Abstract
Early overuse of a lesioned forelimb, induced by immediate immobilization of the intact forelimb after a cortical lesion, has been reported to increase tissue damage and delay functional recovery. To investigate if early training without immobilization of the intact forelimb could increase tissue loss and reduce recovery, the middle cerebral artery was ligated distal to the striatal branches in 25 male spontaneously hypertensive rats. Control rats were housed in standard cages, training rats were transferred to larger cages allowing various activities and received additional special training 1 hour a day starting either 24 hours or 7 days after the ligation. The rats were tested on a rotating pole, in a leg placement test, and in a water maze and they were killed 6 weeks after the ligation. Delayed training resulted in the best overall performance; however, both training groups performed better than standard rats on the rotating pole. The cortical infarct volume was larger in the early training group than in the other two groups (P < .005), possibly related to increased glutamate release and peri-infarct cortical hyperexcitability.
Activation and training starts early after stroke onset in most stroke centers today. Evaluations of stroke units have confirmed that early mobilization reduces secondary complications and mortality and promotes long-term functional outcome (Stroke Unit Trialists' Collaboration, 1997a, b ; Indreavik et al., 1997). To what extent it improves specific motor functions is less clear. Optimal time windows for specific rehabilitation interventions after stroke have not been defined.
That experience and training can promote adaptive plasticity in the healthy brain as well as after brain lesions has been shown in many clinical and experimental studies (Rosenzweig, 1984; Jenkins and Merzenich, 1987; Pons, 1988; Bach-y-Rita, 1990; Kaas, 1991; Pascual-Leone and Torres, 1993; Schieber, 1995; Seitz et al., 1995; Florence et al., 1997; Nicolelis, 1997; Nudo et al., 1997; Steinberg and Augustine, 1997). After forelimb primary motor cortex damage in the rat, functional forelimb recovery is related to reorganization of adjacent areas of the cortex (Castro-Alamancos and Borrell, 1995). Electrophysiologic studies in monkeys with small cortical lesions in the digit representation area have shown that tissue surrounding a cortical lesion in the hand representation undergoes a further territorial loss in the functional representation of the affected body part (Nudo and Milliken, 1996). Training, started within 5 days after the lesion, could prevent the further tissue loss and stimulate functional reorganization in the undamaged surrounding motor cortex (Nudo et al., 1996). A recent extensive study on somatosensory cortex plasticity before and after small cortical lesions have confirmed extensive training-induced multifocal reorganization in the ipsilateral cortex (Xerri et al., 1998). These studies have led to the conclusion that representational substitution can mediate behavioral restitution after focal cortical stroke.
The studies referred to above support the importance of training for optimal functional outcome after focal cortical lesions. However, some reports indicate that intense early training may have a negative effect. After frontal cortical lesions in the rat, adaptive changes with increased dendritic branching have been observed in the intact contralateral hemisphere, changes that could be prevented by immobilization of the intact forelimb, which also prevented functional improvement (Jones and Schallert, 1994). Furthermore, the resulting overuse of the lesioned forelimb increased tissue loss around the lesion (Kozlowski et al., 1996; Humm et al., 1998).
The possibility that early training after stroke could have a negative effect is of obvious clinical concern. Immobilization of the forelimb ipsilateral to the lesion immediately after the surgical procedure, thus forcing the animal to rely on the contralateral limb for postural support and movements, may be stressful for the animal. Although it has been reported that immobilization of the intact arm can improve function in the lesioned arm months or even years after stroke onset (Wolf et al., 1989; Taub et al. 1993), this intervention is not likely to be initiated in the acute phase of stroke. Placing rats in an enriched environment (i.e., large cages with the opportunity for various activities but not forcing the animals to do any specific task) 24 to 30 hours after stroke significantly improves outcome without altering infarct volume (Ohlsson and Johansson, 1995; Grabowski et al., 1995; Johansson and Ohlsson, 1996). What should be considered to be overuse of the lesioned limb? Is there a difference between early and late training if the rats are not prevented from using the intact limb? To answer these questions we have compared morphologic and functional outcome in control rats housed in standard laboratory cages with that of rats exposed to an enriched environment, combined with specific training 1 hour a day 5 days a week, starting either 24 hours or 7 days after ligation of a middle cerebral artery (MCA).
