Abstract
Aims
Colchicine is effective for secondary prevention of major adverse cardiovascular events (MACE), yet its benefit in individuals with diabetes remains uncertain despite their elevated cardiovascular risk. This systematic review and meta-analysis assessed the impact of colchicine on cardiovascular outcomes in individuals with diabetes.
Methods
Databases were searched from inception to November 2024 for randomized controlled trials assessing colchicine for reducing MACE. A meta-analysis was performed using RevMan 5.4 with a fixed-effects inverse-variance model to generate pooled risk ratios (RR, HR). PROSPERO registration: CRD42024575366.
Results
Five RCTs were included, comprising 2977 participants with diabetes (1492 (50%) colchicine; 1485 (50%) placebo/usual care). Most were males over 60 years. Colchicine significantly reduced MACE, HR 0.79 (95% CI 0.67–0.94, p = 0.006). Subgroup analysis of diabetic versus non-diabetic participants yielded a similar effect, HR 0.75 (95% CI 0.67–0.83, p < 0.001). Colchicine did not increase adverse events: composite events RR 0.81 (95% CI 0.68–0.96; p = 0.02), gastrointestinal events RR 1.09 (95% CI 0.98–1.20, p = 0.12), or musculoskeletal events RR 0.77 (95% CI 0.43–1.38, p = 0.39). Benefits were observed with 0.5 mg daily over a median 28.6 months.
Conclusion
Colchicine is associated with a reduction in major cardiovascular events in individuals with diabetes, with consistent long-term benefit at low doses without an increase in adverse effects.
Keywords
Introduction
Non-communicable diseases are a growing global health challenge. An estimated 830 million people globally live with diabetes, predominantly in resource-limited settings, resulting in a substantial burden on global healthcare. 1
Individuals with diabetes are at heightened risk of adverse cardiovascular events, emphasizing the need for effective, accessible interventions to reduce this burden. 2
Colchicine is a widely used, long-established, low-cost anti-inflammatory medication. 3 Its anti-inflammatory activity stems from inhibition of microtubule polymerization, which disrupts neutrophil activation, migration, and inflammasome-mediated interleukin-1β production. 4 This anti-inflammatory action has been used to treat gout and pericarditis. 5
Beyond its conventional indications, emerging evidence suggests that colchicine exerts cardioprotective effects by mitigating inflammation, a key driver of atherosclerosis and thrombosis. 6 This anti-inflammatory mechanism of action may be particularly beneficial for individuals with diabetes, who often have elevated inflammatory markers and oxidative stress. 6
This systematic review and meta-analysis aim to evaluate the impact of colchicine on cardiovascular outcomes in persons with diabetes.
Methods
The review followed the Cochrane Handbook for Systematic Reviews of Interventions and adhered to PRISMA reporting guidelines. 7 A pre-specified protocol was registered in PROSPERO (CRD42024575366).
Inclusion and exclusion criteria
Randomised controlled trials (RCTs) enrolling individuals with diabetes, without age restrictions, were included. Studies were eligible if they focused exclusively on diabetic populations or reported separate outcomes for diabetic subgroups within mixed cohorts. Trials were excluded if they did not involve diabetic participants, failed to report cardiovascular outcomes, lacked a control group, were not in English, or provided insufficient methodological information.
The intervention of interest was low-dose, long-term colchicine therapy (>6 months), used as an adjunct to standard care for the prevention of cardiovascular events. Eligible trials evaluated colchicine alone or in combination with other pharmacological or non-pharmacological strategies. Comparators included placebo, standard therapy, or usual care without colchicine. Studies not assessing colchicine-based interventions were excluded.
The primary outcome was major adverse cardiovascular events (MACE), including myocardial infarction, ischaemic stroke, acute coronary syndrome, coronary revascularisation, and cardiovascular death. Secondary outcomes were adverse drug reactions. N-of-1 trials, non-randomised and pseudo-randomised studies, reviews, editorials, protocols, and qualitative research were excluded.
