Abstract
TESS Research Foundation (TESS) is a patient-led nonprofit organization seeking to understand the basic biology and clinical impact of pathogenic variants in the SLC13A5 gene. TESS aims to improve the fundamental understanding of citrate’s role in the brain, and ultimately identify treatments and cures for the associated disease. TESS identifies, organizes, and develops collaboration between researchers, patients, clinicians, and the pharmaceutical industry to improve the lives of those suffering from SLC13A5 citrate transport disorder. TESS and its partners have developed multiple molecular tools, cellular and animal models, and taken the first steps toward drug discovery and development for this disease. However, much remains to be done to improve our understanding of the disorder associated with SLC13A5 variants and identify effective treatments for this devastating disease. Here, we describe the available SLC13A5 resources from the community of experts, to foundational tools, to in vivo and in vitro tools, and discuss unanswered research questions needed to move closer to a cure.
Plain language summary
SLC13A5 citrate transporter disorder is an ultra-rare, neurodevelopmental disorder that severely impacts cognition and motor control. It is characterized by frequent, intractable seizures that develop hours or days after birth, low tone, global developmental delay, a unique, varied, and difficult to categorize movement disorder, limited expressive verbal capabilities, tooth abnormalities, and increased citrate in both the CNS and serum. Seizures are frequently medically intractable, patients are often on multiple antiseizure medications and have frequent emergency room visits and hospitalizations for status epilepticus. SLC13A5 citrate transporter disorder is caused by mutations in the SLC13A5 gene which encodes a sodium-dependent citrate transporter, NaCT. NaCT is responsible for transporting citrate, a key molecule in cellular metabolism, from the extracellular space into cells, especially in the central nervous system and the liver. NaCT has been extensively studied in multiple animal models and affects lifespan and loss of some transporter activity actually improves metabolic syndrome in all animal species tested so far while causing mild neurological dysfunction in rodents. Although not definitively proven, it is presumed that loss of neuronal cell citrate transporter activity in the brain is the cause of seizures. Since the discovery of the disorder in 2014, there has been a rapid expansion in characterization of the disease. This has been aided by development of multiple models and molecular tools for studying wild type and mutant SLC13A5 making it a tractable candidate for therapeutic development. TESS Research Foundation is dedicated to driving SLC13A5 research and supporting children and families living with the disorder. Here, we describe the available SLC13A5 resources from the community of experts, to foundational tools, to in vivo and in vitro tools, and discuss unanswered research questions needed to move closer to a cure.
Introduction
SLC13A5 citrate transporter disorder is an ultra-rare, autosomal recessive, neurodevelopmental disorder that severely impacts cognition and motor control.1–5 It is characterized by frequent, intractable seizures that develop hours or days after birth, hypotonia, global developmental delay, a unique, varied, and difficult to categorize movement disorder, limited expressive verbal capabilities, tooth abnormalities, and increased citrate in both the CNS and serum.1–12 Seizures are frequently medically intractable, patients are often on multiple antiseizure medications and have frequent emergency room visits and hospitalizations for status epilepticus.3,7,10
SLC13A5 citrate transporter disorder is caused by biallelic, loss-of-function variants in the SLC13A5 gene which encodes a sodium-dependent citrate transporter, NaCT. NaCT is responsible for transporting citrate, a key substrate in cellular metabolism, from the extracellular space into cells, especially in the central nervous system and the liver.5,13–16 NaCT has been extensively studied in multiple animal models; however, disruption of the NaCT ortholog in Caenorhabditis elegans and flies increases lifespan and improves metabolic syndrome in all animal species tested so far while only causing mild neurological dysfunction in rodents.17–21 Although not definitively proven, it is presumed that loss of neuronal cell citrate transporter activity in the brain is the cause of seizures. Since the discovery of the disorder in 2014, there has been a rapid expansion in characterization of the phenotype, genotype, and molecular understanding of the disease. This has been aided by development of multiple models and molecular tools for studying wild type and mutant SLC13A5 in vivo and in vitro making it a tractable candidate for therapeutic development.