MATERIALS AND METHODS
Surgery, housing conditions, and training
The experimental protocol was approved by the local Ethics Committee for Animal Research. Twenty-five male 4 months old spontaneously hypertensive rats were anesthetized with methohexital 50 mg/kg intraperitoneally. After a small craniotomy, the right cerebral artery (MCA) was ligated with a 10–0 monofilament nylon thread distal to the origin of the striatal branches (Coyle, 1982). To keep the operation time as short as possible (20 to 30 minutes) and to avoid problems in the behavioral tests, the animals were not intubated and no catheters were inserted for blood pressure and blood gas control. During the first 24 postoperative hours all rats were kept in individual cages. After that time, eight control rats were housed in standard laboratory cages, four rats in each cage. 550 × 350 × 200 mm until the end of the experiment. Eight early training rats were transferred to enriched environment cages and received, in addition, a special training program as described below 5 days a week. Nine late training rats were transferred to the same treatment starting 7 days after the ligation. The enriched environment cages were 820 × 610 cm × 450 mm, furnished with horizontal and inclined boards and equipped with various items, such as a chain, a swing board, and wooden blocks (Ohlsson and Johansson, 1995; Grabowskiet al., 1995; Johansson and Ohlsson, 1996). Twice a week the space between the boards was changed and some objects replaced with new ones. The training program consisted of the following: three times a week the rats were trained to traverse a rotating pole, hang on to a rope, and keep their balance on an inclined plane. One rat was handled at a time on each test, thus offering the rat some rest before the next test. The total training time for the group of eight rats was about 1 hour. Twice a week, on days not involving the training described above, the rats were placed in a rectangular wooden box, 2000 × 370 × 450 mm, with wire netting roof. The box was connected to the home cage at one end. When put in the box the rats were placed at the end opposite of the home cage. The box contained four platforms connected with sticks, ropes, and a swing arranged in such a way that it became increasingly more difficult to move from one platform to the next. If they missed one passage they would fall down and have to start all over again. To stimulate the rats to explore the cage, some pieces of apples were placed in the distal part most difficult to reach (according to our earlier experience, rats like apples). The rats were taken out of the wooden box after 1 hour whether or not they had reached the end of the box.
All rats were housed in a large well-ventilated room with a constant temperature of 23°C. The temperature within the cages does not differ from the room temperature and we know from other studies that the resting body temperature is the same for rats housed in a standard and in an enriched environment (36.8 to 374°C).
Behavioral tests
The rats were tested on a rotating pole and in a leg placement test 1, 2, and 4 weeks after the operation. The rotating pole measures coordination and integration of movements (Ohlsson and Johansson, 1995). The pole, 45 mm in diameter and 1.5 m in length, rotated alternately to the left or to the right with three or 10 turns per minute. The score was 0 if the rat fell; 1 if the rat was unable to traverse the pole but did not fall; 2 if the rat fell while attempting to cross the pole; 3 if the rat jumped with both hind limbs together, apparently supporting the weak hind limb with the opposite strong limb; 4 if the affected hind limb was used for less than 50% of the steps; 5 if the rat crossed the pole with a few foot slips; and 6 if the rat crossed the pole with no foot slips.
In the limb placement test, modified after De Ryck et al. (1989), the rat was held by the examiner and forelimb and hindlimb placing was noted when approaching a table or table edge from various directions with and without allowing tactile, proprioceptive, and visual support as earlier described in detail (Ohlsson and Johansson, 1995; Johansson and Ohlsson, 1996). The test consists of six parts with forepaw scoring in all and additional hindpaw scoring in two. Each trial was scored as follows: 0 = no placing, 1 = incomplete and/or delayed (>2 seconds) placing, and 2 = immediate and correct placing. Thus, for each body side, the maximum score, i.e., normal performance, is 16.
Spatial navigation abilities were evaluated using a Morris Water Maze (Morris, 1984). The maze consisted of a circular tank,1800 mm in diameter and 450 mm deep, filled with water kept at room temperature and rendered opaque by the addition of powdered milk to prevent the rat from locating the platform in the center of one of the quadrants by sight. The maze was located in a corner of a room with many external cues that were visible from within the pool and thus available for orientation to the rats. To escape from the water the rats could climb to the hidden platform.
