Abstract
Background:
Primary small intestinal lymphoma (PSIL) is a rare malignancy with heterogeneous clinical and pathological features, making accurate diagnosis challenging.
Objectives:
To investigate the clinical value of double-balloon enteroscopy (DBE) combined with endoscopic ultrasonography (EUS) in the diagnosis of small intestinal lymphoma.
Design:
Retrospective cohort study.
Methods:
We retrospectively reviewed 21 patients with pathologically confirmed PSIL who underwent both DBE and EUS at the Department of Small Intestinal Endoscopy, The Sixth Affiliated Hospital of Sun Yat-sen University, between September 2022 and May 2025. Clinical data were collected and analyzed in combination with pathological findings.
Results:
A total of 21 patients were included (12 males, 9 females; median age of 52 years, range 31–87 years. Subtypes of B-cell lymphomas included diffuse large B-cell lymphoma (n = 5), follicular lymphoma (n = 3), and mucosa-associated lymphoid tissue lymphoma (n = 6). T-cell lymphomas included peripheral T-cell lymphoma (n = 7). Endoscopic findings were classified into five categories (1): hypertrophic type (2), exophytic tumor type (3), follicular/polypoid type (4), ulcerative type, and (5) diffusion type. EUS classification included superficial spreading (38.1%, 8/21), diffuse infiltrative (42.9%, 9/21), and nodular (19.0%, 4/21).
Conclusion:
DBE combined with EUS provides complementary morphological and structural information for the diagnosis of PSIL, improving clinical recognition and subtype characterization. Future multicenter studies with larger cohorts are warranted to validate these findings and establish standardized diagnostic protocols.
Introduction
Primary small intestinal lymphoma (PSIL) refers to a malignant tumor originating from the submucosal lymphoid tissue of the duodenum, jejunum, and ileum, accounting for 20%–30% of all primary gastrointestinal lymphomas. 1 The ileum is the most frequently involved site, followed by the jejunum and duodenum. Clinical manifestations include gastrointestinal symptoms such as abdominal pain, melena, hematochezia, and intestinal obstruction, with some patients presenting systemic symptoms like fever, night sweats, and weight loss. Histopathologically, PSIL is predominantly non-Hodgkin lymphoma, mostly of B-cell origin. Diffuse large B-cell lymphoma is the most common subtype, while others include follicular, mantle cell, mucosa-associated lymphoid tissue (MALT) lymphoma, and Burkitt lymphoma. T-cell lymphomas are rare in PSIL, predominantly occurring in the jejunum but associated with a higher perforation rate. Pathological biopsy remains the gold standard for PSIL diagnosis, primarily through endoscopic biopsy or postoperative pathological examination of resected specimens. Compared with gastric and colorectal lymphomas, PSIL presents greater diagnostic challenges due to poor general condition in some patients at onset and technical difficulties in reaching distal small bowel segments via conventional endoscopy. Most PSIL lesions originate from the submucosal layer and muscularis propria, where insufficient biopsy depth or inadequate sampling often complicates pathological diagnosis. Furthermore, the endoscopic morphology of intestinal lymphomas exhibits high heterogeneity across different pathological subtypes, making differentiation from infectious enteropathies and inflammatory bowel diseases challenging. Endoscopic ultrasonography (EUS) enhances diagnostic accuracy by positioning the ultrasound probe directly against lesions through the intestinal lumen, providing detailed visualization of intestinal wall layers and adjacent structures. 2 This technique enables precise characterization of mural architecture. Our center has achieved promising outcomes in lesion evaluation using DBE combined with small intestinal EUS. This article summarizes characteristic features of PSIL through a comprehensive analysis of white-light endoscopy, EUS findings, and pathological classifications.