TESS Research Foundation, a patient founded, 501(c)3 nonprofit organization established in 2015, is the only patient organization dedicated to driving SLC13A5 research and supporting children and families living with SLC13A5 citrate transporter disorder. TESS has partnered with patients, academic researchers, industry, and clinicians to drive patient-centered SLC13A5 research. Over the past 8 years, TESS has distributed over $2 million in grants to 23 different groups around the world. Together with their partners, TESS has developed databases, biological resources, and funded research projects to help understand the basic biology of SLC13A5 and pursue multiple different avenues to produce a cure. 22 Here, we describe the available SLC13A5 resources from the community of experts, to foundational tools, to in vivo and in vitro tools, and discuss unanswered research questions needed to move closer to a cure.
Results
SLC13A5 community
TESS has built an engaged community of patients, researchers, clinicians, and industry members all focused on SLC13A5 citrate transporter disorder. This community fuels TESS and determines success: when SLC13A5 citrate transporter disorder patients can live healthy, independent lives. This community drives the patient-centered research priorities, guides the development of research resources, and is a pillar for therapeutic development.
Supporting and empowering an engaged patient and caregiver community
One of the initial and most important TESS roles is supporting and engaging patients and their families. Rare disease caregivers are lived experience experts with intimate knowledge about disease presentation and often must gather and disseminate information to clinicians and researchers. 23 Patients and caregivers are the experts providing the focus and impetus for the ensuing research. They are passionate advocates for their loved ones and finding a cure for the disease. While TESS strives to achieve a cure, the foundation supports impacted families immediately by fostering community, keeping patients and caregivers informed of the scientific progress, and connecting families with resources. Because SLC13A5 citrate transporter disorder is rare, the affected families are geographically dispersed across the globe. TESS began monthly virtual family support group meetings to bring families together. This hour-long meeting includes scientific updates, as well as family-only discussions in which families discuss relevant topics ranging from medication, recent hospital experiences, insurance, or other clinical care issues. The opportunity to connect consistently with other affected families builds and grows the SLC13A5 community.
TESS creates and collects resources for families posted on the foundation website, a monthly electronic newsletter, and quarterly mail updates with the goal of supporting families in their daily experiences and becoming clinical trial ready. TESS works to make scientific topics accessible to the wider community through their monthly blog, Science Simplified where different authors write short articles explaining scientific or medical topics in lay terms. Altogether, the SLC13A5 family community is an engaged group eager to learn, interact with the research and clinical community, and find new treatments for SLC13A5 citrate transporter disorder.
Convening the community: bringing researchers, clinicians, industry, and patients together
TESS is uniquely suited to drive patient-centered SLC13A5 citrate transporter disorder research – bringing together multiple stakeholders worldwide with a common goal of finding a cure for SLC13A5 citrate transporter disorder. This involves partnering with patients, caregivers, researchers, clinicians, industry partners, and nonprofit research entities (Figure 1). TESS is able to bring the SLC13A5 research community together through monthly, virtual SLC13A5 Research Community Updates, as well as bi-annual International Research Roundtables or Clinical and Family Conferences. TESS has hosted 7 research roundtables growing from a small group of 16 to, in 2022, 97 participants including clinicians, translational researchers, patients and caregivers, biotechnology partners, governmental, and nonprofit partners all working together to share information and develop a research and clinical strategic plan. The clinical and family conferences provide a forum to discuss SLC13A5 basic, translational, and clinical research from labs around the world and most importantly provides an opportunity to meet and learn from patients and caregivers who participate.
