Abstract
Background:
Malignant bowel obstruction (MBO) is a frequent and debilitating complication in advanced abdominal cancers, particularly ovarian, colorectal, and gastric malignancies associated with peritoneal carcinomatosis. Surgery is often not feasible, and conventional decompression carries substantial morbidity. Endoscopic ultrasound-guided enterocolostomy (EUS-EC) with lumen-apposing metal stents (LAMS) has emerged as a minimally invasive alternative, but the evidence remains limited.
Objectives:
To systematically evaluate the feasibility, safety, and clinical outcomes of EUS-EC with LAMS for the palliation of MBO.
Design:
Systematic review and meta-analysis.
Methods:
A comprehensive literature search was performed across PubMed, Embase, Cochrane Library, and Web of Science from database inception through August 2025. Studies were eligible for inclusion if they involved patients with MBO undergoing EUS-EC using LAMS. Statistical analysis was performed using a random-effects model with Hartung–Knapp adjustments. Primary outcomes were technical success (defined as successful stent deployment) and clinical success (defined as relief of obstruction and restoration of bowel function).
Results:
Twenty-three studies (78 patients) were included. Pooled technical and clinical success rates were 96.1% and 88.4%, respectively. The mean hospital stay was 10.6 days, and oral intake resumed after a mean of 3.1 days. Mean post-procedure survival was 91 days, consistent with the advanced disease stage of most included patients. Adverse events were uncommon: diarrhea (6.4%), perforation (3.8%), bleeding (2.5%), and aspiration (1.2%). Stent misdeployment occurred in one cohort (26.6%), but all cases were managed endoscopically without major sequelae. Among 33 deaths reported, most (78.8%) were due to disease progression, with only 6.1% procedure-related.
Conclusion:
EUS-EC with LAMS demonstrates high technical and clinical success, rapid symptom relief, and low procedure-related morbidity, supporting its potential as an emerging palliative option for carefully selected patients with MBO. Evidence remains limited to small retrospective cohorts and case reports, highlighting the need for prospective comparative trials with patient-centered outcomes.
Introduction
Malignant bowel obstruction (MBO) in patients with advanced cancer presents a complex clinical challenge that requires an individualized and nuanced approach. 1 Management decisions are influenced by prior surgical history, tumor burden, the extent of disease dissemination, and overall prognosis. Reported prevalence varies by cancer type and study population, with estimates ranging from approximately 5% to 51% in ovarian cancer, 10% to 28% in colorectal cancer, and 3% to 15% in other malignancies. 2 The wide range, particularly in ovarian cancer, reflects differences in patient cohorts, disease stage, and diagnostic criteria across studies. In a large review of 490 cancer patients at MD Anderson Cancer Center, 68% of obstructions were tumor-related, most commonly involving the small bowel (64%), followed by the colon (20%) and gastric outlet (16%). 3
Treatment of MBO is individualized, aligning with each patient’s prognosis and goals of care. Palliative surgery is not routinely performed due to critical considerations, including the obstruction's location, tumor burden, comorbidities, and patient performance status. 4 Although surgery may extend symptom-free intervals compared to conservative care, it is associated with significant postoperative morbidity in over half of patients and carries a notable risk of mortality from complications. 5 Only one-third of surgically treated patients achieve sustained palliation, with a median survival of just 90 days, often accompanied by substantial treatment-related morbidity. 6 For inoperable MBO, symptomatic management such as gastric venting, cessation of oral intake, intravenous hydration, and parenteral antiemetics may relieve symptoms. However, prolonged nasogastric aspiration can lead to discomfort and complications such as esophagitis, nasal erosion, and bronchoaspiration.4,7 Alternative management for non-resectable tumors, including creation of a permanent stoma, carries high morbidity and mortality and significantly impacts quality of life, increasing caregiver burden.8,9
Recent advances have promoted self-expanding metal stents (SEMS) as an effective endoscopic alternative for proximal small bowel and colonic obstructions. 5 SEMS are well-established for left-sided malignant obstructions, as demonstrated by trials such as CReST and ESCO, which found SEMS significantly reduced stoma formation without compromising long-term survival.10–12 The European Society of Gastrointestinal Endoscopy recommends SEMS as the preferred palliative option and a viable alternative to emergency resection. 13 For right-sided malignant colonic obstruction, evidence remains limited, though meta-analyses suggest SEMS may facilitate minimally invasive surgery without increasing mortality or anastomotic failure. 14
