Abstract
The transient receptor potential (TRP) superfamily of ion channels in humans comprises voltage-gated, non-selective cation channels expressed both in excitable as well as non-excitable cells. Four TRP channel subunits associate to create functional homo- or heterotetramers that allow the influx of calcium, sodium, and/or potassium. These channels are highly abundant in the brain and kidney and are important mediators of diverse biological functions including thermosensation, vascular tone, flow sensing in the kidney and irritant stimuli sensing. Inherited or acquired dysfunction of TRP channels influences cellular functions and signaling pathways resulting in multifaceted disorders affecting skeletal, renal, cardiovascular, and nervous systems. Studies have demonstrated the involvement of these channels in the generation and transduction of pain. Based on the multifaceted role orchestrated by these TRP channels, modulation of the activity of these channels presents an important strategy to influence cellular function by regulating intracellular calcium levels as well as membrane excitability. Therefore, there has been a remarkable pharmaceutical inclination toward TRP channels as therapeutic interventions. Several candidate drugs influencing the activity of these channels are already in the clinical trials pipeline. The present review encompasses the current understanding of TRP channels and TRP modulators in pain and pain management.
Introduction
The transient receptor potential (TRP) superfamily is an assembly of nonselective cation channels that functions as molecular sensors responding to intra- and extracellular environmental alterations.1 -3 They are evolutionary conserved integral membrane proteins. 4 Several TRP channels are polymodal, facilitating cellular signal integration, molecular sensing and coincidence detection. 5 These channels have been documented in many eukaryotes, extending right from single-cell green algae to mammalian systems. 6 TRP channels have been found to be intricated in a variety of biological processes linked to thermo-, chemo-, photo-, and mechanosensation, in addition to maintaining homeostasis.
TRP channels in mammals are grouped into 6 subfamilies namely TRPA (ankyrin), TRPC (canonical), TRPM (melastatin), TRPV (vanilloid), TRPML (mucoliptin), and TRPP (polycystin), on the basis of sequence similarity.4,7,8 Among these channels, TRPC exhibits the nearest homology to the Drosophila TRP, involved in phototransduction. Mammalian TRPC channels are non-selective Ca2+ permeable cation channels that are triggered by downstream activation of phospholipase C cascades through G protein-coupled receptors (GPCRs). These channels are a cluster of 7 diverse proteins subdivided into 4 subgroups including TRPC2, TRPC3,6,7, TRPC1, and TRPC4/5 based on amino acid homologies.9,10 Although knowledge in relation to the vital physiological functions of TRPC channels remains incomplete, their involvement in a variety of pathological pathways and conditions of the cardiovascular system, nervous system, and kidneys is driving the exploration for TRPC channel modulators as potent therapeutic agents. 3
TRP channels dysfunction has been intricated in diverse disease conditions encompassing from overactive bladder (TRPV1) through obesity (TRPV4 and TRPM5), chronic cough (TRPA1, TRPV1) and pain, diabetes (TRPV1, TRPM4), chronic obstructive pulmonary disease (TRPV4) to cardiac hypertrophy (TRPC6), dermatological disorders (TRPV3), cancer (TRPC6, TRPV2 and TRPM8) and familial Alzheimer’s disease (TRPM7).11 -20 Gain-of-function mutations in genes that encode for TRP channels have been associated with human ailments as revealed in familial episodic pain syndrome (TRPA1). 21 Thus, regulating these hyperactive TRP channels by antagonist mediators may prove to be clinically favorable. Besides, loss-of-function mutation like loss of TRPML1 function (type-IV mucolipidosis) is also alarming. 22 The present systematic review highlights the emergence of these TRP channels in regulating diverse biological functions offering novel and promising targets for drug development. The involvement of TRP channels in various organ-specific diseases has been represented in Figure 1.