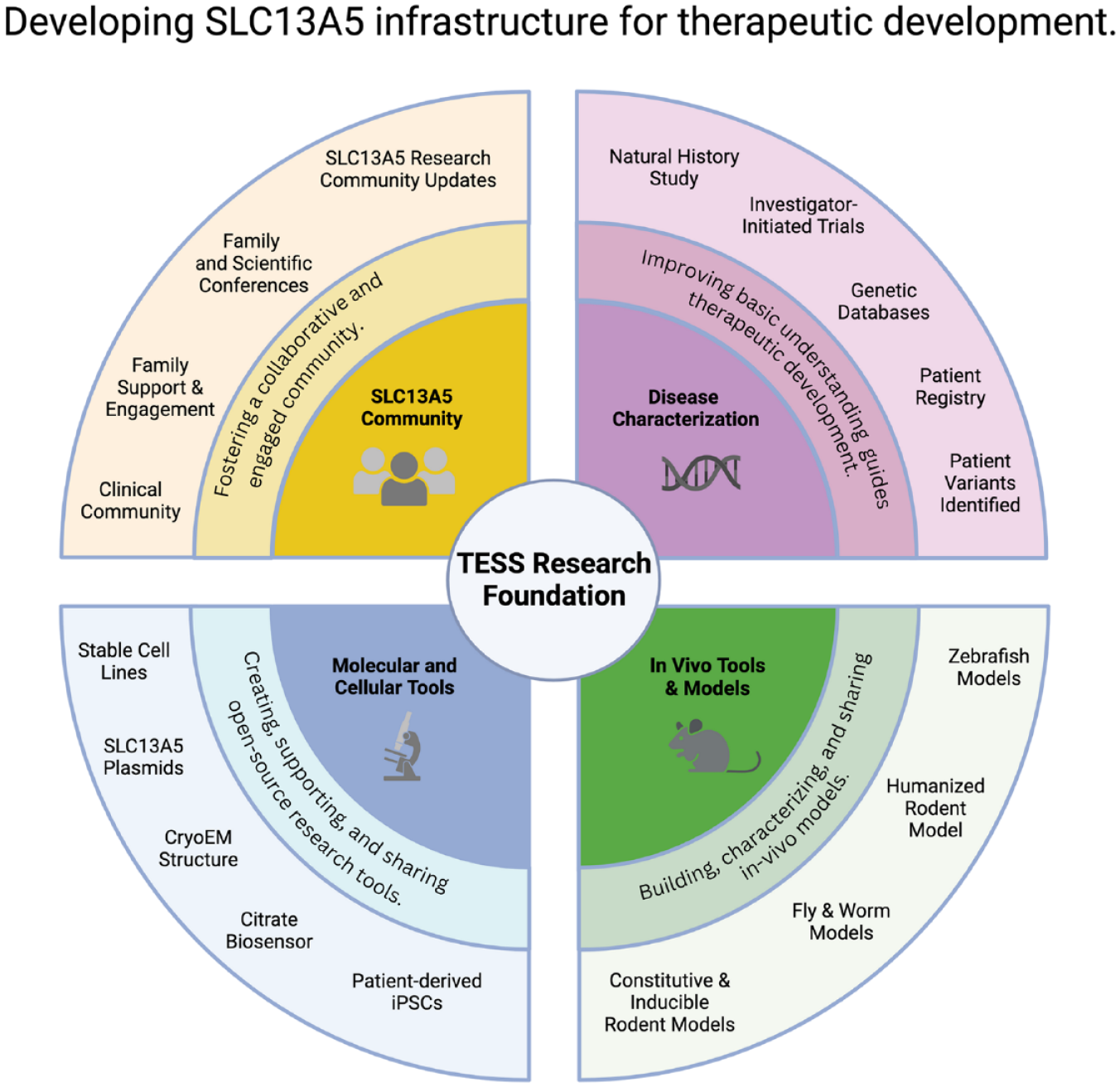
Available resources for research into SLC13A5. Research tools include models of protein activity and structure, patient phenotyping across the lifespan, and biomarker development for future clinical trials. Molecular tools include multiple animal models, iPSCs and other mutant cell-lines. Lastly, TESS brings together the research and patient community with monthly family meetings providing support and research updates, research community talks and annual conferences.