Patients and method
Patients
This was a retrospective study conducted between September 2022 to May 2025 at the Small Bowel Endoscopy Center, The Sixth Affiliated Hospital of Sun Yat-sen University. Medical records and pathological data of 21 patients with PSL were reviewed. The patients’ information was anonymized prior to analysis. Histopathological diagnosis was based on the WHO classification3,4 and performed through morphologic and immunophenotypic analyses of endoscopically biopsied or surgically resected specimens. Patients were included in the study based on the definition of primary gastrointestinal non-Hodgkin’s lymphoma according to Lewin. 5 Those patients who presented with second malignancies or without follow-up information were excluded. In this study, duodenal lesions referred to sites distal to the descending part of the duodenum (beyond the reach of conventional gastroscopy), while ileal lesions referred to sites inaccessible by colonoscopy; therefore, DBE was required for adequate evaluation and tissue sampling. No patients in this study underwent capsule endoscopy.
The diagnostic workup included a detailed medical history and physical examination, complete blood cell count, serum chemistry, computed tomography (CT) scan, multiple endoscopic biopsies, and positron emission tomography (PET) imaging was performed in some patients.
Stage
Musshoff’s variation of the Ann Arbor method for gastrointestinal lymphoma was utilized for staging, 6 and the imaging modality for clinical staging was CT or PET.
Endoscopic features
We classified the endoscopic findings into five categories: (1) hypertrophic type, (2) exophytic tumor type, (3) follicular/polypoid type, (4) ulcerative type, and (5) diffusion type.
For EUS, a 20 MHz microprobe ultrasound system (Endoscopic Ultrasound System IM-02P-202501, InnerMedical Co., Ltd, Shenzhen, China) was introduced through the biopsy channel of the double-balloon enteroscope (DBE). All procedures were performed by endoscopists experienced in DBE, ensuring smooth completion even in anatomically challenging locations such as the jejunum and ileum.
The procedure workflow was as follows: (1) DBE examination: The small intestine was systematically examined using the push–pull technique with alternating balloon inflation/deflation. (2) Lesion detection and characterization: Upon identifying a lesion with white-light endoscopy, warm water (35°C–37°C) was infused through the biopsy channel to distend the lumen and optimize visualization. (3) Small intestinal EUS (SIEUS): A 20 MHz microprobe was advanced through the DBE biopsy channel, and ultrasound parameters (frequency and depth) were adjusted to evaluate the lesion’s layer of origin, internal echo pattern, margins, and relationship with adjacent structures.
Statistical analysis
All analyses were performed using SPSS Statistics 26.0 software (SPSS Inc., Chicago, IL, USA). Age was analyzed as a continuous variable and reported as the median with range. Laboratory parameters, including hemoglobin, albumin, and lactate dehydrogenase (LDH), were dichotomized into “normal” and “abnormal” according to institutional reference values, as these thresholds are clinically relevant for patient assessment. Clinical symptoms, lesion location, and disease stage were analyzed as categorical variables and presented as frequencies and percentages. No missing data were present in the included variables. The reporting of this retrospective study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 7
Results
Clinical features
From September 2022 to May 2025, a total of 21 patients diagnosed with PSIL were included in this analysis. Table 1 summarizes the clinical characteristics of these patients. The median age of the cohort was 52.10 (48.00–66.25) years, with a gender distribution of 12 males and 9 females, and an age range of 31–87 years. The most common symptom was hematochezia (n = 20), followed by abdominal pain (n = 14), abdominal distension (n = 11), diarrhea (n = 5), nausea, and vomiting (n = 2). Of all patients, 3 (14.3%) acquired illness in the duodenum, 5 (23.8%) in the jejunum, 8 (38.1%) in the ileum, and 5 (23.8%) in more than one location.
Demographic and clinical characteristics of primary small intestinal lymphoma.
