Abstract
Background and Purpose
Cerebral venous thrombosis (CVT) is a rare form of stroke. The objective of this study was to investigate the characteristics, outcomes, and prognostic factors of CVT patients.
Methods
This is a five-year retrospective study of CVT patients admitted to our Hospital from April 2016 to April 2021. Medical records were accessed to confirm the diagnosis and obtain the clinical, imaging, laboratory, and treatment data. Further, outcomes of the patients at three months were collected. Binary logistic regression was used to analyse the prognostic factors.
Results
Seventy-three patients with CVT were analysed (35.6% male; mean age, 44.3 years). A poor outcome (modified Rankin scale score, 3–6) was 17.8% of patients, and mortality was 10.9%. Factors associated with poor outcomes in all patients with CVT included the duration of presenting symptoms <24 hours (OR: 3.95, 95% CI: 1.13–13.82), age ≥ 50 years (OR: 8.12, 95% CI: 2.16–30.71), a non-fasting glucose level of ≥ 200 mg/dL (OR: 9.58, 95% CI: 1.90–48.25), impaired consciousness (Glasgow coma scale score, 3–8) (OR: 5.63, 95% CI: 1.39–22.81), male sex (OR: 3.73, 95% CI: 1.07–12.98), and brain herniation (midline shift ≥ 5 mm) (OR: 8.75, 95% CI: 1.94–39.57). Headache and use of an oral contraceptive pill were associated with good outcomes in all of the patients.
Conclusion
The non-fasting glucose level, a modifiable prognostic factor, exhibited the highest correlation with poor outcomes in CVT patients.
Introduction
Cerebral venous thrombosis (CVT) is an uncommon form of stroke. 1 Clinical characteristics of CVT include headache, seizure, focal neurological deficit, and impaired consciousness. 2 There are many precipitating factors of CVT, including oral contraceptive pill (OCP) usage, intra- and paracranial infection, pregnancy, malignancy, and hematologic and autoimmune diseases. 3 CVT is caused by thrombosis of cerebral veins or sinuses, which leads to increased intracranial pressure and occurrence of venous infarction, and might result in cerebral edema and intracerebral hemorrhage (ICH). Shakibajahromi et al. found that 22.4% of patients with CVT had poor outcomes, and 5.2% died. 4 Recent studies have found various prognostic factors related to outcomes in CVT, which include sex, age, impaired consciousness, neurological deficit, seizure, ICH, brain herniation, and intra- and paracranial infection.2–11 However, some results of these studies were inconclusive. In 2004, Ferro et al. found that central nervous system (CNS) infection was associated with poor outcomes, whereas Shakibajahromi et al. found no such association.4, 5 Moreover, there is limited data on the prognostic factors of CVT in Thai patients.
The non-fasting glucose level at admission is a simple, useful, and widely accessible test used to evaluate patients with stroke; it is associated with stroke outcomes.12–15 In patients with non-lacunar stroke, Bruno et al. 12 found an association between a higher admission blood glucose level and worse three-month outcomes. Stead et al. 13 found that hyperglycemia at the onset of non-diabetic acute ischemic stroke was linked with notably more severe results. Zonneveld et al. 14 determined that hyperglycemia at admission was related to post-stroke infection and a worse functional outcome following non-diabetic acute ischemic stroke. According to Snarska et al., 15 hyperglycemia at admission was associated with a worse clinical prognosis and a greater risk of in-hospital mortality in patients with ischemic, and hemorrhagic stroke. However, the data associating non-fasting glucose levels at admission with outcomes in patients with CVT are limited. To the best of our knowledge, only one study has investigated this relationship and found that hyperglycemia at admission is a strong predictor of poor clinical outcomes in patients with CVT. 16
Thus, the objective of this study was to investigate the clinical characteristics, outcomes, and prognostic factors of CVT. Prognostic factors included signs and symptoms, precipitating factors, laboratory findings (including non-fasting glucose level, complete blood count, and coagulogram), radiological findings, and treatment.
