Abstract
Background:
There has been rapid drift of rehabilitation professionals toward the clinical use of technology aided electrical interventions. Brain is a cortical hub of functionally related neural connections. Motor learning entails strong interaction with the cognitive domains. So better outcomes may be expected by optimally targeting functionally correlated areas simultaneously through tDCS.
Aim:
To determine the therapeutic effect of Multi Channel tDCS in combination with Functional electrical stimulation, SaeboFlex and conventional rehabilitation on recovery of Cognitive Domains, Motor Functions of Paretic Hand, and Gait in individuals with subacute Stroke.
Methods:
This is prospective, randomized, double blind controlled clinical trial. Subacute Stroke Survivors with the age Group (40-75 years) will constitute the Population of the study. Participants will be randomly allocated to experimental or control group. Participants of Experimental group will receive Multi channel tDCS, Functional electrical stimulation, Saebo Flex Training and conventional rehabilitation. Participants of the group B will receive FES, training with SaeboFlex, conventional physiotherapy intervention similar to as given to the participants of group A and sham multi channel tDCS.
Outcome Measures:
The primary outcome measures of the study will be Fugl Meyer assessment, Electroencephalogram and secondary outcome measures of the study will be Grip strength, Pinch strength, Nine hole peg test( NHPT), Wisconsin gait scale, Montreal cognitive assessment, Electroencephaloraphy to observe the cortical changes and tDCS adverse effect questionnaire and stroke specific quality of Life scale.
Statistical Analysis:
The primary analysis of the study will be done at the end of 4 weeks. Statistical analysis of data will be done using SPSS Version 22 with the help of a statistician.
Conclusion:
An insight into the therapeutic interventions augmenting, cognitive and motor domains simultaneously may yield better outcomes in the field of stroke rehabilitation thereby improving quality of life of stroke survivors
Keywords
Background
Stroke is the third leading cause of Disability-Adjusted Life-Years (DALYS) 1 and the second leading cause of death across the globe. 2 India, being one of the most populous countries, stands second in having the highest rates of mortality and morbidity due to stroke. 3 A range of signs and symptoms appear following a stroke that varies from altered levels of consciousness to motor, sensory, cognitive, perceptual, and psychological impairments, depending upon the extent of damage in the focal areas of the brain. Among the constellation of symptoms, motor impairment affecting both upper and lower extremities is prevalent in more than 8% of stroke victims. 4 Motor impairment also accounts for difficulty in walking for stroke survivors. 5 Locomotion difficulties occur due to changes in kinematic parameters, that is, decreased hip and knee flexion, increased knee extension and decreased ankle dorsiflexion in stance and swing phase of gait cycle respectively. 6
The primary goal of any rehabilitation is to achieve the highest level of recovery by choosing appropriate interventions while considering factors such as injury characteristics and preserved inter-hemispheric integrity. 7 Re-acquisition of motor functions depends on the integrity of inter-hemispheric motor circuits. In the year 1949, Donald Hebb proposed the rule that “Neurons that fire together, wire together.” Hebb’s rule provided the theoretical base that homosynaptic and heterosynaptic activities, facilitate synaptic formation and consolidation during motor rehabilitation. 8 Due to this interconnected network, motor learning entails strong interactions with cognitive domains like attention, planning, memory and execution control. Cognitive impairment has a great influence on motor outcomes and the functional independence of stroke victims. Hence, cognitive deficits cannot be left neglected while working toward motor rehabilitation after stroke. 9
Traditional motor rehabilitation strategies involve different approaches that improve motor functions either by minimizing the level of impairment or by developing compensatory strategies. But ongoing research has revealed that these rehabilitation strategies promote contra- and ipsilesional plastic changes after stroke, the functional outcomes lack practical significance. Even after completing standard rehabilitation, approximately 50% to 60% of patients still exhibit some degree of motor impairment and require at least partial assistance in activities of day-to-day living. In essence, the model proposes that motor deficits are due to reduced output from the damaged hemisphere and excess inhibition of the damaged hemisphere from the intact hemisphere.26,28 Thus, improvement may be possible by either up-regulating excitability of the lesioned motor cortex or down-regulating excitability in the intact motor cortex. A growing body of evidence from small clinical trials has demonstrated the efficacy of both approaches to induce considerable changes in cortical excitability, which often correlate with relevant clinical gains in motor functions. 10
Electrical interventions in the form of transcranial direct current stimulation (tDCS) are proving to be a promising tool for adding therapeutic benefits in post-stroke outcomes. The brain is a complex, plastic, electrical network operating at multiple scales—neural processing is essentially mediated by functional and structural networks. In most of the trials, single-channel tDCS has been successively applied to different cortical areas targeting motor and cognitive deficits in isolation at different points of time to yield potential outcomes. However, to the best of our knowledge, simultaneous targeting for multiple disconnected brain regions is hard to find in the literature. Transcranial direct current stimulation is suited to improve post-stroke rehabilitation outcomes, but effect sizes are often moderate and suffer from variability. Indeed, the location, extent, and pattern of functional network connectivity disruption should be considered when determining the optimal location sites for tDCS therapies.11,12
The clinical utility of multichannel transcranial direct current stimulation (M-tDCS) in post-stroke rehabilitation needs to be explored. M-tDCS is a network-oriented, neuromodulatory technique, through which multiple areas of the cortex can be targeted simultaneously. The identification of involved networks in a patient-specific context, along with a deeper understanding of interconnected neuronal populations, may yield better outcomes when applied alongside conventional rehabilitation. 12 Hence, looking at the dearth of evidence, this study is an attempt to strengthen the interhemispheric circuitry model by determining the therapeutic benefits of M-tDCS by targeting the motor cortex and cortical area for cognitive functions concurrently at the same time point.