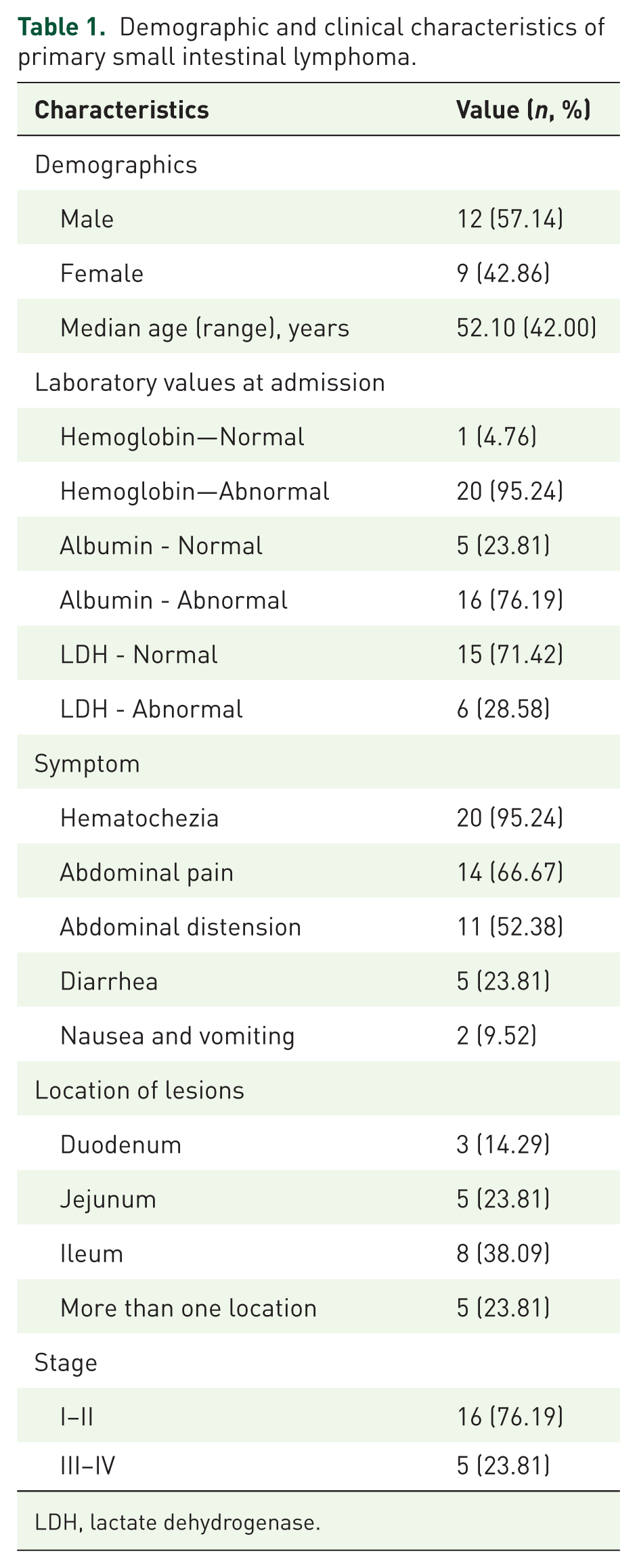
LDH, lactate dehydrogenase.
Among the various clinical variables studied, a higher number of patients exhibited abnormalities in certain indicators such as hemoglobin (HGB), albumin (ALB), and lactate dehydrogenase (LDH). Among the 21 patients with available data, 95.23% had lower HGB levels, 76.19% had lower ALB levels, and 28.57% had higher LDH levels than the standard range.
During CT examination, intestinal wall thickening of varying degrees, space-occupying lesions in the small intestine, and celiac lymph node enlargement were observed in the patients. 16 (76.2%) patients had stage I–II disease, and 5 (23.8%) had stage III–IV disease (Table 1).
Histopathological classification
Among B-cell lymphomas, subtypes included DLBCL in five cases, follicular lymphoma (FL) in three cases, and MALT lymphoma in six cases. Within T-cell lymphomas, peripheral T-cell lymphoma was identified in seven cases (Table 2).
Histopathological classification and endoscopic characteristics of primary small intestinal lymphoma.