Building on these developments, lumen-apposing metal stents (LAMS), originally approved for the drainage of pancreatic fluid collections—have been increasingly adapted for off-label use in transluminal procedures such as cholecystoduodenostomy, gastroenterostomy, and jejunojejunostomy.15–20 Among these, EUS-guided gastroenterostomy (EUS-GE) has demonstrated superior technical success, durability, and clinical outcomes compared with duodenal SEMS for both benign and malignant gastric outlet obstruction.19,21 Extending this principle beyond the upper gastrointestinal tract, endoscopists have developed EUS-guided enterocolostomy (EUS-EC), a technique in which a cautery-enhanced LAMS is deployed to create an anastomosis between the colon and a dilated proximal small-bowel loop upstream from the obstruction. The procedure is typically performed under general anesthesia with broad-spectrum antibiotics: a linear echoendoscope is advanced through the colon, dilated small-bowel loops are identified using ultrasound and fluoroscopy, and the stent is deployed to establish a bypass. Successful case reports include EUS-guided transmural LAMS placement for benign anastomotic strictures in high-risk surgical patients, EUS-guided ileocolonic anastomosis for recurrent SBO in an HIV-positive patient, and EUS-guided ileosigmoidostomy for malignant distal SBO.22–24
EUS-EC with LAMS is a relatively novel intervention that holds promise for reducing reinterventions related to tissue ingrowth, expediting the resumption of nutrition and chemotherapy, and overcoming the limitations inherent in conventional surgical methods. 25 However, the current evidence is derived almost entirely from case reports and small retrospective cohorts, which limits both the strength and generalizability of existing data. To date, no systematic review or meta-analysis has synthesized these fragmented findings to provide pooled estimates of efficacy, safety, clinical outcomes, and patient-centered outcomes. The objective of this review is to provide the first comprehensive synthesis of the available evidence on EUS-EC with LAMS for MBO. By clarifying its effectiveness, safety profile, and impact on patient care, this review aims to inform clinical decision-making in the palliative setting and identify priorities for future prospective and comparative studies.
Methods
Study design
This systematic review and meta-analysis were conducted in accordance with the guidelines of the Cochrane Handbook for Systematic Reviews of Interventions and reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement.26,27 The protocol was registered with PROSPERO (CRD42025636186). Because this investigation utilized data exclusively from previously published studies, no ethical approval or approval from the institutional review board (IRB) was required.
Search strategy
A comprehensive literature search was conducted in four databases, PubMed, Embase (Elsevier), Web of Science, and the Cochrane Central Register of Controlled Trials (CENTRAL), covering all records from each database’s inception through August 28, 2025. Medical Subject Headings (MeSH) terms and free-text keywords were combined using Boolean operators. The primary search terms included “coloenterostomy,” “enterocolostomy,” “endoscopic ultrasound guided,” “anastomosis,” and “bowel obstruction.” Specific search strings for each database, along with additional details, can be found in Supplemental Table 1. All referenced studies were thoroughly examined to identify any additional studies that may have been overlooked by the initial search terms. Only those studies that met the inclusion criteria were incorporated into the analysis.
Eligibility criteria
For the descriptive analysis, all study types that met the inclusion criteria were considered eligible, including case reports, case series, conference proceedings (published abstracts), observational studies (retrospective and prospective), open-label trials, and randomized controlled trials. The study population comprised patients with MBO, defined as obstruction resulting from an underlying malignant pathology, who underwent creation of an anastomosis using a LAMS placed under endoscopic ultrasound (EUS) guidance. All forms of MBO (proximal or distal, complete or partial) were included.
The intervention of interest was EUS-EC, in which a LAMS was used to establish an anastomosis between the small and large bowel, thereby bypassing the obstruction and providing an alternative route for bowel contents. To qualify for inclusion, studies were required to report at least one primary outcome, defined as either technical success or clinical success.
Exclusion criteria were studies with insufficient data, those addressing indications other than MBO, animal studies, unpublished or non-peer-reviewed articles, guidelines, letters, review articles, and duplicates. Patients with functional bowel obstruction (e.g., pseudo-obstruction) were also excluded. When multiple publications reported overlapping data, the most recent and comprehensive study was prioritized. Only studies published in English were considered.
Study selection
All references identified through the search were imported into Mendeley Reference Manager (version 1.19.8) (Amsterdam, North Holland, Netherlands) for duplicate removal. Three independent reviewers (M.S., K.K., and B.B.) screened the titles and abstracts in a blinded manner to identify potentially eligible studies. Full-text versions of these shortlisted articles were subsequently assessed against the inclusion and exclusion criteria. Any disagreements were resolved through discussion; if consensus was not reached, two additional reviewers (I.E. and H.R.) were consulted. The study selection process is visually summarized in a PRISMA flow diagram (Figure 1).