Aim and Objectives
Aim
The primary aim of this study is to determine the therapeutic effect of multi-channel transcranial direct current stimulation (M-tDCS) on the recovery of cognitive domains, motor functions of paretic hand and gait in individuals with subacute stroke.
Materials and Methods
Research design
This is a prospective, 2-group, parallel design, double-blinded randomized controlled clinical trial in which males and females of age group (40-75) years diagnosed with cortical stroke will constitute the population of the study.
Participant recruitment
The participants will be recruited from various Physiotherapy/rehabilitation Centers/Hospitals in Punjab. The recruitment will be done from January 2020 and December 2021. The study will be conducted in the neuro-physiotherapy rehabilitation unit of the department of physiotherapy, Punjabi University Patiala, and from the neuroots- neuro-rehabilitation center in Patiala. Participants will be recruited by referral from eminent neuro physicians and neurosurgeons from different regions in Punjab. Participant information sheets will be given to all the participants and written consent will be obtained from all of them prior to the start of the study. Participant sheets will be provided in the local language. All the participants will be notified and well informed regarding study objectives, procedure, interventions, potential risks and /or expected benefits. They will be informed that their participation will be voluntary and they have full rights to withdraw at any point of time during the study without having any influence on future treatments provided by their site, clinicians, and research team.
Eligibility criteria
Inclusion criteria
Individuals, both males and females, within the age group of 40 to 75 years old, diagnosed with the following:
Subacute stroke (Ischemic type)
Cognitive impairment (MMSE Score:18-23)
Preserved range of wrist extension (≈10°)
Modified Ashworth scale score <2 and
Able to walk able to walk independently with or without the use of walking aid
Exclusion criteria
Individuals not fulfilling the criteria of the study with the following
Diagnosed with Hemorrhagic stroke
Visual analog scale for pain score >4 in the upper extremity/lower extremity.
History of neurological diseases other than stroke
History of musculoskeletal injury/disease affecting upper Extremity and lower extremity motor functions
Diagnosed with Psychosomatic Illness/Disease
Who are medically unstable due to any Cardiovascular or respiratory illness.
Uncontrolled hypertension.
Individuals taking neuropsychiatric drugs
Diagnosed with any Systemic illness
Pregnant women
Metallic Implants
Participants of the other pharmacological & rehabilitation studies during the study period
Hypersensitivity or hypersensitivity disorders.
Non-Cooperative Individuals
Who are not willing to participate in the study.
Randomization, Allocation, and Blinding
A total of 60 participants will be recruited. The randomization schedule for all participants will be established before recruitment by the investigator, using a computer-generated random list. Participants will be randomly allocated into 2 groups,’ that is, experimental group that is, multichannel transcranial direct current stimulation group (M-tDCS) and control group, that is, sham multichannel transcranial direct current stimulation group (sham M-tDCS). In both groups, participants will receive standard physical therapy intervention including the training of the paretic hand with Saebo Flex, functional electrical stimulation on the paretic leg and a conventional bank of exercises. Participant allocations will be placed in opaque, sequentially numbered and sealed envelopes and will be kept with an independent person not involved in the study. Post-screening, the allocation schedule will be revealed to the study investigator. The assessor and participants will be blinded to treatment allocation throughout the study. The schematic Consolidated Standard of Reporting Trials flow chart for the study protocol is displayed in (Figure 1).