Data collection and analysis
A search strategy was developed by identifying relevant subject headings (e.g., MeSH terms) and keywords. Two reviewers independently extracted data and assessed study eligibility using Covidence software, with discrepancies resolved by consensus or third-reviewer adjudication. Extracted data were transferred to Review Manager for analysis. 8 Risk of bias was evaluated independently by two reviewers using Cochrane criteria for randomized trials, with disagreements resolved through discussion or supervisor input.
Measures of treatment effect
Quantitative effect measures (hazard ratios and risk ratios) were analysed, with data extracted separately for diabetic and non-diabetic subgroups. Of six trials identified, five were included in the meta-analysis; one was excluded because it reported odds ratios, which were incompatible for pooling. Adverse event data were pooled according to availability across the included studies. The risk of bias was assessed using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions, “Assessing the risk of bias in a randomized trial.” 9
Statistical analysis
The analysis was conducted using Review Manager, version 5.4, 8 and SPSS, version 27. 10 Effect estimates were analysed using hazard ratios (HRs) and risk ratios (RRs), as appropriate. HRs were log-transformed and pooled using the generic inverse-variance method. RRs were combined using a fixed-effects model with inverse-variance weighting. Publication bias was assessed using funnel plots.
Subgroup and sensitivity analysis
Subgroup analyses compared effects between diabetic and non-diabetic participants using reported subgroup-specific estimates. Sensitivity analyses were performed for composite adverse events by excluding studies at high risk of bias to evaluate the robustness of pooled results.
Results
The five trials included in the meta-analysis involved 14,664 participants. The majority (>60%) of participants were male, and the mean age ranged from 60 to 67 years across the trials.
Characteristics of the included randomised controlled trials.

RCT = randomised controlled trial. The table summarises trial sample size, sex distribution, sample size of participants with diabetes mellitus, key inclusion criteria, comparator groups, mean age (years), duration of participant follow-up, and colchicine dose and frequency.
