Abstract
Myopia is far beyond its inconvenience and represents a true, highly prevalent, sight-threatening ocular condition, especially in Asia. Without adequate interventions, the current epidemic of myopia is projected to affect 50% of the world population by 2050, becoming the leading cause of irreversible blindness. Although blurred vision, the predominant symptom of myopia, can be improved by contact lenses, glasses or refractive surgery, corrected myopia, particularly high myopia, still carries the risk of secondary blinding complications such as glaucoma, myopic maculopathy and retinal detachment, prompting the need for prevention. Epidemiological studies have reported an association between outdoor time and myopia prevention in children. The protective effect of time spent outdoors could be due to the unique characteristics (intensity, spectral distribution, temporal pattern, etc.) of sunlight that are lacking in artificial lighting. Concomitantly, studies in animal models have highlighted the efficacy of light and its components in delaying or even stopping the development of myopia and endeavoured to elucidate possible mechanisms involved in this process. In this narrative review, we (1) summarize the current knowledge concerning light modulation of ocular growth and refractive error development based on studies in human and animal models, (2) summarize potential neurobiological mechanisms involved in the effects of light on ocular growth and emmetropization and (3) highlight a potential pathway for the translational development of noninvasive light-therapy strategies for myopia prevention in children.
Introduction
Myopia results from a mismatch between the axial length of the eye and the power of its refractive components leading images to be focussed in front of the retina and causing blurred vision at distance. 1 The prevalence of myopia exhibits wide geographical variations in the world. In developed nations, the prevalence of the condition among adults ranges from 15% to 49%, 2 and rises up to approximately 69% in 15-year-olds under cycloplegia. 3 In developing countries, the rate of myopia in 15-year-old adolescents is much lower, between 14.7% and 16.2% in Colombia and 5.5% in Africa.3–5 Singapore and East Asian countries like China, Taiwan, Hong Kong and South Korea are the most affected (Figure 1(a)).6–11 While myopia prevalence ranges from 29% in 7-year-olds to 53.1% in 9-year-olds in the school-based population of the Singapore Cohort Study of Risk factors for Myopia (SCORM) (Figure 1(b)), 2 it can reach 69% in 15 years of age with 86% of affected population being Singaporean-Chinese. 3 In young adults, the prevalence of myopia is even higher with 82.3% of Chinese male military conscripts having myopia and 13.1% high myopia. This is particularly worrying as patients with high myopia [more than −5 Dioptres (D)] are at risk of developing pathologic myopia and other associated sight-threatening ocular conditions such as glaucoma, retinal detachment, myopic macular degeneration and choroidal neovascularization.12,13 Pathologic myopia is a major cause of visual impairment and blindness in Asian populations. 14 The risk of developing high myopia increases drastically with the early onset and progression,15,16 especially in Asian populations where myopia progresses faster. 17 Although the precise mechanisms of myopia onset and progression are not completely understood, it is admitted that it involves multiple genetic and environmental factors.

Average myopia prevalence: (a) in young adults of East Asian Countries during 2012–2020 and (b) in Singapore across different age groups during 1999–2001.
