Abstract
Background:
Cerebral venous thrombosis (CVT) is an uncommon cause of stroke in young adults. We aimed to determine the impact of age, gender and risk factors (including sex-specific) on CVT onset.
Methods:
We used data from the BEAST (Biorepository to Establish the Aetiology of Sinovenous Thrombosis), a multicentre multinational prospective observational study on CVT. Composite factors analysis (CFA) was performed to determine the impact on the age of CVT onset in males and females.
Results:
A total of 1309 CVT patients (75.3% females) aged ⩾18 years were recruited. The overall median (IQR-interquartile range) age for males and females was 46 (35–58) years and 37 (28–47) years (p < 0.001), respectively. However, the presence of antibiotic-requiring sepsis (p = 0.03, 95% CI 27–47 years) among males and gender-specific risk factors like pregnancy (p < 0.001, 95% CI 29–34 years), puerperium (p < 0.001, 95% CI 26–34 years) and oral contraceptive use (p < 0.001, 95% CI 33–36 years) were significantly associated with earlier onset of CVT among females. CFA demonstrated a significantly earlier onset of CVT in females, ~12 years younger, in those with multiple (⩾1) compared to ‘0’ risk factors (p < 0.001, 95% CI 32–35 years).
Conclusions:
Women suffer CVT 9 years earlier in comparison to men. Female patients with multiple (⩾1) risk factors suffer CVT ~12 years earlier compared to those with no identifiable risk factors.
What is the current knowledge on CVT?
Cerebral venous thrombosis (CVT) is an unusual cause of stroke, primarily affecting women and with significant mortality and morbidity. However, detailed differences between genders and their risk factors have not been well studied primarily because of the rarity of this condition.
What question did this study address?
What is the age of onset of CVT among genders, and how do established risk factors impact this?
What does this study add to existing knowledge?
The presence of multiple risk factors, specifically gender-specific risk factors, exposes (particularly female patients) to a significantly earlier age of CVT onset by approximately 12 years than women with no identifiable risk factors.
How might this potentially impact the practice of neurology?
Females with established (particularly gender-specific) risk factors should promptly raise concerns regarding a CVT diagnosis following appropriate symptoms. Managing those factors will be important in reducing risk.
Introduction
Cerebral venous thrombosis (CVT) is an uncommon cause of stroke caused by blood clotting in the venous sinuses resulting in impaired venous drainage and raised intracranial pressure, potentially leading to significant morbidity and mortality.1,2 CVT accounts for 0.5%–1% of all stroke and occurs three times more commonly in females and more frequently in young adults, although the number of patients studied for these data have been limited.3,4 In high-income regions such as Europe and Australia, yearly CVT incidence is about 1–2 per 100,000 population.5,6 Moreover, the disease may remain underdiagnosed if clinical correlation or apparent findings on routine neuroimaging do not expedite diagnostic venography.3,7–9
Commonly known risk factors for CVT include pregnancy and puerperium, oral contraceptive use, hormonal imbalance, especially thyroid gland disease, malignancy, connective tissue disorder, severe dehydration, head trauma and neurosurgery.5,8–10 In about 50% of CVT patients, more than one associated risk factor may be present, while 15% of cases are of unknown aetiology.4,9–11 Nevertheless, inherited and acquired thrombophilia like antithrombin deficiency, protein C and S deficiency, mutation of factor V Leiden or prothrombin gene and antiphospholipid antibodies are risk factors of CVT. Furthermore, myeloproliferative neoplasms and prolonged immobilisation may also increase CVT risk.7–12
Diagnosis of CVT can be challenging, requiring a high index of clinical suspicion due to the diversity of risk factors, variable clinical presentations and multiple comorbidities.11–15 While risk factors have been assessed in CVT,1,3,6,9–11 studies seldom evaluated their effect on age differences on CVT onset.
We aimed to analyse data from the multinational BEAST (Biorepository to Establish the Aetiology of Sinovenous Thrombosis) repository to determine the impacts of age, sex and risk factors including gender-specific risk factors on CVT onset.
