Abstract
Background:
The current road to developing treatments for rare diseases is often slow, expensive, and riddled with risk. Change is needed to improve the process, both in how we think about rare disease treatment development and the infrastructure we build to support ongoing science. The National Institutes of Health (NIH)-supported Rare Diseases Clinical Research Network (RDCRN) was established to advance the diagnosis, management, and treatment of rare diseases and to promote highly collaborative, multi-site, patient-centric, translational, and clinical research. The current iteration of the RDCRN intends to build upon and enhance successful approaches within the network while identifying innovative methods to fill gaps and address needs in the approach to the rare disease treatment development process through innovation, collaboration, and clinical trial readiness.
Objective:
The objective of this paper is to provide an overview of the productivity and influence of the RDCRN since it was first established 20 years ago.
Design and methods:
Using a suite of tools available to NIH staff that provides access to a comprehensive, curated, extensively linked data set of global grants, patents, publications, clinical trials, and FDA-approved drugs, a series of queries were executed that conducted bibliometric, co-author, and co-occurrence analysis.
Results:
The results demonstrate that the entire RDCRN consortia and network has been highly productive since its inception. They have produced 2763 high-quality publications that have been cited more than 100,000 times, expanded international networks, and contributed scientifically to ten FDA-approved treatments for rare diseases.
Conclusion:
The RDCRN program has successfully addressed some significant challenges while developing treatments for rare diseases. However, looking to the future and being agile in facing new challenges that arise as science progresses is important.
Plain language summary
The Rare Diseases Clinical Research Network (RDCRN) is a Federally directed research network that targets research to help investigators move closer to treatments for rare diseases. The network supports 20 different groups that study rare diseases. Each group focuses on three or more rare diseases and the research is conducted at multiple sites. Each group works closely with both the National Institutes of Health (NIH) and patient advocacy groups.
The primary focus of the network is clinical trials readiness, which simply means knowing who to treat, when to treat, and how to treat, thus taking some of the risk out of clinical trials. This knowledge is gained through natural history studies.
The network, supported by grants, holds a competition every five years to select groups to participate in the network. The RDCRN is supported by ten different institutes at the NIH.
To date the RDCRN has published numerous manuscripts in topics ranging from findings from natural history studies and case reports to practice guidelines and clinical trials. To date the RDCRN has been involved in work that has led to ten treatments being approved by the Food and Drug Administration (FDA).
Introduction
‘Rare is not rare’ is a phrase embraced by the rare disease community. While estimates vary, there are between 5000 and 10,000 rare diseases that impact the lives of approximately 30 million people in the United States, and 350 million people worldwide (Figure 1). 1 Treatments for rare diseases, however, are rare, with less than 5% of rare diseases having an FDA-approved treatment. It has been estimated that it can take as long as 10–15 years to get a drug from discovery to market and this lengthy journey may cost as much as 2.6 billion dollars.2,3 There are about ~30–50 new drugs/biologics for rare diseases approved each year in the United States. While this is laudable, most of these approvals are for rare diseases that have existing treatments. The rate of approval for diseases that have no prior approved treatment, or a new condition, is only about ~3–5 new drugs per year. Assuming the rate of approval of new treatments for previously untreated rare diseases remains constant, in the next 10 years treatments would become available for only ∼600 of the known rare diseases. 4 The slow pace has been attributed to many causes, some of which are inherent to rare diseases themselves, such as the small numbers of patients and clinicians for any one condition and their dispersed geographic distribution. The lack of in-depth knowledge of disease progression, range of disease manifestation, and a lack of adequate clinical or biological markers to support the clinical development of new therapeutics also contribute to slow development. In addition to the hurdles slowing down the pace of rare disease research, once a treatment gets to development, there is no guarantee that the treatment will make it to the patients. It has been estimated that less than 12% of all drugs entering development make it to market, with failure most often attributed to a lack of efficacy (56%) or safety issues (28%). 5

The pace of disease gene discovery as cataloged by the OMIM Morbid Map Scorecard. 6
It is evident that the current road to the development of treatments for rare diseases is slow, expensive, and riddled with risk. Change is needed to improve the process, both in the way we think about rare disease treatment development and the infrastructure that we build to support ongoing science. Austin and colleagues suggested change in the current process could be achieved through a more efficient development process driven by radically new approaches including utilizing common standards across distinct research fields, sharing best practices, creating sustainable business models, and redefining the regulatory environment. 4 While one program cannot solve all the problems faced in the current treatment development pipeline, it can facilitate meaningful changes in many areas, and make a significant difference. One program that was established to address such problems is the Rare Diseases Clinical Research Network (RDCRN).
