Abstract
Proton pump inhibitors (PPIs) have transformed the way acid-peptic diseases are treated, offering improved patient tolerance, safety, and treatment outcomes. This article explores the critical issue of co-administering PPIs with dual-antiplatelet therapy (DAPT) for cardiac patients, particularly those at risk of gastrointestinal bleeding due to DAPT. Understanding the pharmacokinetics of individual PPIs, including omeprazole, pantoprazole, lansoprazole, and rabeprazole, is essential. Variations in hepatic metabolism can impact clinical efficacy and contribute to drug interactions. Of particular concern is the potential for PPIs to counteract clopidogrel’s antiplatelet effects, thereby influencing major adverse cardiovascular events (MACE). This review sheds light on the intricate interplay between PPIs and antiplatelet agents. It emphasizes the necessity for cautious consideration when co-prescribing PPIs to cardiac patients on DAPT. This article advances our understanding of these interactions and their implications for cardiac safety, offering valuable insights for clinicians and researchers.
Introduction
Over the years, proton pump inhibitors (PPIs) have progressively established as the primary treatment option for acid-peptic disease. Compared to the previous generation of drugs such as synthetic prostaglandin analogs, anticholinergics, and histamine-2-receptor antagonists (H2RAs), PPIs have been reported to have excellent safety, improved patient tolerance, and better outcomes in the acid-peptic disease treatment. 1 PPIs are also indicated for a subset of patients with cardiac conditions, these are patients who require dual-antiplatelet therapy (DAPT), usually comprising of aspirin and clopidogrel/ticagrelor/prasugel. Among these, the clopidogrel and aspirin combination is the most widely used. DAPT is prescribed for long-term use to prevent thrombotic complications related to acute coronary syndrome (ACS) and coronary artery disease (CAD). In patients with non-ST-segment elevation myocardial infarction (NSTEMI) or ST-segment elevation myocardial infarction (STEMI) who require oral therapy, it is also prescribed to prevent stent thrombosis following coronary artery bypass grafting (CABG) or percutaneous coronary intervention (PCI)2, 3. The rationale for co-prescribing PPIs to such patients with cardiac conditions is to prevent gastrointestinal (GI) bleeding, which is one of the commonly observed side effects of DAPT.4, 5
PPI Guidelines for Patients with Cardiovascular Issues
The 2010 Expert Consensus from the “American College of Cardiology Foundation (ACCF)/American College of Gastroenterology (ACG)/American Heart Association (AHA)” recommends that PPIs should be prescribed only to patients at high-risk of bleeding.6, 7 In 2010, ACCF foundation task force, in collaboration with ACG and AHA, created guidelines for managing GI risks associated with NSAID and antiplatelet therapy. For those with a history of upper GI bleeding and for those on antiplatelet therapy who are at higher risk of GI bleeding, they recommended PPI therapy. In contrast, all the patients receiving DAPT were advised to routinely utilize PPIs by the 2018 ESC guidelines. The ESC emphasized the importance of taking all possible measures to minimize the risk of bleeding complications during DAPT. This discrepancy in clinical recommendations is primarily due to differing interpretation of the COGENT trial, a major randomized controlled trial that revealed a pharmacologic interaction between omeprazole and clopidogrel, although it did not show a clinically significant impact on adverse cardiovascular events.6, 8, 9 The 2016 “ACCF/ACG/AHA” guidelines recommend prescribing a PPI as needed for patients on DAPT who have several risk factors for bleeding in the GI tract. However, in patients with less risk for bleeding in the upper GI tract, the use of PPIs or H2RAs has shown only marginal benefit. 7 The 2016 ACC/AHA guidelines emphasized that patients who are at high-risk should be the main recipients of PPIs. A newly developed risk assessment tool, the “DAPT score,” derived from the DAPT study, assists in deciding whether to extend, continue, or prolong DAPT in patients who have undergone coronary stent implantation. “A score of ≥2 indicates a favorable benefit/risk ratio for prolonged DAPT, whereas a score of <2 suggests an unfavorable benefit/risk ratio.” 