Abstract
Pigmented lesions of the oral mucosa encompass several benign and malignant conditions that may be a matter of concern under both clinical and histopathological views. We reported a case of a 62-year-old woman, presenting with an asymptomatic, deeply pigmented lesion on the soft palate. On examination, it appeared asymmetrical, with irregular borders and an area of ulceration. A biopsy, taken to rule out melanoma, revealed a pigmented carcinoma in situ. Throughout the tumor thickness, numerous interspersed melanocytes were found that did not extend to neighboring epithelium. These were large, richly dendritic, and presented abundance of melanin granules and small nuclei. Mild melanin incontinence was found. Scanty transfer of pigment to dysplastic epithelial cells was found through Fontana Masson staining. On immunohistochemical analyses, there were pancytokeratin-stained tumor epithelial cells; increased cell proliferation throughout the entire thickness of the tumor was emphasized by Ki-67 immunomarking. P16 was negative. The dendritic cells were selectively stained for S-100, HMB45 and Melan A. Wide spectrum in situ hybridization for human papillomavirus (HPV) was negative. Unfortunately, following diagnosis, the patient refused any treatment option. Pigmented squamous cell carcinoma with melanocyte colonization must be taken into account in the differential diagnosis of pigmented lesions of the oral cavity.
Introduction
Carcinoma in situ is characterized by full cytological and architectural epithelial dysplastic changes, but without breach in the basement membrane. 1 According to the World Health Organization (WHO), carcinoma in situ in the oral cavity is considered synonymous with severe dysplasia. Squamous cell carcinoma (SCC) is the most common malignant neoplasm of the oral cavity and oropharynx. 2 Carcinoma in situ and SCC show the same etiology and clinical features. They occur predominantly in alcohol and tobacco-using adults in their fifth and sixth decades of life. Clinically, they can present in a diverse manner, including red or mixed red and white lesions, white plaques, ulcers with indurated raised margins, and exophytic, ulcero-proliferative, or verrucous growths. 2
Melanocytes are normally present in the basal layer of the mucosa and may proliferate in reactive, benign, or malignant lesions. The most common types of pigmented lesions found in the oral cavity include melanotic macule, melanoacanthoma, pigmented nevi, and melanoma.2,3 Melanin pigmentation in other entities is infrequent. One rare lesion, in which this association has been described, is pigmented squamous cell carcinoma (PSCC).4-7 The present report describes the clinical and histopathologic features of a case of pigmented carcinoma in situ of the oral cavity.
Case Report
An otherwise healthy edentulous 62-year-old Brazilian woman sought a dental school outpatient clinic for prosthetic treatment. Intraoral examination revealed a 40 mm × 20 mm painless, asymmetric, darkly pigmented macule on the soft palate, with irregular borders and focal ulceration (Figure 1A). The patient reported no prior knowledge of the lesion. There were no palpable cervical lymph nodes. The clinical diagnosis was melanoma; hence, an intralesional biopsy was performed (Figure 1B).
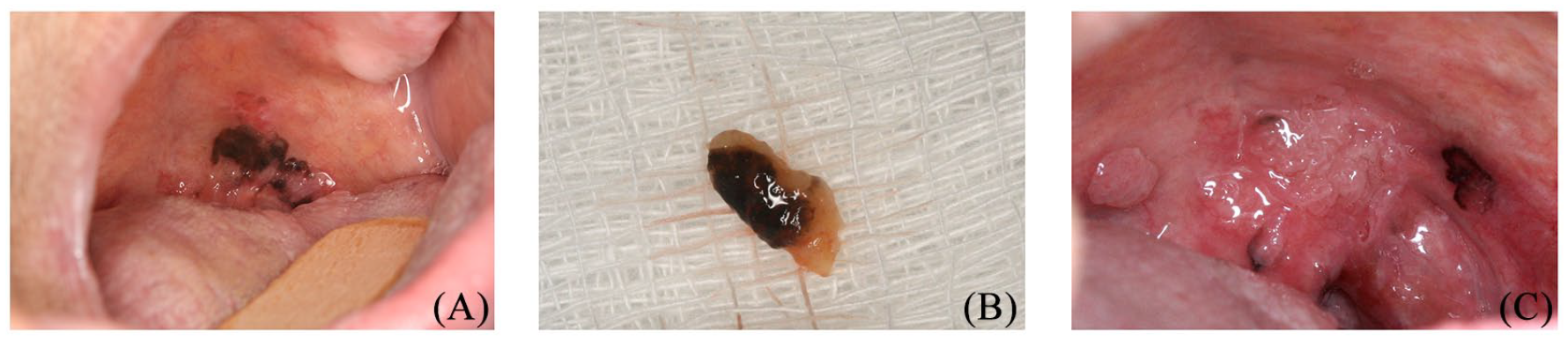
(A) Oral examination at presentation: dark pigmented macule of the soft palate associated with an area of ulceration. (B) Biopsy specimen macroscopic view. (C) After 1 year, the lesion had increased in size.