Methods
Study Design and Patients
We conducted a retrospective study on patients with CVT who were admitted from April 2016 to April 2021 at our Hospital, a 900-bed regional hospital. All admission medical records with International Classification of Diseases 10th edition diagnostic codes of G08, O22.5, I67.6, I63.6, and O87.3 were searched. 17 We included patients who had a definitive diagnosis of CVT using standard neuroimaging (magnetic resonance venography or computed tomographic venography) that was confirmed by certified radiologists. Patients with incomplete medical records were excluded from the study.
Data Collection
The data collected included demographic data, underlying disease, signs and symptoms at presentation, precipitating factors, laboratory and radiological findings, treatment, and outcomes. Data collected on neurological signs and symptoms at presentation included duration of presenting symptoms, blood pressure, headache, nausea/vomiting, impaired consciousness (Glasgow coma scale [GCS] score, 3–8), seizure, hemiparesis/hemiplegia, and hemisensory loss. Data on the precipitating factors included pregnancy, use of OCP, malignancy, antiphospholipid syndrome, intra- or paracranial infection, nephrotic syndrome, antithrombin III deficiency (<79% of normal activity), protein C deficiency (<70% of normal activity), and protein S deficiency (<60% of normal activity). Laboratory findings included non-fasting glucose levels at admission and data obtained by complete blood count and coagulogram. Non-fasting glucose at admission was measured using a point-of-care glucose test of capillary blood at the emergency department or on the ward within 24 hours of admission. Radiological findings included the location of the thrombosis, number of thrombosis sites, brain herniation, and ICH. Treatment-related data included decompressive surgery, anticoagulant use, and time from door to the first anticoagulant dose. Finally, outcomes were measured three months after hospital discharge, evaluated using the modified Rankin scale (mRS), and divided into good (mRS score 0–2) and poor (mRS score 3–6).
Sample Size
The sample size was calculated to assess the percentage of patients with CVT who had poor outcomes. Previous reports found that 22.4% of patients with CVT had a poor outcome. 4 The sample size calculation used an estimation of an infinite population proportion with an alpha level of 0.05, a delta level of 0.1, and a Z level of 1.96. This resulted in an estimated study size of 67 patients with CVT.
Statistical Analysis
Data analysis was performed using SPSS (version 19; IBM, Armonk, NY). Continuous variables are presented as mean and standard deviation (SD), whereas categorical variables are presented as numbers and percentages. The independent paired t-test, chi-squared test, and Fisher’s exact test were used to compare the differences between the groups. Binary logistic regression was used to analyse the prognostic factors related to the outcomes. Statistical significance was set at p < .05.
Results
Among the 87 CVT patients in our database, 14 had incomplete data. Consequently, we conducted an analysis using information from 73 CVT patients, with 35.6% being male (see Table 1). The mean (SD) age was 44.3 (13.9) years. The common symptoms and signs included headache (71.2%), seizure (60.3%), and hemiparesis/hemiplegia (49.3%). Common precipitating factors included protein S deficiency (68.5%), OCP use (37.0%), and antithrombin III deficiency (30.2%). There were 10 patients (13.7%) with intracranial/paracranial infection-related CVT. Of these, 6 patients experienced a good outcome, while four had a poor outcome. Common locations of thrombosis included the superior sagittal sinus (69.9%), right transverse sinus (42.5%), and right sigmoid sinus (28.8%). Within the group affected by deep CVT, all five patients with internal cerebral vein thrombosis had a good outcome. Among the five patients with a vein of Galen thrombosis, four experienced a good outcome, while one had a poor outcome. Moreover, 58.9% of the patients had ≥ 3 sites of thrombosis. ICH was present in 58.9% of patients, whereas brain herniation (midline shift ≥ 5 mm) was observed in 12.3%. A poor outcome was seen in 17.8% of the patients, and 10.9% of patients died. Using binary logistic regression, we found that six factors were associated with poor outcomes in all patients with CVT (Table 2) including
Duration of presenting symptoms < 24 hours Age ≥ 50 years Non-fasting glucose level ≥ 200 mg/dL Impaired consciousness (GCS score, 3–8) Sex (male) Herniation (Midline shift ≥ 5 mm)
Characteristics of 73 Patients with Cerebral Venous Thrombosis.