3 However, the ESC highlighted the increased morbidity and mortality linked to bleeding events following successful PCI, and thus recommended prescribing PPIs to all patients on DAPT. 8 Additionally, for patients on single or DAPT for cardiovascular prophylaxis with a history of ulcer bleeding, PPI therapy was recommended in the 2020 International Consensus guidelines for treating non-variceal upper gastrointestinal bleeding. 10 However, since there are not enough randomized controlled trials (RCTs), this was only recommended as a “conditional recommendation” based on “low-quality evidence.” These recommendations also took into account the possible higher risk of death or myocardial infarction (MI) linked to using PPIs and DAPT at the same time. 10 If GI risk factors exist, the “Italian Association of Hospital Gastroenterologists (AIGO) and the National Association of Hospital Cardiologists” (ANMCO) recommend prescribing PPIs to patients on a single antiplatelet agent. 11 They suggest that patients with history of peptic ulcer disease who are on antiplatelet therapy, and concurrently using another “antiplatelet agent, vitamin K antagonists (VKAs), direct-acting oral anticoagulants (DOACs), non-steroidal anti-inflammatory drugs (NSAIDs), or steroids,” should be taken into account when using PPIs because of the higher risk of bleeding. Having GERD, being over 65, and experiencing dyspeptic symptoms are additional risk factors.
Pharmacokinetics of Individual PPIs and their Metabolic Differences
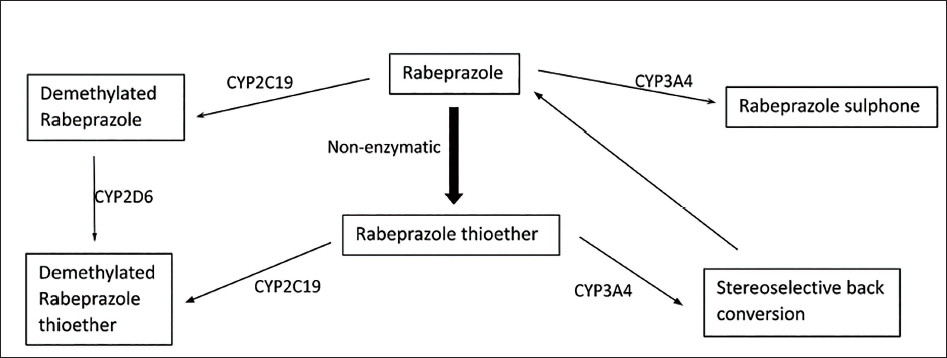
The main pharmacokinetic parameters of different drugs in the class of PPIs like omeprazole, pantoprazole, lansoprazole, and rabeprazole, are not significantly different. Variations in the hepatic metabolism of these drugs can cause interpatient variability in acid suppression, increased possibility of pharmacokinetic drug interactions, and, in turn, in clinical efficacy. Pantoprazole, omeprazole, and lansoprazole are significantly metabolized by several CYPs, most predominately by CYP3A4 and CYP2C19.12, 13 omeprazole is primarily metabolized through the formation of 5-hydroxyomeprazole via CYP2C19. Additionally, it is converted to omeprazole sulphone by CYP3A4. These are further metabolized to omeprazole hydroxy sulphone. Even though CYP3A4 catalyzes the reaction to form the omeprazole sulphone, omeprazole has a 10-fold lower affinity for CYP3A4 compared to CYP2C19. Consequently, omeprazole is more likely to interact with and affect the metabolism of substrates for CYP2C19 than those for CYP3A4. 14 Similarly, CYP3A4 and CYP2C19 are the main enzymes that break down lansoprazole to form 5-hydroxy-lansoprazole and lansoprazole sulfone, respectively. The major metabolite is lansoprazole sulfone. CYP2C19 primarily metabolizes pantoprazole via O-demethylation, which is followed by sulfate conjugation to yield pantoprazole sulfone, which is the main metabolite in the plasma. Pantoprazole sulfone is produced by CYP3A4, following metabolic pathways similar to those of lansoprazole and omeprazole. 15 All PPIs are significantly metabolized in the liver. The cytochrome P450 (CYP) isoenzyme CYP2C19, which is involved in PPI metabolism, has significant genetic polymorphism, and it has been demonstrated that this polymorphism significantly increases plasma levels of pantoprazole, lansoprazole, and Omeprazole but not of Rabeprazole. Despite having a higher degree of structural similarity with other PPIs in its class, rabeprazole distinguishes itself. 16 Despite being a substituted benzimidazole sulfoxide, rabeprazole has a very different metabolic profile. It is primarily metabolized with only minor involvement of CYP3A4 and CYP2C19 through non-enzymatic reduction to a thioether compound.12, 13 Thus, CYP2C19 plays a significant role in determining the pharmacokinetic profiles of pantoprazole, omeprazole, and lansoprazole, but has a lesser impact on that of rabeprazole.