The Rare Diseases Clinical Research Network
The RDCRN was established via The Rare Diseases Act of 2002 (Public Law 107-280) which directed the National Institutes of Health (NIH) to support regional ‘Rare Disease Clinical Centers of Excellence’ (RDCRCs) for clinical research, career enhancement, and demonstration of diagnostic, prevention, control, and treatment methods for rare diseases. The RDCRN is a network of consortia that each study at least three different rare diseases. The RDCRN has been continually funded, through competitive grant cycles every 5 years since 2003 (RDCRN1, RDCRN2, and RDCRN3) and is currently in its fourth funding cycle (RDCRN4, 2019–2023; Figure 2). Scientific oversight for the RDCRN has been provided by the National Center for Advancing Translational Sciences (NCATS) working collaboratively with other NIH institutes and centers including the National Institute of Neurological Disorders and Stroke (NINDS); the National Institute of Allergy and Infectious Diseases (NIAID); the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK); the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD); the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS); the National Heart, Lung, and Blood Institute (NHLBI); the National Institute of Dental and Craniofacial Research (NIDCR); the National Institute of Mental Health (NIMH); the Office of Dietary Supplements (ODS); and the NIH Office of the Director (OD). Each consortium is funded by two or more NIH institutes/centers reflecting that rare diseases often span the missions of multiple institutes.

Rare Diseases Clinical Research Network Consortia and Funders 2018–2024.
The underlying approach of the RDCRN has always been broad, as dictated by The Rare Disease Act of 2002, to include ‘regional centers of excellence for clinical research into, training in, and demonstration of diagnostic, prevention, control, and treatment methods for rare diseases’ (Public Law 107-280). As a program tasked to address these challenges, we ask, ‘how can we work faster, amplify our efforts by sharing resources, and have strong data from rigorous research’? One method is embracing the principles of translational science (Figures 3 and 4), an approach that aims to accelerate the process of turning biomedical research discoveries into real-world applications. 7 Why this approach? While it is often thought that the only roadblocks to developing treatments for rare diseases are scientific, other barriers including operational, financial, and administrative significantly impede the rate of scientific progress and add significant expense to the discovery process. These principles promote ‘out-of-the-box’ thinking and can be generalized to multiple different disciplines within the rare disease drug development pipeline. The principles include the following:
Crosscutting solutions for many of the persistent challenges
Emphasis on creativity and innovation
Leveraging cross-disciplinary teams
Enhancing the efficiency and speed of translational research
Utilizing boundary-crossing partnerships
Using bold and rigorous research approaches

The field of translational science aims to accelerate the translation of research discoveries into solutions to improve human health, by transforming the way that translational research is done.

Translational science principles.
Rare disease research usually focuses on one disorder at a time. An area of emphasis in RDCRN4 is to collaborate to identify common threads, common mechanisms, and common solutions for common problems across the consortia within the RDCRN. For example, problem-solving could reach across consortia by shifting focus from disease-specific challenges to identifying commonalities across broader groups, such as neurologic disorders, metabolic disorders, immunologic disorders, or mendelian disorders (Table 1). Similarly, the consortia could focus on common mechanisms or common organ systems (Table 1). Experts from various disciplines are encouraged to look beyond the confines of their own specific rare diseases for expertise to solve challenges that are faced by multiple research teams. Consortia are encouraged to discuss not only the science underlying rare diseases but also to share best practices for recruitment, diversity outreach, data analysis, and innovative clinical trial design.
Examples of commonalities across RDCRN consortia.