PRISMA flow diagram of study selection.
Outcomes
The primary outcomes of this study were meticulously defined to ensure a comprehensive understanding of the procedure’s effectiveness. These outcomes included technical success was defined as the accurate placement of a LAMS, creating a connection between the colon and a dilated segment of small intestine located upstream from the obstruction. 10 Clinical success referred to relief of the bowel obstruction following the procedure, demonstrated by improvement in obstructive symptoms, restoration of bowel movements, and removal of any nasogastric or nasojejunal tube when present. 10
Secondary outcomes included the time to resume oral intake, length of hospital stay (measured in days), survival duration post-procedure (also measured in days), adverse events, and overall mortality rates. Causes of death included factors such as progressive cancer under comfort care or hospice, sepsis, and any complications related to the procedure. Additionally, the study explored adverse events of interest, such as diarrhea, perforation, bleeding, and aspiration associated with the procedure.
Data extraction
Two reviewers (K.K. and B.B.) systematically and independently extracted data using a predefined Excel form. Extracted information included critical study characteristics—such as authors, publication year, country, and study design—as well as patient demographics, such as age, gender distribution, and sample size. Details of the intervention, including the location of the obstruction, type of LAMS used, balloon dilation after LAMS placement, periprocedural antibiotic use, and techniques, were documented, along with all other relevant outcomes as outlined in the eligibility criteria. Any discrepancies that arose were resolved either through consensus or by consulting a third reviewer (M.S.) to ensure the accuracy and integrity of the data.
Quality assessment
The quality and risk of bias of the included cohort and case-control studies were assessed using the Newcastle–Ottawa Scale (NOS) for observational studies. 28 This tool evaluates studies based on three domains: selection of study groups, comparability of groups, and outcome assessment. The selection domain assessed representativeness of the exposed cohort, selection of the non-exposed cohort, ascertainment of exposure, and demonstration that the outcome was not present at the start of the study. The comparability domain evaluated whether the study controlled for confounding factors. The outcome assessment domain considered the assessment of outcome, follow-up duration, and adequacy of follow-up. Studies were awarded a maximum of nine points. Scores of 7–9 were considered high quality, 4–6 indicated fair quality (moderate to high risk of bias), and ⩽3 denoted low quality (very high risk of bias).
For the case reports, methodological quality was appraised using the JBI Critical Appraisal Checklist for Case Reports, which includes eight items assessing reporting of patient demographics, clinical history, condition on presentation, diagnostic tests and results, intervention details, post-intervention outcomes, adverse or unanticipated events, and key takeaway lessons.
Data analysis
Descriptive statistics were used to summarize the data. Categorical variables were presented as frequencies and percentages, while continuous variables were reported as means with standard deviations (SDs) or confidence intervals (CIs), as available. Meta-analyses of single-arm continuous outcomes were performed using the meta package (version 8.2.1) in R (version 4.5.1, R Foundation for Statistical Computing, Vienna, Austria).29,30 Pooled estimates were calculated using an inverse-variance random-effects model, with the between-study variance (τ2) estimated by Restricted Maximum Likelihood (REML). To provide more reliable inference in the setting of a limited number of studies, Hartung–Knapp adjustments were applied for the confidence intervals. Because the outcomes of interest (length of stay, days to oral intake, and survival time) are measured in days and restricted to positive values, analyses were performed on the log-normal scale and subsequently back-transformed, ensuring pooled estimates and confidence intervals remained clinically interpretable and non-negative. For sensitivity analyses, raw-scale models were also examined. Between-study heterogeneity was assessed using the Cochran Q test (p < 0.10 considered significant) and quantified with the I2 statistic, with thresholds of 25%, 50%, and 75% interpreted as low, moderate, and high heterogeneity, respectively. In addition to pooled means and 95% CIs, 95% prediction intervals were reported to indicate the range of effects expected in future comparable settings.
For subgroup analyses, cohort studies were analyzed separately from case reports, and tests for subgroup differences were performed. When SDs were missing or reported as zero, they were imputed using the median SD from other included studies, and this assumption was explicitly reported. Given the small number of studies (k < 10 for all outcomes), publication bias was not formally assessed, as funnel plot asymmetry and Egger’s regression test are unreliable under these conditions.