The rats were trained in the water maze for four trials a day over 5 consecutive days 3 weeks after the MCA ligation. On each trial the rat was placed at one of the four starting positions and given 60 seconds to find the submerged platform and climb onto it. After 15 seconds rest on the platform the rat was placed at the next randomly predetermined starting point. The latency and the distance swam to find the hidden platform and the swim speed were recorded by a computer-based video tracking system (Paul Fray Ltd, Cambridge, U.K.). After the last trial on the 5th day the platform was removed and the rat placed at one of the starting positions and allowed to swim freely for 60 seconds. The swim path was plotted, and the time spent in each of the quadrants recorded by the computer, together with the number of crossings over concentric annuli with their center at the site where the platform would have been.
Perfusion, histology, and volumetric estimates
Six weeks after the arterial, ligation the rats were anesthetized with methohexital 50 mg/kg intraperitoneally and transcardially perfused with a solution of 4% paraformaldehyde in 0.15 mol/L S® rensen phosphate buffer (pH 7.4) for 15 to 20 minutes after an initial rinse with 0.9% sodium chloride. The brains were post-fixed in the same fixative, embedded in paraffin, and serially sectioned in 5-μm thick sections. The cross-sectional area was measured on 12 sections, 900 μm apart. The infarct volumes were calculated from the cross-sectional areas and the distance between the sections according to the Cavalieri principle (Gundersen et al., 1988). The tissue loss was calculated as the difference between cortex in the infarcted and contralateral cortex. Thalamic atrophy was evaluated by measuring thalamic area on the level of bregma -1.8, -2.3, -2.8, and -3.3 (Paxinos and Watson, 1986).
The image analyzing system used for the volume determinations consisted of a light board with constant light (Northern light model B90 Imaging Research Inc., Ontario, Canada), a video camera (CCD72, Dage MTI, Michigan City, Indiana, U.S.A.) fitted with a Nikon lens (f 55mm), a digitizing unit attached to the video camera (Dage MTI, see above), and a Macintosh computer equipped with a video card (Image Grabber, Neotech Ltd, Eastleigh, Hampshire, U.K.). To capture the digitized images, the software Image Grabber 2.03 (Neotech Ltd) was used. For further processing we used the Image/MG 1.44b (NIH, Bethesda, MD, U.S.A.).
Statistical analysis
Data from tests based on scoring systems are presented as median values with 25% upper and lower percentiles. The Kruskal-Wallis nonparametric analysis of variance with a multiple comparison post hoc test at 95% significance level was used for determination of differences between the groups. Oneway parametric analysis of variance with Scheffé's post hoc procedure at 95% significance level was used to determine group differences in weight and infarct volume and the water maze test and data represented as mean ± SD.
RESULTS
One rat in the standard group was found dead in the cage 2 days after the operation. Two rats, one from the early rehabilitation and one from the standard group were excluded because of unsuccessful ligation of the MCA. The final analysis was accordingly based on six standard, seven early rehabilitation, and nine late rehabilitation rats. There was no difference in body weight among the three groups in the postoperative period (Table 1).
Body weight (g) before and after ligation of a middle cerebral artery distal to the striatal branches
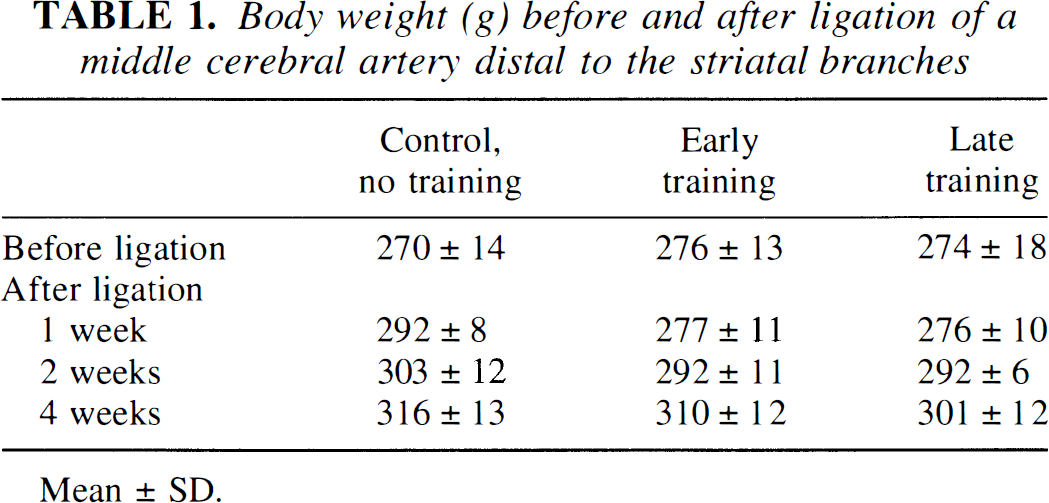
Mean ± SD.