The schematic Consolidated Standards of Reporting Trials flow chart for the study protocol. M-tDCS Multichannel Transcranial Direct Current Simulation, muscle; s-M-tDCS, Sham Multichannel Transcranial Direct Current Simulation.
Enrollment and Baseline Measurements
The initial screening of participants will be done with a “Comprehensive Neurological Assessment” Perfroma copyrighted under Divya Midha and Narkeesh Arumugam copyright office of the Government of India with unique registration No. “L-80961/2019.” Measures regarding the demographic profiles of the participants, that is, name, age, gender and contact information and the measures regarding the characteristics of stroke will also be documented including the type of stroke, duration of stroke, paretic side, and level of impairment caused by the stroke by National Institutes of Health Stroke Scale Score (NIHSS). After the initial screening, the objective measurement will be taken for all the primary and secondary outcome measures of the trial. The schedule of patient participation will be decided as per the guidelines provided by the Standard Protocol Items: Recommendations for Interventional Trials statement (Figure 2). 13

Standard Protocol Items: Recommendations for Interventional Trials schedule for patient participation.
Sample Size
G* Power tool (Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany; http://www.gpower.hhu.de/) was used for sample size calculation for the present trial(12)(13). Based on the parameters, that is, effect size (0.5); power (80%), alpha error (0.05), the sample size calculated for the study is 56. Given a reasonable attrition rate of 5%, the final sample size calculated was 60 with n = 30 in each group. The level of significance will be set at 0.05. The participants will have to spend at least 50% of the time of the session in order to be called successful performers; failing to achieve this will be included in the category of non-performers and will be excluded from the study.14,15
Interventions
After initial screening and baseline assessment, participants will be randomly allocated into either of the intervention group, that is, Experimental Group A, Multichannel Transcranial direct current stimulation group, that is, (M-tDCS Group) and Control Group B, sham—Multichannel Transcranial direct current stimulation group (sham-M-tDCS Group)
Multichannel Transcranial Direct Current Stimulation Procedure
The M-tDCS Group will receive multichannel transcranial direct current stimulation on the underlying scalp, over points of the dorsolateral prefrontal cortex (DLPFC) and primary motor cortex (PMC) of the lesioned hemisphere prior to the start of physical therapy. M-tDCS will be applied via a pair of sponge electrodes soaked in normal saline water connected to a 9-volt battery-operated direct current stimulator device. Points of stimulation corresponding to DLPFC and PMC will be selected based on the measurement via the 10 to 20 EEG international classification system. A cathode electrode will be placed on the contralateral hemisphere. The size of the electrodes will vary between 25 and 35 cm2. 12
Locating PMC and DLPFC Areas via 10 to 20 EEG International Classification System
Participants will be seated in a comfortable chair. Four anatomical landmarks will be located for the positioning of the electrodes; nasion, that is, point between the forehead and the nose; inion: the lowest point of the skull from the back of the head, indicated by a prominent bump; Pre auricular points (A1 and A2): indentation directly above the zygomatic notch. Opening the mouth makes it easier to locate this point. All the measurements will be done with a tape measure. Measurements will be taken in “cm” as shown in Figure 3.
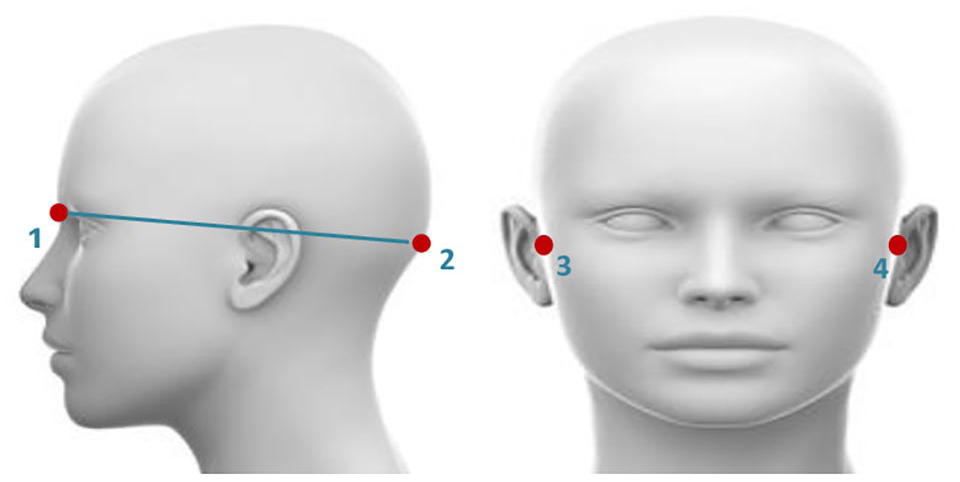
Anatomical Landmarks: (1): Nasion; (2): Inion; (3): A1-Right Pre Auricular Point; (4): A2-Left Pre Auricular Point.