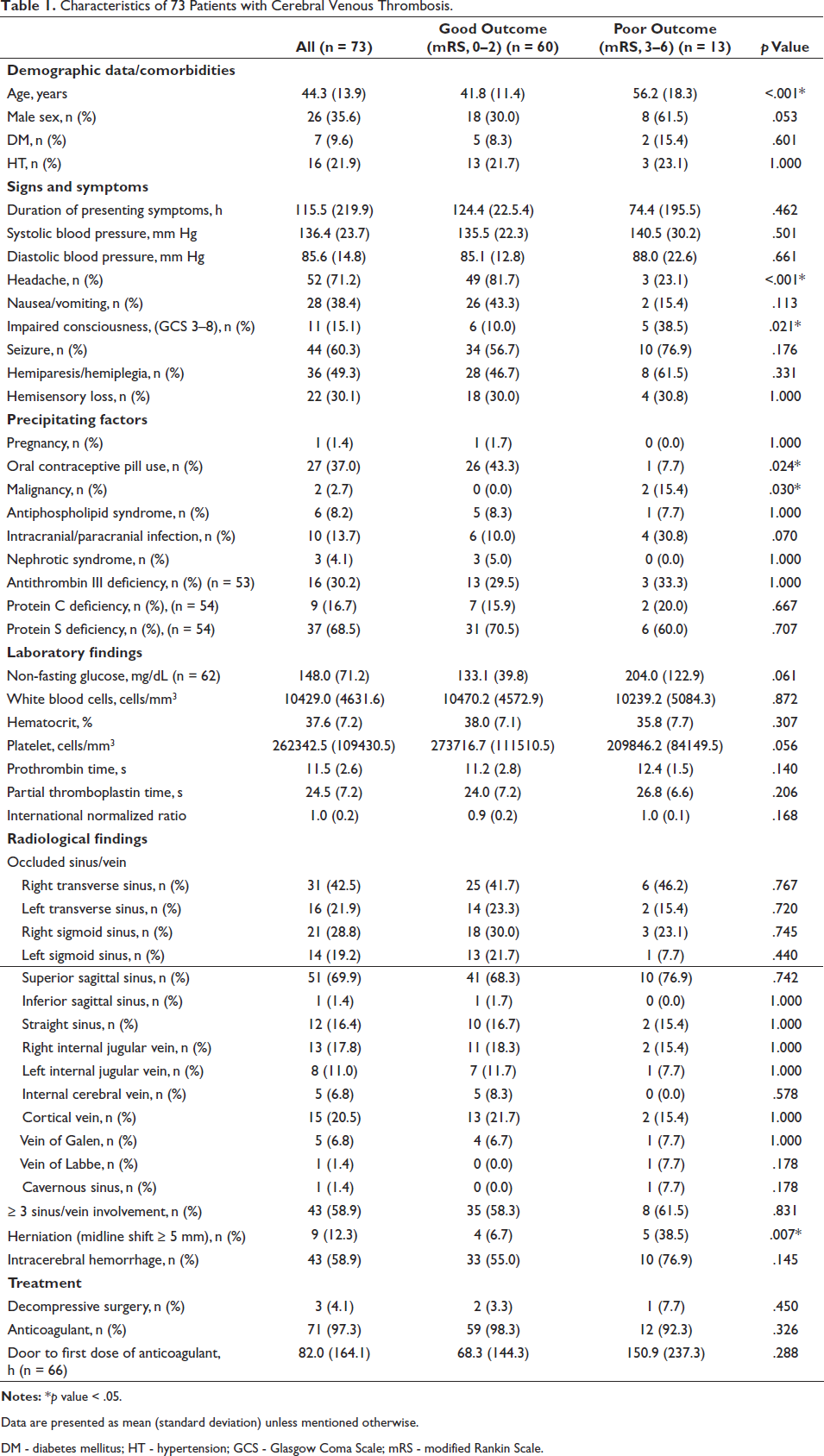
Data are presented as mean (standard deviation) unless mentioned otherwise.