Infarct volumes and thalamic atrophy
The infarcts did not extend into the striatum in any of the rats. The loss of cortical tissue in the right infarcted hemisphere was significantly larger in the early training group than in the standard and late rehabilitation groups (Fig. 1). The thalamic atrophy was also most pronounced in the early training group with significant difference from the late training group (Fig. 1). The tissue reduction was most obvious in the sections Bregma -2.8 and -3.3 (Paxinos and Watson, 1986).
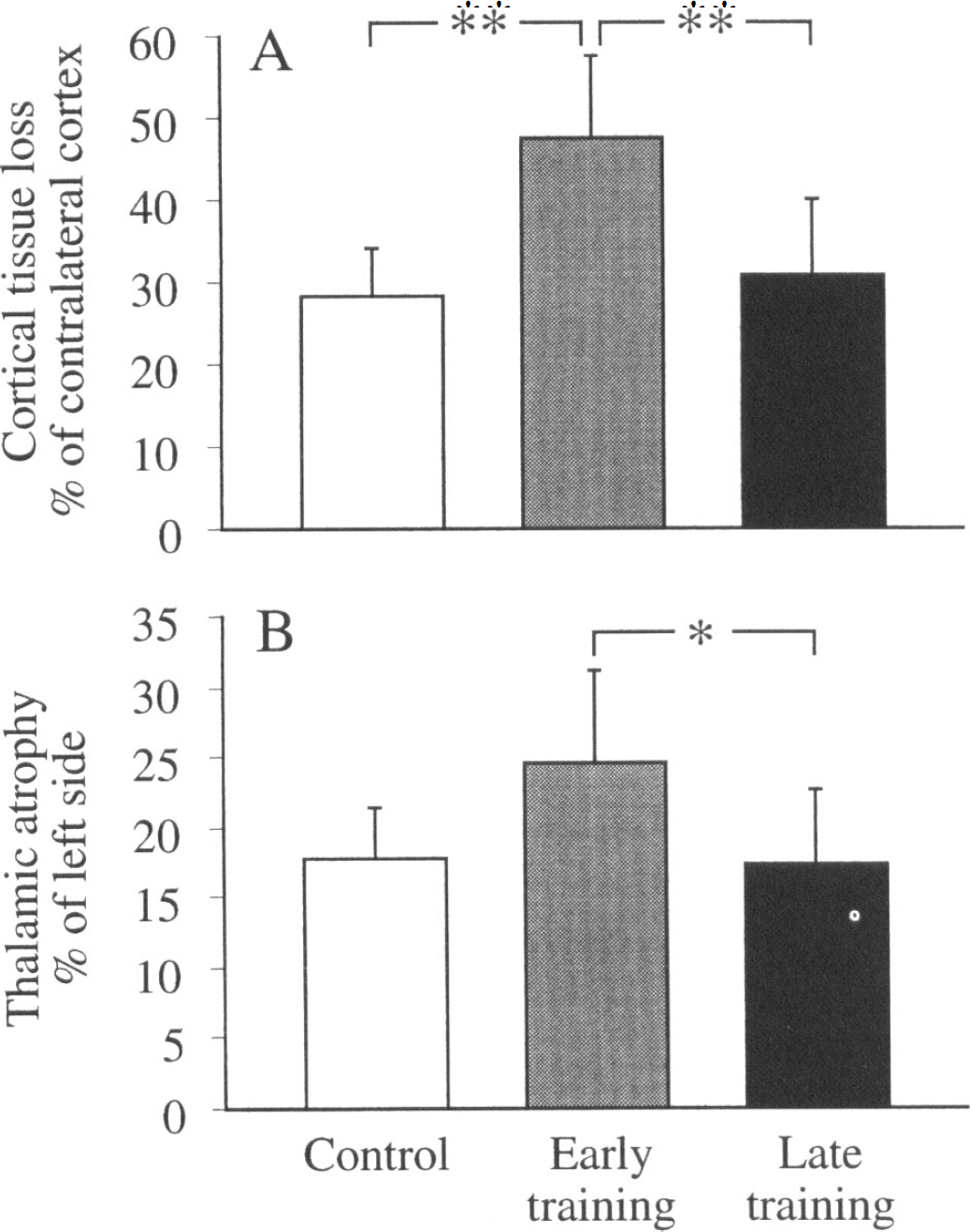
Cortical
Behavior
The rats were easy to handle during the training and did not show any signs of stress such as urinating or defecating. Both late and early training rats performed significantly better than rats in the standard group 2 and 4 weeks after the arterial ligation (Fig. 2). The leg placement scores for the left paws were similar in the three groups at 2 weeks; however, at 4 weeks the trend was the same as in the pole test, although the ranges were large and the differences were not significant. Median values (25 and 75 percentiles in brackets) were 5 (4.75 and 9.25), 11 (6.50 and 13.5), and 13 (5.25 and 14.5) in the standard, early, and late training groups, respectively. The leg placement score on the intact right side was 16 (full score) at all times.
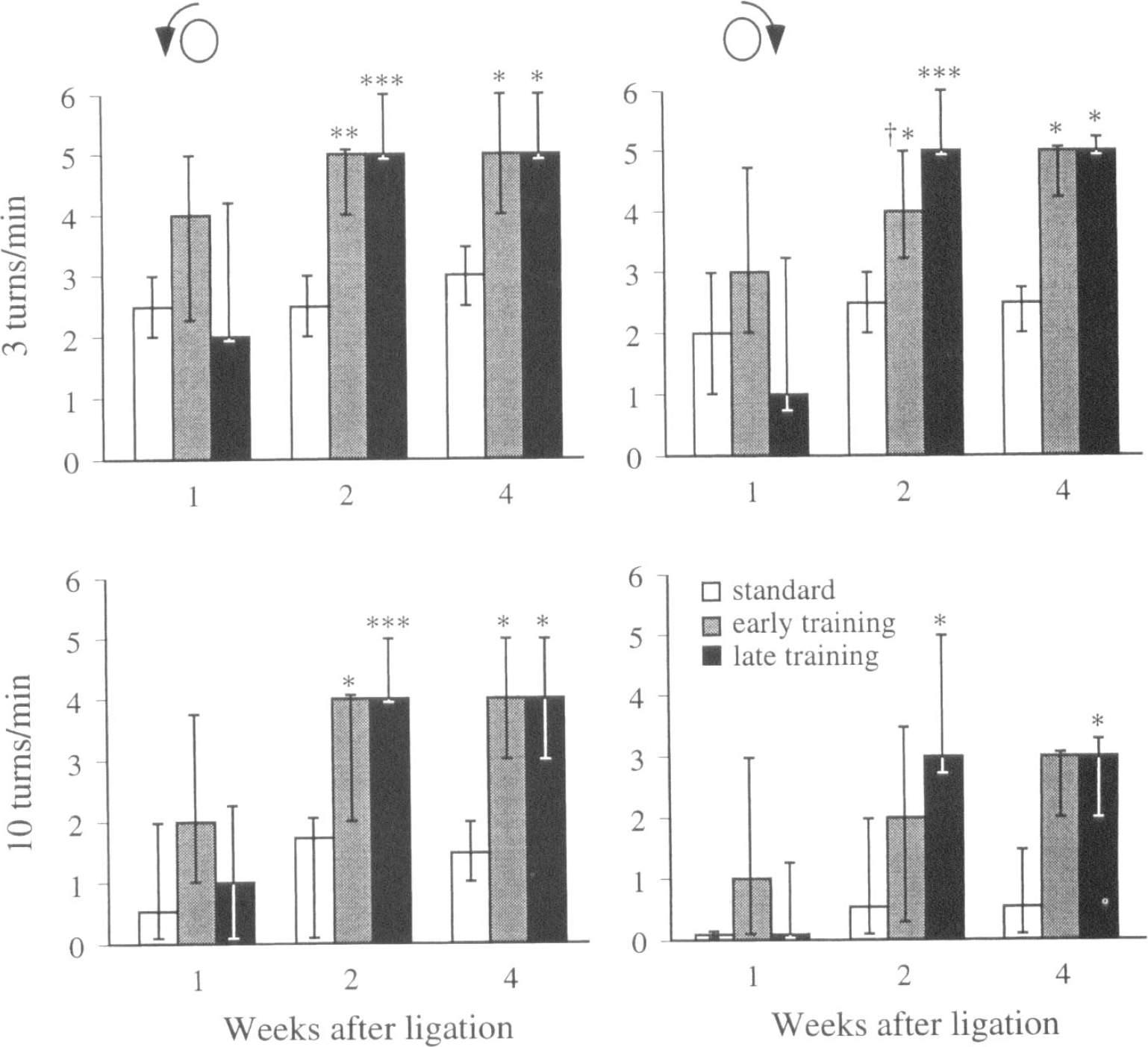