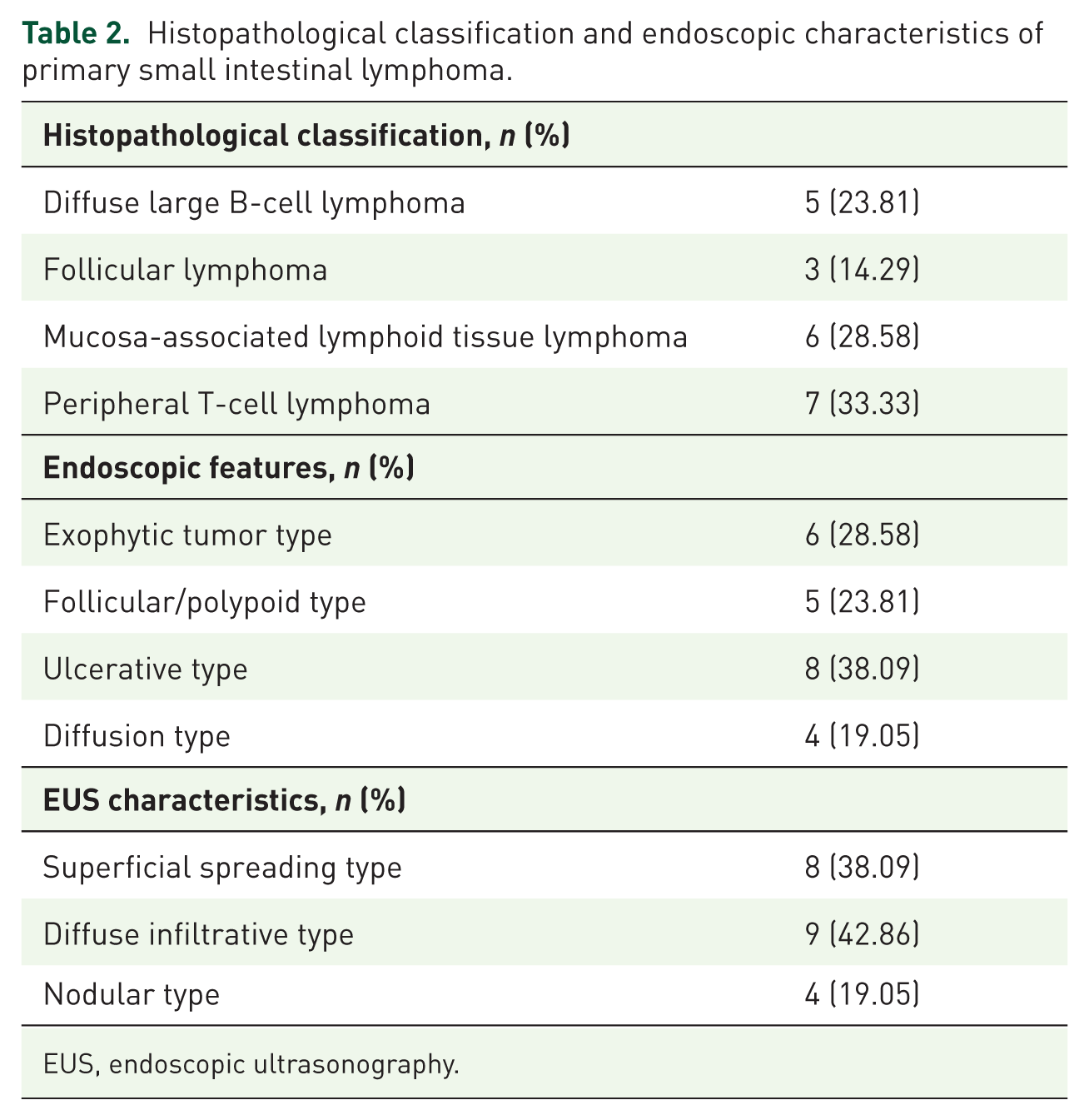
EUS, endoscopic ultrasonography.
Enteroscopic features
During hospitalization, all patients underwent an enteroscopy examination. The endoscopic features were divided into the following groups: six patients had an exophytic type (Figure 1(a)), characterized by tumor-like lesions such as a large, friable, nodular mass with signs of hemorrhage, surface erosion, or moss cover; five patients had a follicular/polypoid type (Figure 1(b)), characterized by multiple lymphoid follicular eminences or multiple polypoid lesions; eight patients had an ulcerative type (Figure 1(c)), characterized by single ulceration or multiple ulcerative lesions; and four patients had a diffuse type (Figure 1(d)), characterized by mucosal erosion, punctate or patchy bleeding, mucosal rough hyperplasia, and other endoscopic manifestations. Among these patients, two patients had two or more endoscopic features simultaneously (Table 2).