DM - diabetes mellitus; HT - hypertension; GCS - Glasgow Coma Scale; mRS - modified Rankin Scale.
Factors Associated with Poor Outcomes in Patients with Cerebral Venous Thrombosis.
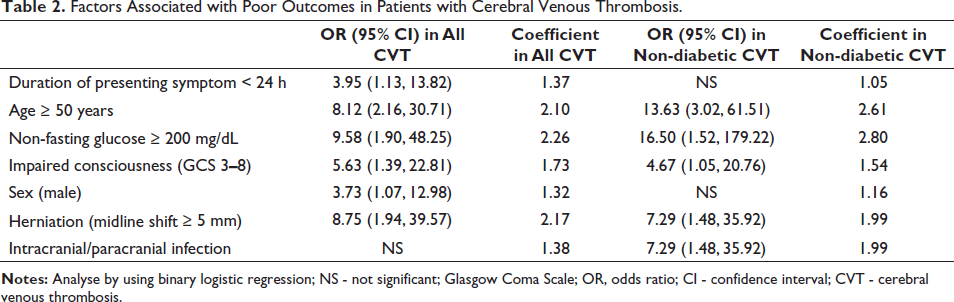
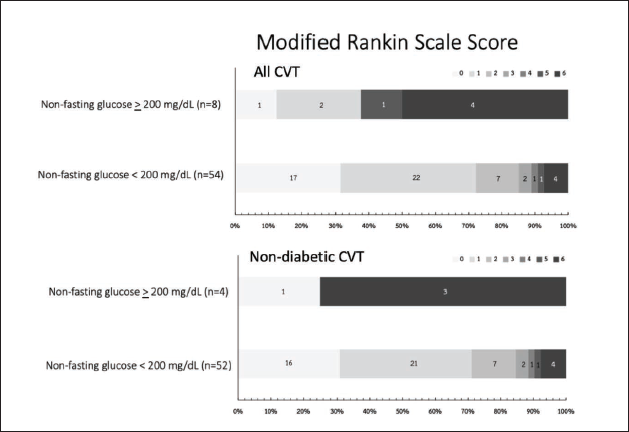
We found an association between poor outcomes and severe hyperglycemia (Table 2, Figure 1), which might be due to underlying diabetes mellitus. Therefore, we excluded patients with diabetes to evaluate the effect of non-fasting glucose levels in patients with CVT and without diabetes. In these patients (n = 66), factors associated with poor outcomes (Table 2) included age ≥ 50 years, non-fasting glucose levels ≥ 200 mg/dL, impaired consciousness (GCS core 3–8), brain herniation, and intra- or paracranial infection (Table 2, Figure 1). Headache and OCP use were associated with good outcomes in all the patients with CVT and those with CVT and without diabetes (Table 3). For the treatment, decompressive surgery was performed in 4.1% of patients. Anticoagulant therapy was administered to 97.3 % of patients. Only two patients did not receive anticoagulants because of CNS infection-related CVT.
Factors Associated with Good Outcomes in Patients with Cerebral Venous Thrombosis.
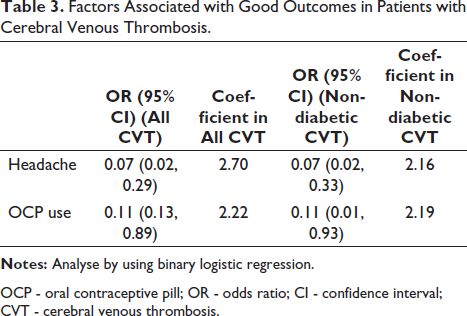
OCP - oral contraceptive pill; OR - odds ratio; CI - confidence interval; CVT - cerebral venous thrombosis.