When studies reported medians instead of means, Wan et al.'s method was used to estimate means and SDs. 31 Log transformation was applied where appropriate to normalize skewed distributions before conversion. 32
Results
Study selection and population characteristics
From an initial 7540 records identified via database searches, 23 studies ultimately met the inclusion criteria after full-text review. This review included 78 patients drawn from five retrospective studies (60 patients) and 18 case reports (18 patients).22–24,33–46 The retrospective studies comprised one single-center series, two multicenter cohorts, and two additional case series conducted between 2018 and 2025 at tertiary centers across Europe, the United States, and Asia, with a majority of male patients (53%–56%) and ages ranging from 26 to 92 years. Malignant obstructions were most commonly observed in the distal small bowel (70.5%), with 3.8% at the ileocolonic valve and 20.5% in the colon. When deployment data were available, LAMS placements were most frequent in the ileum (29.5%) and jejunum (17.9%), with less frequent placements in the duodenum (1.3%) and in various colonic segments (ascending 5.1%, transverse 12.8%, descending 14.1%, sigmoid 19.2%). Metastatic cancer accounted for the majority of cases. Other identified etiologies included rectal carcinoma and peritoneal carcinomatosis. Significant comorbidities (e.g., COPD, hepatic metastases, and pelvic recurrence) and prior surgeries (e.g., hemicolectomy and cystectomy) often rendered surgical intervention unsuitable. Failed decompression attempts were reported in 41.8% of cases with nasogastric tubes, 12.1% with enteral stents, 27.8% with venting PEG or jejunostomy tubes, and 38.2% with surgical debulking. These cases subsequently underwent EUS-guided LAMS placement. A detailed breakdown of the characteristics of the included case reports is provided in Tables 1 and 2, and patient population data for all included studies are presented in Table 3.
Study characteristics and demographics of patients undergoing EUS-guided enterocolostomy.

COPD, chronic obstructive pulmonary disease; EUS, endoscopic ultrasound; LAMS, lumen-apposing metal stent; NR, not reported; SCC, squamous cell carcinoma; NGT, Nasogastric Tube.
Clinical characteristics and outcomes of patients undergoing EUS-guided enterocolostomy in reported cases.

CI, confidence interval; COPD, chronic obstructive pulmonary disease; CT, computed tomography; EUS, endoscopic ultrasound; FOLFOX, folinic acid, fluorouracil, and oxaliplatin (Chemotherapy Regimen); HIPEC, hyperthermic intraperitoneal chemotherapy; LAMS, lumen-apposing metal stent; NR, not reported; PEG, percutaneous endoscopic gastrostomy; pT4N0R0, Tumor Stage Pathological Classification; RR, risk ratio; SBO, small-bowel obstruction; SCC, squamous cell carcinoma; Tx, Treatment.
Baseline characteristics of the study population undergoing EUS-guided enterocolostomy.

Ileocolonic Anastomosis, Surgical Connection Between Ileum and Colon; n/N, Number of Patients with the Feature/Total Number of Patients; N, Total Number of Patients; PEG, percutaneous endoscopic gastrostomy.
Procedural characteristics
Anastomoses created using LAMS were largely unspecified (39.7%), followed by ileocolostomy (29.5%), ileosigmoidostomy (11.5%), jejunocolostomy (17.9%), and duodenocolostomy (1.3%). Among the stents used, 48.7% were 15 × 10 mm AXIOS, while 51.3% were 20 × 10 mm AXIOS, with selective use of HOT SPAXUS reported in individual cases. Balloon dilation was performed in 59% of procedures, and 47% of patients received periprocedural antibiotics where data were available. Two primary techniques were employed: wire-guided methods in 46% of cases and freehand approaches in 51% of cases. A detailed breakdown of procedural characteristics is presented in Table 4.
Procedural characteristics of EUS-guided enterocolostomy.

EUS, endoscopic ultrasound; F/U, Follow-Up; LAMS, lumen-apposing metal stent.
Quality assessment
The NOS assessment rated all four retrospective studies as moderate quality, highlighting strengths in cohort representativeness, exposure ascertainment, and follow-up adequacy. A detailed account is presented in Table 5. For the case reports, all 18 clearly described patient demographics, clinical presentation, diagnostic evaluation, intervention performed, and post-intervention outcomes. Overall, all case reports were deemed suitable for inclusion. A detailed account of the JBI assessment is provided in Supplemental Table 2.
NOS evaluation of included studies.

NOS, Newcastle–Ottawa Scale.
Outcomes
A detailed account of outcomes is provided in Table 6.
Clinical outcomes of EUS-guided enterocolostomy.
