Abstract
Persistently high-raised eosinophils are the hallmark of the rare heterogeneous group of illnesses known as idiopathic hypereosinophilic syndrome (HES). Eosinophil-mediated multiorgan damage is often the result of chronic illness. Although it can affect any organ, the heart, lungs, brain, spleen, and skin are frequently impacted. It may occasionally be necessary for management to use a multidisciplinary team approach. Cardiac involvement in the form of endomyocardial fibrosis or thromboembolic complications is frequent in the active disease. In HES, native and prosthetic mitral valve involvement is common; aortic valve involvement is extremely uncommon.
This case represents an unusual instance of prolonged aortic valve thrombosis in a male patient in his early fifties diagnosed with HES. Appropriate administration of blood-thinning agents and immunosuppressive treatment promotes effective management of thrombotic complications and associated symptoms, which eventually prevents further embolic problems and preserves the native valve.
Keywords
Introduction
Eosinophils are granulocytes that are a subset of myeloid cells. They have cytoplasmic granules with cytotoxic proteins inside of them, and these can destroy tissue directly when released. Eosinophils can circulate in the peripheral circulation; however, they are mostly found in mucosal tissues that are exposed to the environment. A heightened eosinophil count of more than 500 microliters (µL) is known as eosinophilia. A vast array of clinical disorders can arise from eosinophilia or it can remain asymptomatic. 1 In 1968, researchers Hardy and Anderson introduced the designation “hypereosinophilic syndrome” (HES) to describe a group of illnesses marked by organ damage and eosinophilia that is chronically severe. The pathogenesis of this illness is heterogeneous and might include neoplasms, autoimmune disorders, myeloproliferative diseases, allergies, and nonparasitic infections. The following criteria were established in 1975 as empirical diagnostics for the idiopathic hypereosinophilic syndrome (IHES): (a) a peripheral blood eosinophil count exceeding 1,500 cubic millimeters (mm3) for more than six months; (b) organ damage or dysfunction; and (c) the lack of identifiable causes for eosinophilia and eosinophilic blasts in the peripheral blood. The IHES cannot be confirmed without meeting all three requirements because it is an exclusion diagnosis. 2 From an epidemiological perspective, the illness primarily affects men and has a bimodal distribution in age between 20 and 50 (but it can also strike children). 2 Based on data from the SEER (Surveillance, Epidemiology, and End Results) program, the age-standardized occurrence rate averaged roughly 0.036 cases per 100,000 individuals between 2001 and 2005. 3
Any organ system including the skin, lungs, digestive, central, and/or peripheral nervous system can potentially be involved. In 50%-75% of cases, there is cardiac involvement, which can show up as restrictive cardiomyopathy, valve damage, and/or heart failure. There is a higher chance of thromboembolic consequences due to the disease’s intrinsic hypercoagulable status. 2 The relationship between blood eosinophilia and endomyocardial disease has been established in several clinical entities since 1893 when Reinbach initially reported a right ventricular endocardial mural thrombus in a patient with eosinophilia. These conditions encompass Löffler’s fibroplastic endocarditis, African endomyocardial fibrosis as described by Davies, eosinophilic leukemia, and connective tissue disorders associated with eosinophilia. 4 The initial acute phase of HES is marked by the accumulation of eosinophils within the subendocardial layer and is usually asymptomatic. However, in rare cases, fulminant myocarditis with widespread necrosis and quickly progressing heart failure may transpire. Patients run the danger of systemic embolization when intracavitary (mostly ventricular) thrombi occur in the second stage. Gradually, subendocardial fibrosis develops as a result of ongoing eosinophilic inflammation. 5 Later on, thrombi may organize as valvular or intraventricular vegetations, which would indicate an endocarditis known as Löffler’s endocarditis. Damage to the valves is often caused by fibrotic valvular thickening and/or decreased subvalvular apparatus movement, which most often results in aortic valve regurgitation. In rare instances, valve thickening and fibrosis can lead to aortic or mitral stenosis. 5
Thus, a rare but well-known side effect of HES is Löffler’s endocarditis. A review of the 26 case reports revealed that the mitral valve (65%) was the most often damaged cardiac structure, followed by the tricuspid valve in 42% of cases, the left ventricle in 23% of cases, and simultaneous involvement of both the valves in 35% of reports. 6 Therefore, there have been cases previously described of diffuse endocardial involvement with frequent valvular lesions affecting the mitral and tricuspid valve, although aortic valve involvement is still uncommon.