Methods
The BEAST study: The BEAST protocol has been published in detail elsewhere. 7 However, briefly; the international BEAST consortium is a multicentre prospective observational study and has recruited extensive phenotypic clinical data and DNA samples from CVT populations aged ⩾18 years from 11 different tertiary care centres located in Finland, Sweden, Greece, Italy, Portugal, UK, Belgium, France, Netherlands, Mexico and USA (white non-Hispanic). The study recruited between 2000 and 2018. Age at CVT onset and extensive haematological investigations, including thrombophilia testing (antithrombin deficiency, protein C and S deficiency, lupus anticoagulant, factor VIII mutation, factor V Leiden and prothrombin G20210A mutation and plasma homocysteine levels) were obtained on hospital admission. The diagnosis of CVT for BEAST was confirmed by angiography, either conventional, computed tomography venography (CTV), magnetic resonance (MR) imaging or dedicated venography, as previously described. 7
The study was granted ethical approval from all participating institutions from local institutional review boards. Informed written consent was obtained for all patients, and data was encrypted. Established risk factors for CVT from the BEAST data set were analysed based on the presence or absence of each risk factor in the male and female populations separately.
Statistical analysis
Results were analysed using SPSS V25.0 statistical software for windows, and descriptive statistics were summarised using median with interquartile range (IQR) for continuous variables and proportion for categorical variables. The Chi-square and Mann-Whitney U tests were used for the single-factor analysis of categorical and continuous variables, respectively. A Box and Whisker plot was utilised to demonstrate the median age distribution of CVT onset; the Mann-Whitney U test determined the significance of age difference among the study groups. A composite factor analysis (CFA) was undertaken to assess the effect of multiple risk factors on the age of CVT onset. CFA is a clustering procedure where potential risk factors are classified into two groups (‘0’ and ‘⩾1’ – based on the number of significant risk factors) to observe the population distribution and age of CVT onset. Statistical significance was defined as p ⩽ 0.05 throughout.
Results
The BEAST study recruited 1309 CVT patients (males n = 323; females n = 986), and the overall median (IQR-Interquartile Range) age of CVT onset was 46 (35–58) and 37 (28–47) years for males and females, respectively, (p < 0.001) (Figure 1). Approximately 50% of the CVT population initially tested positive for lupus anticoagulant, protein C and S deficiency, antithrombin deficiency, factor V Leiden mutation and prothrombin gene mutation in both males and females. Although positive thrombophilia cases developed CVT earlier than those who tested negative, age differences were not significant (p > 0.05) between positive and negative cases in both males and females (Tables 1 and 2).
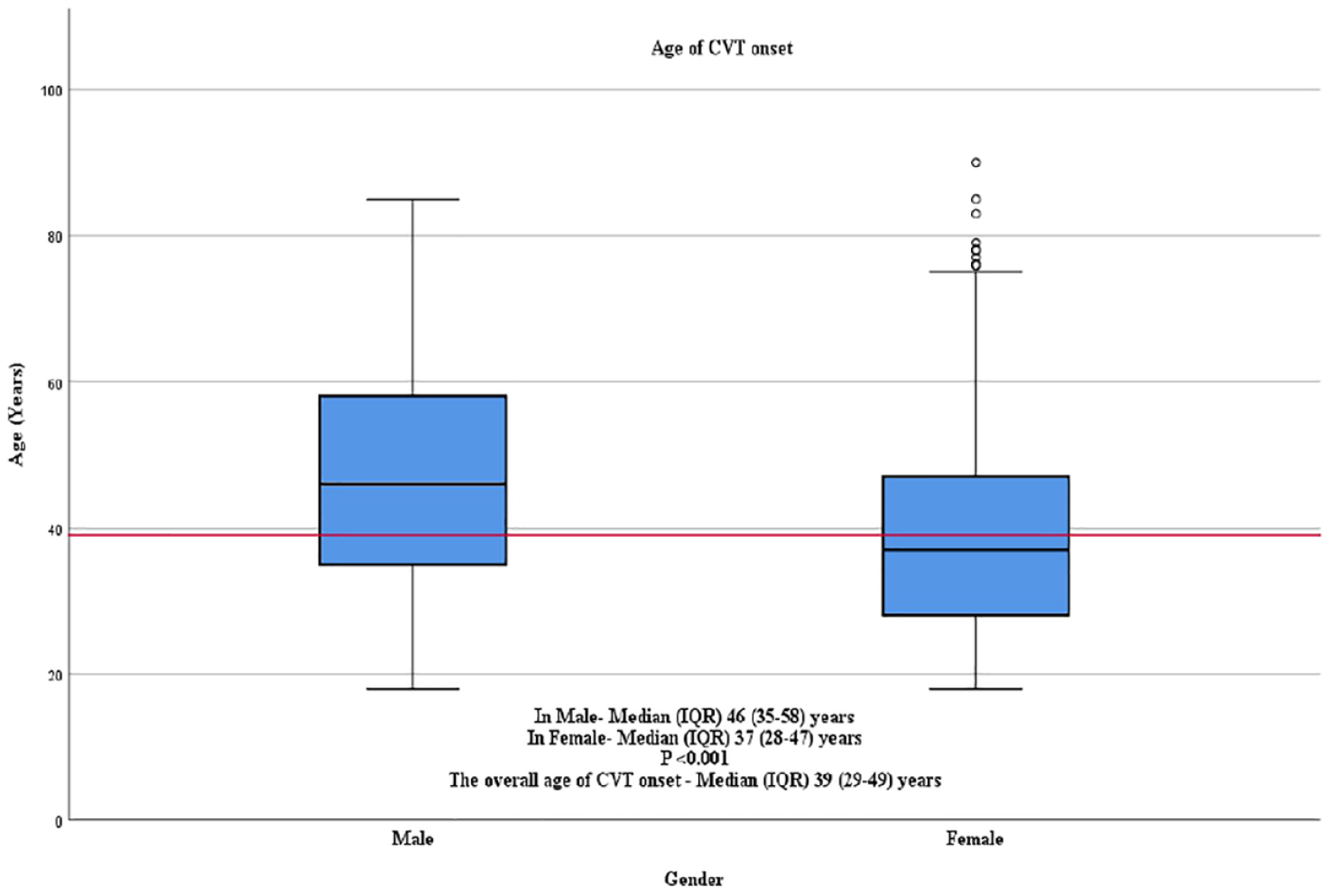
Box and Whisker plot demonstrating age distribution among the study population; Red line represents the overall median age of CVT onset.
Distribution of risk factors among the male population (N = 323).