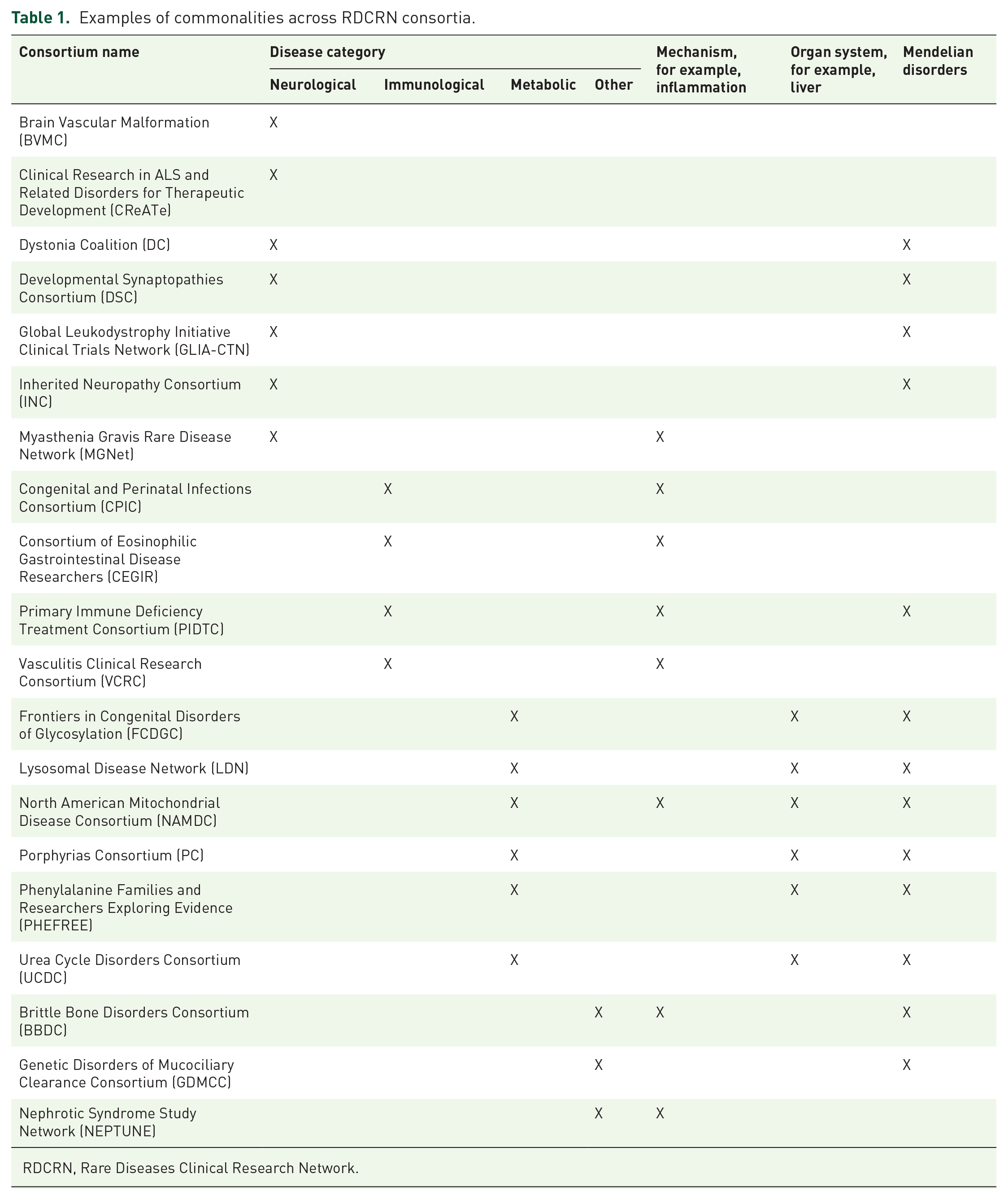
RDCRN, Rare Diseases Clinical Research Network.