In addition to serving to bridge the research, clinical, and patient communities, TESS provides seed funding for projects that can then seek out larger and more sustainable funding through entities such as the National Institute of Health and Rare Diseases Clinical Research Network. The foundation grants support projects to collect preliminary data, thus de-risking projects. To date, TESS has distributed over $2 million in research funding to 23 different researchers. 22 Seed funding from TESS Research Foundation has supported research leading to NIH funding, including three R01s, one R21, an R13, a K99, and an F99. 24 Seed funding has supported basic, translational, and clinical studies including NaCT structural studies, the development of potential therapeutics including an SLC13A5 gene therapy and rational drug design, as well as ongoing Natural History Studies. 22 Additionally, grants and awards from the Chan Zuckerberg Initiative Rare As One Project and the Patient-Centered Outcomes Research Institute supports TESS internal salaries and projects. For example, in their patient registry, TESS aggregates phenotypic and genotypic data and has the most extensive collection of known pathogenic mutations in SLC13A5. TESS also identifies and links potential collaborators and provides letters of support for grant applications. Most significantly, TESS has engaged patients as partners throughout this research process.
Governance, operations, and strategy: growing the SLC13A5 community through the board of directors, scientific advisory board, and staff
TESS maintains two volunteer boards that advise, guide, and implement TESS’ mission. These boards are champions for SLC13A5 citrate transporter disorder and a major reason why the understanding of the disorder has made rapid progress since 2014. The board of directors is composed of volunteers who donate their time, expertise, and resources to advancing the work of TESS Research Foundation. Directors have expertise in law, communications, technology, finance, and fundraising, ensuring that TESS remains a viable, highly efficient nonprofit in good standing, obeying applicable laws and regulations and following our bylaws. The majority of our board members have a direct connection to a person with SLC13A5 citrate transporter disorder, ensuring our mission remains patient-centered and instilling a sense of urgency. The board approves all major funding decisions, implements best practice policies, and expands the SLC13A5 network by inviting their connections to learn about SLC13A5 citrate transporter disorder and TESS Research Foundation.
The TESS Scientific Advisory Board (SAB) is composed of clinical, scientific, and pharmaceutical industry experts who donate their time and expertise to assist the TESS board of directors. The SAB establishes research priorities, reviews grant proposals, and guides the basic, translational, and clinical research toward therapeutic development. The SAB also champions SLC13A5 research to the broader scientific community. The TESS boards have had a major impact on the development of tools needed for understanding the underlying disease mechanisms, drug discovery, and clinical trial readiness.
As TESS has grown and matured, so has its internal capacity. TESS currently has a mixture of paid and volunteer staff, including a full-time salaried scientific director, operations manager, and part-time development director. Volunteer staff includes a full-time executive director and part-time communications director and family outreach coordinator. These paid positions were made possible by grants supporting projects from the Chan Zuckerberg Initiative Rare as One Project and PCORI. This small but mighty staff operationalizes the boards’ directives and executes in day-to-day progress.
