Abstract
Hypereosinophilic syndromes are a heterogeneous group of rare diseases characterized by eosinophil-related organ damage and peripheral blood hypereosinophilia. Hypereosinophilic syndromes may occur secondary to a variety of clinical entities, for example, drug hypersensitivity reactions, parasitic infections, autoimmune disorders, and malignancies. Toxocariasis is a parasitic infection caused by the larval stage of the Toxocara species. It is usually a childhood disease and invades organs such as the liver but can affect any organ. Hypereosinophilic syndrome and multiorgan involvement are very rare manifestations of Toxocara infection, especially in adults. However, the disease may be underdiagnosed because of different factors, including a lack of laboratory infrastructure in some countries, a lack of uniform case definitions, and limited surveillance infrastructure, with its estimation constituting a challenge. We, therefore, present a probable case of hypereosinophilic syndrome with multiorgan involvement secondary to infection with Toxocara canis in a 79-year-old Swiss female patient with a medical history of ischemic cerebrovascular insult and a curatively resected non-small-cell bronchial carcinoma, successfully treated with albendazole and steroids.
Introduction
Hypereosinophilic syndrome (HES) refers to a group of rare hematologic disorders. HES is characterized by blood and tissue eosinophilia and eosinophil-induced tissue damage with associated dysfunction. HES may occur secondary to a variety of clinical entities, for example, drug hypersensitivity reactions, parasitic infections, autoimmune disorders, and malignancies.1,2 Parasitic diseases such as helminth infections, including toxocariasis, are classically associated with eosinophilia which is usually most pronounced in the early stages of the infection, representing the tissue infiltration by the larva and then slowly decreases over time.3,4 However, eosinophilia in patients with toxocariasis can be particularly notable, with reported eosinophil counts as high as 15,000–100,000/μL, and can take years to fully resolve. Moreover, asymptomatic cases with marked eosinophilia persistent for a long period of time after ingesting a very low inoculum are also reported.4–7 Although parasitic infections represent one of the main causes of eosinophilia worldwide,2,8,9 it is a rather rare cause of eosinophilia in Switzerland. 10 We, therefore, report the case of HES in a Swiss adult patient, who presented massive eosinophilia, severe pruritic urticarial exanthema with eosinophilic infiltration, hepatitis, interstitial pneumopathy, and infiltrative cardiomyopathy due to infection with Toxocara canis. Written informed consent was obtained from the patient for publication of the details of her medical case.
Learning points
- Hypereosinophilia is usually associated with drug-associated hypersensitivity reactions, hematologic diseases, or parasitic infections.
- Toxocariasis occurs through animal contact and the consumption of contaminated vegetables and raw meat.
- Toxocariasis represents one of the most common helminth diseases in industrialized countries.
Case report
In early 2020, a 79-year-old female patient was referred by her general practitioner due to a severe pruritic urticarial rash (Figure 1), diarrhea for 2 months, dyspnea, and worsening of her general medical condition. The patient’s personal history included an ischemic cerebrovascular insult in 2010 and a curatively resected non-small-cell bronchial carcinoma also in 2010. Furthermore, the patient had a known penicillin allergy, and new medications were not taken at this time point. No recent foreign travel had been undertaken, and the environmental history was not remarkable. The patient had no pets of her own but had occasional contact with a dog belonging to an acquaintance.