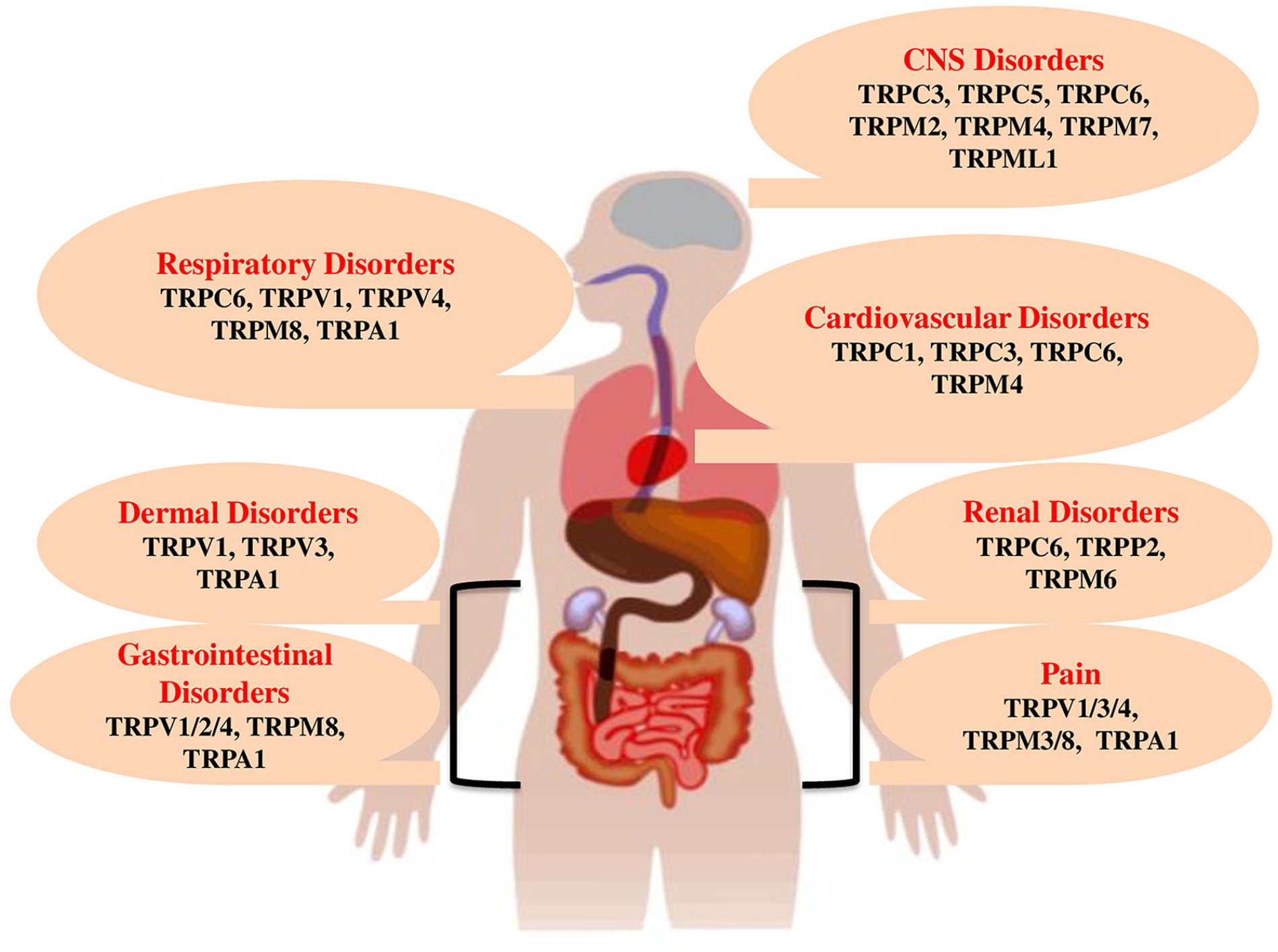
TRP channels and associated organ-specific etiologies.
Pain and Its Types
Chronic Pain is a debilitating indicator of diverse ailments resulting in notable disability and reduction in the quality of life. Effective pain management strategies are in their infancy thereby posing a global unmet challenge for health. The complexity in the understanding and treatment of pain has paved the way for extensive and prolonged overuse of morphine/opioids manifestation leading to serious adverse events.