The histopathological features were full-thickness epithelium atypia and a minimal keratinization tendency toward the surface (Figure 2A to C). Epithelial cells presented relatively scant cytoplasm, high nuclear-to-cytoplasmic ratio, larger and dark nuclei with unapparent nucleoli, and scattered mitoses. Several basal and suprabasal melanocytes were interspersed throughout the lesion. These exhibited large, richly dendritic cytoplasm, full of melanin granules and small nuclei, without atypia. The melanocytes were distributed in a uniform way and did not extend to neighboring epithelium. Fontana Masson staining revealed little transfer of pigment to dysplastic epithelial cells (Figure 2F). Mild melanin incontinence was found. Immunohistochemical staining within the tumor epithelial cells was positive for pancytokeratin (Figure 2E). Increased epithelial cells proliferation throughout the entire tumor thickness was highlighted by Ki-67 immunomarking (Figure 2D). The dendritic cells were selectively stained for S-100, HMB-45, and Melan-A (Figure 2G and H). The details of the stain and antibodies used for immunohistochemistry are in Table 1. Investigation for human papillomavirus (HPV) has been performed by immunohistochemical staining for p-16 and wide spectrum in situ hybridization (ISH) for HPV, covering 6, 11, 16, 18, 31, 33, 35, 45, 51, and 52 genotypes (Dako, GenPoint HPV, Biotinylated DNA Probe, Code Y1443). Both tests were negative for HPV. The diagnosis of pigmented carcinoma in situ with benign melanocytes colonization was rendered.

Histopathology of the pigmented carcinoma in situ. (A-C) In situ carcinoma with several dendritic melanocytes interspersed throughout the tumor (H&E stain, original magnification ×400, respectively). (D) Increased cell proliferation in the entire epithelial thickness (immunohistochemistry, Ki-67, original magnification ×100). (E) Tumor epithelial cells were positive for pancytokeratin (original magnification ×100). (F) Fontana Masson staining revealed little transfer of pigment to dysplastic epithelial cells (original magnification ×100). (G) and (H) Single dendritic melanocytes are immunostained, respectively, for Melan A and HMB-45 (original magnification ×400).
Details of the antibodies used for immunohistochemistry.