RDCRN consortia
Each individual consortium within the RDCRN must meet the following criteria:
Consists of multiple clinical sites
Studies three or more rare diseases
Conducts clinical studies, one of which is a natural history study or longitudinal in nature
Includes patients and patient advocacy groups that are integrated into the consortium in a meaningful manner
Provides career enhancement for new and upcoming rare disease researchers
Conducts pilot studies
Each consortium promotes highly collaborative, multi-site, patient-centric, translational, and clinical research with the intent of addressing unmet clinical trial readiness needs that will move the field forward from its current state. Simply stated, the goal of clinical trial readiness is to lessen or de-risk the treatment development process. Within the context of the RDCRN, the ‘de-risking’ process is intended to begin early on the road to treatment development, in parallel to basic and preclinical research. Examples of clinical trial readiness within the RDCRN include studies that validate clinical research tools, including biomarkers or clinical outcome assessment measures that are fit-for-purpose within a defined context of use relevant to clinical trials. Studies also include projects that expand the knowledge of disease natural history necessary for clinical trial design and studies identifying characteristics for stratification or determining inclusion and exclusion criteria; the stage of disease progression that may be responsive to treatment; and data needed for determining sample size. It is important to emphasize that the science conducted within the RDCRN must adhere to NIH principles of rigor and reproducibility, with investigators providing evidence of preclinical efficacy and the level of effect of any treatment that is being evaluated to ensure that clinical testing is warranted.
An important component of each consortium and the network is the involvement of the Coalition of Patient Advocacy Groups (CPAG). It is a requirement of the network for the consortium to have a meaningful partnership with a patient advocacy group and/or patients. The relationship must extend beyond a letter of support for a grant application or a yearly update from the investigators. CPAG members are invited to participate as partners in consortium activities, including monthly calls and meetings. CPAG members are active in both the consortia and network by providing insight, input, and feedback to clinicians, researchers, and the NIH. Currently, there are 163 active Patient Advocacy Groups (PAGs) within the network. Individual consortia have between 1 and 60 PAGs partnering in a variety of activities at differing levels of effort. The average number of PAGs within a consortium is 9, with a median of 4.
The importance of developing the next cohort of clinicians and researchers in rare diseases is emphasized through the career enhancement core. This core of each consortium facilitates critical education of future generations of rare disease clinician investigators who will carry on the important clinical trial readiness and therapeutic development initiated by the RDCRN.
The CPAGs have an active steering committee that meets monthly with a focus on research. The groups share ideas, information, experiences, and resources with the primary focus being research. They also have representatives who serve on the RDCRN Executive Leadership team alongside the scientific steering committee chairs.
The RDCRN has continued to grow and develop since the first 10 consortia were funded in 2003–2004 (Table 2). Between 2003 and 2018 (RDCRN1, RDCRN2, and RDCRN3), 28 individual consortia were funded. The accomplishments of the first two cycles of the RDCRN have been previously documented in the literature.8–11 In this paper, we will focus on the continued progress of the RDCRN in its third cycle of support (RDCRN3, 2014–2018), as well as provide updates to the RDCRN structure and research priorities in the most recent fourth cycle (RDCRN4) that was initiated in 2019 (through 2024).
Funded rare diseases Clinical Research Network Consortia 2003–2024.

RDCRN, Rare Diseases Clinical Research Network.
Objectives
The objective of this paper is to provide an overview of the productivity and influence of the RDCRN since it was first established 20 years ago. Bibliometric and network analyses were used to provide a quantitative, informed description of research productivity, citation impact, and the scope of research collaboration from RDCRN-supported articles during the period 2003–2020.12,13 Although bibliometric analysis does not provide a complete picture of all scientific communication necessary for translation, most of the biomedical knowledge and discoveries that lay the foundation for clinical practice will pass through the academic literature in some form. Thus, examining bibliometric patterns of how research findings are documented and shared in the literature is one method of charting the progress of translational science. In addition, we describe six case studies that demonstrate the practical application of translational science principles that have directly contributed to clinical trial readiness.