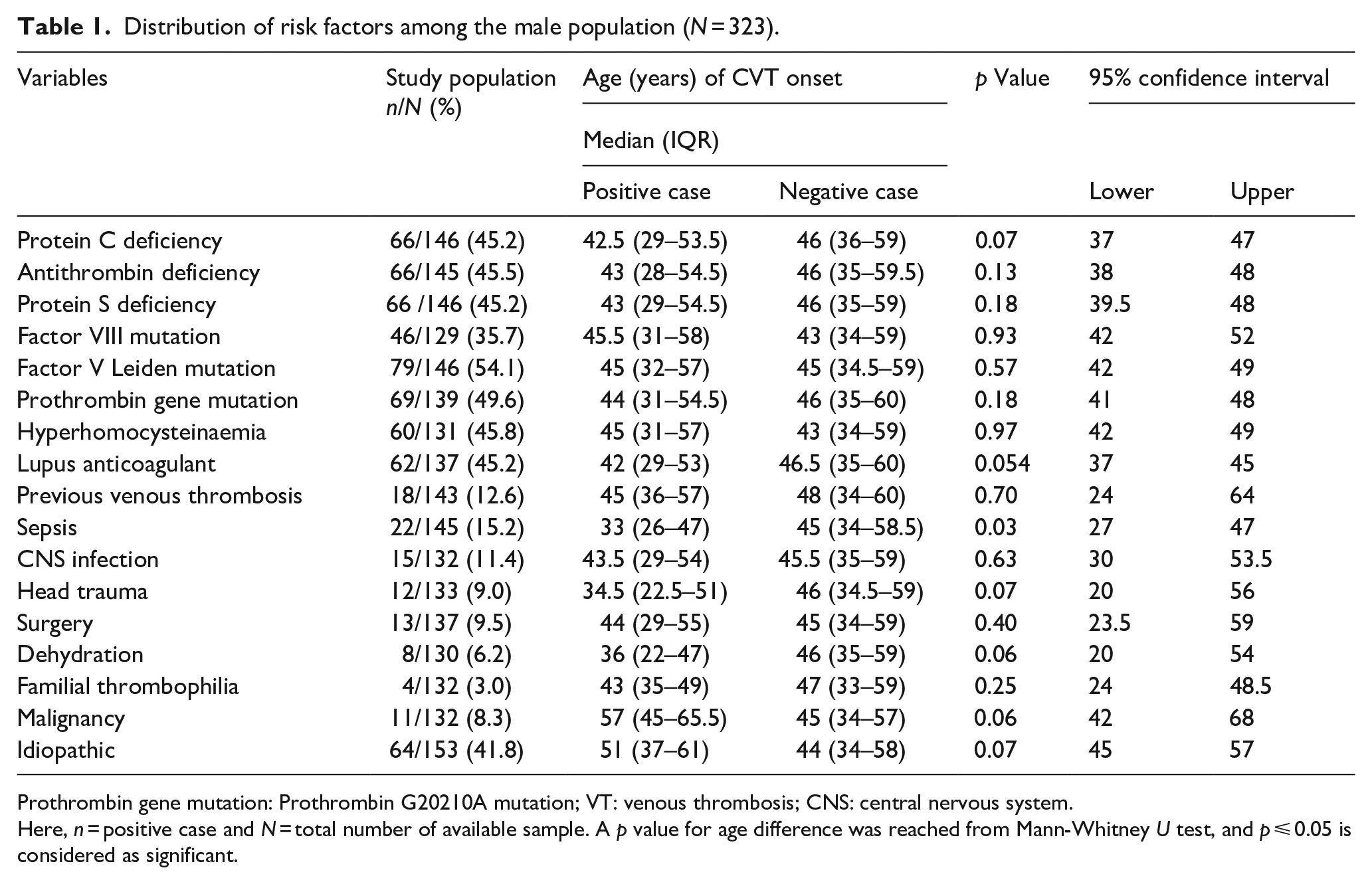
Prothrombin gene mutation: Prothrombin G20210A mutation; VT: venous thrombosis; CNS: central nervous system.
Here, n = positive case and N = total number of available sample. A p value for age difference was reached from Mann-Whitney U test, and p ⩽ 0.05 is considered as significant.
Distribution of risk factors among the female population (N = 986).