Primary Motor Cortex (PMC), Location of (C3), and (C4) Points of Stimulation
Anteroposterior distances between nasion and inion will be documented. The midpoint of the distance between nasion and inion will be documented as (Cz). Followed by which, 2 points will be marked at the distance of 10% of the total distance between 2 preauricular points, that is, 10% up from the left auricular point (A1) named as (T3) and 10% up from the right-sided preauricular point (A2) named as (T4). The distance between (T3) to (Cz) and (T4) to (Cz) will be measured and (C3) will be located at the midpoint of the total distance between (T3) to (Cz) on the scalp over the left hemisphere whereas (C4) will be located at the midpoint of the total distance between (T4) to (Cz) on the scalp over the right hemisphere (Figure 4).

Location of Primary Motor Cortex and Dorsolateral Prefrontal cortex via 10 to 20 EEG Classification System.
Dorsolateral Prefrontal Cortex (DLPFC), Location of (F3), and (F4) Points of Stimulation
Two points will be marked at the distance of the total length measured from nasion to inion at the front and back of the head. The point at the 10% of the distance of the total length measured from nasion to inion up from the nasion is named as (Fpz) on the front side 10% up from the inion as (Oz) on the back of the head. Tape will be encircled and the distance will be measured from (Fpz) to (Oz) on both sides. (FP1) and (Fp2) points will be located at 5% of the total circumference of the head across (Fpz) and (Oz) points on the left and right sides respectively. The point at 10% distance of the total circumference of the head from (Fp1) down toward the left ear will be named as (F7) and toward the right ear will be designated as (F8). At the midpoint between (Fz) and (F7) will be designated as (F3) whereas at the midpoint between (Fz) and (F8) will be designated as (F4) points (Figure 4). 12
Application of M-tDCS
Prior to the start of the application, the participant will be screened for absolute contraindications of tDCS. He/she will be made to sit comfortably on a chair and be awake during the stimulation. Anode electrodes (red-colored) will be placed over the underlying scalp region of the lesioned hemisphere and cathode electrodes (black-colored) will be placed over the underlying scalp of the unaffected hemisphere with the help of a tDCS cap or velcro straps. M-tDCS will be employed over the primary motor cortex, that is, C3/C4 and the dorsolateral prefrontal cortex, that is, at F3/F4 points measured via 10 to 20 EEG international classification system. M-tDCS will be given with 2 mA intensity for a period of 20 minutes, 5 sessions/week for a total of 4 weeks. 11
Sham Multichannel Transcranial Direct Current Stimulation Procedure
Participants of the Sham-M-tDCS group will be made to sit comfortably relaxed and awake throughout the procedure. Contraindications for tDCS will be checked. The measurement procedure will be followed similarly to mentioned above. Points for electrode placement will be located via 10 to 20 EEG international classification system. Electrodes will be placed over (C3/C4) for targeting the primary motor cortex and (F3/F4) points for targeting the dorsolateral prefrontal cortex. Electrodes will be hydrated with normal saline prior to the placement in order to ensure appropriate hydration. Stimulation will be started at 2 mA intensity but it will be ramped off to zero after 30 seconds of the start of stimulation but the electrodes will be kept at the same points till 20 minutes of the total stimulation time. The intervention will be given for 5 days /week for 4 weeks with a total of 20 sessions of stimulation.
Standard Physical Therapy Intervention
SaeboFlex Training for Hand Training
SaeboFlex is a dynamic, custom fabricated wrist, hand, finger orthosis. SaeboFlex will be worn by the participant which consists of a forearm support attached to a dorsal hand platform that anchors 2 spring attachments. The SaeboFlex features a spring-loaded finger extension system. These springs provide resistance to the muscles that are used to grasp and assist in opening the hand and fingers for release. It also supports the wrist, hand and fingers in a functional position Individual finger sleeves are attached to the springs by a high tensile polymer line to provide assistance with finger extension. Spring tensile strength can be adjusted for the appropriate amount of finger extension assistance needed. Each finger sleeve is attached to the springs by a high-tensile polymer line in order to provide assistance with finger extension. Spring tensile strength can be adjusted for the appropriate amount of finger extension assistance needed, and the orthosis has no motor or electrical parts.