Drug Interactions of PPIs
Studies have reported data, backed with strong evidence about the interaction between antiplatelet agents and PPIs. The risk of reinfarction increased with the use of PPIs in a population-based study on the interactions between PPI and clopidogrel treatment. 17 According to meta-analyses published in 2019, taking individual PPIs like omeprazole, esomeprazole, pantoprazole, and lansoprazole with or without concurrent clopidogrel increases the chance of cardiovascular disease (CV) by a significant amount. 18 Kwok et al. found, in a subgroup meta-analysis of observational studies that PPIs were associated with increased cardiovascular risk, even independent of clopidogrel. 19 In addition, Pang et al. in a comprehensive review and meta-analysis of 15 RCTs including 50,366 patients reported that the risk of MI recurrence, stent thrombosis, MACE, stroke, and target vessel revascularization (TVR) was reduced for individuals in the clopidogrel-treated non-PPI group than the PPI with clopidogrel group, though the two groups had similar cardiovascular death, bleeding events, and all-cause death. 20
Risk of MACE with Individual PPIs
The results from trials on the risk of major adverse cardiovascular events (MACE) with the combined use of DAPT and PPIs have been controversial. Several extensive clinical studies, such as ADAPT-DES, and trials like BASKET and CAPRIE, suggested that the combined use of PPIs and clopidogrel was linked to a higher incidence of MACE.21–23 However, other large-scale prospective studies, including PRINCIPLE-TIMI 44 and PRODIGY, as well as trials like COGENT, CREDO, and TRITON-TIMI 38, presented contrasting results. These studies found that using PPIs and clopidogrel together did not elevate the risk of NACE, and instead, was associated with a reduced risk of gastrointestinal bleeding.9, 24, 25 In 2019, a randomized controlled trial was conducted by Moayyedi et al. with 17,598 patients who had peripheral artery disease and stable CV to evaluate the effects of PPIs. Of the total participants, 8,791 were assigned to the PPI group. The patients were split into three treatment groups: rivaroxaban alone (5 mg twice daily), aspirin alone (100 mg), or rivaroxaban (2.5 mg twice daily) combined with aspirin. Participants were monitored for three years. The analysis found no difference that is statistically significant in cardiovascular outcomes—MI, stroke, or cardiovascular death—between the PPI and placebo groups. The PPI most studied for interaction with DAPT is omeprazole, and has been shown to have an inhibitory effect on clopidogrel in several studies.26–28 The evidence about other PPIs concerning their interaction with clopidogrel is inconsistent. In a meta-analysis including 21 studies, Niu et al. found that those with CAD who concurrently use clopidogrel and specific PPIs, including omeprazole, lansoprazole, esomeprazole, and pantoprazole, had a higher risk of developing MACE (OR: 1.42; 95% CI: 1.30-1.55). In contrast, three studies investigating rabeprazole showed no major heterogeneity and no significant increase in risk (OR: 1.03; 95% CI: 0.55-1.95), indicating that rabeprazole may be safer compared to other PPIs. 29 However, an analysis of observational studies by Guo et al. found an irregular relationship between the usage of different PPI subtypes and an elevated risk of MACEs. 