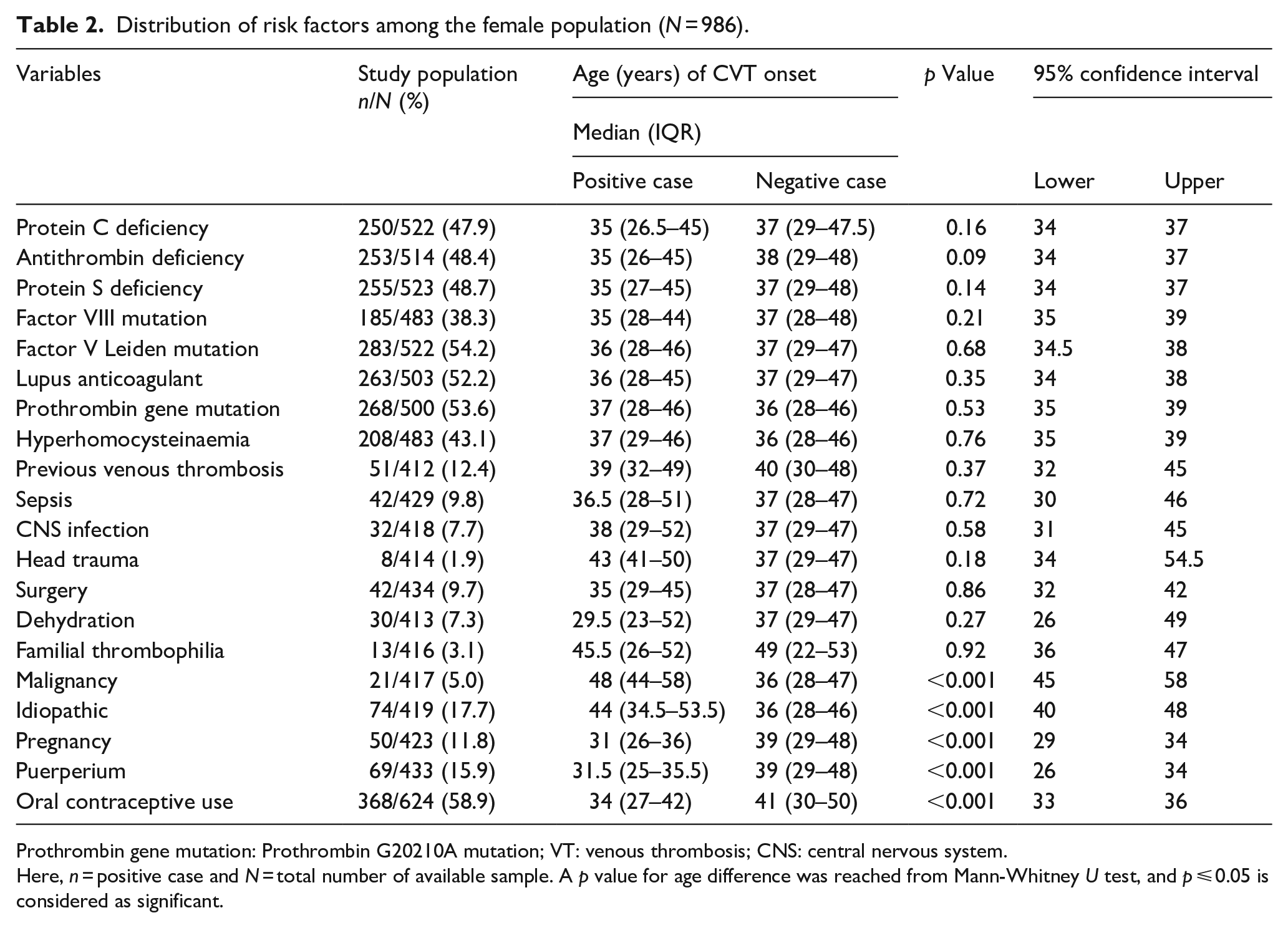
Prothrombin gene mutation: Prothrombin G20210A mutation; VT: venous thrombosis; CNS: central nervous system.
Here, n = positive case and N = total number of available sample. A p value for age difference was reached from Mann-Whitney U test, and p ⩽ 0.05 is considered as significant.
In males, the most common risk factors associated with CVT were previous venous thrombosis (~13%), antibiotic requiring sepsis (~15%), CNS infection (~11%) and head trauma (9%). However, the age of CVT onset between the presence or absence of risk factors was insignificant (p > 0.05) in males except for systemic infection (p = 0.03), where the median (IQR) age was 33 (26–47) compared to 45 (34–58.5) to those without systemic infection (Table 1).