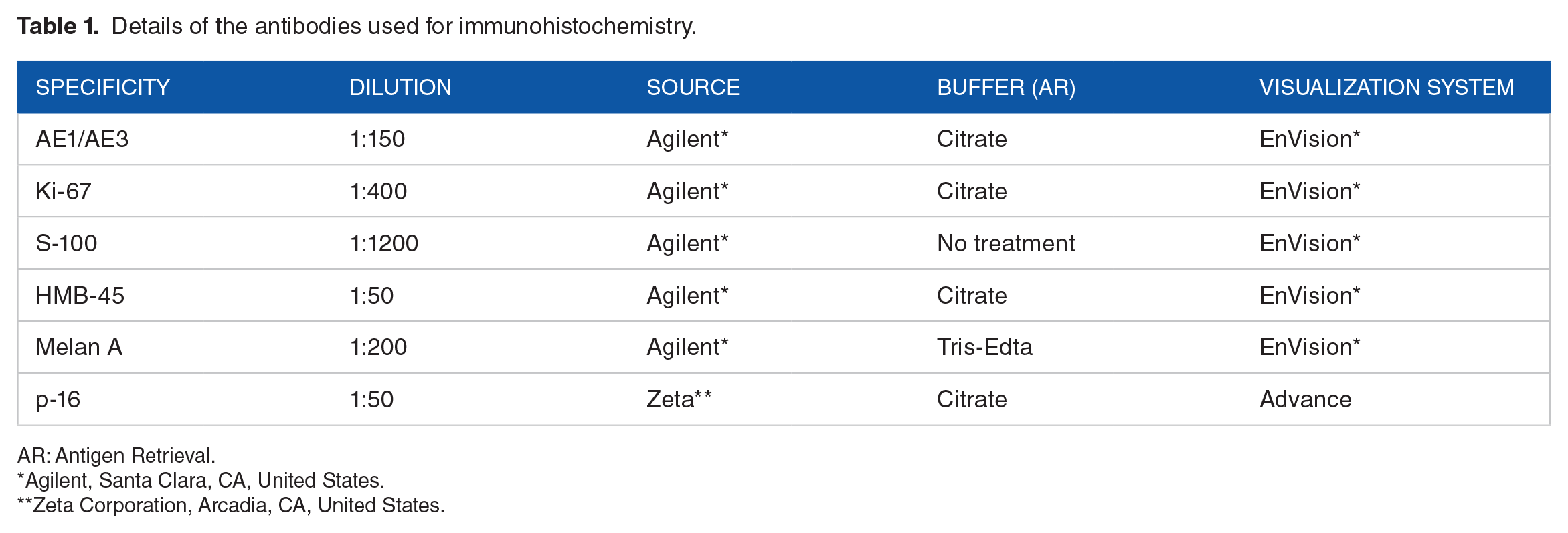
AR: Antigen Retrieval.
Agilent, Santa Clara, CA, United States.
Zeta Corporation, Arcadia, CA, United States.
The patient was then referred to the Head and Neck Surgery Department for standard surgery and/or radiation therapy; however, she refused any treatment options, including making an appointment with a head and neck surgeon. The patient only accepted doing a prosthetic dental treatment. After a 1-year follow-up, the lesion had increased in size (Figure 1C).
Discussion
About 0.01% to 7% of cutaneous and non-cutaneous SCCs are pigmented. 8 They may contain abundant melanin within the cytoplasm of the tumor cells, but pigmented SCCs have few dendritic melanocytes scattered beyond the epithelium basal layer. Few benign and malignant tumors were described to present melanocyte association.3-8 In the oral cavity, taking into account the present report, only 19 cases of carcinoma in situ or SCC colonized by melanocytes have been published in English literature (Table 2).4-7,9-15 In 14 lesions, the pigmentation was clinically discernible,4-7,9-15 but in 4, it was not.7,10 The majority were pigmented SCC4,6,7,10,12-14; only 6, including the current case, were in situ.5,7,9,11,15
Reported cases of oral pigmented squamous cell carcinoma (PSCC).

_____ No information.
According to the literature (Table 2), when considering gender, the lesion is slightly more frequent in males (11 cases), with the typical patient being a 55-year-old male (age range, 30-81 years). The most commonly affected sites are the tongue (7), followed by the gingiva (2), floor of the mouth (2), soft palate (2), hard palate, retromolar pad, tonsil, oropharyngeal, maxillary alveolar ridge, lower lip, and buccal mucosa (one case each), with the clinical aspects being similar to those found in standard SCC.4-7,9-15
The clinical differential diagnosis of pigmented lesions in the oral cavity is broad. It includes melanocytic lesions (nevus and melanoma), melanotic macule, melanoacanthoma, and nonmelanocytic lesions (such as trauma, foreign body tattoo, pigmented SCC, or pigmented carcinoma in situ, and vascular lesions, such as Kaposi sarcoma, hemangioma, among others). As these lesions’ prognoses are different, obtaining a biopsy is very important to set the diagnosis and rule out malignancy. In cases of vascular lesions, the clinicians should do a diascopy and aspiration before performing a biopsy. Thus, clinicians should have a low threshold to biopsy and be aware of pigmented carcinoma in situ.4,7,14,15 Although rare in the oral cavity, melanoma was included in the initial clinical differential diagnosis for the present report due to the clinical presentation.