Wearing Saeboflex on the hemiparetic side, a session of 9 different task-oriented activities will be done by the participant for 1 hour. Tasks included grasping and picking up,7.6 cm diameter sponge ball (weighing <60 g) and releasing it into (28 cm × 32 cm × 40 cm) sized target container. Training activities include: (a) while attaining the sitting position, the participant will be asked to move the soft ball from the side of the affected foot to the table; (b) moving the soft ball from the normal extremity to the affected extremity diagonally;(c) moving the soft ball from the affected extremity to the normal extremity diagonally while standing;(d) moving a soft ball from the left to the right side on the table while standing; (e) moving a soft ball from a box, situated at knee height on the affected side, to a table while standing;(f) moving a soft ball through the target from the left to right side while standing;(g) grasping and releasing a soft ball to forward and backward transfer on the table while standing; (h) grasping and releasing a soft ball to diagonally forward and backward transfer on the table while standing;(i) moving a soft ball from a cup to a cup on the table while standing. Each task was performed for about 5 to 6 minutes. Training will be given 5 times per week for 4 weeks (20 sessions). 17 The location of the target container will be changed according to the rehabilitation needs of the individual participant.
Functional Electrical Stimulation
Functional electrical stimulation will be given to the lower extremity of the paretic side. Stimulation will be given with a battery-powered current stimulator. Functional electrical stimulation will be given via surface electrodes, the anode will be applied over the motor point of the tibialis anterior and the cathode will be applied behind the head of the fibula over the peroneal nerve. Stimulation will be given in the surged mode as well as walking mode. The surged mode will be used for ensuring appropriate electrode placement. Stimulation parameters will be 40 Hz frequency, with a pulse width of 0.3 ms. The stimulation will be delivered with maximum tolerance intensity (20-60 mA), For walking mode, a foot sensor attached behind the heel will control the stimulation by activating ankle dorsiflexors when the foot will be off the ground during the swing phase of the gait cycle, whereas stimulation will be switched off when the foot will be grounded. 16 The participant will be provided with gait training along with the FES stimulation in the walking mode which includes walking in a corridor; forward walking, obstacle walking, stepping up activity, stepping down activity, weight shifts in different directions. The intensity and complexity of gait training activities will be modified according to the individual rehabilitation need of the participant. The FES will be given for 10 minutes/session. 18
Conventional Rehabilitation
The standard physiotherapy treatment will be tailored to the individual participants, the number and intensity of the exercises will be guided by the research therapist based on the strengths and limitations of the paretic extremity of the participant. The exercises guided by the investigator are described in Figure 5. A tailored exercise session will be given for 60 minutes for 5 days/week for a total of 4 weeks. 19 A total of 20 sessions of conventional rehabilitation will be given to participants of both groups.
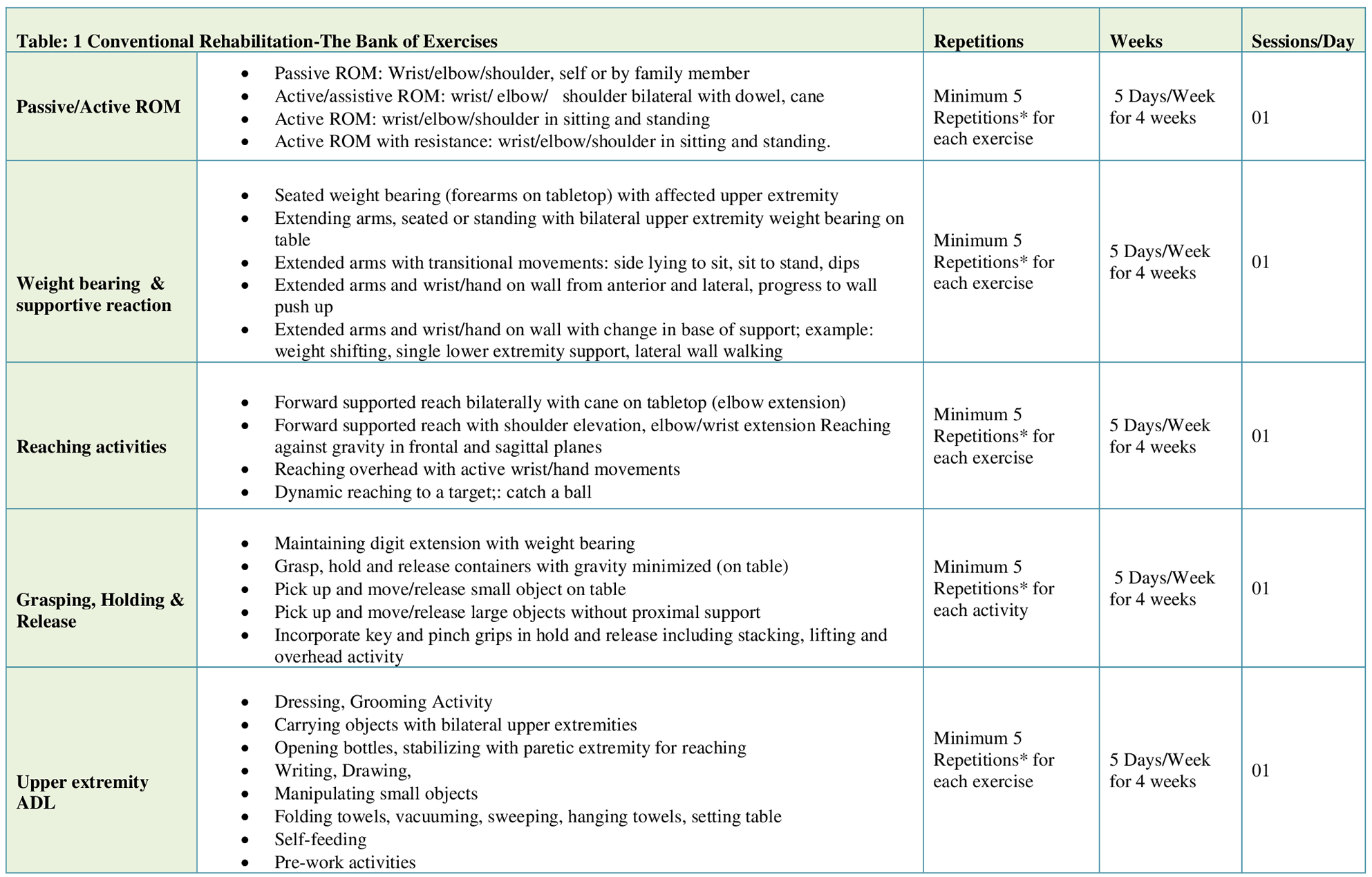
Conventional rehabilitation protocol.
Outcome Measures
All the assessments will be done at the beginning (day 0) followed by day 15 to day 30 post-intervention by an independent person who will be blinded to the intervention. Participants will be assessed on day (0), day (15), and day (30). The investigator will ensure that the instruments used in the assessment and examination have good validity and reliability.
Primary Outcome Measures
Based on the primary objective of the trials the primary outcome measures of the study will be a Fugl-Meyer Assessment for evaluation of motor domains of paretic extremities, the Montreal Cognitive Assessment for evaluation of cognitive domains and the Wisconsin Gait Scale (WGS) for evaluation of locomotor functions of the stroke participants and electroencephalography for observing cortical changes.