Discussion
In our study, demographic characteristics, signs and symptoms, radiological findings, treatment, and outcomes were similar to those reported previously.2–11 However, our results showed that 68.5% of the patients with CVT had protein S deficiency, which was higher than that previously reported by Karsy et al. 8 (10.7%) and Pan et al. 9 (55.5%). We provide two explanations for the difference in the percentage of patients with a protein S deficiency: (a) differences in genetic predispositions, or (b) the protein S level was obtained at the time of CVT diagnosis, before the initiation of anticoagulant treatment; therefore, the lower protein S level might be attributed to the effects of acute thrombosis. Additionally, due to the retrospective study design, most patients with a low protein S level did not have their results retested after withholding anticoagulants. For antithrombin III deficiency, our data were comparable to those documented in a previous report in Thailand by Rojanapitayakorn 18 (30.2% vs. 40%). This might be explained by the similarity in the genetic predisposition of the patients included in the study. Twenty-seven patients had CVT related to OCP use. Among them, three patients were older than 50 years. Compared to a previous report in China, 9 our study revealed nearly four times the proportion of CVT-related OCP use (37% vs. 9.5%). This might indicate that some patients used OCPs without any indication—OCPs are available over-the-counter in Thailand without a physician’s prescription, potentially leading to OCP overuse. Headache and OCP use were associated with good outcomes. Patients with CVT with headaches usually did not have impaired consciousness, and according to the study by Aarju et al., headaches in CVT might be associated with isolated intracranial hypertension, which was associated with a good outcome. 11 Further, OCP use induced thrombosis by increasing factor VII, factor X, and fibrinogen levels, which might be associated with the good outcome observed in our study since it is a precipitating factor that can be reversed by stopping OCP use, unlike some kinds of malignancies or genetic prothrombotic factors. Most prognostic factors related to poor outcomes have been described in previous reports,2–11, 16 except for the duration of the symptoms. We found that the duration of presenting symptoms of < 24 hours was associated with poor outcomes. This may be due to the presence of a more severe disease leading to patients with CVT presenting with symptoms earlier.
Our study also demonstrated that a non-fasting glucose level of ≥ 200 mg/dL at admission was associated with poor outcomes at three months in all patients with CVT (Table 2, Figure 1). We set the cutoff point for hyperglycemia at 200 mg/dL because this level is associated with hyperglycemic emergencies, including diabetic ketoacidosis. In 2012, Zuurbier et al. 16 found that the non-fasting glucose level at admission was associated with poor outcomes in non-diabetic patients with CVT; however, they did not analyse the data of all patients with CVT. In 2020, Wu et al. 19 found that an elevated fasting glucose level was a predictor of poor outcomes in patients without diabetes who had CVT. The evidence from this and the previously mentioned studies indicate that blood glucose level is associated with the outcome of patients with CVT. We posit four explanations for this association: (a) The severity of CVT activates the hypothalamic–pituitary–adrenal axis, resulting in cortisol release and consequently causing hyperglycemia; (b) Hyperglycemia promotes thrombosis progression, as supported by the Hermanides et al. 20 clinical study and pathophysiological processes that hyperglycemia induces coagulation via upregulation of tissue factor, endothelial glycocalyx damage, increased plasminogen activator inhibitor-1, increased von Willebrand factor, decreased protein C level, and increased oxidative stress.21–27; (c) hyperglycemia may lead to increased severity of CVT by exacerbating brain edema and ICH;28, 29 and (d) these three processes occur in a vicious cycle. Fortunately, blood glucose is an adjustable factor that can be managed with oral hypoglycemic medication or insulin treatment. The identification and management of hyperglycemia in CVT patients with or without diabetes would thus likely lead to a more positive prognosis. Our study has limitations, such as the small sample size, single-center design, and retrospective study design, which may introduce bias and errors. Future prospective research conducted at multiple centers, involving a larger number of patients, should investigate the impact of strict glucose control in hospitals on outcomes for CVT patients. The utilization of multivariate logistic analysis could potentially enhance the accuracy of the association.
Footnotes
Acknowledgment
The abstract of this manuscript was presented at the 39th Annual Meeting of the Royal College of Physicians of Thailand, held April 21-23, 2023, in Pattaya City, Thailand.
Data Sharing Statement
We state that the data supporting the study’s results are available.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
This study was approved by the Ethics Committee of Buriram Hospital (Institutional Review Board: BR0032.102.1/45).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
Not applicable due to the retrospective study design.
