontal gyrus (dorsolateral part), putamen, and cingulate gyrus, with the largest cluster of 22 973 voxels. The next largest cluster (5731 voxels) was mainly demonstrated in regions of the left brain's middle temporal gyrus and hippocampus. The surrounding areas of the right brain side, including the insula and middle frontal gyrus, thalamus and lenticular nucleus, dorsolateral and medial parts of the superior frontal gyrus and cingulate gyri indicated substantially lower WM in AD-CVD than in controls. Besides, relative to controls, the AD-CVD group demonstrated a lower WM in the left parahippocampal gyrus, bilateral cingulate gyrus, right lobules (4, 5, 6) of the vermis and left paracentral lobule. Further, as shown in Table 6 and Figure 2d to f, small clusters (>100 voxels) were noticed. No significant differences were noticed for the opposite t-contrast (AD-CVD > controls).
Regions of lower white matter (WM) volume in AD-CVD compared with controls (
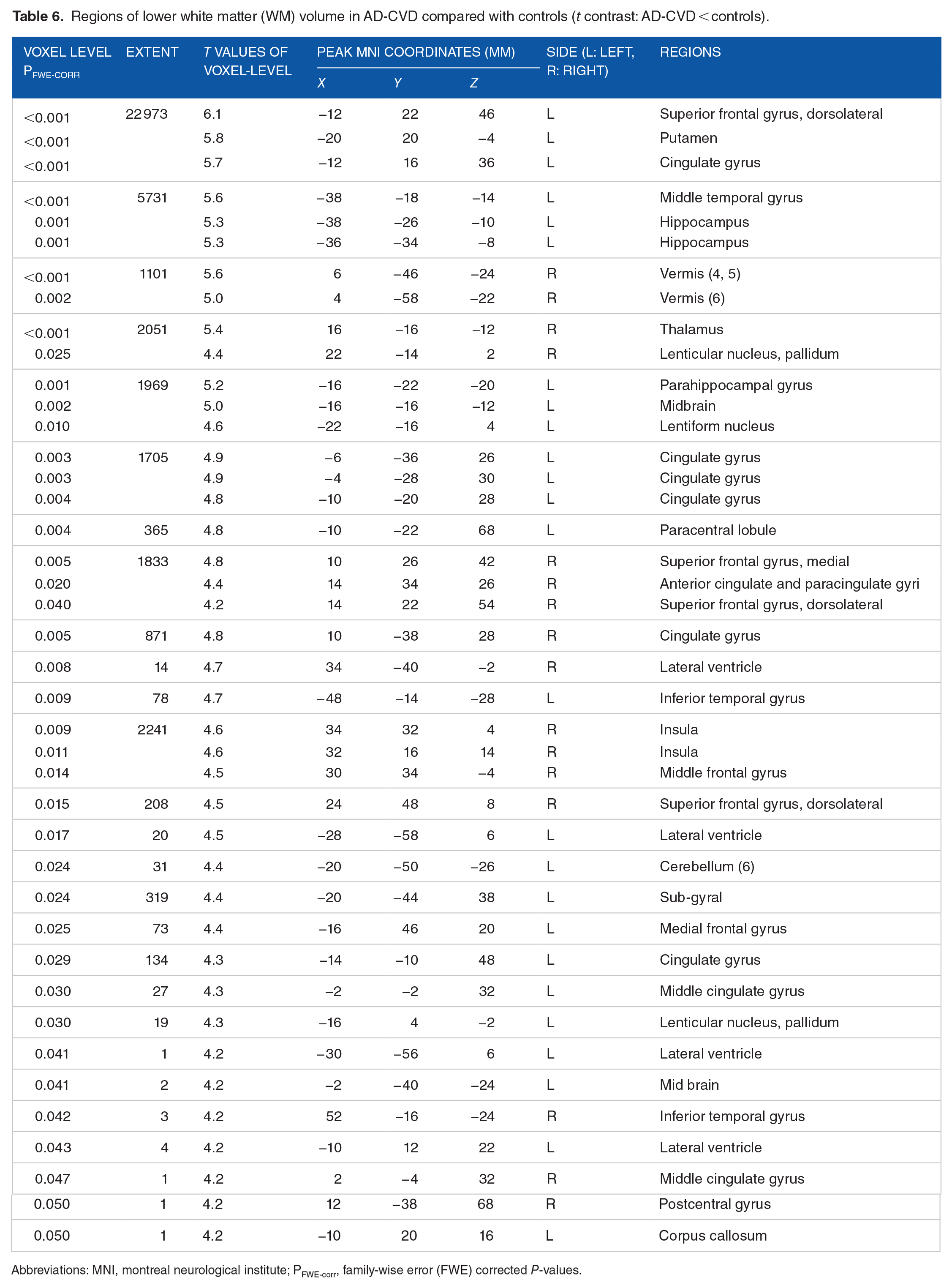
Abbreviations: MNI, montreal neurological institute; PFWE-corr, family-wise error (FWE) corrected
In comparing AD and AD-CVD using the uncorrected thresholding of
Discussion
In this study, we applied VBM to explore the trend of possible differences in GM and WM of AD, AD-CVD and age and sex-matched controls. Both dementia groups (AD, AD-CVD) relative to controls presented an overall lower GM and WM volume in the brain.