Anticoagulant therapy to prevent further clot formation and control of the eosinophil count is the major goal of therapy to stop organ damage from getting worse. 2
Diagnosis and Treatment Plan
In 2012, a 53-year-old male patient was unexpectedly found to have HES during his visit to a community health center (CHC) but did not receive any treatment at that time. He now presents with non-progressive dyspnea for the previous two months without chest pain, palpitations, or pedal edema. Informed and written consent was received from the patient. However, ethical committee approval was not required as the patient was treated for symptoms with a routine procedure, and no new treatment was required and this case report is not based on formalized data collection systems, such as registries, clinical trials, patient use programs that are named after approval, patient support and disease management programs, patient or healthcare provider surveys, or data collection on patient compliance or efficacy.
The patient had never used any medication in the past. First and second heart sounds were normal during the clinical examination, and there was a grade 2/6 ejection systolic murmur at the second right intercoastal space in the parasternal region. Additional systemic examinations turned up no noteworthy anomalies.
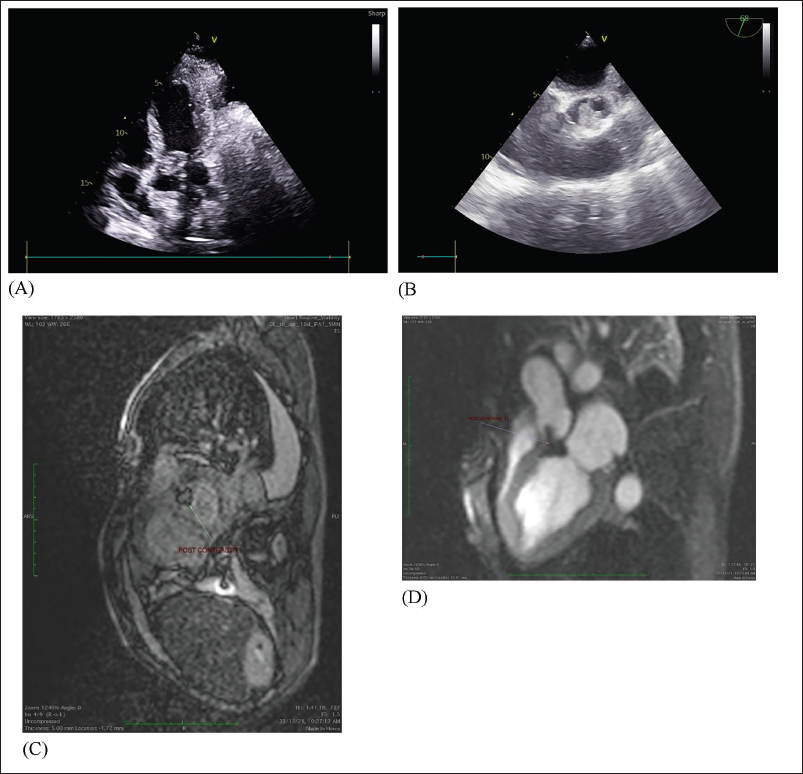
On routine blood investigation, it was found that the patient had an absolute eosinophil count of 7,210/cumm, and the remaining blood parameters were within normal limits. Transthoracic 2D echocardiography (ECHO) showed an irregular echogenic mass of approximately 2 × 2 cm attached to the aortic valve which was extending and creating an obstruction to the left ventricular outflow tract (LVOT). Mitral and tricuspid valves were normal. The ejection fraction was normal without any features of regional wall motion abnormality. On transesophageal 2D ECHO, it was confirmed that the attachment of the echogenic mass was at the right aortic cusp with extension to the LVOT (Figure 1). Infectious endocarditis was ruled out by the negative results of blood cultures, serological tests, and clinical characteristics; inflammatory markers, autoimmune and thrombophilic evaluations were also negative. X-rays and computed tomography (CT) thorax were done previously, which were normal for this patient and hence pulmonary causes for his non-progressive dyspnea were also ruled out. Cardiac magnetic resonance imaging (MRI) was performed to improve visualization and assess tissue characteristics. The imaging showed normal morphology and function of both ventricles, supported the diagnosis of the aortic valve thrombosis, as no enhancement was observed following contrast administration.
(A) Transthoracic 2D ECHO Showing Echogenic Mass Attached to Aortic Valve. (B) Transesophageal 2D ECHO Showing Echogenic Mass Attached to a Right Coronary Cusp (RCC). (C and D) Cardiac MRI Showing No Post-contrast Enhancement Suggestive of Thrombus.
The management was not streamlined in this case. Due to the rarity of this condition, management often requires a multidisciplinary approach involving a cardiologist, a hematologist, and possibly a cardiac surgeon. A specialist multidisciplinary discussion felt that there was no clear indication for surgical intervention unless there was a significant deterioration in the patient’s symptoms. Initial management included intravenous heparin, followed by a transition to oral anticoagulation therapy. The patient was also started with prednisone therapy. A structured plan for ongoing clinical assessment and ECHO surveillance was established. Close monitoring and ongoing management are essential to prevent complications and optimize outcomes for patients with this rare presentation.