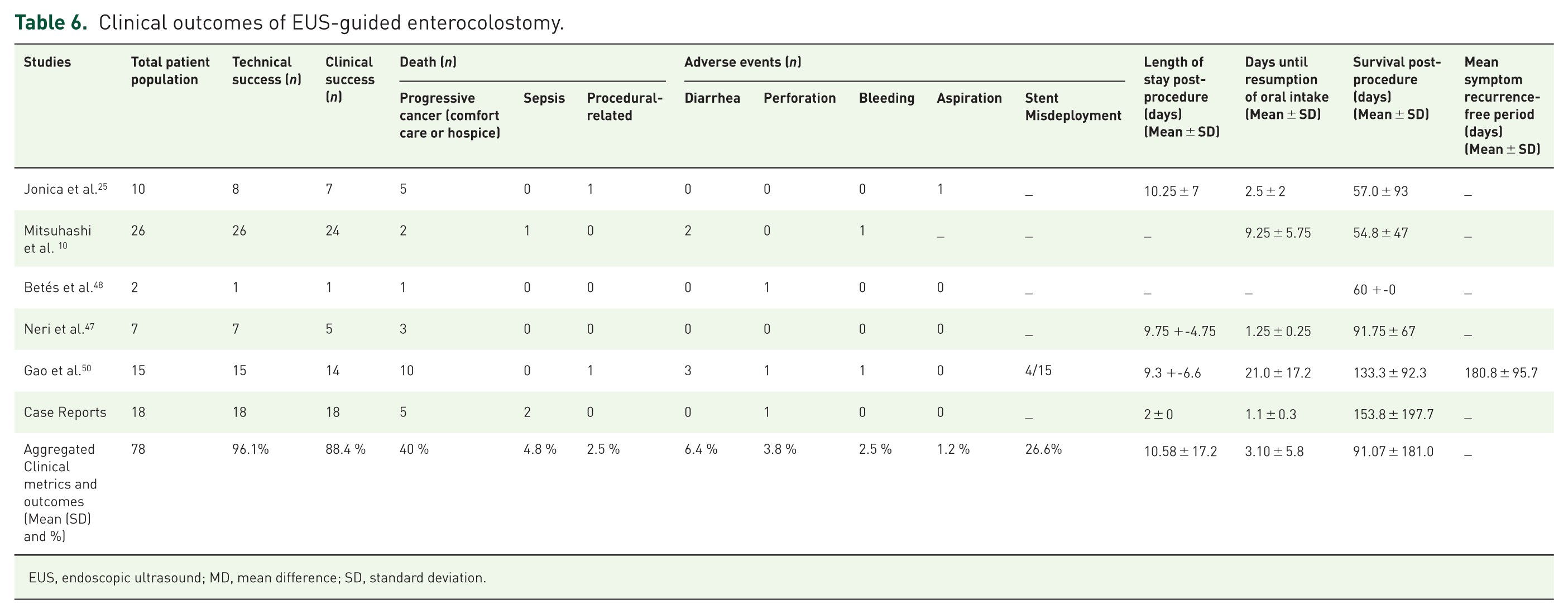
EUS, endoscopic ultrasound; MD, mean difference; SD, standard deviation.
Primary outcomes
The primary outcome included a technical success rate of 96.1% and a clinical success rate of 88.4%, the latter defined as symptom relief and restored bowel function.
Secondary outcomes
The pooled mean hospital stay following the procedure was 10.58 days (95% CI: 3.20–34.97; prediction interval: 1.47–76.04; I2 = 77.8%, p = 0.004) (Figure 2). In subgroup analysis, the mean was 12.74 days (95% CI: 6.80–13.99) among cohort studies and 2.00 days (95% CI: 0.40–10.07) among case reports, with a statistically significant difference between subgroups (Q = 4.62, p = 0.0315).

Forest plot of mean hospital stay following EUS-EC.
The pooled mean survival after the procedure was 91.07 days (95% CI: 56.15–147.72; prediction interval: 29.03–285.74; I2 = 71.5%, p = 0.004) (Figure 3). Subgroup analysis demonstrated a mean survival of 81.21 days (95% CI: 53.42–157.60) across cohort studies and 153.80 days (95% CI: 84.93–278.52) among case reports, with subgroup difference approaching significance (Q = 3.20, p = 0.0736).