Genetic factors have been mainly related with the finding that children with myopia have myopic parents.7,18,19 Genome-wide association studies (GWAS) and whole-exome sequencing studies on European populations have identified over 200 genetic loci associated with refractive error and myopia.20,21 Genome-wide meta-analysis for refractive error on European and Asian cohorts revealed 24 additional loci (BICC1, BMP2, BMP3, CACNA1D, CD55, CHD7, CHRNG, CNDP2, GRIA4, CYP26A1, GJD2, KCNJ2, KCNQ5, LAMA2, MYO1D, PCCA, PRSS56, RASGRF1, RDH5, RORB, SIX6, TOX, ZIC2 and ZMAT4) associated with myopic features.22 Some of these novel loci are known to be implicated in the development of eye, ion transport, retinoic acid metabolism, neurotransmission and extracellular matrix production. 22 Conversely to the rising myopia prevalence worldwide, genetic predisposition has not significantly changed over the past few decades, which implies that environmental factors, potentially interacting with genetic traits, are mainly at the origin of the ongoing myopia epidemic. 23
Environmental factors influencing myopia onset include, but are not limited to, level of education, near work and time spent outdoors.24–28 Increased time outdoors has been emphasized as an important modifiable environmental factor for myopia control.29,30 Irrespective of physical activity, increased time outdoors is associated with a reduced odds ratio of myopia, even when children perform a high amount of near work.31,32 In addition, Donovan et al. 33 have found myopia progression to be slower during the summer, possibly because of increased outdoor exposure. The exact protective feature(s) of the outdoor environment against myopia are still unclear, but may include variations in accommodation due to uniform dioptric space, increased pupil constriction, increased retinal focus and decreased blur as well as increased spatial frequency and changes in the characteristics of light exposure. 34
While epidemiological studies can only highlight associations between bright light exposure outdoors and myopia prevention,34–38 substantial evidence from animal studies support a protective effect of bright light on experimental myopia development.39–41 In addition, interventional studies in humans have also shown a beneficial effect of both outdoor35,42 and indoor (classroom) exposure 43 to increased but not so intense light levels. According to Rucker, 44 the different patterns in luminance, but also colour contrast, has a significant effect on the refraction and accommodation. In accordance with findings in humans and animal models, a recent meta-analysis of GWAS comprising 160,420 participants of cross ethnicity (European and Asian) revealed 140 genetic associations linked with light-dependent pathways which include genes associated with novel pathways such as anterior-segment morphology (TCF7L2, VIPR2 and MAF) and angiogenesis (FLT1). Furthermore, genes involved in glutamate receptor signalling (GNB3 and CLU) and dopaminergic pathway (DRD1) were identified as key genes in the light-dependent retina-to-sclera signalling cascade potentially controlling ocular growth. 45
In this narrative review, we focus on the literature investigating light-driven modulations of ocular growth and refractive error development in humans and animal models. We will also summarize the current knowledge on neurobiological and photoreceptoral mechanisms involved in the putative effect of light against myopia onset and highlight a potential pathway for the translational development of noninvasive light-therapy strategies to halt or delay myopia onset in children.
Light exposure and myopia in humans
Time outdoors and myopia
Increased time outdoors has been shown to prevent or delay myopia onset in several studies.17,46–50 The protective impact of increased time outdoors against myopia applies even in children performing higher amounts of near work, 17 and is predominantly attributed to intermittent exposure to high levels of sunlight and independent of physical activity.29,30,35,47 Furthermore, increased outdoor time has been shown to have a protective effect on the cumulative incidence rate of myopia in children enrolled in randomized clinical trials in China and Taiwan.29,30,35 On the other hand, increased near-work time and reduced outdoor activities have been suggested to be at the origin of the increased myopia prevalence in older children. 51 In the SCORM study, however, participants who spent more time outdoors were less likely to be myopic while the amount of near work did not predict outdoor activity. Therefore, outdoor activity may be an independent factor and not merely the reciprocal of near work. 46 Interestingly, increases in refractive error progression, axial growth rates and less power loss which occur before myopia onset also seem to be influenced by reduced time spent outdoors. 50 According to Lingham et al., 52 the potential protective effect of outdoor light against myopia is most likely due to one or both of the following factors which are suboptimal in indoor lighting: (1) increased light intensity and (2) favourable spectral composition of light. Although little has been established on the involvement of the spectral composition of light on ocular growth in humans, it is interesting to note that individuals with colour vision red/green colour vision deficiency were reported to be less myopic than individuals with normal colour vision. 53
Bright light and myopia
Epidemiologic research indicates that greater average daily light exposure is associated with a reduced axial elongation during childhood. 38 A study cluster-randomized intervention-controlled trial conducted in Taiwan showed that exposure to outdoor light leads to less myopic shifts, reduced axial elongation and a 54% lower risk of myopia progression. 35 Cross-sectional studies using objective methods (wearable light sensors) to quantify illuminance have shown that Australian myopic children aged 10–15 years had lower average light exposure and lower amount of outdoor time compared with emmetropic children. 54 Furthermore, comparisons between Australian and Singaporean children aged 10–12 years showed that light exposure patterns are of shorter durations and lower intensities in Singaporean children, who tend to have a higher risk of developing myopia. 36 Short exposures of spurts of light in Singaporean children are mostly seen during the periods 9 a.m.–10:30 p.m., 12 p.m.–1 p.m., or 3 p.m.–4 p.m., possibly due to child’s travel time to their school and home in morning and evening. 55 In Australian children, the peaks in outdoor light occur at similar timings, but with greater duration of exposure per hour (10 min or more) when compared with Singaporean Children. 36 Patterns of light exposure are known to be influenced by seasons. Myopia progression and axial length elongation are slower in summer compared with winter.33,56 A study in the United States has shown that children aged 7.6 ± 1.8 years spent more time outdoors during summer time, compared with spring and fall. 57 The light exposure pattern was correlated with the parents’ pattern, suggesting that educational programmes promoting the increase of time outdoors must start with parents.