The mechanisms of pigmentation in PSCC remain unclear. Some authors have reported that the malignant proliferation of keratinocytes stimulates an increased synthesis of pigment, as well as an increase in the number of melanocytes. 7
Satomura et al 6 demonstrated that SCC cells may produce stem cell factor and endothelin-1-stimulating melanin synthesis and melanocytic proliferation. Even in rare cases, cancers can produce factors that stimulate the proliferation of melanocytes, as reported in PSCC of the skin. 16
Some authors have speculated that the pigmentation of skin PSCC might be acquired from a surrounding pigmented lesion, such as lentigo, melanocytic nevus, or a pigmented solar keratosis.17-19 In the current report, it was difficult to know if there was a pre-existing pigmented lesion, as the patient was unaware of the lesion.
Other authors have suggested that due to the random distribution of melanocytes in the population, pigmentation of carcinomas may, in fact, be incidental.9,20
Finally, some HPVs are thought to be able to induce melanogenesis; however, the mechanism is still unknown. Longitudinal melanonychia is demonstrated to be the most characteristic manifestation of HPV type 56-associated Bowen disease. 21
Oropharyngeal squamous cell carcinoma is increasingly recognized as distinct among head and neck SCC because of the common association with HPV, because it has different demographics, a characteristic molecular profile, and a distinctly better prognosis. 22 Our patient had no history of alcohol or smoking habits. Nevertheless, the location and pigmentation favored HPV-related SCC, and it did not stain for P16 or ISH for HPV. Nevertheless, HPV-related SCC cannot be ruled out. The best method for HPV detection remains controversial. Many recommend the use of both p16 immunohistochemistry and HPV ISH. P16 expression is up-regulated when there is a high-risk HPV infection; strong and diffuse p16 immunoreactivity is found in cases of HPV-related SCC. In situ hybridization for high-risk HPV is more practical in a clinical setting and is quite specific but has limited sensitivity. False negatives and positives are reported with each of these techniques. Polymerase chain reaction (PCR) has been reported to be more sensitive, potentially amplifying the contaminant HPV.22,23 Although HPV is found, its presence cannot lead to being able to claim a causal relationship with SCC.
The most important differential diagnoses to be ruled out were melanoma associated with SCC and oral melanoacanthoma. Melanomas can colonize mucosal carcinoma in situ. 24 However, no cytologic and/or architectural atypia was found within the melanocytic component. Oral melanoacanthoma, which typically affects the mucosal surfaces susceptible to trauma, is believed to be a reactive process. It features hyperplasia of both spinous cells and melanocytes, and none of the 2 components is neoplastic. 24
Once the patient refused any treatment, the carcinoma could have progressed to SCC. The biologic behavior of PSCC has not yet been well defined due to a low number of reported cases. While some authors have suggested that the prognosis of a PSCC is similar to that of SCC,6,14 Mikami et al reported a better prognosis for PSCC than for conventional SCC. The clinical behavior of pigmented carcinoma in situ resembles that of a conventional carcinoma in situ. 4
The rationale behind the idea of publishing this case was the fact that, among pigmented oral mucosal lesions, particularly malignant neoplasms, the prognosis of PSCC is significantly better than that of a melanoma, hence the importance of making both clinicians and pathologists aware of this differential diagnosis. Furthermore, the clinicians should be aware of PSCC for clinical diagnosis.
Footnotes
Acknowledgements
The authors wish to thank Jeruza Pinheiro da Silveira Bossonaro and Nadir Freitas for their excellent technique, expertise, and assistance.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author’s Note
Luiz Alexandre Thomaz is also affiliated with Faculdade de odontológia da APCD_ FAOA, Brazil.
Author Contributions
All authors had the same contribution to the article.
Ethical Approval
The study has been approved by the institutional research ethics committee and has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed Consent
Informed consent was obtained from the participant included in the study.