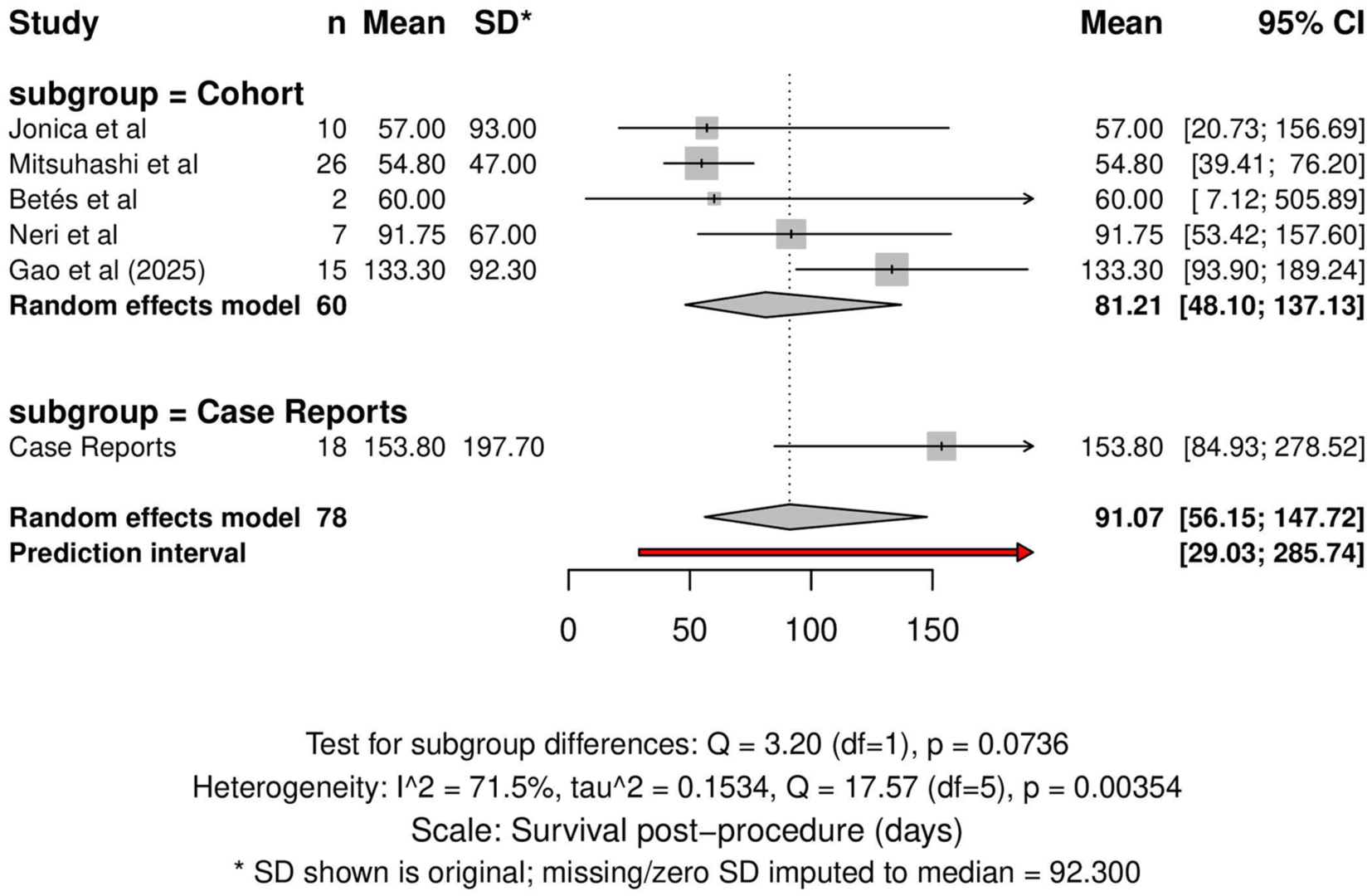
Forest plot of mean survival after EUS-EC.
The average time to resumption of oral intake (Oral intake refers to any enteral intake; diet stage was inconsistently specified) was 3.10 days (95% CI: 0.85–11.38; prediction interval: 0.13–73.50; I2 = 98.8%, p < 0.0001) (Figure 4). In subgroup analysis, based on 58 patients from retrospective cohorts and 10 patients from case reports with reported data, the pooled mean was 4.05 days (95% CI: 1.52–4.10) for retrospective cohorts and 1.10 days (95% CI: 0.97–1.25) for case reports, with a statistically significant subgroup difference (Q = 6.63, p = 0.01).

Forest plot of mean time to oral intake resumption.
As an exploratory outcome, symptom recurrence-free survival was reported in Gao et al. 50 Using Wan’s method to convert the median (165 days, range 40–353, n = 12) to mean ± SD, the estimated mean recurrence-free survival was 180.8 ± 95.7 days.
Adverse events
Adverse events were relatively uncommon. The most frequently reported complications were diarrhea (6.4%), perforation (3.8%), bleeding (2.5%), and aspiration (1.2%). Stent misdeployment occurred in four cases (26.6%), reported exclusively in Gao et al., 50 but all instances were promptly recognized and successfully managed endoscopically without major sequelae.