To date, however, much remains unanswered regarding the characteristics of outdoor light exposure necessary to circumvent myopia in humans. For instance, what is the minimal required outdoor light intensity (threshold) to avoid the myopia onset (e.g. 1000 lux, 10,000 lux)? How long should the exposure to outdoor light be (e.g. 40 min, 2 h per day)? Can the exposure be intermittent or cumulative over time (e.g. 5000 lux for 1 h/day or 1000 lux for 5 h/day)? Addressing these questions in humans, in longitudinal studies using objective wearable light-tracking strategies is essential for the development of effective outdoor programmes against myopia.
Outdoor programmes for myopia prevention
Outdoor preventive measures are vital to control and lower myopia progression in children. For children at risk of developing myopia, preventive interventions should be initiated before the onset of this ocular condition. Trackers that record and quantify light levels 55 and outdoor time have been proposed to encourage outdoor activities among children with daily goal of 2 h per day and at least 14 h per week. 49 According to French et al. 58 Australian children with baseline refraction of + 1D at age 6 years should be targeted as an at-risk group in prevention programmes for myopia, with the goal of maintaining a slightly hyperopic refraction. Intervention in East Asia might need to be earlier than 6 years of age due to the high number of early-onset myopes. According to a meta-analysis performed by Ho et al. 59 on the outdoor research methods in Asian children aged between 4 and 14 years, outdoor exposure slows myopic refraction by 32.9% and axial elongation by 24.9%.
Outdoor programmes in the schools and community can be developed based on the longitudinal data to increase time outdoors. Additional classes involving outdoor activities can be added to each school day or children can be encouraged to go outside for outdoor activities during recess and after school. 59 Organizing community-based outdoor programmes on weekdays may also be beneficial. Importantly, sun-protective strategies such as tree shade, hat and sunglasses can still allow high levels of light to reach the eye and can potentially protect from the myopia development. 60 Nevertheless, given the competitive nature of schooling systems in Asia and sometimes weather and pandemic constraints, increasing time outdoors remains challenging. These restrictions emphasize the need to rethink indoor-based light-therapy strategies for the prevention of myopia. Without a clear understanding of the anatomical, physiological and neurobiological impact of distinct light features (intensity, spectrum, timing, frequency) on ocular growth and myopia development, the development of adequate artificial light-therapy strategies remains challenging.
Experimental research on light and myopia
While epidemiological investigations over many decades highlighted the protective effect of outdoor bright light exposure on myopia, 47 studies in various animal models have scrutinized the impact of various intensities and spectro-temporal modulations of light regimens on ocular growth and refractive error development. 61 These studies, performed in controlled experimental conditions, have tried to elucidate the underlying mechanisms of the protective impact of light against myopia.
Animal models for myopia
The use of animals for studying mechanisms underlying refractive error development dates back to the mid-1970s after Hubel et al., 62 investigating the cortical effects of monocular visual deprivation in young macaques, serendipitously reported that after eyelid suture for many months, the eyes of animals developed high levels of myopia. These findings were afterward established in young tree shrews 63 and chickens. 64 The initial procedures to induce myopia by suturing the eyelids have been replaced with (1) form deprivation myopia (FDM), via reduction of quality (e.g. sharpness and contrast) of retinal image formation using frosted goggles or (2) introducing controlled hyperopic defocus (minus lenses), termed as lens-induced myopia (LIM). Conversely, inducing myopic defocus (plus lenses) leads to lens-induced hyperopia (LIH).