Disease characterization
Despite substantial research on NaCT, its role in human health and disease is not well understood. When SLC13A5 citrate transport disorder was first described there were few scientific tools available for studying the disorder. 1 Since 2014, a 2 Å cryo electronmicroscopy (cryo-EM) 3D structure of NaCT was resolved. 25 TESS maintains a database of all patient-identified variants, and all tested to date have a loss of citrate transport function.5,25,26 Described variants include pathogenic, likely pathogenic, and variants of uncertain significance (VUS) and continue to be added in the scientific literature and through direct communication with affected families to the TESS database (Figure 2, Supplemental Table 1). TESS has collected a comprehensive list of pathogenic and likely pathogenic variants from around the world, along with the frequency at which they are found in this growing cohort. This data have proven critical for estimating disease incidence and determining which allele-specific therapies might target the greatest number of patients. 27 A remaining challenge is functionally characterizing the large number of SLC13A5 variants and the RESOLUTE and REsolution consortiums, European public private partnerships, for de-orphanizing solute carrier proteins, have made significant headway in this area. 28
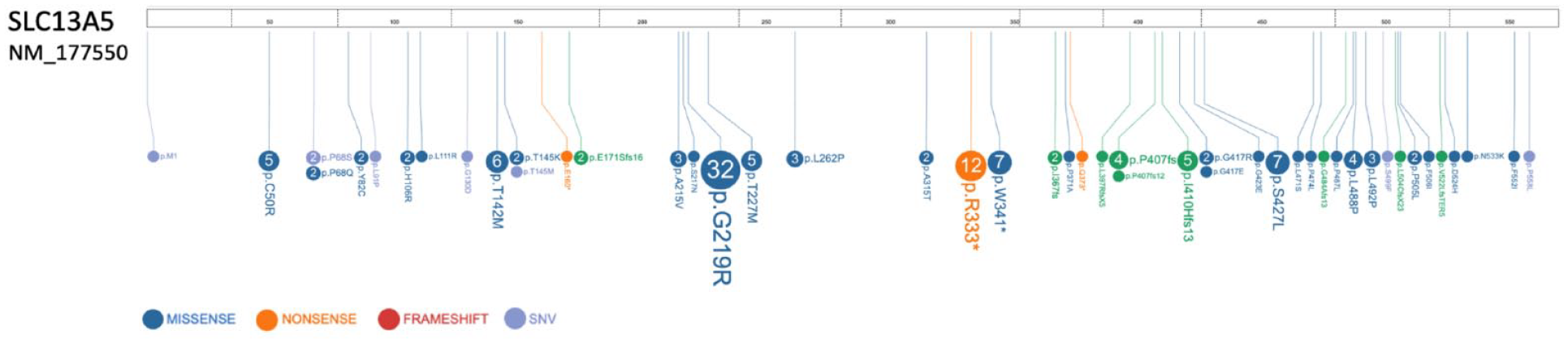
SLC13A5 patient variants. SLC13A5 variants are found throughout the SLC13A5 protein (NaCT). Numbers in bubble reflect the the number of patients in the TESS Research Foundation patient registry with at least one copy of variant. Image was created using ProteinPaint.
TESS grantees and research partners have conducted extensive studies on citrate transporter activity as well as measuring citrate of patients’ serum and cerebrospinal fluid (CSF) demonstrating increased citrate levels in all subjects studied to date.2,4,5,9,26,29,30 Thus, elevated citrate in serum and CSF appears to be an orthogonal, functional molecular marker of the disease and helpful to further confirm the disease etiology in those suspected of SLC13A5 but with VUS on genetic testing. Of note, a recent study using genetic and metabolomics data was able to show that plasma citrate level is a plausible biomarker for SLC13A5 function. 31 Future studies are focused on understanding the relationship between citrate levels and disease severity as well as the use of serum and CSF citrate as an outcome measure for clinical trials.
Understanding SLC13A5 citrate transporter disorder through Natural History Study
SLC13A5 citrate transporter disorder is a rare disease with significant impacts on all aspects of the patients’ and caregivers’ lives.4,8 This impact is reflected not only quantitatively, via disease impact focus groups, and quality of life measurements, but also qualitatively in the caregivers’ conversations, and the need for frequent hospital visits, the long lists of medications and therapies. TESS Research Foundation currently sponsors and partners in three natural history studies initiated in 2020 including (1) an In-person, prospective Natural History Study with academic partners at Stanford University, University of Texas, Southwestern, and Brown University open to patients in the United States, (2) a prospective, remote Natural History Study involving telehealth visits and questionnaires open to patients outside the United States, and (3) a retrospective Natural History Study analyzing de-identified medical records of SLC13A5 citrate transporter disorder patients utilizing the Ciitizen platform (Figure 3).32–34 The age of enrolled patients range from infants to adults, and the studies assess seizure burden, movement, global and cognitive development, overall health, and the impact on quality of life for caregivers and patients. These studies are critical for establishing baseline cognitive and motor ability, symptom progression or regression, biomarker discovery, and outcome measures for future clinical trials (Figure 3). NHS provides a more complete view of the disorder to facilitate specific caregiver counseling and information for treating physicians.4,7

SLC13A5 citrate transporter disorder clinical phenotype. (A) SLC13A5 citrate transporter disorder is a disorder that affects multiple regions of the body with a prominent neurological phenotype, dentition issues, a severe movement disorder, and citrate elevation. (B) Ongoing SLC13A5 NHS includes retrospective and prospective studies.