Pain can be majorly categorized into 3 types: nociceptive, nociplastic and neuropathic. Nociceptive pain is the most common chronic pain that arises from the activity in neuronal cascades encompassing majorly spinal pain and arthritis. Nociplastic pain, on the other hand, is caused by the abnormality in the processing of pain signals lacking the involvement of damage or disease pathophysiology. About 15% to 25% cases of chronic pain belong to neuropathic pain that arises due to an injury or disease targeting the somatosensory nervous system characteristically linked with sensory anomalies like allodynia and numbness. It also includes postherpetic neuralgia, diabetic neuropathy and radiculopathy. Allodynia and hyperalgesia are common symptoms of the disorder and may be beneficial adaptations to prevent susceptible organs from damage. However, elevated sensitivity for pain may persist prolong after the initial cause for pain. For addressing the challenges associated with pain management and improving the quality of life, a better understanding of the mechanisms underlying the pain machinery is warranted so as to come out with advanced treatment modalities.23 -27
Nociceptive Pain
The generation of nociceptive pain is orchestrated by specific nerve endings called nociceptors that possess a comparatively greater activation threshold in relation to those that are involved in the sensation of taste, smell, sound, and light. Nociceptors are potent sensors for noxious thermal stimuli, chemical irritants, strong mechanical stimuli and those stimuli that can potentially cause tissue damage reaching the threshold to trigger nociceptive nerve termini generating action potentials, which later are transmitted and apparent as pain signals. These stimuli are then transformed into electrical signals and transduced as action potentials with the involvement of several channels and receptors situated on free synaptic terminals and nerve endings. Together as a class of cation-permeable polymodal channels involved in cell depolarization, TRP channels finds its association with various physiological roles, including nociception.28 -30
In the recent decade, extensive research has been carried out on mechanisms underlying the molecular machinery in chronic pain, thereby rendering striking avenues for novel, safe, and efficacious pharmacological interventions. In agreement with this, TRP ion channels offer attractive targets to combat pain.
Nociception and TRP Channels
Pain particularly in the trigeminal regions is orchestrated by nociceptive trigeminal afferents. Exogenous and endogenous ligands activate transduction molecules including nonspecific cation channels TRPV1 and TRPA1 that are articulated by a noteworthy population of trigeminal nociceptors distributed across meningeal tissues. The majority of these nociceptors possess vasoactive neuropeptides namely substance P and calcitonin gene-related peptide (CGRP). 31 The nociceptive pathways are triggered during pathological conditions through neuropeptides, augmented trafficking and expression of TRP channels, phosphorylation and other associated mediators. Such nociceptor stimulation could be a remarkable pathophysiological machinery linked to both central and peripheral sensitization of the trigeminal nociceptive cascade contributing to the pathophysiology of nociceptive pain.32-34
The functional characterization of nociceptive neurons is according to the ion channels and the type of sensory receptors present on the plasma membrane across the somata and nerve fibers. These channels and/or receptors play a pivotal role in the detection of diverse noxious stimuli.33,34 The nociceptive neurons convert these noxious signals into electrical signals and finally transmit the information to the central nervous system. Nociceptive channels and/or receptors of the TRP family is comprised of a wide group of molecular transducers/detectors. Depending on their capability in the transduction and detection of explicit nociceptive modalities TRPV, TRPA and TRPM have been categorized as “nociceptive TRP channels.” Stimulation of these channels upon pain-inducing and/or specific noxious stimuli serves as the chief mode in the transduction and detection of pain during physiological and pathological situations. Additionally, modulation of the trafficking properties and/or channel functionality together with the alterations in gene expression profile of the nociceptive TRP channels are regarded to be extremely crucial in impacting nociception and pain perception during diverse pathophysiological conditions.35,36
Members of the TRP ion channel family offer attractive targets to combat pain sensation at the root level. Among them, importantly 3 TRP channels, TRPV1, TRPA1, and TRPM3 are of significant interest, as they were deciphered as sensors of chemical and heat-induced pain in nociceptor neurons. A growing body of recent findings using mice models demonstrated that noxious heat sensing relies on these 3 channels. Furthermore, these channels share a partly overlapping expression profile, especially in nociceptor neurons, and are involved in hypersensitivity and pathological pain signaling, offering challenging targets for analgesic drug development.29,37 -39
On similar lines, TRPV4, a polymodal, non-selective cation channel that senses mechanical, thermal, and environmental signals has also attracted researchers attraction owing to its association with a range of diverse physiological functions. The consequences of chronic TRPV4 activation and gain-of-function genetic mutations indicate that TRPV4 may also be a potent therapeutic target for peripheral neuropathies, neurogenic inflammation, and impaired wound healing. Endogenous stimuli namely lipid binding and osmotic stress are recognized TRPV4 activators. G protein-coupled receptors (GPCRs) utilize TRPV4 as an effector protein to augment inflammatory and nociceptive signaling. Additionally, in pathophysiological conditions like pulmonary edema, neuropathies, and, arthritis, GPCRs also mediate the stimulation of kinase signaling pathways to enhance TRPV4 phosphorylation, leading to enhanced neuronal excitability and sensitization. Besides, phospholipase activity also contributes to the generation of polyunsaturated fatty acid lipid mediators that directly trigger TRPV4.40,41
TRP Channels (TRPV1, TRPA1, TRPM8): Promising Targets for Drug Development
Irrespective of the TRP channels being highly conserved evolutionarily, their sensitivity to extrinsic stimuli exhibits remarkable species-linked disparities. Owing to this fact, recent research has been inclined toward deciphering the clinical implications of TRP channels in diverse diseases. The findings of these studies have revealed 4 types of TRPV channels (TRPV1-4) majorly expressed in afferent nociceptors, pain-sensing neurons, and participate in the transduction of thermal and chemical stimuli. 42 Prominently, TRPV1-3, and TRPM8 act as thermoreceptors, and TRPA1 and TRPV4 act as mechanoreceptors. These ion channels associated with thermal, chemical, and mechanical sensations can therefore be promising targets to combat chronic pain.