Across all included studies, a total of 33 deaths (42.3%) were documented. The majority were attributed to progression of underlying malignancy (26 deaths, 78.8%). Other causes included sepsis (three deaths, 9.1%), procedure-related complications (two deaths, 6.1%), and other causes such as delayed digestive hemorrhage and pulmonary abscess (two deaths, 6.1%). Only a small fraction of deaths (6.1%) were directly procedure-related, whereas nearly four-fifths reflected the natural progression of advanced disease.
Discussion
Our systematic review and meta-analysis found that EUS-EC using LAMS is a safe, feasible, and effective palliative option for MBO in appropriately selected patients. The procedure demonstrated consistently high rates of technical and clinical success across heterogeneous study designs and was generally associated with rapid symptom improvement, early resumption of oral intake, and relatively short hospital stays. Adverse events were infrequent and mostly minor, and instances of stent misdeployment, although occasionally reported, were effectively managed endoscopically without major sequelae. Mortality across studies was primarily related to the progression of advanced malignancy rather than procedural complications.
For comparison, SEMS remain the benchmark therapy for malignant obstruction. Reported technical and clinical success rates are 97%–99% and 80%–89% for duodenal obstruction, 95%–100% and 84%–100% for jejunal obstruction, and 81%–95% and 76%–91% for left-sided colonic obstruction, respectively.51–55 Adverse events occur in 10%–22%, most often migration, occlusion, or perforation, and are usually manageable.51–56 By contrast, surgical palliation has lower success rates, with diverting stoma, internal bypass, and resection achieving technical success in 80%, 78%, and 63% of cases, respectively. 57 Clinical improvement rates vary from 32% to 100%, with diet resumption between 45% and 75%, 58 but surgery carries a higher perioperative risk, with complication rates of 7%–37% and mortality ranging from 6% to 32%.58–60 PEG tube placement provides decompression in selected patients, with technical success of 89%–100% and symptom relief of 77%–96%, though oral intake is inconsistently restored.1,61–63 Median survival after SEMS for duodenal obstruction is 93–116 days,53,54 and colonic SEMS achieves outcomes comparable to colectomy. 64 Against this backdrop, the pooled technical and clinical success of EUS-EC appears comparable to SEMS and surgery, with the additional advantage of facilitating oral intake.
The success of EUS-EC is underpinned by real-time imaging that permits precise stent deployment, thereby minimizing the risk of misplacement. 65 In addition, the flanges on either side of the LAMS provide strong anchoring platforms for maintaining anastomotic patency. The shorter design of LAMS may also reduce the risk of kinking or collapse, given its more rigid structure. Traditional SEMS are prone to tumor ingrowth due to placement adjacent to malignant tissue. EUS-EC circumvents this issue by bypassing the affected tumor segment.66–68 The inherent design of LAMS reduces the likelihood of migration and occlusion, as evidenced by a low reported reintervention rate of 4%.66,67 EUS-EC effectively addresses challenges in patients with multifocal strictures or peritoneal carcinomatosis, where traditional endoluminal stents may fail. The procedure’s versatility and feasibility across various bowel locations further support its potential applicability.
Efficiency is reflected in a mean hospital stay of 10.6 days and oral intake resumption within 3.1 days. Minimizing hospital stay reduces morbidity, infection risk, and mortality, particularly in this patient population. 69 By comparison, surgical palliation typically requires longer hospitalization (12.5–31 days) and delayed diet resumption, varying by technique, with one study reporting 5 days for surgical gastrojejunostomy versus 3 days for laparoscopic gastrojejunostomy.58,69–71 Patients undergoing palliative surgery for MBO due to peritoneal carcinomatosis had a mean hospital stay of 20.0 ± 23.1 days, with oral intake resumption as a prerequisite for discharge. However, the study did not explicitly specify the time to oral intake resumption postoperatively. 72 In one report, surgical patients spent 61% of their remaining life in the hospital. 73 EUS-EC also reduces procedure time: approximately 36–53 min compared with a median of 152 min for surgery.25,50,74 Although surgical approaches can restore bowel continuity, their comparable success comes at the cost of longer recovery, which often conflicts with the palliative goal of optimizing quality of life. Mean survival after EUS-EC was approximately three months (91 days), highlighting that long-term outcomes such as stent patency and delayed adverse events remain underexplored.