30 Yasu et al. evaluated the safety and efficacy of rabeprazole in patients undergoing drug-eluting stent (DES) implantation-related DAPT with clopidogrel and aspirin, comparing them to patients receiving DAPT alone. The study found that patients in the rabeprazole group had more severe coronary artery lesions, necessitating a greater number of DES. Nevertheless, there was no correlation between co-administration of rabeprazole and a higher risk of stent thrombosis. The incidence of MACE was not increased by rabeprazole usage. 31 Similarly, in a meta-analysis of 28 studies involving 131,412 patients, Lee et al. reported that patients on pantoprazole or lansoprazole had a higher risk of MACE than those not using PPIs. The pooled HR for adjusted MACE events indicated that the increased risk was consistent across four classes of PPIs, except for rabeprazole. Notably, the only PPI that did not markedly raise the risk of MACE was rabeprazole. 32 Among patients aged more than 60 years undergoing PCI and administered DAPT, those on rabeprazole experienced a considerably less severe upper gastrointestinal hemorrhage than those taking only DAPT, and there were no significant differences in the incidence of MACEs between the groups. PPIs are primarily metabolized by CYP2C19, but the degree of enzyme inhibition varies across different PPIs. They are categorized according to the affinity they have for CYP2C19; some have notable inhibitory potential, while others have low capacity. Rabeprazole is metabolized through both “CYP-mediated and non-CYP-mediated” processes, with non-CYP-mediated metabolism being predominant. Rabeprazole has less of an impact on the metabolism of clopidogrel than other PPIs and is a less potent competitive inhibitor of CYP2C19. Additionally, while taking most PPIs after a meal, the plasma concentrations drop by up to 40%, with the exception of rabeprazole. PPIs like omeprazole, lansoprazole, esomeprazole, and pantoprazole are primarily metabolized through the enzymatic pathway in the liver by CYP2C19 and CYP3A4.3, 33 Since PPIs lead to significant and prolonged elevation of gastric pH, they can interfere with the absorption of concurrent medications. Further, PPIs can weaken the antiplatelet effects of clopidogrel by competing for CYP2C19 and interfering with the formation of the active metabolite of clopidogrel. Multiple studies have shown that the combination of a PPI with clopidogrel reduces the ability of clopidogrel to prevent cardiovascular events.
A 12-month study assessed the risk of hospitalization for MACE—“defined as myocardial infarction, unstable angina, stroke or transient ischemic attack, urgent TVR, or cardiovascular death”—in participants taking clopidogrel alongside a PPI. The risk associated with omeprazole was 39% higher (p < .0001), the risk associated with esomeprazole was 57% higher (p < .0001), the risk associated with pantoprazole was 61% higher (p < .0001), and the risk associated with lansoprazole was 39% higher (p < .0004). Niu et al. study revealed significant (OR: 1.42; 95% CI: 1.30-1.55) increase in the risk of MACE occurrence. However, a subgroup analysis assessing the effect of individual PPIs reported that omeprazole (OR: 1.40), lansoprazole (OR: 1.51), esomeprazole (OR: 1.59), and pantoprazole (OR: 1.52) showed a significantly higher risk of MACE, but, rabeprazole (OR: 1.03) proved to be the only PPI with no significant increase in MACE risk when combined with clopidogrel in the management of CAD as it is predominantly metabolized through the non-enzymatic pathway to rabeprazole thioether, and to demethylated rabeprazole and rabeprazole sulphone as shown below.