Methods
Data analysis tools
iSearch
iSearch (Version 2.6) (NIH Office of Portfolio Analysis) is a suite of tools available to NIH staff that, through a single interface, provides access to a comprehensive, curated, extensively linked data set of global grants, patents, publications, clinical trials, and FDA-approved drugs. The iSearch Publications tool utilizes the National Library of Medicine PubMed database and adds to its records from the NIH Scientific Publication Information Retrieval and Evaluation System (SPIRES). The SPIRES database contains verifiable mappings between scientific publications and NIH grant numbers.
iCite
iCite (Version 2.0) (NIH Office of Portfolio Analysis) is a web application that provides traditional citation metrics, along with the Relative Citation Ratio (RCR) and other metrics developed by the NIH Office of Portfolio Analysis. 14 RCR is a time-sensitive metric that uses an article’s co-citation network to field-normalize the number of citations it has received. 15 An article’s co-citation network consists of all other articles it was cited with during each instance of the article being cited by another publication. The RCR compares the analyzed article’s citations per year with citations per year received by other NIH-funded articles in the same field and year.15,16
VOSviewer
VOSviewer (Version 1.6.18) (Leiden University, Leiden, The Netherlands) is a free software tool for constructing and visualizing bibliometric networks. These networks may for instance include journals, researchers, or individual publications, and they can be constructed based on citation, bibliographic coupling, co-citation, or co-authorship relations. 17 VOSviewer also offers text mining functionality that can be used to construct and visualize co-occurrence networks of important terms extracted from a body of scientific literature. 18
Data collection
A query was executed in the iSearch Grants module to identify U54 grants awarded under Funding Opportunity Announcements (FOA) RR03-008, OD08-002, or TR13-002. Results were transferred to the iSearch Publications module and filtered on the publication period 2004–2020. The publications and associated metadata were exported as an Excel spreadsheet, or directly to iCite for further analysis.
Data analysis and visualization
Bibliometric analysis
The following data were produced using iCite: total number of publications, mean number of publications per year, number of citations per year (maximum, mean, standard error of the mean, and median), RCR (maximum, mean, standard error of the mean, and median), and weighted RCR. The RCR is a new metric developed within the Office of Portfolio Analysis that represents a citation-based measure of the scientific influence of one or more articles. It is calculated as the cites/year of each paper, normalized to the citations per year received by NIH-funded papers in the same field and year. A paper with an RCR of 1.0 has received the same number of cites/year as the median NIH-funded paper in its field, while a paper with an RCR of 2.0 has received twice as many cites/year as the median NIH-funded paper in its field.
Co-author analysis
PMIDs were used to search PubMed and the data were exported in the PubMed format. The authors and affiliations of each included publication were extracted, cleaned, and standardized using Excel and the VOSviewer thesaurus function. Co-authorship networks were generated using VOSviewer and the fractional counting method, for authors or organizations with at least five publications.
Co-occurrence analysis
VOSviewer was used to create a co-occurrence density map of all authors and MeSH keywords that occurred at least 20 times. Unrelated words (i.e. generic terms, regional words) were excluded and repetitive words (i.e. singular and plural forms, abbreviations, and full name) were standardized and merged using Excel and the VOSviewer thesaurus function.
Results
Bibliometric analysis
One useful way to understand the impact of a research program is through bibliometric portfolio analysis. Although publications are not themselves an end goal for translational endeavors, bibliometrics does describe a pivotal early stage in the process of translating new scientific discoveries to clinical use.
Publications linked to RDCRN grants were identified using the NIH internal data platform iSearch and analyzed using iCite. 14 RDCRN investigators who were funded during the first three cycles have been highly productive and produced 2763 publications in 644 journals from 2004 to 2020, with an average of 162 publications per year [Figure 5(a)]. Individual RDCRC produced an average of nine papers per year (range: 2–28). As of mid-2022, publications were cited a total of 108,643 times, with an average of 40 citations per publication [range 0–1306; Figure 5(b)]. Overall, there were 519 reviews, 355 studies (clinical, comparative, multicenter, or observational), 227 case reports, 166 trials (clinical or randomized controlled), 64 editorials, 19 meta-analyses, and 11 practice guidelines, as described by the PubMed article type.

RDCRN publication impact from 2004 to 2020. (a) Number of publications per year, (b) total number of citations by year cited, and (c) median RCR for each RDCRC.