For females, oral contraceptive use was most prevalent (~60%), followed by puerperium (~16%), pregnancy (~12%), previous venous thrombosis (~12%) and sepsis (~10%) (Table 2). However, significant age differences were only observed for malignancy (p < 0.001) and gender-specific risk factors (GSRF) like pregnancy (p < 0.001), puerperium (p < 0.001) and oral contraceptive use (p < 0.001) between positive and negative cases among the female population. Cases with risk factors, that is, CVT with malignancy and idiopathic factors, were found in older age groups compared to those with no risk factors, in males (57 years vs 45 years; p = 0.06, and 51 years vs 44 years; p = 0.07) but significantly in females (48 years vs 36 years; p < 0.001, and 44 years vs 36 years; p < 0.001) populations, respectively (Tables 1 and 2).
Composite factor analysis (CFA) based on significant (p ⩽ 0.05) risk factors (generalised model) and gender-specific risk factors (GSRF model) recruited from Table 2 observed that ~72%, and ~60% of females had had multiple risk factors in the generalised and GSRF CFA model, respectively. Furthermore, both generalised and GSRF-based CFA found that patients with multiple risk factors have CVT significantly (p < 0.001) earlier than those with no identified risk factor group (Table 3).
Impact of multiple risk factors on the age of CVT onset among the female population based on Composite Factor Analysis (CFA).
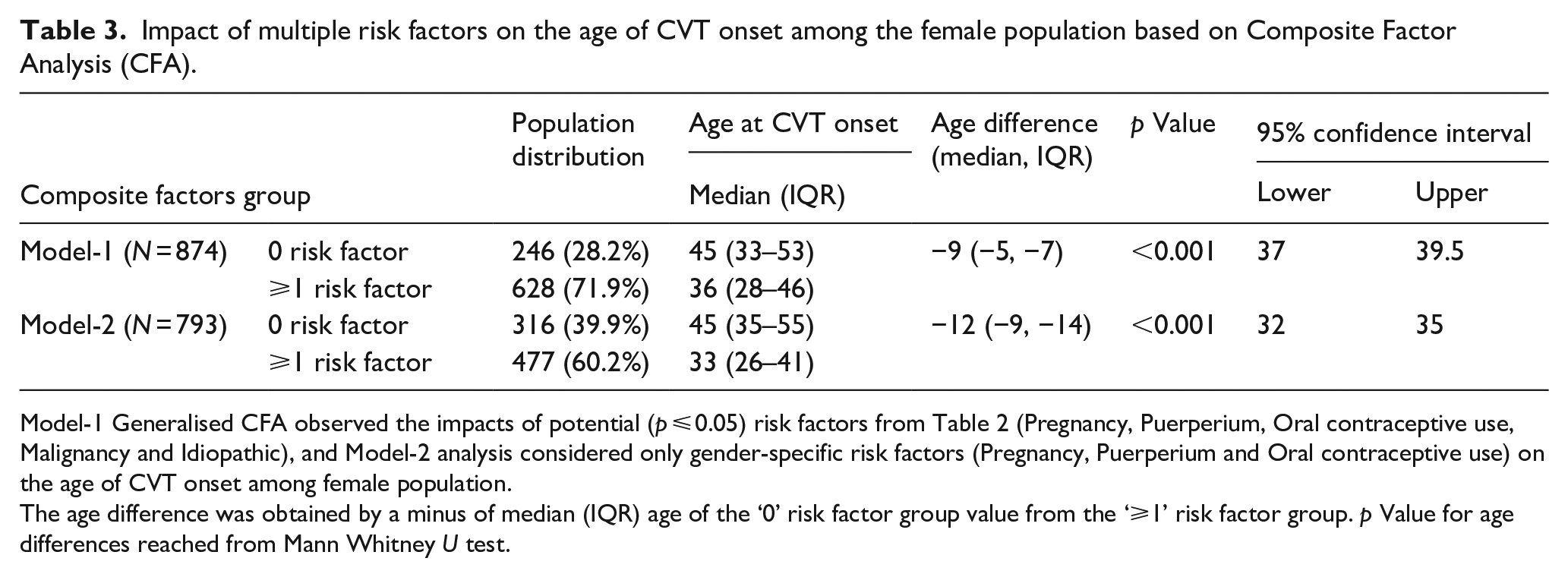
Model-1 Generalised CFA observed the impacts of potential (p ⩽ 0.05) risk factors from Table 2 (Pregnancy, Puerperium, Oral contraceptive use, Malignancy and Idiopathic), and Model-2 analysis considered only gender-specific risk factors (Pregnancy, Puerperium and Oral contraceptive use) on the age of CVT onset among female population.