Unique metabolic Pathway of Rabeprazole
Rabeprazole has a distinct catabolic pathway, which makes it less susceptible to the effects of CYP2C19 genetic variations. Ieiri et al. studied the effect of CYP2C19 genetic polymorphic metabolism on the disposition of rabeprazole (10 mg), following dosing for 8 days. The results revealed that none of the pharmacokinetic parameters of rabeprazole were affected by metabolism in homozygous or heterozygous genotypes. The increase in the AUC for rabeprazole on Day 8 compared to Day 1 was within a two-fold range, even in poor metabolizer genotypes. Overall, clinical evidence suggests that rabeprazole exposure is less affected by genetic or drug-induced changes in metabolism compared to other PPIs.
Activation by cytochrome P450 isoenzymes is necessary for clopidogrel, the most frequently employed prodrug in DAPT. 31 Previous research has demonstrated that CYP2C19 and CYP3A4 are essential for the metabolic activation of clopidogrel. 4 Consequently, clopidogrel and PPIs can compete for the metabolism of CYP3A4, CYP450 enzyme families, and CYP2C19 raising concerns that PPIs might reduce clopidogrel’s antiplatelet effect and potentially increase the risk of MACE. 5 Additionally, genetic polymorphisms have been identified that may influence the response to clopidogreland, in theory, heighten the likelihood of CYP450-mediated drug interactions. 9 Moreover, the release of other drugs from their dosage forms, including aspirin, which is a crucial component of DAPT, may be impacted by the increased gastric pH caused by PPIs. This alteration can affect the drug’s absorption and metabolism. 34 Concerns arose about the potential interaction between PPIs and clopidogrel, particularly regarding the possibility that PPIs that inhibit the CYP2C19 pathway may be specifically affected by this interaction. 35 rabeprazole is metabolized in two ways: “CYPmediated and non-CYP-mediated,” with the latter taking precedence. 32 Its metabolism is primarily non-enzymatic, with metabolites being eliminated through the kidneys. CYP3A4 and CYP2C19 contribute only to a small portion of its enzymatic metabolism. 36 As a result, in comparison to other PPIs, rabeprazole is a weaker inhibitor of CYP2C19 and has minimal effect on clopidogrel’s metabolic pathway. 32
Hence, for patients with genetic polymorphism of CYP2C19, and patients who are drugs whose metabolism depends on CYP450 isoenzymes, especially through CYP3A4 and CYP2C19, rabeprazole may be among the safest PPIs. Therefore, based on evidence, if clopidogrel and PPIs do interact with each other, it appears to be agent-specific rather than “class-specific.” 37
Conclusion
PPIs have become the standard treatment for acid reflux over the years. The goal of co-prescribing PPIs to such cardiac patients is to decrease gastrointestinal bleeding, which is a typical adverse effect of DAPT. Pantoprazole, omeprazole, and lansoprazole are extensively metabolized in the liver by several CYPs, most notably CYP3A4 and CYP2C19. Despite having a higher degree of structural similarity with other PPIs in its class, rabeprazole distinguishes itself. A prior study demonstrated that CYP2C19 and CYP3A4 are crucial for the metabolic activation of clopidogrel. Concerns have been raised that PPI could hinder clopidogrel’s antiplatelet activity, raising the risk of MACE. Since rabeprazole is not as effective as the other PPIs as a competitive inhibitor of CYP2C19, no significant increase in risk and no significant heterogeneity was found. Thus, rabeprazole is safer than other PPIs and did not raise the risk of MACE, demonstrating its cardiovascular safety.
Footnotes
Acknowledgement
Assistance for conducting the research, developing the model and drafting the manuscript was provided by Shalaka Marfatia of pharmEDGE and her team.
Authors Contribution
J.D, K.P: Prepared and reviewed the manuscript. S.D: Prepared, reviewed, and edited the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The paper does not involve animal and human studies, so no ethical clearance was obtained.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
This paper does not involve human studies, so patient written informed consent was not obtained.