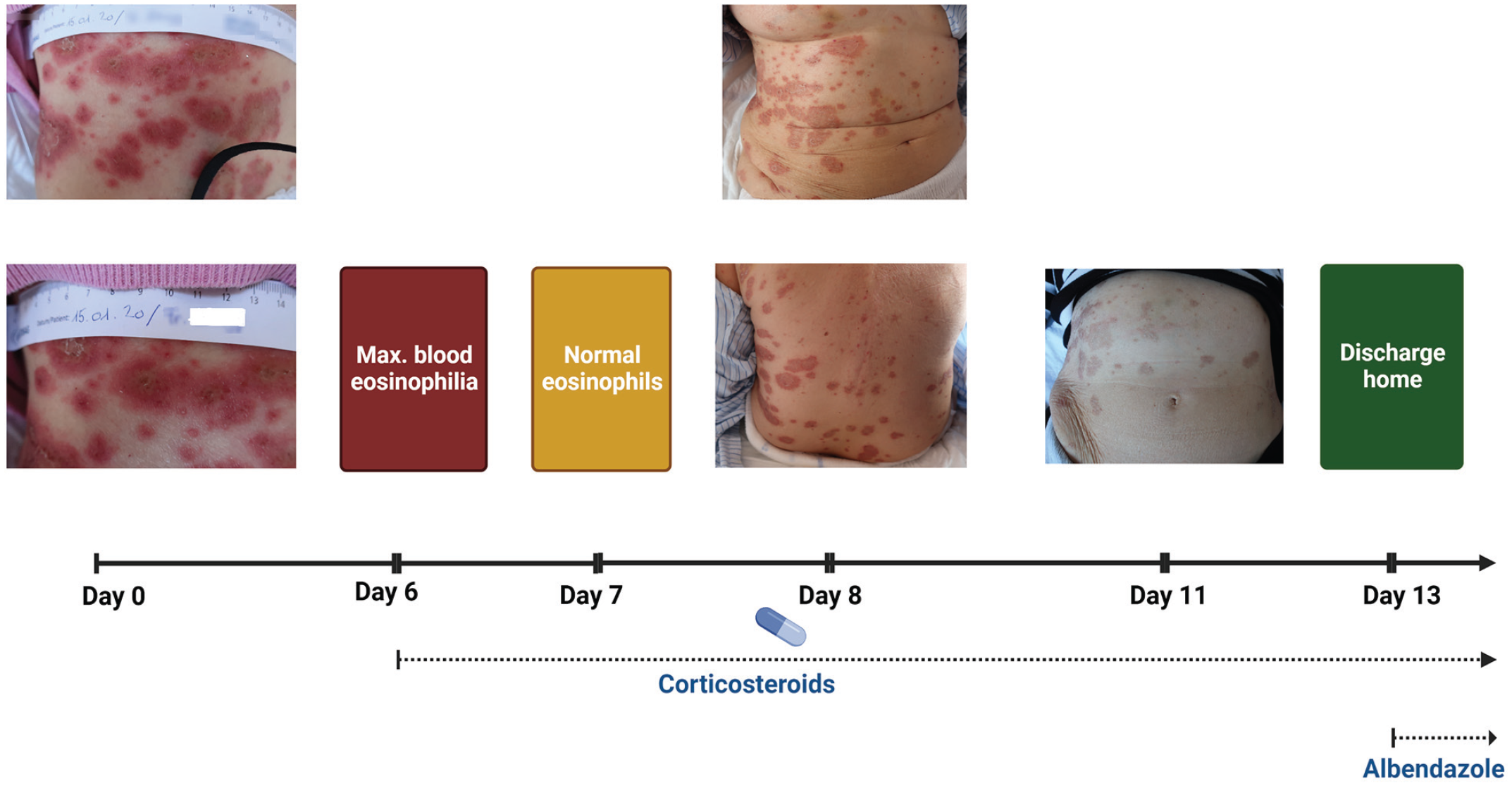
Timeline of the patient’s clinical course including clinical events and medication (Figure 1 created with BioRender.com).
Clinical examination revealed an erythematosquamous erythema on all extremities and trunks without affecting palms and soles (Figure 1). Laboratory analysis revealed leukocytosis (30.04 G/L), eosinophilia (1.11 G/L), and an elevated C-reactive protein level (182.5 mg/L) (Table 1). Chest X-ray was not suggestive of pneumonia or other pathologies. Urinalysis was not remarkable, as several blood cultures were taken without pathogen detection. An empiric antibiotic treatment with ceftriaxone for possible bacterial infection was initiated. A skin biopsy was performed.
Laboratory data on admission, hospital day 3, hospital day 5, and discharge home.

ASAT: aspartate aminotransferase; ALAT: alanine aminotransferase; GGT: gamma-glutamyl transferase; ALP: alkaline phosphatase; CRP: C-reactive protein.
Subsequently, the pruritic exanthema was progressive, and diarrhea persisted. Furthermore, there was an increasing need for oxygen, while the eosinophil count continued to rise to a maximum value of 11.79 G/L within 5 days.
Diagnostics
Extended investigations for an underlying systemic disease of autoimmune, neoplastic, or myeloproliferative type proved negative. Computed tomography (CT) of the thorax and abdomen showed interstitial alterations of the lungs—potentially of eosinophilic origin, whereas abdominal findings were not remarkable. Similarly, gastro- and colonoscopy revealed no morphological evidence of malignancy, colitis, or parasitosis. Villous relief in the small intestine was unremarkable. Histologically, there was no evidence of eosinophilic colitis. A transthoracic echocardiography revealed hypertrophic cardiomyopathy potentially in the context of eosinophilic infiltration. Serologically, there was no evidence of vasculitis or collagenosis with negative antinuclear antibodies and anti-neutrophil cytoplasmic antibodies. Skin biopsy revealed perivascular lymphoplasmacytic inflammation with an admixture of eosinophilic granulocytes.
Due to the clinical suspicion of a parasitic disease, we collected appropriate stool samples and performed serological analyses. The toxocariasis enzyme-linked immunosorbent assay (ELISA) was found positive twice. Serologies for other tissue helminth infections (i.e., trichinellosis, fasciolosis, and strongyloidiasis) and stool tests were unremarkable.