The COLCOT and LoDoCo2 trials reported subgroup analyses including participants with type 2 diabetes.11,12 The Deftereos et al. trial included exclusively participants with type 2 diabetes, 13 whereas the remaining studies did not specify the type of diabetes.
Meta-analysis in persons with diabetes
The pooled hazard ratio (HR) was 0.79 (95% confidence interval [CI] 0.67–0.94; p = 0.006), indicating that colchicine significantly reduced the incidence of cardiovascular events (cardiovascular death, myocardial infarction, stroke, and coronary revascularisation) among individuals with diabetes compared with control over time. Heterogeneity was low (I2 = 7%) (Figure 1). Forest plot of the meta-analysis in individuals with diabetes. Overall, five studies were included. The hazard ratios of trials were pooled using fixed-effects model. Vertical line at HR = 1 representing no effect, squares for individual study HRs, and a diamond for the pooled HR. I2 is the percentage of total variation across studies due to heterogeneity. CI, confidence interval.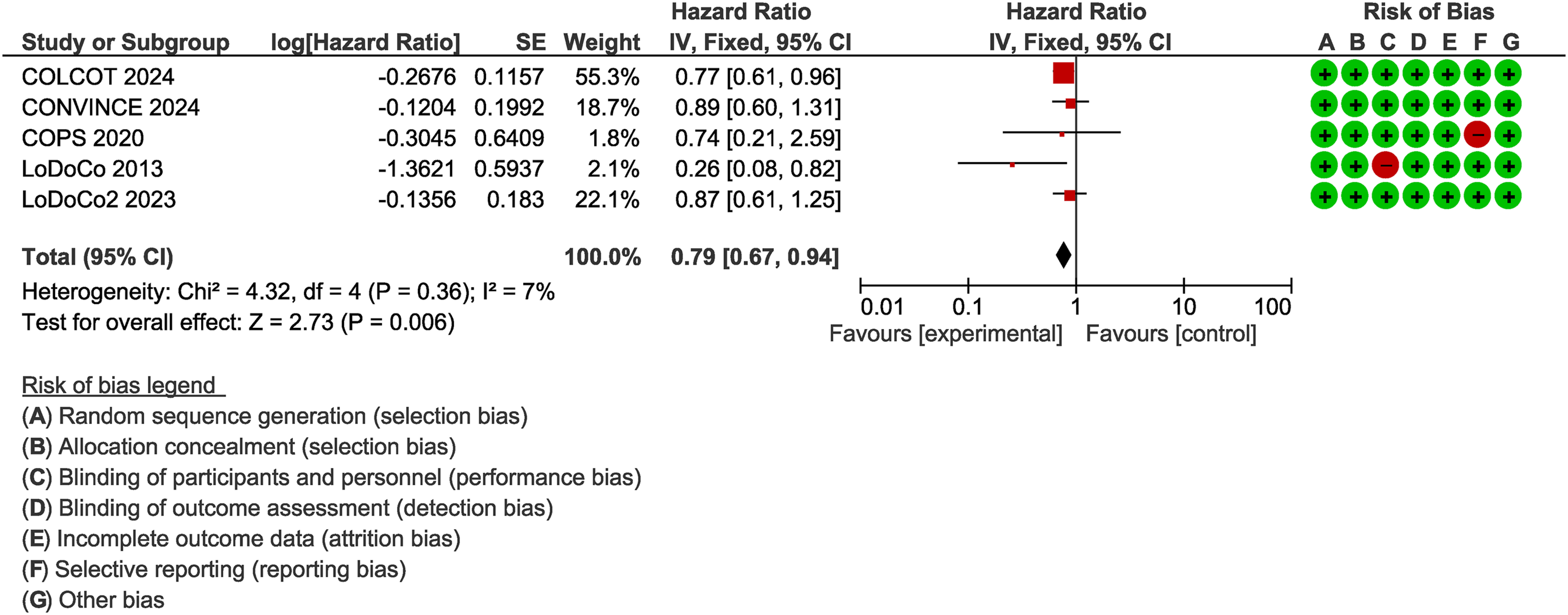
Subgroup analysis
Subgroup analysis of diabetic and non-diabetic participants showed a pooled HR of 0.72 (95% CI 0.63–0.83; p < 0.001) in the non-diabetic subgroup. The overall pooled HR across subgroups was 0.75 (95% CI 0.67–0.83; p < 0.001).
Risk of bias
Figure 1 presents the risk-of-bias assessment of the included studies. One RCT was open-label and therefore judged to have a high risk of performance bias due to lack of blinding. The COPS 2020 trial demonstrated an increased risk of detection bias. 14
Adverse events
Gastrointestinal adverse events (RR 1.09, 95% CI 0.98–1.20; p = 0.12) and musculoskeletal adverse events (RR 0.77, 95% CI 0.43–1.38; p = 0.39) were not significantly increased in the colchicine group compared with placebo or usual care.
Sensitivity analysis was performed excluding two trials with a high risk of bias (COPS 2020 and LoDoCo 2013).14,15 Composite adverse events were significantly reduced compared with placebo or usual care (RR 0.81, 95% CI 0.68–0.96; p = 0.02), indicating a lower relative risk of composite adverse events with colchicine treatment.
Overall, pooled analyses of composite adverse events, gastrointestinal adverse events, and musculoskeletal adverse events did not demonstrate a significant increase in risk associated with colchicine compared with control. Publication bias appeared minimal, as studies were symmetrically distributed in the funnel plot.
(Refer Supplemental file for forest plots).