In vivo tools and models
Animal models are critical for understanding NaCT function as it pertains to the multiple systems impacted by loss of citrate transport and for the development of therapeutics. Multiple animal models that can recapitulate some of the disease phenotypes are available for studying SLC13A5 including constitutive gene knockouts in flies, worms, and mice (Table 1). In flies and worms, mutation or knockdown of NaCT – also known as I’m Not Dead Yet (INDY) in flies – extends lifespan in a similar manner to calorie restriction.17–19 Interestingly, rodent SLC13A5 gene deletion models recapitulate a milder neurologic phenotype of the human disorder.18,20 SLC13A5 constitutive knockout mice show reduced body weight, arterial blood pressure, heart rate, and mice are protected against development of dietary and age-related diabetes.18,35 This has led to interest in targeting SLC13A5 in the liver to help prevent metabolic syndrome and type II diabetes.36–40 Although subtle, these mice were also observed to have seizures beginning at 7 weeks, and the mice showed lower seizure threshold when challenged with chemoconvulsants. 20 Loss of NaCT also leads to reduced enamel formation in rodents and human.41–43 In contrast to loss of function (LOF) models, overexpression of SLC13A5 in the forebrain of mice lead to autistic-like behaviors and disrupted white matter integrity while generalized overexpression leads to a progeria phenotype.44,45
Tools available to study SLC13A5. Tools include multiple animal models, iPSCs, including isogenic controls, neural precursor cells, biosensors as well as multiple inhibitors.

Although confounding, one hypothesis for the milder neurological phenotype in the constitutive SLC13A5 knockout mice is the significant interspecies differences in the specificity of the NaCT for citrate.25,29,50 While bacterial, rodent, and dog NaCT transport citrate with a high affinity and low capacity, human NaCT is a low affinity, but high-capacity transporter and more highly selective for citrate, while rodent NaCT transports citrate and succinate equally well.25,29,52–54 While important progress has been made using rodent loss of NaCT models, these species-specific differences indicate that studying the human NaCT is particularly relevant for understanding SLC13A5 citrate transporter disorder. TESS Research Foundation received a grant from the Orphan Disease Center Jumpstart program for the development of a humanized SLC13A5 mouse model. This model is currently available at the Jackson Laboratory and demonstrates a tangible example of how Patient Advocacy Groups can help drive research through supporting the development of multiple different disease models. Funding provided by the TESS Research Foundation, National Research Council of Canada, and NIH has supported the development and characterization of additional models: including SLC13A5fl/fl mice available for conditional knockout by crossing with mice harboring different floxed promoter as well as mice with two common pathogenic SLC13A5 mutations seen in patients, mouse G222R equivalent to human G219R, and mouse T230M equivalent to human T227M (personal communication). 43
Molecular and cellular tools
Multiple molecular and cellular tools exist to study SLC13A5. TESS funded the development of seven induced Pluripotent Stem Cell lines (iPSCs), four from SLC13A5 citrate transporter disorder patients with compound heterozygous SLC13A5 variants, of which one currently has isogenic (mutation corrected) control and additional isogenic controls under development, and three lines from SLC13A5 heterozygous carriers as additional controls. 22 These include the most common mutation (G219R) as well as two individuals with a heterozygous deletion of the gene which ensures all introduced mutations are hemizygous. Multiple groups are developing additional iPSCs (personal communication). Additional tools include multiple SLC13A5 encoding plasmids, siRNA, and a genetically encoded citrate biosensor.39,47–49 TESS continues to support the development of molecular and cellular models by funding patient-derived iPSCs characterization and cryo-EM analysis of mutations. 22 Solving the cryo-EM structure of human NaCT provides important understanding of NaCT, as well as the ability to characterize SLC13A5 variants based on the variant location and predicted biophysical mechanism.5,25 These studies established two types of pathogenic mutations: Type I variants affect NaCT cellular localization and protein function, while Type II variants are targeted to the cell membrane, but lack citrate transport activity. Localizing the variants onto the 3D structure has allowed further refinement of variant classification.5,25,50
Additional molecular and cellular tools are currently in development, including small molecule NaCT inhibitors (Table 1).39,40,51 Antibodies that provide specificity for NaCT have been lacking, and TESS is supporting the development of nanobodies. Additionally, TESS Research Foundation is also partnering with COMBINEDBrain to collect multiple patient tissues as a part of the COMBINEDBrain Biobank. 55 Importantly, part of the goal of TESS Research Foundation is to make all of these tools, from the molecular and cellular tools to in vivo models, accessible to researchers in both academia and industry.