Ligands bind to these channels (TRPV1, TRPA1, and TRPM8
TRPV1 as a Drug Target
TRPV1 is a very good example of a TRP channel exhibiting diversity in function and expression. It is prominently expressed on primary sensory neurons as an important inducer of neurogenic inflammation and a key integrator of painful stimuli. 44 In addition, sensory neurons expressing TRPV1 have been associated with itching and heat sensing. The pioneering work of David Julius using capsaicin, a natural pain inducer revealed the TRPV1 ion channel as a heat-activated nociceptor in the peripheral nervous system. For this breakthrough discovery, the Royal Swedish Academy of Sciences awarded the 2021 Nobel Prize in Physiology and Medicine to David Julius. 45 Neuronal TRPV1 in the viscera stimulates reflex cascades including intestinal peristalsis, cough, micturition, and heart rate. Brain nuclei and non-neuronal cells also express TRPV1 but at lower levels. 46
TRPV1 agonists, MRD-652 and olvanil (NE19550) orchestrating different activation kinetics of the receptor have been developed. Both the compounds exhibited promising results in animal models of pain, however, their clinical manifestations are yet to be carried out. 47 Resiniferatoxin, an ultrapotent capsaicin analog, is undergoing the phases of clinical trials to check its efficacy as a permanent analgesia in cancer subjects with severe intractable pain, and in chronic osteoarthritic pain. It has been tested in a small-scale study involving females with cervical cancer, and the outcomes have revealed promising results. 48 Recent studies have demonstrated vitamin D playing the role of a partial agonist of TRPV1 at physiologically relevant free plasma levels.49,50
Parallely, several TRPV1 antagonists have also been deciphered, that have been developed as clinical lead candidates. However, their efficacies exhibited variations in preclinical models of pain. Few of them were found to be effective in neuropathic and inflammatory pain models, while the rest showed efficacy only in the inflammatory pain models. The majority of the first-generation TRPV1 antagonists induced unwanted on-target adverse consequences, because of which they were withdrawn from clinical trials. GSK’s SB-705498 was the first TRPV1 antagonist that progressed to clinical development in the year 2005 and later failed to execute the desired effects. Recent findings have however discovered (S)–N-(3-isopropylphenyl)-2-(5-phenylthiazol-2-yl) pyrrolidine-1-carboxamide to be a powerful and brain-penetrant TRPV1 antagonist.51,52
Animal model studies employing TRPV1 antagonist, GRC-6211 suggested therapeutic potency in suppressing chronically inflamed bladder hyperactivity, although clinical studies pertaining to their implication in human subjects remain to be established. Antagonist like MK-2295 was withdrawn owing to the burn injuries and AMG-517 due to febrile reactions. Moreover, some of these antagonists namely AZD-1386 that entered into phase II of clinical trials were terminated as they failed to execute the analgesic action in osteoarthritic pain. Other terminated antagonists in clinical trials include PHE-575 and GRC-6211.52 -54 TRPV1 antagonist variability may be attributed to the fact that a partial agonist can orchestrate its action either as an agonist or antagonist, based upon the occurrence of additional ligands.