Fugl-Meyer Assessment
The Fugl-Meyer Assessment is a reliable, multi-item, performance-based impairment index that evaluates the physical performance of an individual following stroke. It is categorized into 4 different domains, that is, motor function, joint range of motion, sensory function, balance and joint pain. Each domain contains multiple items scored on a three-point ordinal scale. The motor domain items evaluate movement coordination and reflex activity of the upper extremity and lower extremity joints. The upper extremity motor score ranges from 0 to 66, the lower extremity motor score range varies from 0 to 34 (total motor score of 100 points). Light touch and pain constitute sensory domains with the total score ranging between 0 and 24 (0 -12 for upper and lower extremity each), passive range of motion and joint pain scores range from 0 to 44 (0 -24 for upper extremity & 0-20 for lower extremity)20,21
Montreal Cognitive Assessment
The Montreal cognitive assessment is a brief screening tool for cognitive domains providing a quick assessment of the global cognitive state of an individual in a short period of time. It includes the assessment of short-term memory, executive functions, visuospatial abilities, language, attention, concentration, working memory, and temporal and spatial orientation. Memory testing is done by a delayed recall of 5 nouns learned in 2 trials. An alternation task, a phonemic fluency task and 2 verbal abstraction tasks will be given for the evaluation of executive functions. Visuospatial skills will be tested by a copy task of a 3-dimensional cube and clock drawing test. Language function assessment will be done by asking the patient to name 3 animals, repeating exactly 2 syntactically complex sentences and by the phonemic fluency task used previously. Forward and backward, a sustained attention task, and a serial subtraction task will be given for assessment of attention, concentration and working memory. Orientation to time and place will be assessed by asking the participant regarding the date, month, year, day, place & city he or she is in. The global score will be obtained the points of each successfully completed task, in a range from 0 to 30 points, with higher scores indicating better cognitive performance. 22
Wiscosin Gait Scale (WSG)
The Wisconsin Gait Scale (WGS) is a stroke-specific scale used for the objective measurement of gait performance in individuals with stroke. It quantifies changes in the spatial and temporal parameters and gait kinematics. It includes 14 elements further divided into sub-scales as per phases of gait cycle. That is, stance phase, toe-off, swing phase, and heel strike. The first 5 elements are included in the stance phase of the affected leg. The second part of the scale is the toe-off phase of the affected leg and includes 2 sub-categories: The third subscale is the swing phase of the affected leg including 6 subcategories. The last part of the scale is the heel strike of the affected leg and includes only one sub-category: 14-initial foot contact of the affected leg. The minimum and the maximum score range is 13.5 to 42(21) In a study conducted on assessment of internal consistency and test-retest reliability, authors stated that WGS is a promising tool for a qualitative, observational analysis of gait in post-stroke survivors and helps the clinicians in planning and modification of treatment plan. WGS has a high internal consistency with Cronbach’s α coefficient ranging between 0.85 and 0.88. and high test-retest reliability.23,24
Secondary Outcomes
Nine Hole Peg Test (NHPT)
The Nine Hole Peg Test (NHPT) is one of the commonest, standardized time-based quantitative measures of manual dexterity. It consists of a square board with 9 holes, approximately 2.54 cm apart. The participant will be instructed to pick 9 pegs out of a shallow container, 1 by 1, as quickly as possible thereby putting them into 9 holes simultaneously and removing them from the holes to place them back in the shallow container. 25
Electroencephalography (EEG)