The age difference was obtained by a minus of median (IQR) age of the ‘0’ risk factor group value from the ‘⩾1’ risk factor group. p Value for age differences reached from Mann Whitney U test.
Following a quality control subgroup analysis for missing and non-missing values, we found no significant differences in the distribution of risk factors and age difference in CVT onset between missing and non-missing cases for each risk factor (Supplemental Table 1).
Discussion
We present a multicentre international study on 1309 CVT patients and confirm that females suffer CVT ~9 years younger than their male counterparts. Females with gender-specific risk factors such as pregnancy puerperium and oral contraceptive use suffered CVT significantly earlier (~12 years) than those with no risk factors. In our study population, females were affected three times more commonly compared to males.
Most cases in our study population were female (~75%), as previously recognised.3,8–10 However, Coutinho et al. 11 also observed that females were significantly (p < 0.001) younger compared to males, 34 (25–47) years versus 42 (33–57) years, respectively, although that study was smaller and from a limited geographical region. In a recent small Iranian study, Shakibajahromi et al. 14 also found that 73.6% of CVT patients were female, and the overall age was ~38 years. However, in a South Asian study, Wasay et al., 15 Kalita et al. 16 and Narayan et al. 17 observed that ~59%, ~52% and ~46% of patients were females, and the overall age of CVT onset was 31, 29 and ~31.3 years, respectively. Indian and South Asian patients suffered CVT at an earlier age with a lower prevalence of affected females compared to European study findings, possibly due to sampling bias and sociocultural barriers associated with access to treatment.15,18–22
We find that females are affected by CVT significantly earlier than males and prominently in reproductive age because of a higher number of familial thrombophilia and gender-specific factors, a result supported by existing, albeit small published literature.8–13,23–25 In a European study, sepsis (21%), malignancies (11%) and trauma (8%) were higher among men, which is comparable to our study results. 11 Moreover, Coutinho et al. 11 also observed that septic sinus thrombosis commonly develops in males because of a greater incidence of ear, nose and throat infections among males. In a recent study, Shakibajahromi et al. 14 observed that ~75% of female CVT patients were associated with pregnancy, postpartum period or OCP use, similar to a previous study findings (~65% female) 11 but significantly higher than our current study. Furthermore, studies from Mexico26–28 find lower prevalence of oral contraceptive use (18%–20%) but a higher (~30%–47%) association of pregnancy and puerperium with CVT onset, similar to a Romanian study where the authors found ~38% of the CVT cases were associated with GSRF. 29 Although several previous studies have identified CVT risk factors,11,14–17,30 these did not evaluate the impacts of multiple risk factors versus no identifiable risk factor on the age of CVT onset. We report that females presenting with multiple (⩾1) risk factors suffered CVT 12 years earlier, which is the characteristic finding of this current study.
We present a large multicentre international study on CVT, but despite its sample size, some limitations need to be acknowledged. This observational study evaluates only adult CVT patients, so our results are not applicable to children. Although participants’ ancestry was primarily based on self-identification, previous studies have demonstrated that self-identification of origin is reliable.8,9 Not all centres measured every blood test, and lab testing variability at different institutions may lead to bias between reporting study centres. However, the distribution of the thrombophilia abnormality was broadly consistent with existing papers among European populations. Although we demonstrate a significant association between age, sex and risk factors among the CVT population, our study was not designed to determine its pathophysiological mechanism. Further, there is a risk of selection bias as patients were included based on the availability of DNA samples, although all patients approached consented. Finally, we did not analyse treatment or prognostic outcomes for which a longer-term follow-up study would be required.