Enteroscopic features of primary small intestinal lymphoma. (a) Exophytic tumor type. (b) Follicular/polypoid type. (c) Ulcerative type. (d) Diffusion type.
EUS characteristics
In the EUS classification, PSIL can be categorized into three types: superficial spreading type, diffuse infiltrative type, and nodular type. The distribution in this cohort was as follows: superficial spreading type in eight cases (Figure 2(a)), diffuse infiltrative type in nine cases (Figure 2(b)), and nodular type in four cases (Figure 2(c)). The EUS characteristics of each type are described below: Superficial spreading type: Focal thickening of the submucosa and muscularis propria with hypoechoic alterations and ill-defined layer demarcation, Diffuse infiltrative type: Transmural thickening of the intestinal wall with complete loss of layer demarcation, Nodular type: Localized wall thickening accompanied by nodular protrusions, showing hypo- to anechoic structures within the nodules (Table 2).

EUS characteristics of primary small intestinal lymphoma. (a) Superficial spreading type. (b) Diffuse infiltrative type. (c) Nodular type.
Discussion
PSIL is a rare disease, but its incidence has been increasing worldwide in recent years. Because patients often present with nonspecific systemic or gastrointestinal symptoms, establishing a timely diagnosis remains difficult. DBE plays a pivotal role in this process by allowing direct visualization of mucosal abnormalities, precise localization of lesions, and targeted biopsies for histopathological confirmation. 8 More recently, the introduction of EUS has further advanced diagnostic accuracy, combining the strengths of endoscopy and ultrasound. When used alongside biopsy sampling, EUS provides valuable information that aids in both the detection and differentiation of PSIL. 2 In this single-center retrospective cohort, we demonstrate the clinical value of combining DBE with EUS in the diagnosis of PSIL. DBE enabled direct visualization and biopsy of lesions throughout the small bowel, while EUS enhanced diagnostic accuracy by characterizing the depth, morphology, and echo features of lesions. Together, these methods provided complementary information that improved correlation with histopathological findings.
The manifestations of PSIL are nonspecific and may mimic benign ulcers or adenocarcinoma under conventional white-light endoscopy. Because PSIL frequently originates in the submucosal layer rather than the mucosa, routine biopsies often fail to capture diagnostic tissue, contributing to high rates of misdiagnosis. 9 Therefore, when PSIL is suspected, multiple deep biopsies, excision of large mucosal pieces, and routine immunohistochemical staining are essential.
Our cohort revealed diverse endoscopic morphologies that correlated with histological subtypes. DLBCL, the most common and aggressive intestinal B-cell lymphoma, 10 typically presents as a mass, ulcerative, polypoid, diffuse infiltrative, or mixed form. MALT lymphoma, arising from mucosal lymphoid tissue, 11 showed solitary or multiple masses, polypoid changes, diffuse infiltration, or ulcerative lesions, often with sessile or semi-pedunculated protrusions and normal-appearing mucosa; ulcers could be annular or serpiginous. Consistent with previous reports, EUS findings further categorize MALT lymphoma into superficially spreading, diffusely infiltrative, mass-forming, and mixed types, with the superficially spreading and diffusely infiltrative forms recognized as distinctive patterns of low-grade disease. 12 Mantle cell lymphoma (MCL), a monomorphic mature B-cell neoplasm, 13 displayed diffuse, nodular, or mantle zone growth, and endoscopically appeared as polyps, masses, ulcers, or nodules, with multiple polyps ranging from a few millimeters to several centimeters. In line with previous studies, MCL often presents as submucosal lesions, supporting its characteristic growth pattern beneath the mucosa rather than confined to the surface. 14 FL, an indolent subtype, 15 mainly manifested as multiple small polypoid lesions, but mass-like submucosal tumors and ulcerations with peripheral edema were also observed. Consistent with these findings, another study using EUS has demonstrated homogeneously hypoechoic thickening of the submucosal layer at sites where mucosal abnormalities were observed, further supporting the submucosal origin of these lesions. 