EEG signals will be acquired with a 64-channel EEG active electrode system. In this study, CAR was used for reference with the average of whole EEG channels for each individual EEG channel. EEG recording will be done in a separate room with noise-proof environment to avoid any artifacts. Ag/AgCl electrodes were positioned at Fz, Cz, Pz, Fp1, Fp2, F3, F4, F7, C3, P3, T3, T5, O1, F4, F8, C4, P4, T4, T6, O2 in accordance to the international 10 to 20 system, with electrode impedances all less than 5 kΩ. The analog to digital conversion rate was 250 Hz for all channels using a 16 bit AD converter. EEG was recorded for a minimum duration of 15 minutes in participants in an eyes-closed state with the patient awake and alert. The EEG recording was done on day 0 (baseline assessment), day 15 to day 30 post intervention. 26
Stroke Specific Quality of Life
Stroke-specific quality of life is a standardized, reliable (reliability coefficient 0.92), validated scale that specifically measures the quality of life of individuals that have had a stroke. The scale consists of 12 items grouped into physical and psychological subscales and 49 items, a 1 to 5 point range is used for scoring each item. The total range of scores vary from 49 to 245. The lowest score indicates poor quality of life. 11
Grip strength measurement
Grip strength of the paretic hand of the participant will be measured by JamarTM Hand
Dynamometer. The participant will be instructed to sit in an upright and relaxed position without resting/holding on to something. The participant will be directed to hold his/her arm in a 90° angle keeping the wrist in a neutral position with the thumb positioned up toward the ceiling. Handle width will be adjusted according to the size of the participant’s hand. For the required grip the middle finger bone (phalanx) of the middle finger needs will be positioned at an angle of 90° while covering the handle. The “peak-hold” needle of the handgrip dynamometer will be ensured to be at zero. The participant will be instructed to press as hard as possible and hold it for a period of 5 seconds. Three trials will be taken. The highest value of the measurements will be used for analyses. 27
Pinch strength measurement
Pinch Strength measurement will be taken by using Jamar hydraulic Pinch Gauge. The position of the participant was chosen based on the norms suggested by the American Society of Hand Therapists’ (ASHT) arm position during grip strength tests. The participant will be seated in a comfortable position on a chair with shoulder adducted and neutrally rotated, the elbow flexed to 90°, and the forearm and wrist in a neutral position.” The participant will be asked to press the pinch gauge as hard as possible and release it. Dual scale readout measures of the pinch gauge measure strength in both pounds and kilograms, up to 45 lbs., where a peak will hold the needle and retain the highest reading until reset. Measurements will be taken in kilogram (Kg) units for tip to tip pinch (tip of thumb and tip of index finger), key pinch (thumb pad to the lateral aspect of middle phalanx of index finger), 3 jaw chuck pinch (thumb pad and pad of middle and index fingers) and pulp pinch (thumb pad and index finger pad) of the paretic hand. Three trials of measurements will be done and the best one will be documented. 26
Data analysis
After successful completion of data collection, statistical analysis of data will be done using the IBM SPSS statistical software ver. 22.0. (IBM Corp., Armonk, NY, USA) with the help of a statistician. Baseline characteristics of eligible participants will be presented using descriptive statistics. Prior to the commencement of statistical analysis, a statistician will check for normal distribution with the help of Kolmogorow–Smirnow test. Based on the normality of data, descriptive statistics will be expressed as mean ± standard deviation or median and intra-quartile range. Data will be collected at 3 points of time for each participant and within-group comparisons will be conducted using one-way ANOVA. Post hoc testing will be done to identify the actual differences within group scores at different points of time. A between-group comparison (intervention group vs control group) will be done using an independent t-Test. The level of significance will be set at 0.05 for all analyses.