Relative to controls, the regions with the most significant cluster of GM difference in both disorders (AD, AD-CVD) were mainly associated with the right sides of the inferior temporal gyrus and parahippocampal gyrus. The inferior temporal gyrus is involved in verbal fluency, and the impairments in these functions are early markers of AD.48,49 The parahippocampus encloses the hippocampus and contributes to the encoding and retrieval of memory. 50 AD and AD-CVD groups also demonstrated a substantially lower GM surrounding the regions of the left thalamus, bilateral gyrus rectus, and parts of the superior frontal gyrus. Other cortical and deep subcortical structures also showed a significantly different GM volume in both disorders compared to controls. It is common to observe a widespread pattern of GM difference in AD individuals compared to controls; 51 a previous meta-analysis on VBM reported GM changes at 334 coordinates in people with AD. 43 Furthermore, the VBM on WM revealed that both dementia groups (AD, AD-CVD), relative to the control group, had WM differences in diverse brain areas. Nineteen different-sized clusters of WM differences in AD compared to controls were shown in VBM analysis covering a large area in the brain, whereas 27 clusters with varying sizes were shown in AD-CVD. Previous studies.21,52 -54 on AD or cognitive impairment individuals showed the WM vulnerability of many of the regions that are reported in this study. In either GM or WM, the patterns of volumetric differences in AD and AD-CVD than in the control group overlap in most regions. It suggests that both dementia groups, despite their different levels of CVD, might have common pathologies of neurodegeneration, such as amyloid plaque and tau deposition.
In our comparison of two groups of dementia, VBM did not present a significant distinction between AD and AD-CVD in GM using thresholding of
Another finding of this study was to observe the tendency of lower GM of the AD-CVD group, particularly in the cerebellar regions compared to the AD group (Table 3). The cerebellar lobules (6, 7b, 8) that showed the GM difference in this study are from the cerebellum’s posterior lobules, engaged in cognitive and sensorimotor tasks. 60 Given that our AD-CVD group (mean age = 76, 2 out of 37 subjects < 65 years) was relatively older than the AD group (mean age = 73.1, 16 out of 83 subjects < 65 years) as well as aging alone could lessen the Purkinje cells of the cerebellum and its volume, reported in an animal study. 61 We thus postulated that there might have been an age effect on our AD-CVD subjects for having lower GM in the cerebellar regions compared to AD.
In the WM analysis to compare two dementia groups, we observed a WM difference in AD compared to AD-CVD, mainly in the right thalamus. In contrast, one large cluster (1534 voxels) of difference was shown in the right brainstem of the AD-CVD compared to AD. The regions, that is, cerebellum and brainstem, that respectively showed lower GM and WM in AD-CVD participants compared to AD are known to be associated with the control of balance. It is noteworthy that the incidence of falls was reported to be higher, albeit not significantly, in VaD compared to AD. 62 This might indicate the contribution of CVD by affecting the brain regions that are involved in the regulation of balance. Another finding is that the trend of differences between AD and AD-CVD was found to be evident in VBM analysis on GM rather than WM.