Based on the clinical picture, the presence of hypereosinophilia (HE), a strongly elevated total serum immunoglobulin (Ig) E, hepatitis, intestinal pneumopathy, infiltrative cardiomyopathy, the skin biopsy with eosinophilic infiltration, and positive serological diagnostic, we hypothesized the diagnosis of visceral toxocarosis with HES.
Therapy and course
We started a high-dose corticosteroid therapy, under which the leukocytosis, including the eosinophil count, rapidly regressed, as also did the cholestatic enzymes and the transaminases (Table 1). Alongside these observations, the patient showed a rapid clinical improvement: the erythema regressed (Figure 1), the diarrhea stopped, and oxygen supplementation could be discontinued. In addition to steroid therapy, which was tapered gradually, the Toxocara infection was treated with albendazole 400 mg bid for 5 days.
In the following controls, the patient remains asymptomatic until today (2023), while follow-up laboratory analysis showed the absence of eosinophilia.
Discussion
HES refers to a heterogeneous group of rare diseases characterized by eosinophil-related organ damage and peripheral blood HE (eosinophils > 1.5 × 109) measured at least two times within a time interval of 4 weeks. However, the assumption of the time limit is not required for the definition of HES in the case of acute establishment of organ dysfunction. Moreover, histologically evident tissue deposition of eosinophils also serves as a criterion for the diagnosis of HES. 1
The pathophysiology of eosinophil-associated tissue and organ damage is complex and cannot be only explained by isolated eosinophil accumulation. Indeed, various clinical and experimental studies indicate that pro-inflammatory and tissue-damaging properties of eosinophils considerably contribute to the pathogenesis of many eosinophil-related diseases and HESs. 11 Accumulating data additionally suggest that eosinophils have a cardinal involvement in the development of tissue remodeling and fibrosis, more specifically, through their potent elaboration of remodeling and fibrogenic growth factors, such as interleukin (IL) 3, IL-5, and granulocyte-macrophage colony-stimulating factor.12–14 The enhanced production of eosinophilopoietic cytokines leads to increased differentiation and uncontrolled proliferation, which, in turn, magnifies migration, adhesion, and activation of the eosinophils.1,11 Moreover, the intensive monoclonal proliferation of eosinophils from myeloid progenitor cells related to gene reorganization of oncogenic tyrosine kinase receptors may also play a fundamental role in the pathophysiology of HE and HES. 15
Eosinophilic endomyocardial fibrosis was first described by Loeffler in 1934, 16 and since then, the classification of HESs has had significant progress. Recently, HE and HES have been classified into six proposed, clinically defined subtypes, including familial or hereditary HE/HES, myeloid HES, lymphocytic variant HSE, overlap HES, idiopathic HES, and secondary or associated HES. The latest encompass HE/HES with a known underlying etiology requiring special management and treatment. Indeed, a variety of clinical entities (e.g., drug hypersensitivity reactions, parasitic infections, autoimmune disorders, and malignancies) are associated with secondary, nonclonal, reactive HE/HES, representing the majority of the HE/HES cases. Parasitic infections are the most frequent cause of eosinophilia worldwide, with tissue-invasive helminthic infection being the most prevalent.8,9
Upon the evaluation of eosinophilia, especially in areas with limited exposure to parasites, other secondary causes should be excluded. These include allergies and medication-related hypersensitivity reactions, representing the dominant cause of eosinophilia in these areas.4,17 However, there are many other conditions known to cause secondary eosinophilia, such as disorders of the immune system or malignancies (i.e., lung carcinoma, sarcomas, lymphomas), which should be taken into account if no obvious cause of eosinophilia is recognized. 18
Toxocariasis, a prevalent zoonotic helminth infection, is commonly observed in industrialized countries. 19 It is primarily caused by ingesting eggs from nematode parasites belonging to the Toxocara species. The dog roundworm T. canis is the predominant source of transmission, while the cat roundworm Toxocara cati contributes to a lesser extent.20,21 Seroprevalence rates of toxocariasis exhibit global variation, with tropical and subtropical regions displaying the highest prevalence, surpassing 50%. 19 For Europe, its estimated impact appears to be around 18%. Switzerland represents one of the European countries with low seropositivity, ranging between 1.7% and 6.3%, probably due to climate conditions eliminating the larval development and survival of Toxocara in the environment. 19 Transmission to humans typically occurs through the direct fecal–oral route, resulting in toxocariasis and associated complications, including allergic and neurological disorders. Ingestion of encysted third-stage larvae found in raw or undercooked meat or organs from paratenic hosts, such as sheep, cattle, and chickens, can also lead to human infection.22–25 Notably, consuming inadequately cooked or raw liver from infected animals has been linked explicitly to toxocariasis.22,23 However, it is important to note that research on Toxocara transmission to humans through food consumption remains limited, 26 emphasizing the need for a comprehensive and thorough evaluation of this mode of transmission.