Treatment Outcome
After continued therapy, ECHO revealed complete resolution of the thrombosis after four weeks, and the additional follow-up sessions revealed a symptom-free condition in the patient. The eosinophil count was within normal limits after four weeks. Prompt anticoagulation therapy not only led to thrombus resolution but also prevented future embolic complications and ultimately preserved the aortic valve. Therefore, it can be concluded that the patient responded well to oral steroids and anticoagulation therapy, which were subsequently prescribed as part of his long-term management plan.
Discussion
According to literature data, valve thrombosis usually concerns native and prosthetic mitral valves in HES, while aortic valve involvement is very rare, especially on the right aortic cusp with extension to the LVOT. Nevertheless, the involvement of the aortic valve was confirmed with the help of cardiac MRI and ECHO. Eosinophil buildup in tissues is a direct cause of organ damage in eosinophilic disorders. When there is cardiac involvement, eosinophils are first deposited in myocardial cells. After being locally activated, they generate and secrete cytotoxic proteins that damage the endothelium of the heart and capillaries. However, the quantity of eosinophils degranulating in tissues appears to be more closely linked to the severity of problems than peripheral blood eosinophilia. 2
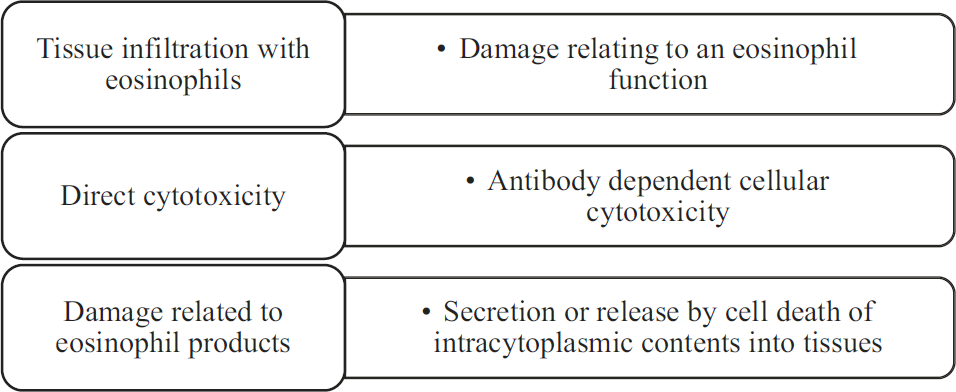
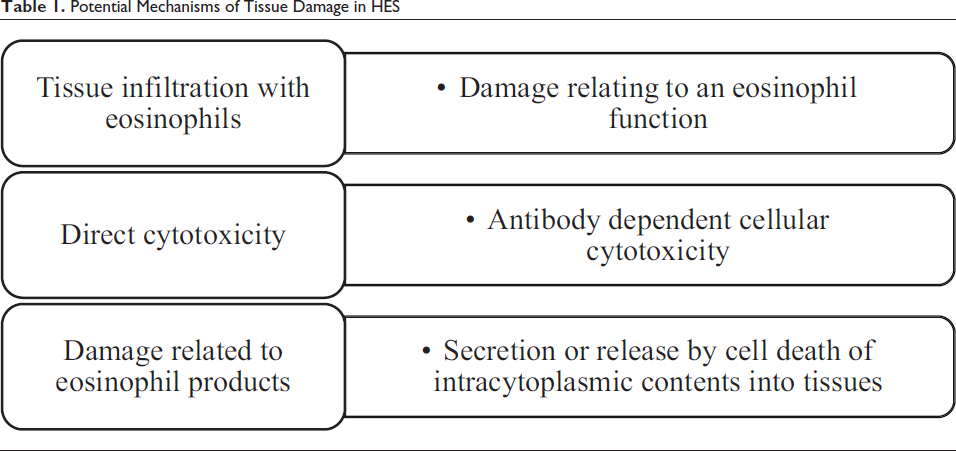
Effectively, three criteria are being used for establishing the diagnosis of idiopathic HES7, 8: persistent eosinophilia of 1,500 eosinophils/mm3 for at least six months or death before six months with signs and symptoms of HES disease; lack of evidence for parasitic, allergic, or other recognized causes of eosinophilia (Table 1) despite careful evaluation; and signs and symptoms of organ system involvement or dysfunction either directly related to eosinophilia or unexplained in the given clinical setting. Various potential mechanisms of eosinophil-related tissue damage in the syndrome have been appreciated recently (Table 1). Eosinophils have potential to infiltrate any organ system involved in the HES.