To assess the impact of these papers, we used the metric RCR. 15 Developed at the NIH, the RCR represents the field- and time-normalized citation rate and is benchmarked to 1.0 for a typical (median) NIH paper in the corresponding year of publication. The median RCR score of all RDCRN-supported publications was 1.25, indicating that these publications were cited, on average, higher than 59% of comparable NIH-funded papers. Twenty-five out of twenty-eight consortia had an RCR greater than one [range 0.77–2.99; Figure 5(c)].
To measure the level of collaboration between RDCRN members, we constructed co-authorship networks using the open-source software platform VOSviewer. 17 There were 52 unique authors that contributed to at least five RDCRN-supported publications from 2004 to 2010. This increased to 340 authors for 2004–2010 and 946 authors for 2004–2020 (Table 3). Figure 6 represents the co-authorship networks of the largest connected set of authors. The number of links and total link strength (TLS) for 2004–2010 were 95 and 115, respectively. These increased to 2100 links and a TLS of 1551.50 for 2004–2015 and 15,067 links and a TLS of 5505 for 2004–2020. Despite the large increase in the size of the network, the number of clusters was the same for 2002–2015 and 2002–2020 (n = 22) demonstrating that the network growth was within established collaborations.
Co-authorship metrics of authors of RDCRN-supported publications 2004–2020.

RDCRN, Rare Diseases Clinical Research Network.

Co-authorship network of authors of RDCRN-supported publications 2004–2020.
RDCRN-supported papers were published by authors at 362 organizations from 18 countries from 2004 to 2020 (Table 4, Figure 7). Figure 8 represents the co-authorship networks of the largest connected set of organizations. The initial network of 2004–2010 consisted of six large US academic institutions with 15 links and a TLS of 4. In 2015, the network had expanded to include 113 organizations in 10 countries (1584 links, TLS = 514.5) and further in 2020 to include 362 organizations in 30 countries (15,102 links, TLS = 3726). International collaboration increased greatly from 2010 to 2020 and in 2020 47% of the organizations were outside of the United States (Figure 7).
Co-authorship metrics of organizations of RDCRN-supported publications 2004–2020.

RDCRN, Rare Diseases Clinical Research Network.

Co-authorship organizations of RDCRN-supported publications 2004–2020.

Co-authorship network of organizations of RDCRN-supported publications 2004–2020.
A co-occurrence visualization map was constructed using all authors and MeSH keywords 18 (Figure 9). Keywords appearing more than 20 times were included in the map (n = 229). The most common keywords were ‘mutation’ (400), ‘phenotype’ (226), ‘treatment outcome’ (201), ‘retrospective studies’ (183), ‘biomarkers’ (n = 182), ‘brain’ (171), ‘severity of illness index’ (168), and ‘cohort studies’ (155). The map reflects commonalities across topics of focus for the consortia that are universal including outcome measures, biomarkers, and study design.

Keyword co-occurrence visualization map of RDCRN-supported publications 2004–2020.
Clinical studies
A key feature of the RDCRN is the requirement for longitudinal natural history studies in rare diseases. The FDA issued a draft guidance, Rare Diseases: Natural History for Drug Development 19 to inform the design and implementation of natural history studies that can be used to support the development of safe and effective drugs and biological products for rare diseases. Since its inception, the RDCRN has conducted 85 natural history studies. In addition to natural history studies, the RDCRN also conducts pilot studies and clinical trials. Clinical trials are critical to developing and evaluating new treatments for rare diseases. To date, the RDCRN has directly supported 81 clinical trials, predominantly small early-phase studies of repurposed drugs, diets, supplements, procedures, devices, and some novel drugs. RDCRN-supported research has also contributed to many other larger phase II/III clinical trials of novel and repurposed drugs funded by industry, NIH Institute-specific grants, FDA, universities, and patient advocacy groups. RDCRN investigators’ contributions include disease phenotype, patient population, clinical sites, endpoints, biomarkers, as well as early phase safety and efficacy studies. The RDCRN contributions have led to the FDA approval for ten treatments for rare diseases, four of which are illustrated in the six case studies in the following section.