Discussion
The evidence from the available research has generated both support for and opposition to the relationship between colchicine use and diabetes in secondary prevention of major adverse cardiovascular events (MACE). Our findings indicate that low-dose colchicine is equally effective in preventing cardiovascular events in both non-diabetic and diabetic individuals, suggesting that diabetes status does not significantly modify the relationship between colchicine and MACE risk. The present findings are consistent with two meta-analyses evaluating the relationship between colchicine and coronary heart disease risk, which reported that colchicine reduces the risk of MACE in patients with or without diabetes or hypertension.16,17 However, some studies suggest that colchicine may confer greater cardiovascular benefit in diabetic populations than in non-diabetic groups, supporting the anti-inflammatory properties of the drug in the context of diabetes-associated inflammation. A meta-analysis reported that the absolute risk reduction with colchicine was notably greater in individuals with diabetes than in those without diabetes. 18 The COLCOT trial findings further suggest that colchicine may provide enhanced cardiovascular protection in individuals with diabetes compared with non-diabetic participants, supporting the hypothesis that its anti-inflammatory effects are particularly beneficial in states of heightened inflammation. 11 These findings are especially relevant for individuals with diabetes, who often exhibit elevated inflammatory activity contributing to increased cardiovascular risk. 11 Importantly, no significant adverse effects related to diabetes status or glycaemic control were reported, reinforcing the safety of colchicine in diabetic populations.
LoDoCo2 trial demonstrated that patients with chronic coronary artery disease and type 2 diabetes mellitus have a higher risk of major adverse cardiovascular events than those without diabetes. Nevertheless, colchicine consistently reduced recurrent MACE irrespective of diabetic status. The incidence of new-onset type 2 diabetes mellitus was numerically lower in the colchicine group. 12
Although colchicine has been widely used for various inflammatory conditions, its use has been associated with adverse events, most commonly gastrointestinal symptoms. 19 Clinical trials indicate that serious adverse effects are rare and are typically associated with overdose. 20 In the present study, pooled analyses showed no significant increase in adverse effects at a low colchicine dose of 0.5 mg. Similarly, a meta-analysis of randomised controlled trials confirmed that low-dose colchicine has a favourable safety profile. 19
Treatment duration is an important consideration, as colchicine is often prescribed long term. In the included trials, colchicine was administered for more than 6 months, with a median treatment duration of 23 months. These findings are supported by previous meta-analytic evidence demonstrating that the risk of gastrointestinal adverse events and treatment discontinuation is not significantly increased when colchicine is used at low doses (0.5 mg daily) or for longer durations (>6 months). 19
Limitations
Separate analyses by diabetes type, duration, level of glycaemic control, or individual MACE components could not be performed due to limited data availability. In addition, adverse event profiles were not reported specifically for diabetic participants and were instead presented for overall trial populations. The limited number of studies focusing exclusively on diabetic cohorts restricted a more detailed evaluation of colchicine’s cardiovascular effects in this population, underscoring the need for further research in more diverse and representative cohorts.
Conclusions
Colchicine is associated with a reduced risk of major adverse cardiovascular events in individuals with diabetes. This benefit appears consistent irrespective of diabetes status, with comparable effects observed in non-diabetic populations. These effects were observed at low doses with sustained long-term treatment and without a significant increase in adverse events.
Supplemental material
Supplemental Material - Effect of colchicine for secondary prevention of cardiovascular diseases in individuals with diabetes: A meta-analysis of randomized trials
Supplemental Material for Effect of colchicine for secondary prevention of cardiovascular diseases in individuals with diabetes: A meta-analysis of randomized trials by Niludi Ranwanee Yasaratna, Danushi Wijekoon, Jean-Claude Tardif and Christopher Reid in Diabetes & Vascular Disease Research
Footnotes
Author note
Presentations: Poster presentation at 73rd Annual Scientific Meeting of the Cardiac Society of Australia & New Zealand (CSANZ) 2025.
Acknowledgements
We acknowledge all the participants and investigators of the included trials.
Ethical considerations
Ethics approval was not required as this study is a systematic review and meta-analysis of previously published data.
Author contributions
Two reviewers independently extracted data from the included studies and cross-checked their extracted data to ensure accuracy and consistency. Discrepancies in data extraction were resolved through discussion among the two reviewers, and the consensus was made by the third reviewer under the guidance of the supervisor.
Funding
The authors received financial support for the publication of this article from the Monash University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data analysed during this study are included within the published articles identified in the systematic review. Additional data are available from the corresponding author upon reasonable request.
Registration
This review was prospectively registered in PROSPERO (CRD42024575366).
Supplemental material
Supplemental material for this article is available online.