Public databases
The rapid growth of information in publicly accessible genetic and patient databases as well as biobanks has opened unprecedented avenues for disease research to conduct in-depth analyses for genetic variants, metabolites and clinical data, uncover novel correlations, and gain profound insights into the underlying mechanisms of a particular disease (Table 1). For SLC13A5 citrate transporter disorder, these public databases can be used to further characterize known or to identify new genetic variants and linking these to disease-related outcomes and biomarkers. Two recent studies utilized data from Ciitizen, a patient-facing platform that streamlines the generation of regulatory-grade clinical data from unstructured sources. These studies characterized the overall growth patterns, laboratory tests, diagnostic codes, neurodevelopmental time course, imaging and electroencephalogram observed in patients with SLC13A5 disorder within the United States.4,7 In two other studies, genetic variants in the SLC13A5 genes within the UK Biobank leveraged a Mendelian randomization paradigm to gain insight into the effects of SLC13A5 inhibition on health-related outcomes and biomarkers.31,46 The UK Biobank is a large-scale biomedical database and research resource containing de-identified genetic, lifestyle, and health information and biological samples of approximately 500,000 volunteers at the mean age of 57 years (https://www.ukbiobank.ac.uk). There is evidence that reduced SLC13A5 function due to genetic variation in the SLC13A5 locus is causally linked to higher citrate and calcium levels in plasma, lower fasting blood glucose as well as improved kidney function measured by blood urea nitrogen, creatinine, and cystatin C. 31 The second study showed that single nucleotide variants which are linked to reduced SLC13A5 function lowered the osteoporosis risk in the middle-aged population of the UK biobank. 46
Discussion/Conclusion
TESS Research Foundation is a motivated partner working to drive therapeutic development
Patient Advocacy Groups can be powerful partners for researchers and industry. TESS Research Foundation is working to drive therapeutic development from basic science through to clinical trials. Supported in part by the Chan Zuckerberg Initiative Rare as One Project and the Patient-Centered Outcomes Research Institute (PCORI) Engagement Awards, two awards focused on capacity building for small Patient Advocacy Groups, TESS continues to drive therapeutic development for SLC13A5 citrate transporter disorder. TESS Research Foundation provides seed funding for research projects and brings together multiple stakeholders to develop therapies for SLC13A5 citrate transporter disorder. TESS Research Foundation is highly motivated to develop and nurture a strong research community and provides a platform of collaboration and open science through monthly SLC13A5 Research Community Updates, a patient registry, multiple natural history studies, as well as developing and providing SLC13A5 patient and family community input for researchers.