The supplementary file presented VBM results for comparing dementia groups (AD, AD-CVD) and controls (Supplemental Tables S1-S3, Supplemental Figures S1-S2) after smoothing the modulated GM and WM images by 12 mm FWHM Gaussian kernel. There were differences in VBM results of GM and WM between using 8 and 12 mm smoothing kernels for comparing AD versus controls and AD-CVD versus controls; in particular, these VBM results differed in the size and number of clusters of differences and the locations of peak MNI coordinates. Nevertheless, two different smoothing kernels did not considerably affect the VBM results in comparing AD and AD-CVD groups; these two VBM results seemed similar in most distinct areas (see Table 4 and Supplemental Table S2).
A few studies using other MRI sequences showed a potential abnormality in the WM fiber of the AD-CVD cohort. For example, Jang et al, 27 using diffusion tensor imaging (DTI), reported a more extensive change of WM integrity in subcortical VaD and AD-CVD groups; however, the alternation was minimal in the pure AD group, compared to controls. Ji et al 30 hypothesized that the AD-CVD subjects might have higher free water values, representing more disruption in WM than the AD without CVD. A recent pilot study Lee et al63 reported the potential abnormalities in WM tissue in the AD-CVD cohort.
This study has strengths as well as limitations, which suggest future works. 3T MRI data for all three groups were used in this study. VBM, an unbiased, automatic, and widely used tool for group-wise volumetric comparisons, 64 reduced any potential bias associated with imaging analysis. We had diagnostic reports with brain imaging findings for 69 participants from their neurologist or neuropsychiatric; for the rest, we estimated the HIS scores based on the available information regarding their medication, medical history, and caregiver comments for grouping them as AD and AD-CVD. We also acknowledged our small sample size in this study. Nevertheless, the brain areas that showed a trend of potential differences between AD and AD-CVD encouraged conducting further analysis on large samples; they might also be informative in future studies for generating hypotheses in specific regions of interest for separating AD and AD-CVD.
Conclusion
VBM exhibited a significant volumetric difference in GM and WM of AD and AD-CVD groups compared to control subjects. AD and AD-CVD also showed a trend of possible differences in GM and WM, and these preliminary findings demand further analysis in a larger population.
Supplemental Material
sj-docx-1-exn-10.1177_26331055231225657 – Supplemental material for Gray and White Matter Voxel-Based Morphometry of Alzheimer’s Disease With and Without Significant Cerebrovascular Pathologies
Supplemental material, sj-docx-1-exn-10.1177_26331055231225657 for Gray and White Matter Voxel-Based Morphometry of Alzheimer’s Disease With and Without Significant Cerebrovascular Pathologies by Chandan Saha, Chase R Figley, Zeinab Dastgheib, Brian J Lithgow and Zahra Moussavi in Neuroscience Insights
Footnotes
Acknowledgements
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
Abbreviations
AD, Alzheimer’s disease; AD-CVD, AD with cerebrovascular disease; ADNI, Alzheimer’s Disease Neuroimaging Initiative; CAT, Computational Anatomy Toolbox; CSF, cerebrospinal fluid; CVD, cerebrovascular disease; DTI, diffusion tensor imaging; FWE, family-wise error; GM, gray matter; HIS, Hachinski Ischemic Score; MNI, Montreal Neurological Institute; MPRAGE, magnetization prepared rapid acquisition gradient-echo; MRI, magnetic resonance imaging; rTMS, repetitive transcranial magnetic stimulation; SPM, Statistical Parametric Mapping; SVD, small vessel disease; TIV, total intracranial volume; VD, vascular dementia; NINDS-AIREN, National Institute of Neurological Disorders and Stroke-the Association Internationale pour la Recherche et l’Enseignement en Neurosciences; WM, white matter; WMHs, white matter hyperintensities.
Authors’ Note
Cognitively normal subjects’ data used in the preparation of this article were obtained from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database (adni.loni.usc.edu). As such, the investigators within the ADNI contributed to the design and implementation of ADNI and/or provided data but did not participate in the analysis or writing of this report. A complete listing of ADNI investigators can be found at: content/uploads/how_to_apply/ADNI_Acknowledgement_List.pdf.
Author Contributions
CS analyzed the MRI data, ran statistical analysis and drafted the manuscript. CRF contributed to MRI analysis and writing the paper. ZD contributed to the participants’ grouping and discussion. BL contributed to writing the results and discussion. ZM supervised the entire study and reviewed the paper and contributed with editing the paper.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Weston Brain Institute.
Data Availability Statement
The MRI datasets for our AD and AD-CVD participants can be available upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