The disruption of visual input, especially retinal sharpness and contrast, is considered to be a significant factor in driving the development of myopia in children, particularly during the early postnatal period.65,66 Within that framework, FDM is presented as an open-loop model, where ocular growth has no defined endpoint. Conversely, LIM relies on feedback control using visual signals and is classified as a closed-loop condition, where aberrant ocular growth ceases when the growth signal has been neutralized. 67 LIM and FDM involve different mechanisms of action where optic nerve section reduces LIM 68 but not FDM,69,70 yet in both processes, the levels of retinal dopamine (DA) or vitreal DA metabolites are reduced,71,72 while DA or its agonists can inhibit myopia induction through stimulation of the D2-receptor.73–76 According to Norton, 77 induced development of myopia, in addition to the normal refractive and ocular development, in most animal species appears to mimic that in human, wherein it is characterized mainly by an abnormal enlargement of the post equatorial segment of the eye with a significant increase in axial length. Irrespective of the animal model, induced refractive error is also characterized by key factors such as ocular vitreous chamber elongation, thinning of the choroid,78,79 and thinning of the fibrous sclera. 80 All these features are also observed in the myopic human eye.81,82
Commonly used animal models in experimental myopia
The most commonly used experimental animal models for myopia research are chickens, guinea pigs, tree shrews, mice and some nonhuman primates (NHP). 83
Chickens The chicken model is the most commonly used model in experimental myopia research, owing to the animals’ rapid eye growth (100 µm per day), diurnal activity, and the reproducibility of experimental paradigms. 83 In addition, the chicken eye is relatively large (8–14 mm), has an excellent optical system and responds quickly to a variety of environmental factors including defocus, blur, and photic stimulations. Despite its unique photoreceptoral complexity, the overall spectral sensitivity to human-visible light in chickens is not very different from humans. 84 Furthermore, differentially expressed genes and proteins involved in either myopia or hyperopia in chickens significantly overlap with those implicated in the pathogenesis of sight-threatening secondary disorders in humans. 85 On the other hand, chickens display many anatomical differences in ocular structures (e.g. cartilaginous and fibrous sclera, lack of fovea, etc.) compared with humans. 86 Furthermore, the well-developed circadian system in chickens is sensitive to constant moderate light intensity and has a significant impact on refractive development. These findings of impact of light on circadian rhythms are not extrapolatable to rhesus monkeys and mice models.87–89 Findings on the impact of light on ocular growth and emmetropization in the chicken model may not be easily/necessarily translatable to humans.
Guinea pigs First presented as a model for experimental myopia by Howlett and McFadden, 90 guinea pigs are diurnal dichromatic mammals with retinas comprising rods, and middle- and short-wavelength cones. The cone proportion in guinea pig retinal photoreceptors is high (8%–17%) in comparison with other species. 91 The guinea pig model has been identified as a convenient model for studying refractive error development, 92 given advantages such as easiness to maintain and breed, in addition to their large eyes (axial length around 8.0 mm) and pupils. Furthermore, these small mammals, respond well to form deprivation 92 and lens-induced defocus. 93 On the other hand, guinea pig retinas lack fovea and the induction of myopia is at times challenging with strain variability. Also, studies requiring lens mounting for long periods of time are challenging as guinea pigs tend to scratch and remove the Velcro base holding the lens.
Tree Shrews Owing to its close association with primates and rodents, tree shrews are widely used for studying refractive error, and understanding neurophysiological mechanisms underlying emmetropization. 78 The ocular morphology of tree shrews is similar to humans; however, these animals lack a fovea, have a thicker lens, and thinner choroid void of choriocapillaries unlike in humans. 77 The tree shrews can develop myopia 94 and can actively compensate for defocus and exhibit a single layer sclera similar to humans. These animals possess dichromatic retinas composed of ~95% of cones. 95
Mouse Given its readily available whole-genome sequence, which is 85% homologous to the human genome, the mouse model has always been a popular model for studying the visual system.96,97 Both FDM and LIM in the mouse can be achieved by mounting diffuser or lens (goggles) to eyes either by means of stitching around the eye and reinforcing with glue or by mounting custom-made assembly to hold the lenses intact. On the other hand, the mouse model lacks a fovea, possesses poor visual aptitudes, and has a small eye (axial length of 3.3 mm) making anatomic assessment troublesome. Nevertheless, under photopic conditions, mice still retain adequate spatial vision to respond to LIM and FDM. 98 Despite the concerns for using the mouse as a model for myopia, it has been established as a useful model for pharmacological and genome manipulation studies in the field of myopia. 99
Rhesus monkeys Among NHPs, rhesus macaques, belonging to the old-world monkeys, constitute one of the most suitable models for refractive error studies. The visual physiology of rhesus monkeys is identical to that of humans with a rod-based retina and a cone-based fovea. 100 Raviola and Wiesel 101 have demonstrated the myopia induction in rhesus macaques. The average axial length of 21-day-old baby rhesus macaques is 14.15 mm, very much close to a human baby which is 17.3 mm. 102 Conversely, ethical concerns, logistics, high operational cost, seasonal breeding, low reproductive rate, difficulties in handling infant monkeys, having a customized myopia-inducing helmets/devices adaptable for monkeys and prolonged experimental procedures to obtain myopic shifts make it more challenging to use rhesus macaques for myopia research.