Interestingly, besides these terminated candidate drugs, some promising developments have been noticed. For instance, NEO6860 exhibited an analgesic pattern but failed to outshine the placebo statistically without impacting heat pain perception and body temperature.55,56 Similarly, SB-366791 succeeded in attenuating dental pain in rats and was also found to be efficacious in preventing alveolar bone mass loss in a periodontal disease rat model. 57 Additionally a second-generation compound mavatrep (JNJ-39439335) revealed remarkable success when compared to placebo in stair climbing-provoked pain in subjects suffering from knee osteoarthritis. 58 Clinical trial studies with ABT-102, a polymodal antagonist, were also aborted owing to a clinical risk of burns. However, so far these molecules or other thermoneutral antagonists namely GRTE16523 could not orchestrate substantial analgesic efficacy clinically. Ongoing phase III clinical studies with PAC-14028, a TRPV1 antagonist, have been found to relieve pruritus in human subjects with atopic dermatitis and improve skin barrier functions.53,59 -61
Various pharmaceutical companies faced similar challenges in the pursuit for TRPV1 antagonists. Neurogen and Merck collaboratory development of MK-2295/NGD8243 revealed impaired sensitivity to noxious heat. 62 Besides, antagonists like V116517 demonstrated a safety status in humans but a dosage-dependent rise in body temperature was evident in the rat model. Further, clinical studies so far with V116517 have not been registered. Few antagonists like GRC-6211 have been terminated without providing specifications. Promising polymodal TRPV1 antagonists, JTS-653 and DWP05195 were unable to progress beyond phase II clinical trials. 63 Presently, a small interfering RNA Tivanisiran (SYL1001) is under clinical development against dry eye disease, wherein the nociceptive channel is believed to be associated with ocular inflammation and pain. Topical use of this siRNA has been found to be efficacious with no tolerability concerns. Irrespective of notable investments, to date no TRPV1 antagonist has been reported for clinical use. Moreover, multiple endogenous compounds released under conditions of oxidative stress and inflammation have been described to activate TRPA1, evidently pointing toward a role of the channel in nociception and pathological pain.
TRPA1 as a Drug Target
TRPA1 channels have caught the attention of researchers as an alternative drug target to TRPV1. These channels are reported to be stimulated by a wide range of irritants namely allyl isothiocyanate, allicin, and cinnamaldehyde.64,65 Furthermore, an array of endogenous molecules released during inflammation and oxidative stress have been found to be involved in TRPA1 activation, evidently highlighting a potential role of this channel in pain and nociception. A growing body of evidence employing animal models suggests the therapeutic potency of TRPA1 antagonists namely HC-030031, A-967079, and AMG0902 in human subjects undergoing neuropathic pain.47,66 The combined supplementation of CHEM-5861528 and streptozotocin to rats was found to prevent intraepidermal nerve fiber loss and attenuated pain. So far, GRC-17536 developed by Glenmark is the only TRPA1 antagonist to complete the phase II clinical trials. Even though it noticeably curtailed the pain in non- denervation patients having painful diabetic polyneuropathy with no adverse effects, GRC-17536 failed in terms of pharmacokinetics and bioavailability due to which the molecule was terminated before entering phase III trials. Also, it exhibited poor pharmaceutical characteristics in terms of lipophilicity, bulky structure, and poor solubility. 67 Amgen developed AMG9090, a partial agonist at rTRPA1 and an antagonist of hTRPA1, which was one of the candidate molecules among a class of species-specific TRPA1 modulators. ODM-108, developed by Orion Corporation, which was a negative allosteric modulator of TRPA1 could not progress to phase II clinical trials owing to its complexity in pharmacokinetic outcomes. 54 Interestingly HC-030031, a topical gel effectively reversed cold and mechanical allodynia in mice model with burn injuries induced by exposure to ultraviolet B rays. This indicates the therapeutic potential for TRPA1 antagonist gels/creams for subjects suffering from heat injury and/or sunburn pain.68,69 Furthermore, patents for small molecule candidates targeting TRPA1 channels have been filed by several drug-developing pharma companies including Eli Lilly, Genentech/Roche, Algomedix Inc., Glenmark, Boehringer Ingelheim, Almirall S.A., Orion Corp., EA Pharm Co. and Mandom Corp.