Ability to traverse a pole, rotating with 3 or 10 turns per minute, after distal ligation of the right middle cerebral artery in rats housed four and four in standard laboratory cages; or subjected to enriched environment combined with training 24 hours (early training) or 7 days (late training) after the arterial ligation *P < .05, **P < .01, P < .001 for difference from standard rats. †P < .05 for difference from the late training group. Kruskal-Wallis nonparametric analysis of variance with a multiple comparison post hoc test at the 95% significance level. Score 0 = the rat falls down; score 6 (optimal performance) = the rat crosses the pole with no foot slips.
In the water maze, rats in the late training group swam a shorter distance and found the platform earlier than the other groups during the first test day; however, at later times the other groups had improved to the same level (Fig. 3). The speed did not differ; it was 0.28 ± 0.02, 0.30 ± 0.01, and 0.31 ± 0.03 meters per second in the standard, early and delayed group, respectively, at day 1; and 0.34 ± 0.4, 0.36 ± 0.04, and 0.35 ± 0.04 meters per second, respectively, at day 5. When the platform was removed after the last test and the rats were allowed to swim freely for 60 seconds, the percent of time spent at the site where the platform used to be did not differ (54 ± 17, 54 ± 16, and 53 ± 12% for the standard, early, and delayed groups, respectively).
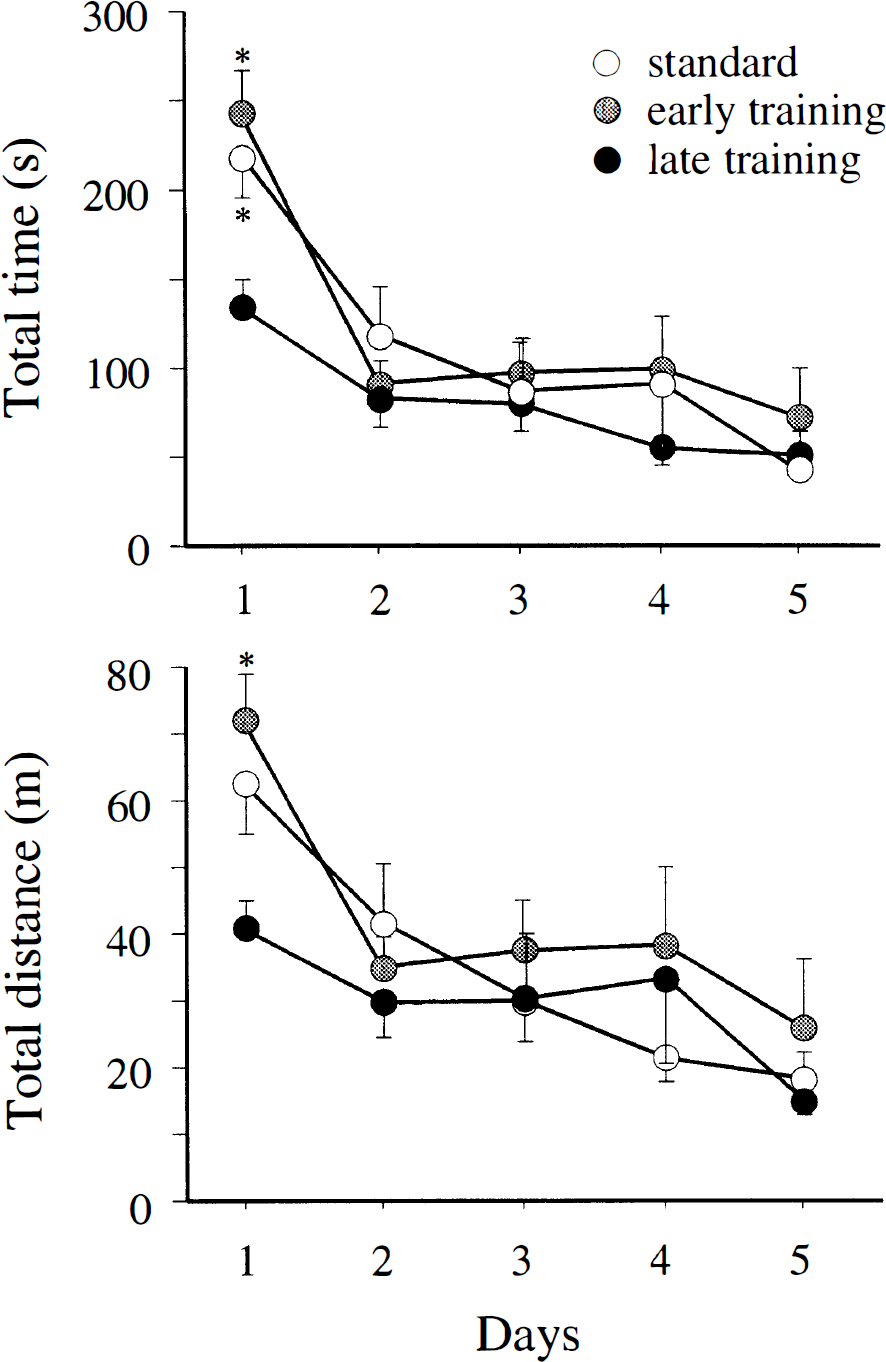
Water maze performance. Rats in the late rehabilitation group swam a shorter distance and found the platform earlier than the other groups on the first day in the water maze test. Analysis of variance with Scheffé's post hoc test at the 95% significance level.