Case studies
Researchers from the Brittle Bone Disorders Consortium (BBDC) translated mechanistic findings into clinical research for osteogenesis imperfecta (OI). Their prior work demonstrated that transforming growth factor-β (TGF-β) is upregulated in bones and connective tissues of mouse models of common forms of OI, suggesting a common signaling pathway in OI and a potential therapeutic target. 20 Researchers used a multiomic approach analyzing RNA and protein expression in human bone samples from OI patients to reveal that TGF-β was upregulated when compared to non-OI bone. 21 To translate these findings to the clinic, the BBDC team tested fresolimumab, a monoclonal antibody therapy that neutralizes TGF-β in a phase I clinical trial. 21 The data showed that treating people with moderate OI using fresolimumab could improve their bone mass. 21 Sanofi has launched a larger phase Ib clinical trial to test safety, tolerability, and impact on bone density (NCT05231668).
Investigators from the Inherited Neuropathy Consortium (INC) demonstrated that mutations in the SORD gene resulting in loss of the enzyme sorbitol dehydrogenase (SORD) function, and consequent intracellular sorbitol accumulation, are responsible for disease in a subset of patients previously diagnosed with Charcot-Marie-Tooth disease (CMT2) or distal hereditary motor neuropathy (dHMN). 22 Interestingly, the SORD mutations were hidden from the gene analysis software most researchers use, and INC researchers found the relevant variations hidden behind a ‘pseudogene’ called SORDP2. 22 A pilot study with AT-007, an aldose reductase inhibitor, in patients with SORD Deficiency, showed that sorbitol level correlated with disease severity, and AT-007 treatment substantially reduced sorbitol levels by a mean of 66% from baseline. 23 These proof-of-concept results provided the impetus for a registrational phase III study of AT-007 by Applied Therapeutics that involves INC sites (NCT05397665). In a pre-specified interim analysis, AT-007 reduced sorbitol levels by a mean of 52%, or 16,000 ng/mL, over a 90-day period, which was highly statistically significant versus placebo (p < 0.001). 24
Research conducted by the Porphyrias Consortium (PC) played a role, through the natural history collected within the consortia, 25 to help Alnylam Pharmaceuticals develop and test the drug givosiran (Givlaari®) for the treatment of acute hepatic porphyria. The consortium’s six sites and some of its satellite research centers were among the study locations for Alnylam’s recent phase III clinical trial (NCT03338816). In addition, the consortium patient advocacy group, the American Porphyria Foundation, did much of the trial recruitment. This clinical trial concluded that patients with acute hepatic porphyria, who received givosiran, had a significantly lower rate of porphyria attacks and better results for multiple other disease manifestations than those who received the placebo. 26 This contributed to the FDA approval of this drug for adults with acute hepatic porphyria in November 2019. 27 The PC also worked closely with another pharmaceutical company, Clinuvel, on clinical trials to evaluate the safety and efficacy of an implant under the skin containing afamelanotide (Scenesse®) for treating erythropoietic protoporphyria (EPP). These trials found afamelanotide increased the amount of pain-free time people with EPP could spend in sunlight, as well as their overall quality of life. 28 The treatment was approved by the FDA in October 2019 – the first agent available to help people with EPP experience pain-free sun exposure. 29
The Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR) collected longitudinal clinical data and biopsy specimens from patients with eosinophilic esophagitis (EoE), an allergic inflammatory disease that damages the esophagus. 30 Researchers placed a special emphasis on developing patient-reported outcomes, along with more objectively measured clinical outcome metrics, and developed a core outcome set for therapeutic studies in EoE 31 which were used as endpoints in the pivotal studies of duplimab (Dupixent®; NCT03633617). Findings showed that the therapy improved symptoms and endoscopic, histologic, and molecular features of the disease. The FDA granted dupilumab priority review and breakthrough therapy designations for EoE, and on 20 May 2022 granted the approval of Dupixent® to Regeneron Pharmaceuticals, Inc. as the first treatment for EoE. 32