TRPA1 antagonists HC-030031 and GRC-17536 are effective anti- tussive mediators.70,71 Recent findings have revealed GDC-0334, a TRPA1 antagonist to possess better target engagement in human subjects. In preclinical studies involving respiratory disease models, GDC-0334 exhibited inhibition toward edema formation, smooth muscle hyperreactivity, and cough response.72,73 Drug discovery strategies have been challenged owing to the limited bioavailability of TRPA1 antagonists.
TRPM8 as a Drug Target
In humans, reduced expression of the TRPM8 gene has been linked with a decreased risk for migraine and decreased cold pain sensitivity. Owing to this fact, TRPM8 antagonists can be employed as a therapeutic intervention in the treatment and management of migraine and also cold-induced pain. Notably, TRPM8 orchestrated a basal activation tone similar to that of TRPV1. Antagonists to TRPM8 channels have been shown to induce a mild hypothermic response, which never restricted them for their use in clinical practice. Two TRPM8 antagonists, PF-05105679 and AMG-333 are already in the clinical studies pipeline which has been found to reduce pain sensation in the cold pressor test. Few of the clinical trial participants experienced a “hot feeling” in the perioral region with the administration of PF-05105679 whereas AMG-333 orchestrated grade 1 side effects in a few volunteers.74,75 In addition, animal studies using KRP-2529 and RQ-00434739 (TRPM8 antagonists) indicated therapeutic efficacy in suppressing chronically inflamed bladder hyperactivity, though clinical studies pertaining to their usage in human subjects is yet to be established. 47 Besides TRPM8 antagonists, TRPM8 agonists like DIPA (di-isopropyl-phosphinoyl-alkane) and WS-12 also possess an analgesic potential. DIPA has been found to augment impulsive painful contractions in the distal colon in humans. TRPM8 agonists have also been reported to maintain the moisture content in the cornea of patients suffering from dry eye disease.75 -77
Recent studies have been carried out to design, synthesize and biologically evaluate new thiazole scaffolds as potent antagonists of TRPM8. 78 Also, an assay has been developed for the rapid identification of TRPM8 antagonists employing automated patch clamp electrophysiology together with virtual screening. With this approach, nebivolol was found to be a potential TRPM8 inhibitor, and the structural modification of its structure can lay a foundation for developing new TRPM8 blockers. 79 Table 1 represents the progress of various TRP modulators in the clinical context.43,80 -88
Agonist and antagonists of TRPV1, TRPA1 and TRPM8.

Knockout Animal Models and TRP Pain Connection
Thermal place preference tests with TRPM8-/- mice suggest reduced sensation across the innocuous low-temperature range. Although colder temperatures revealed a return of temperature preference and cold plate paw withdrawal responses were found to be normal in TRPM8-/- mice some attenuation of thermal preference was observed at temperatures as low as 5°C. Likewise, the flicking responses owing to evaporative cooling from acetone were notably seen to be reduced in TRPM8-/- mice. In the spinal nerve ligation (SNL) neuropathic pain model, research findings have demonstrated that antisense knockdown of TRPA1 by intrathecal delivery, but not TRPM8, resulted in alleviated cold hyperalgesia (Carlos Fernández-Peña and Felix Viana*). Moreover, spinal TRPV1 knockdown using antisense oligonucleotide exhibited concomitant results.89 -92
Conclusion
Management of pain has always been a challenging task and provides an interesting scientific area for clinical attention. With the advent of understanding the machinery of nociceptive TRP channels, a wide range of safe and potent analgesics have been developed. The majority of the ligands have orchestrated pain relief but also have many negative side effects. Progress in TRPV1 antagonist development has largely been influenced by the clinical candidate molecules that led to hyperthermia and elevation of the heat pain threshold. On a similar line, TRPA1 channel-targeted drug development has also been influenced by pharmacokinetic and pharmacological complications. Albeit, several scientific evidences have helped in understanding the molecular machinery of pain, the biology of nociception is still in its infancy. Therefore, deep insights into the knowledge of nociception undoubtedly may lead to the development of more efficacious and potent drugs to tackle multiple pain pathologies.
Footnotes
Acknowledgements
The authors acknowledge the Department of Biotechnology, School of Applied Sciences, REVA University for providing the necessary facilities.
Author Contributions
Both the authors have contributed equally in all the aspects.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