DISCUSSION
The major observation in this study is that training initiated 24 hours after permanent focal brain ischemia can increase cortical tissue loss. We have shown earlier that housing animals in an enriched environment with the opportunity to engage in various activities without specific training significantly improves functional outcome and does not increase tissue loss (Ohlsson and Johansson, 1995; Grabowski et al., 1995; Johansson and Ohlsson, 1996). Thus, it is evident that it is not the different housing conditions but the additional training that is responsible for the increased neuronal damage.
Immobilization of the intact forelimb starting immediately after a cortical lesion, thus forcing the animal to use the weak forelimb, can expand cortical lesions (Kozlowski et al., 1996; Schallert et al., 1997; Humm et al, 1998). In the present study the training was initiated 24 hours after the onset of the ischemic insult and the rats could freely use the intact limb in all training situations, excluding an effect of immobilization on outcome. However, immobilization might have some additional effect, as immobilization of the intact forelimb from 7 days after the lesion had no effect on lesion size but nevertheless interfered with restoration of function (Humm et al., 1998).
Despite the larger tissue loss in the early training group, those rats improved more than standard rats. In the absence of postischemic interventions, a good correlation between behavioral tests and infarct size has been observed in many but not all studies. The tests used and the postischemic timing may be of importance (Johansson, 1995, Yonemori et al., 1996). In animals housed in an enriched environment, the correlation between infarct volume and functional outcome is poor (Ohlsson and Johansson, 1995; Grabowski et al., 1995), an observation that is consistent with the hypothesis that plastic changes in the remaining nervous tissue can influence outcome. The better outcome in the early training group than in standard rats may be related to compensatory adaptation in the contralateral hemisphere (Jones and Schallert, 1994; Schallert et al., 1997) or in other brain regions distant from the infarct area. Furthermore, we must consider the training effect as pole crossing was included in the training program that standard rats did not receive. Even if the increased tissue loss did not correspond to poorer outcome in the test used in our study, it could make the rats more vulnerable to other insults, pharmacologic challenges, or aging. In support of this view, symptoms reappeared at an older age in rats with early lesion-induced sensorimotor impairment and initial recovery of function (Schallert, 1983). Similarly, it has been observed in war veterans that penetrating head injury in young adulthood exacerbates cognitive decline in later years (Corkin, 1989).