16 Enteropathy-associated T-cell lymphoma (EATL), though rare (around 5% of GI lymphomas), is highly aggressive, 17 presents with ulcerative, epithelial mass, diffuse infiltrative, or nodular patterns; ulcers were often deep, polymorphic, and widely distributed, frequently accompanied by strictures, villous atrophy, and mucosal edema. Previous studies have reported somewhat different endoscopic features, including innumerable fine granularities (mosaic mucosal patterns) and diffuse mucosal thickening with semicircular shallow ulcerations in the small bowel, while colonic findings were nonspecific and unable to distinguish EATL type II from other conditions. 18 Taken together, we classified the small bowel EUS appearances of lymphoma into three patterns. The superficial spreading type is characterized by focal thickening of the submucosa and muscularis propria with hypoechoic changes and poorly defined layer boundaries; this pattern is often seen in MALT lymphoma and FL. The diffuse infiltrative type shows transmural thickening of the intestinal wall with complete loss of layer demarcation, a pattern observed in T-cell lymphoma, aggressive B-cell lymphoma, DLBCL, and MALT lymphoma. The nodular type presents as localized wall thickening with nodular protrusions containing hypo- to anechoic structures, most commonly associated with MALT lymphoma. 2
Based on our center’s experience, the EUS features of small bowel diseases can be summarized as follows. Small bowel mesenchymal tumors usually present as masses growing both intraluminally and outward, with larger lesions often showing hemorrhage, necrosis, or cystic degeneration. 19 On EUS, they appear as well-demarcated, round, hypoechoic lesions, typically originating from the muscularis propria, consistent with the prior report by Polkowski et al. 20 Small bowel adenocarcinomas are more localized, often occurring in the proximal bowel, with features of wall stiffness, luminal narrowing, and, in some cases, vascular encasement leading to heterogeneous echogenicity and serosal rupture. Crohn’s disease typically shows varying degrees of wall thickening, most prominent in the submucosa and muscularis propria, with poorly defined but distinguishable layers and occasional hypo- or hyperechoic changes, in line with findings by Maconi et al. 21 Intestinal tuberculosis is characterized by mucosal loss at ulcer sites, wall thickening mainly in the muscularis propria, thinning of the submucosa, and reduced echogenicity. 22
This study is retrospective and single-center with a small sample size, which may limit generalizability and preclude robust subtype-specific analyses. Capsule endoscopy was not used during the study period. Inter-observer agreement for EUS patterning was not assessed, and longitudinal outcomes were not analyzed. Prospective, multicenter studies with standardized DBE/EUS protocols and pathological correlation are warranted. Nevertheless, the clinical presentations, endoscopic features, and histopathological correlations we observed are consistent with patterns reported in previous studies of PSIL. Thus, our results may be generalizable to similar clinical settings, particularly in centers with access to DEB and EUS. However, multicenter studies with larger and more diverse populations are warranted to further confirm the applicability of these findings across different healthcare systems and patient groups.
Conclusion
This article systematically characterizes PSIL through white-light endoscopy, SIEUS, and histopathological patterns to enhance clinical recognition. However, management of PSIL remains suboptimal due to: Low incidence limiting prospective randomized controlled trials, absence of evidence-based guidelines, and knowledge gaps in standardizing diagnosis/therapy across subtypes. Critical unmet needs include: Developing protocols to improve diagnostic accuracy, establishing subtype-specific management frameworks, and reducing misdiagnosis through multidisciplinary approaches.
Supplemental Material
sj-docx-1-cmg-10.1177_26317745251392785 – Supplemental material for Evaluating the diagnostic value of double-balloon enteroscopy combined with endoscopic ultrasonography for primary small intestinal lymphoma
Supplemental material, sj-docx-1-cmg-10.1177_26317745251392785 for Evaluating the diagnostic value of double-balloon enteroscopy combined with endoscopic ultrasonography for primary small intestinal lymphoma by Liu Zhongcheng, Su Ning, Lou Xiaoying, Li Wenru, Xiao Ting, Wu Haili, Wang Zhiyue, Chen Meiying, Zhang Yan and Guo Qin in Therapeutic Advances in Gastrointestinal Endoscopy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