Data management
Data will be collected and simultaneously will be entered and stored in the Microsoft Office Excel 2010 (Microsoft Corp., Redmond, WA, USA). Throughout the course of the study, confidentiality of data will be maintained. A copy of data will also be stored in a password-protected non-rewritable compact disc read-only memory. The primary investigator will be authorized for the access of data and no other person will be given the authority.
Handling of missing data
All possible efforts will be taken for the collection of data at specified points of time. However, at the time of data missing on the primary endpoint, it will be assumed that data are missing at random (MAR). Statistical analysis will be done by list-wise deletion of records with missing values in order to ensure unbiased estimations.
Study ethics
The study has been approved by the institutional ethics committee of Punjabi University, Patiala Punjab (Ref Number: 152/IEC-2019, Dated 03/12/2019). The study has also been registered with the clinical trial registry of India (Reg. Number CTRI/2020/01/022998). The study will be done in accordance with the ethical guidelines of the Indian Council of Medical Research (2017) for biomedical and health research involving human participants in accordance with the principles ofgGood clinical practice, the medical research involving human subjects act and the declaration of Helsinki. (Revised 2013)
Study approval, patient information, and informed consent
The informed consent will be obtained from all the participants prior to the commencement of the trial. Participants will be informed regarding the aims, objectives of the trial, type of constraints, foreseeable risks of the trial. Information will be provided verbally and in written form in the participants’ language. Participation of the individual will be voluntary and no individual will be obliged to take part in the trial. Participants will be authorized to ask anything regarding the study. Participants will be allowed to withdraw their participation anytime during the trial. They will be aware about the investigator, observers and regulatory authorities monitoring the medical records and documentation to the extent permitted to them via regulatory bodies. The informed consent will be signed by the participant and countersigned by the investigator. It will also include demographic details including their contact details.
Data anonymity
Throughout the course of the study, confidentiality of data will be maintained. Data collection will be done on paper-based case report forms (CRFs). Any information capable of identifying participants will be removed. Participants will be given an anonymous code to protect confidentiality. After completion or discontinuation of the study, all records will be kept for a minimum of 15 years at local sites. Access to data during and after the study will only be granted to the designated research team member. Under no circumstances may encoded names and contact information will be revealed and it will be kept subject to professional secrecy.
Data archiving and document source access rules
All the documents pertaining to the trial will be archived as per the good clinical practice guideline for a minimum period of 15 years. The protocol will be retained by the investigator including the original copy of signed informed consent forms for confidentially. No removal or destruction may be carried out. All the documents will be subject to inspection.
Discussion
The existing rehabilitation models, based on noninvasive brain stimulation have been established with improved outcomes as mentioned in the evidence in the form of clinical trials or reviews. But the most of these intervention strategies are centered toward the rectification of motor deficits in isolation without augmenting cognitive domains. Cognitive processes such as memory, orientation, attention, executive functions govern motor performances to a great extent and appropriate execution of motor tasks requires strong interaction between planning, attention, executive functions, memory, and motor learning. Hence, an insight into the therapeutic interventions augmenting, cognitive, and motor domains simultaneously may yield better outcomes in the field of stroke rehabilitation thereby improving the quality of life of stroke survivors. This clinical trial attempts to determine the changes in the cortical network, motor and cognitive outcomes through multi-target stimulation of cortical areas by application of M-tDCS in stroke survivors.
Conclusion
An insight into the therapeutic interventions augmenting, cognitive, and motor domains simultaneously may yield better outcomes in the field of stroke rehabilitation thereby improving the quality of life of stroke survivors. The authors of the study anticipate the significant findings with respect to the improved functions of upper and lower extremies with M-tDCS application.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Arumugam Narkeesh gave substantial contributions to the conception or design of the work; Drafting the work or revising it critically for important intellectual content. Midha Divya prepared the initial draft of the article. Both the authors contributed sufficiently in the work to take public responsibility for the content, including participation in the concept, design, analysis, writing, or critical revision of the manuscript and with mutual agreement gave Final approval of the version to be prepared and published.