Future directions: building a research roadmap for SLC13A5 citrate transporter disorder
TESS Research Foundation has developed a research roadmap that reflects our efforts to develop new treatments (Figure 4). This is possible due to the volunteer SAB, the individual researchers stretching seed funding or donating their resources, industry partners taking on a rare disease indication, and the affected families sharing their lived experiences and donating biosamples. This is also made possible by the TESS staff and volunteers, a small but mighty team working to support SLC13A5 research, community, and education. SLC13A5 research continues to make great strides, but there are multiple fundamental questions about the disorder that are unanswered. Pathogenic SLC13A5 variants lead to a loss of cell membrane citrate transport, as well as elevated citrate in the CSF and serum in LOF animal models.2,4,30 However, surprisingly, in SLC13A5 knockout mice, tissue-specific citrate data are contradicting with citrate decrease in tissues from the parahippocampal cortex but not in hippocampus, 20 and increased in other cell types such as osteoblasts, suggesting a potential compensatory mechanism possibly via mitochondrial citrate production under some but not all conditions. 43 Since there are some key species differences in the NaCT protein between humans and other species,15,16,25,29,50 is this compensatory mechanism also found in human tissue? Furthermore, human SLC13A5 is most highly expressed in the liver and to a much lower extent in the brain, yet LOF leads to a severe neurological phenotype.13,16,38 These findings lead to multiple basic unanswered research questions:
(1) How does cell membrane citrate transport contribute to normal cellular function?
(2) What is the role of citrate in neurological symptoms?
Is it increased extracellular citrate versus decreased intracellular citrate or do both contribute?
(3) What organs and cell types contribute to the disorder?
(4) At what developmental stage(s) is loss of NaCT detrimental?
(5) Why are the kidneys not able to sufficiently excrete the excess serum citrate into the urine?
(6) Does aberrant protein folding of the NaCT mutants and inappropriate subcellular targeting contribute to the disease phenotype?

TESS works in a patient-centered manner to collaborate with all stakeholders to develop treatments for SLC13A5 citrate transporter disorder. This process is an iterative and collaborative process requiring sharing of unpublished and published resources.
There are additional areas of research inquiry raised by the patients. Preliminary patient data indicate a universal onset of seizures in the first days of life and a reduction in seizures in later childhood or adulthood.3,9,10 It remains to be determined whether the seizure reduction is due to use of effective antiseizure medications or the natural progression of the disease. The severe movement disorder in patients has not been reported in animal models. 4 Additional aspects of the disorder that need further characterization include detailed seizure diaries, metabolic assessments, neurocognitive assessments, and quality of life measures in patients and their caregivers. Genetic testing is important for diagnoses, yet only a small fraction of identified variants have been tested for citrate transport activity.5,9,26 Thus, it will be important to functionally test all identified SLC13A5 variants to determine variant pathogenicity and determine if there are phenotype genotype correlates.
Currently, there is no cure for SLC13A5 citrate transporter disorder. Patients continue to rely on symptom management with most requiring multiple antiseizure medications as well as physical, occupational, and speech therapies. One adeno-associated virus (AAV) gene therapy is currently in preclinical development to address the lack of citrate transport in the central nervous system by providing a healthy copy of the SLC13A5 gene, development of additional therapies are needed including repurposed drugs, small molecule development, or metabolic interventions. Importantly, patients may benefit from a combination of therapeutic approaches.
By continuing to fund high-priority research and partner with patients, researchers, clinicians, and industry members, TESS Research Foundation will continue to drive patient-centered SLC13A5 research. With an expansive research toolbox and motivated partners, SLC13A5 citrate transporter disorder is primed for significant research progress moving closer to successful treatments for this rare disease.
Supplemental Material
sj-docx-1-trd-10.1177_26330040241263972 – Supplemental material for The growing research toolbox for SLC13A5 citrate transporter disorder: a rare disease with animal models, cell lines, an ongoing Natural History Study and an engaged patient advocacy organization
Supplemental material, sj-docx-1-trd-10.1177_26330040241263972 for The growing research toolbox for SLC13A5 citrate transporter disorder: a rare disease with animal models, cell lines, an ongoing Natural History Study and an engaged patient advocacy organization by Tanya L. Brown, Matthew N. Bainbridge, Grit Zahn, Kim L. Nye and Brenda E. Porter in Therapeutic Advances in Rare Disease
Footnotes
Appendix
Acknowledgements
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