Adverse events with EUS-EC were infrequent: diarrhea (6.4%), perforation (3.8%), bleeding (2.5%), and aspiration (1.2%). Diarrhea is expected due to the bypass between the small and large intestines, resulting in rapid transit of intestinal contents. Rapid diet resumption may increase the risk of aspiration, which can be mitigated by a gradual increase in oral intake. The overall mortality rate of 42.3% must be interpreted in context: nearly 79% of deaths were due to disease progression, 9% to sepsis, and 6% to procedural complications. Only a minority was directly attributable to the procedure. By comparison, procedure-related mortality with SEMS is 1%–5%,53,54,75 while surgery is associated with higher complication rates (7%–37%) and mortality of 6%–32%.58,59,60 Infection rates for surgery vary depending on location and approach, ranging from 4.5% to 32%.76–78 PEG placement carries aspiration risks of 1.5% and infection rates of 5.4%–30%.62,79,80 However, this is not commonly observed in EUS-EC, given the minimized tissue trauma from LAMS placement compared with the suturing and stapling required in surgical resection. Overall, palliative surgical resection for MBO carries higher complication rates compared with curative resection, further supporting EUS-EC as a preferred alternative.76,77 Thus, the mortality observed after EUS-EC largely reflects advanced cancer biology rather than procedural risk.
Cost and resource utilization remain important considerations. LAMS are more expensive than SEMS and require anesthesia, linear echoendoscopes, and advanced expertise, restricting use to tertiary centers.54–56,81 While EUS-EC provides rapid symptom relief and shorter hospitalization in selected patients, these benefits must be weighed against higher device and procedural costs. SEMS have been shown to be more cost-effective than emergent surgery for malignant colonic obstruction, with lower costs, fewer permanent stomas, and improved QALYs.56,64,75,81,82 No large-scale cost-effectiveness studies have compared LAMS directly with SEMS or surgery in MBO, and available data are limited to small retrospective series from expert centers.53–55 The broader financial feasibility of EUS-EC in palliative care, therefore, remains uncertain.
This study has several limitations that should be considered when interpreting the findings. The current evidence base is primarily composed of small retrospective studies and individual case reports, with no randomized trials available to date. This reliance on lower-level evidence limits both the precision and generalizability of the results. Considerable variability in patient selection, procedural technique, and outcome reporting introduces heterogeneity that reduces the reliability of pooled estimates. Follow-up durations were generally short, with an average post-procedure survival of approximately 3 months, preventing adequate assessment of long-term stent patency, delayed adverse events, or sustained clinical benefit. Incomplete reporting of baseline variables, outcome measures, and denominators, such as diet type and recovery timelines, further weakens the evidence. Patient-centered outcomes, including quality of life, functional recovery, and symptom burden, were infrequently reported. In addition, most studies originated from high-volume tertiary centers, where operator expertise and institutional experience may have influenced outcomes, limiting applicability to lower-volume settings. Publication bias toward favorable or technically successful cases cannot be excluded.
Future research should focus on generating higher-quality evidence through multicenter prospective registries and randomized controlled trials. Comparative studies evaluating EUS-EC against SEMS and surgical palliation are needed to establish relative efficacy, safety, and durability. Standardization of procedural techniques, outcome definitions, and reporting criteria will enhance comparability across studies and allow for more reliable meta-analytic assessment. Longer follow-up periods are necessary to evaluate stent patency, reintervention rates, and delayed complications. Future studies should also incorporate patient-reported outcomes, nutritional recovery, and quality-of-life measures to better capture clinical benefit. Finally, inclusion of economic analyses and participation from centers with varying procedural volumes will improve understanding of real-world feasibility, cost-effectiveness, and generalizability of EUS-EC in the palliative management of MBO.
Conclusion
EUS-guided enterocolostomy with LAMS achieves high technical and clinical success, rapid symptom relief, and low procedure-related morbidity, supporting its role as a viable palliative option for MBO in selected patients. Outcomes appear comparable to SEMS and superior to surgery in terms of recovery, although current evidence is limited to small retrospective series and case reports. Larger prospective studies are needed to confirm efficacy, define patient selection, and evaluate long-term safety, cost-effectiveness, and quality of life.
Supplemental Material
sj-docx-1-cmg-10.1177_26317745251400665 – Supplemental material for EUS-guided enterocolostomy with lumen-apposing metal stents for palliative management of malignant bowel obstruction: a systematic review and meta-analysis
Supplemental material, sj-docx-1-cmg-10.1177_26317745251400665 for EUS-guided enterocolostomy with lumen-apposing metal stents for palliative management of malignant bowel obstruction: a systematic review and meta-analysis by Muhammad Shahzil, Abdulmalik Saleem, Syeda Kanza Kazmi, Bharosha Bhattarai, Muhammad Saad Faisal, Muhammad Hashim Faisal, Hassam Ali, Andrew Ofosu, Hadie Razjouyan and Ikponmwosa Enofe in Therapeutic Advances in Gastrointestinal Endoscopy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