Conclusion
Women of reproductive age suffer CVT ~9 years earlier than men. Moreover, compared to those with no risk factors, females with multiple established (particularly gender-specific) factors suffer from an earlier onset of CVT by up to 12 years.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873221148267 – Supplemental material for Age of onset of cerebral venous thrombosis: the BEAST study
Supplemental material, sj-pdf-1-eso-10.1177_23969873221148267 for Age of onset of cerebral venous thrombosis: the BEAST study by Redoy Ranjan, Gie Ken-Dror, Ida Martinelli, Elvira Grandone, Sini Hiltunen, Erik Lindgren, Maurizio Margaglione, Veronique Le Cam Duchez, Aude Bagan Triquenot, Marialuisa Zedde, Michelangelo Mancuso, Ynte M Ruigrok, Brad Worrall, Jennifer J Majersik, Jukka Putaala, Elena Haapaniemi, Susanna M Zuurbier, Matthijs C Brouwer, Serena M Passamonti, Maria Abbattista, Paolo Bucciarelli, Robin Lemmens, Emanuela Pappalardo, Paolo Costa, Marina Colombi, Diana Aguiar de Sousa, Sofia Rodrigues, Patrícia Canhao, Aleksander Tkach, Rosa Santacroce, Giovanni Favuzzi, Antonio Arauz, Donatella Colaizzo, Kostas Spengos, Amanda Hodge, Reina Ditta, Thang S Han, Alessandro Pezzini, Jonathan M Coutinho, Vincent Thijs, Katarina Jood, Turgut Tatlisumak, José M Ferro and Pankaj Sharma in European Stroke Journal
Footnotes
Acknowledgements
This study utilised the Bio-Repository to Establish the Aetiology of Sinovenous Thrombosis (BEAST) data, and we are grateful to the patients for their participation and collaboration.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants awarded to P.S. from the Stroke Association (UK) and the Dowager Countess Eleanor Peel Trust (UK). P.S. was funded by a Department of Health (UK) Senior Fellowship at Imperial College London for part of this study. The cohort of 231 French cases was constituted during a hospital protocol of clinical research approved by the French Ministry of Health; the biological collection was kept and managed by INSERM CIC-CRB 1404, F-76000 Rouen, France. The controls from Belgium were genotyped as part of the SIGN study. R.L. is a senior clinical investigator of FWO Flanders. T.T. is the recipient of funding from the Sigrid Juselius Foundation (Finland), Helsinki University Central Hospital (Finland), Sahlgrenska University Hospital (Sweden) and the University of Gothenburg (Sweden). The Swedish Research Council (2018-02543) and the Swedish Heart and Lung Foundation (20190203) also supported the study. J.M.C. has received funding from the Dutch Thrombosis Foundation. This material results from work supported by resources and the use of facilities at the VA Maryland Health Care System, Baltimore, Maryland, and was also supported in part by the National Institutes of Health (U01NS069208, R01NS105150 and R01NS100178).
Ethical approval
Ethical approval for this study was obtained from Local/Institutional Ethics/Research Boards in each centre and country.
Informed consent
Written informed consent was obtained from all subjects prior to recruitment.
Guarantor
PS
Contributorship
PS conceived the overall study and directed the work. RR undertook the initial analysis and wrote the first draft. GKD oversaw statistical analysis. RR, GKD, IM, EG, SH, EL, MM, VLCD, ABT, MZ, MM, YMR, BW, JJM, JP, EH, SMZ, MCB, SMP, MA, PB, RL, EP, PC, MC, DAS, SR, PC, AT, RS, GF, AA, DC, KS, AH, RD, TSH, AP, JMC, VT, KJ, TT, JMF and PS contributed to patient recruitment, data acquisition and analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