Motor activity stimulates the release of glutamate and catecholamines (Vanderwolf and Cain, 1994). One possible explanation for the increased tissue loss might be that hyperexcitability of the surrounding tissue in the early postischemic period makes the surrounding neurons vulnerable to excitation. Cortical networks adjacent to a focal brain ischemia are hyperexcitable because of an imbalance between excitatory and inhibitory synaptic function with increased N-methyl-
Physical exercise can also increase body temperature. An exponential relationship between increases in core temperature and plasma growth hormone, prolactin, and catecholamines during exercise has been observed in man with a thermal threshold of approximately 38°C (Randomski et al., 1998). As recently reviewed by Ginsberg and Busto (1998), hyperthermia can significantly increase tissue damage in stroke and glutamate release is one of the proposed mechanisms. In contrast to a marked aggravating effect of hyperthermia in transient focal ischemia, the infarct resulting from permanent MCA occlusion was not significantly affected by hyperthermia in the study by Morikawa et al. (1995), although hyperthermia increased tissue loss after a combined occlusion of MCA and ipsilateral carotid artery (Chen et al., 1991). In global brain ischemia, even mild hyperthermia can have a negative effect 24 hours after the insult. In transient focal brain ischemia, the threshold appears to be much higher. The brain temperature had to be increased to 40°C for a 3-hour period to induce further tissue loss 24 hours after a 60-minute transient occlusion of the MCA (Kim et al., 1996). In a recent clinical study when the temperature was measured on admission and then every second hours for 3 days (Castillo et al., 1998), only body temperature within the first 24 hours from stroke onset significantly correlated to outcome. Body temperature may not accurately reflect brain temperature under ischemia (Kim et al., 1996) and large differences between brain and body temperature were observed in a clinical study on patients referred to intensive care because of high intracranial pressure and large infarcts (Schwab et al., 1997). If such large differences may occur in alert freely moving rats or during exercise is less clear. In female gerbils exposed to small-maze sessions 4 to 6 days after global ischemia, the brain temperature increased about 1°C (Colbourne et al., 1998); the body temperature was not reported.
Although a negative effect of delayed brain hyperthermia in permanent focal ischemia in the rat is not documented it is an important issue that needs to be further studied. We have measured body temperature before and after a training session in previously untrained rats and observed an increase up to 0.8 to 0.9°e. We cannot exclude that this moderate increase in temperature could have a negative effect during a vulnerable period. Whether the increased tissue loss is due to an activity-induced increase of excitatory transmitters, an indirect effect induced by hyperthermia, a combination of the two, or some unrelated events cannot be determined from our data. However, a relevant clinical question is whether corresponding events may happen during rehabilitation sessions in stroke patients in the early postischemic phase.
The window for the critical period when physical activity may add to damage must be determined. Treatments that are of value to rescue brain tissue in the acute stage can have a negative effect on compensatory mechanisms in the rehabilitation phase. Glutamate and NMDA receptors are important for brain plasticity, and cortical map reorganization in the primary somatosensory cortex can be prevented by blockade of NMDA receptors (Hess and Donoghue, 1994; Hess et al., 1994; Kano et al., 1991; Garraghty and Muja, 1996). In addition to glutamate, motor activity is linked to release of serotonin, norepinephrine, and acetylcholine in sensorimotor cortex (Vanderwolf and Cain, 1994), substances known to modify brain plasticity processes (Goldstein, 1998; Kilgard and Merzenich, 1998).
It is well established that early mobilization is essential to reduce secondary thromboembolic events, pneumonia, and mortality in acute stroke (Stroke Unit Trialists' Collaboration, 1997a,b; Indreavik et al., 1997). Earlier experimental data have shown that general activation starting 24 hours after an ischemic event promotes functional outcome without increasing tissue loss (Ohlsson and Johansson, 1995; Grabowski et al., 1995; Johansson and Ohlsson, 1996). However, there might be some advantage to refrain from intense motor training during the first days after large cortical infarcts. Further studies are needed to determine the window for a possible risk for activity-induced additional peri-infarct neuronal loss.