Potential Mechanisms of Tissue Damage in HES
However, majorly two major organ systems show the highest impact from HES, the heart, and the nervous system, exhibiting varying levels of tissue damage, functional impairment, or both that suggested mechanisms other than mere infiltration of eosinophils.7, 8 In HES, heart involvement is very common and remains leading cause for mortality. Initial acute necrosis, followed by thrombosis, and later fibrosis modeling are the three stages that have historically been distinguished in the cardiac pathology of HES. The eosinophils penetrate the endocardium, initiating the process of acute necrosis. The initial and primary target is the endocardium. After endocardial damage, platelet thrombi formation occurs over the injured or denuded endocardium, and a mural thrombus is propagated. Such thrombi commonly involve the posterior leaflet of the mitral or tricuspid valves leading to the atrioventricular valvular insufficiency, being characteristic of the endocardiopathy of the HES. Aortic semilunar valve involvement is uncommon in HES. 9 Peripheral embolization would readily follow in the course of the disease after the deposition of mural thrombi. Moreover, elevated eosinophil cationic protein in serum may contribute to hypercoagulation, which further exasperates the thromboembolic events. 10 The progression concludes with fibrotic thickening of the endomyocardium, often leading to restrictive cardiomyopathy.11, 12
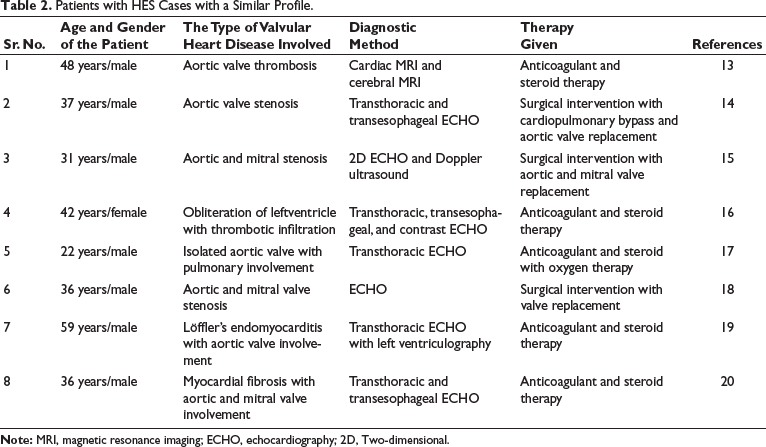
In HES, cardiac involvement may present as heart failure, thrombus formation within the heart—frequently affecting both ventricular chambers and adjoining structures—along with myocardial ischemia, arrhythmias, pericarditis, and transient loss of consciousness. Valve thrombosis often affects prosthetic and mitral valves; aortic valve involvement is extremely uncommon, according to literature data. 13 This case stands out due to the unusual presentation of extensive aortic valve thrombosis in a patient with HES, despite the absence of notable valvular dysfunction. Although rare, aortic valve involvement in HES can give rise to complications including thrombus formation, narrowing (stenosis), or backflow of blood (regurgitation). The best possible outcomes for patients with this uncommon manifestation of HES require early diagnosis and therapy. Numerous reports have documented similar cases of HES (Table 2). In our study, transthoracic 2D ECHO, transesophageal 2D ECHO, and cardiac MRI were utilized for diagnosis which was also the preferred method for the cases described in Table 2.
Patients with HES Cases with a Similar Profile.
Timely initiation of both anticoagulation and corticosteroid treatment resulted in the swift and complete resolution of thrombotic lesions, preserving the native valve and effectively preventing further embolic complications, all without causing significant valve dysfunction. Anticoagulants, such as heparin, work by inhibiting the clotting process, while steroids, such as prednisone, help reduce inflammation and suppress the production of eosinophils, the white blood cells implicated in the pathogenesis of HES. 13 The rapid and complete resolution of thrombotic lesions observed with this treatment approach underscores the importance of early intervention and aggressive management in patients with HES. In most studies presented in Table 2, the preferred method of intervention was anticoagulant therapy along with steroids. However, in a few cases, surgical intervention was required.
This case highlights the importance of early use of multimodal imaging in patients with HES to identify structural abnormalities contributing to symptoms. Early diagnosis, vigilant monitoring, and timely initiation of therapy can alter disease progression and help prevent complications.
Conclusion
The case presented here highlights the importance of vigilance in patients with HES, particularly regarding the rare occurrence of native aortic valve thrombosis. Early detection through ECHO and prompt initiation of therapy are crucial in preventing complications and preserving cardiac function. Long-term management should also focus on maintenance therapy to prevent disease recurrence and minimize associated complications. This underscores the necessity for treating physicians to remain cognizant of such possibilities and to closely monitor patients for any signs of valvular involvement or endomyocardial fibrosis.
Authors’ Contribution
SG helped in study concept, design, data collection, and drafting of the manuscript.
AT and SM contributed to data collection and drafting of the manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.