Rett syndrome is a genetic neurodevelopmental disorder that occurs almost exclusively in females and has a typically degenerative course. The Rett-related Disorders Consortium natural history study was a groundbreaking observational study that tracked the natural course of Rett syndrome in more than 1000 individuals from 2006 to 2021. This study generated extensive information on the spectrum of clinical involvement and correlated genotype–phenotype over a broad spectrum of phenotypes.33–38 The data are helping to validate diagnostic criteria39,40 and develop consensus guidelines across the lifespan. 41 In addition, information from this natural history study, combined with other large disease databases, 42 has been instrumental in establishing clinical trial readiness through the development of outcome measures43,44 and identifying putative biomarkers.45,46 Recent clinical trials47–49 culminated in the May 2023 FDA approval of trofinetide (Daybue™) as the first treatment for Rett syndrome. 50
Discussion
As the RDCRN approaches its 20th anniversary, the consortia within the network continue to mature scientifically, and expand, not only nationally but also internationally. The network is yielding a robust and growing body of influential research findings of consistently high impact. Findings from the RDCRN have contributed to the approval of ten treatments for rare diseases by the FDA. However, as with any longstanding program, while it is important to embrace success, it is equally important to look toward the future and attempt to anticipate challenges yet to come.
Many of the strengths within the RDCRN program are important to maintain and may be useful for some programs to emulate. A primary example of this includes the emphasis on studying more than one disease at a time. This not only allows for the exploration of commonalities across diseases but also allows multiple groups to tackle broad universal problems (e.g. recruitment, small sample size) together. Another asset of the RDCRN is the inclusion and significant partnership of the PAGs within consortia and across the network. Treatments for patients are the central premise of the network and by including PAGs at such a comprehensive level all parties benefit.
The most recent cycle of the RDCRN has focused on sharing resources across consortia. Centralized shared resources are provided to the network by NIH and managed by the DMCC. This approach is not only prudent but also provides a platform on which network-wide data standards can be established. The standards developed have focused on the FAIR principles enabling the data to be interoperable with many other existing rare disease data platforms. The network is also in the process of building a secure data-sharing environment managed by NCATS that will provide controlled access to the data to researchers and the community.
While the program has come a long way, there are still challenges that we face. One such challenge is the sheer number of rare diseases that exist and that need treatments. The RDCRN provides rare disease researchers the opportunity to establish a foundation that can be leveraged to expand ongoing clinical research. To provide consortia ample time to establish a foundation for research groups may compete for up to three cycles of support. Conversely, at the end of three cycles of support, the consortia must graduate from the program to provide an opportunity for other rare disease research groups to establish themselves.
Conclusion
It is also important to highlight the need to enhance diversity, equity, inclusion, and accessibility for both diagnosis and treatment. Special attention also needs to be paid to the increased development of genomic technologies that offer the hope of gene-targeted therapies for numerous rare disorders by directly targeting the causative molecular defect in genetic disease, and the potential to identify babies at or before birth.51,52 However, there are significant challenges in moving gene-targeted therapies from the research environment to a public health environment, including ethical, financial, and infrastructure considerations. 51 In the future, it may be possible to have treatments for a specific disorder and not be able to identify the individuals that could benefit from treatment until it is too late, necessitating periodic queries of a person’s genome throughout the life course. 53 Building robust data-sharing systems will speed drug discovery, optimize trial design and execution, and enable long-term follow-up of treated patients to assure unbiased assessments by all stakeholders of the relative efficacy and safety of new treatments. 54 Leveraging the data systems through artificial intelligence and machine learning will lead to further insight into and across multiple rare diseases. Finally, the value of considering groups of conditions together, despite regulatory challenges, rather than the current system of focusing on one rare disease at a time. 55
Footnotes
Acknowledgements
The authors would like to acknowledge patients and their families, RDCRN researchers and their teams, patient advocacy groups, NIH program officers, and NCATS colleagues.
Correction (May 2024):
Article updated to correct the number of FDA-approved treatments for rare diseases. For further details, please see 10.1177/26330040241256262.
Declarations
Availability of data and materials
Not applicable.