The clinical spectrum of toxocariasis can be divided into four distinct disease entities depending on the clinical presentations and the affected organ and symptoms, that is, visceral larval migrans (VLM), ocular toxocarosis, neurotoxocariasis, and common or covert toxocariasis. The severity of the disease is associated with a load of parasites, the duration of infection, and the age- and immune-mediated responses of the affected individuals.
Although VLM is mainly a childhood disease, adults can also be affected. Visceral toxocarosis presents a variety of clinical symptoms, including general symptoms, for example, fever, anorexia, fatigue, weight loss, pallor, and specific symptoms associated with the affected organs. The liver is predominantly affected in VLM, and its infestation is presented with the formation of granulomatous lesions, hepatomegaly, vomiting, diarrhea, and hepatitis.27,28 Moreover, hepatic larva migrans presenting as upper gastrointestinal bleeding secondary to a hepatic artery pseudoaneurysm have also been described, but this represents an absolute scarcity.29,30 Although there is no pathognomonic feature of VLM, certain imaging findings, in combination with the clinical picture and laboratory findings, may raise suspicion of the disease. Multiple variable sized and shaped, indistinct, confluent focal lesions, eosinophilic granulomas, and abscesses are the most common imaging findings representing pathophysiologically the tissue migration and infiltration of organs by larvae.31–34 Manifestations from other organs, such as the heart and lungs, are less common and include cough, wheeze, dyspnea, bronchospasm, asthma and myocarditis, pericarditis, endocarditis, heart failure, and endocardial thrombosis, respectively.27,28 Of note, lung symptoms are observed in the vast majority of patients with VLM (approximately 80%).35,36 Moreover, VLM is associated with the development of secondary chronic eosinophilic pneumonia.35,37 Similarly to the hepatic VLM, findings on a CT scan include multifocal subpleural nodules with halo or ground-glass opacities and ill-defined lesions.35,38 Interestingly, despite that it is hypothesized that the larvae do not approach other tissues and organs, such as the liver and the lungs, through the celomic cavities after being disseminated hematogenously, several experimental studies have presented that the larvae can migrate through the tissues and celomic cavities of mice and monkeys.39–41 However, recently, and based on imaging findings, a rare case of migration of the toxocariasis larvae from the liver to the lung through a celomic cavity has been described. 42 Moreover, it is well established that patients with VLM may manifest numerous skin lesions (e.g., urticaria, chronic prurigo, pruritus, atopic dermatitis, and non-atopic eczema). 27 Laboratory findings include hyperleukocytosis, elevated immunoglobulin E, hepatic cytolysis or cholestasis, and HE. 43 Our patient presented extended urticaria, fatigue, chronic diarrhea, dyspnea, cough, hepatitis, leucocytosis with HE, intestinal pneumopathy, and hypertrophic, probably infiltrative cardiomyopathy.
In general, diagnosis is based on serological detection by T. canis ES antigen ELISA (i.e., anti-Toxocara IgG and IgE). A positive test result should be clarified for cross-reactivity by antibodies of other tissue helminth infections.28,44
Albendazole at 400 mg twice a day for 5 days is the suggested therapy for VLM. In severe infestations, a combination with prednisone to limit severe inflammatory reactions in the context of larvae death during the antiparasitic therapy dying is indicated. In cases with cardiac involvement, higher doses and longer duration of therapy are strongly recommended.28,44
Conclusion
In conclusion, we report a probable rare case of HES secondary to visceral toxocariasis in an adult Swiss patient, being successfully treated with albendazole and steroids. Indeed, although it is very difficult to reach a definitive diagnosis of toxocariasis, the resolution of HES and the fact that the patient remains asymptomatic for many years enhances the hypothesis that eosinophilia was a secondary phenomenon. Our patient illustrates that, despite toxocariasis being considered mainly a childhood disease, adult patients may also be affected. Given the associated potentially fatal complications, early clinical suspicion and diagnosis are critical.
Footnotes
Author’s contributions
The study was designed by MZ. MZ was involved in the diagnosis and management. KH, TPC, and MZ searched the articles and drafted the manuscript. All authors read and approved the final manuscript.
Data Availability
Data sharing is not applicable to this article as no data sets were generated or analyzed during the present study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Publication costs for this article were funded by the author’s institutions.
Ethics approval
The project did not meet the definition of human subject research. Ethical approval was not required for this case report in accordance with local or national guidelines. Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for publication of the details of her medical case and any accompanying images.