The impact of light on refractive error development
The protective effect of outdoor light exposure against myopia could be attributed to multiple factors, 17 also including light intensity, pattern, and spectrum but also to reduced peripheral retinal defocus and increased visual spatial frequency. This section summarizes the current knowledge about light modulation and ocular growth based on controlled studies in animal models. Please see Table 1 for more details.
Intensity of light
Findings from animal studies support the notion that higher light levels, similar to those encountered outdoors, are predominant factors for myopia prevention. In chickens, dim ambient lighting of 50 lux delivered as a 12 h/12 h light–dark cycle is deleterious to emmetropization, 103 while exposure to high illuminances of light (15,000 lux) for periods of 5 or 6 h per day delays the development of FDM by 60%. 39 This protective impact of light on FDM is dose-dependent, with exposure to 40,000 lux of light-emitting diode (LED) light for 6 h providing comprehensive protection against the onset of FDM. 104 These protective effects of bright light against myopia have been associated with DA release and the D1 receptor signaling pathway (see ‘Light, dopamine and refractive error regulation’ section for more details).61,105 Similarly, bright light exposure can also reduce, but not overcome, the rate of compensation for monocularly fitted negative lenses (−7D) and enhance the rate of compensation for positive lenses (+7D). 40 Alike chickens, tree shrews exposed to bright light (16,000 lux) for 7.75 h/day for 11 days display a reduced development rate of FDM and LIM, 106 while form-deprived eyes of rhesus monkeys reared under 18,000–28,000 lux of metal halide light (4200K) for 6 h a day over ~150 days are less myopic than those reared in normal light. 41 Interestingly, in rhesus macaques, 25,000 lux of bright light for 6 h per day was not sufficient for stopping LIM, suggesting dissimilarities in mechanisms responsible for FDM and LIM. 107 In guinea pigs, bright light (10,000 lux) reduced the myopic shift induced by form deprivation compared with normal lighting (500 lux). 108 While in mice, bright light exposure (2500–5000 lux) for 6 h/day for 4 weeks prevented FDM and presented a hyperopic shift and reduction in ocular elongation compared with normal lighting (100–200 lux). 105 Analogously to bright light, albeit through different mechanisms, short periods (~3 h/day) of de-focusing lens removal or normal vision per day, even in moderate light levels, can compensate for LIM in chickens. 109 Surprisingly, and contrary to earlier studies in chickens, 103 a recent study in infant rhesus monkeys raised under dim light (~55 lux) showed a hyperopic shift when compared with the monkeys raised under normal light (~504 lux). 110 These differences in response to between dim light chickens and monkeys, may be due to differences in the sensitivity of the circadian system between birds and mammals. 111
Timing and duration of bright light
Prevailing evidence on the impact of light intensities on myopia in animal models has raised the question of whether the intensity of light and timing of exposure are interlinked. This has gained more attention with the notion supporting the role of circadian rhythms in ametropia. 112 Recently, Nickla et al. 113 reported that myopic defocus in chickens raised under light levels of 500 lux was more effective at reducing ocular growth when lenses were worn during the evening compared with when lenses were worn in the morning. These moderations were attributed to alterations in the amplitude of the axial length rhythm. On the other hand, constant daily light exposure (2000 lux) was reported to be more effective at inhibiting myopia than a 2 h dose of bright light (10,000 lux) delivered either in the morning, mid-day or evening. 114 Within that same study, however, 2 h of bright light (10,000 lux) delivered midday was more efficient in inhibiting ocular growth than the same light protocol delivered in the evening. 114 Moreover, chickens exposed to ambient light (700 lux) at night (between 12:00 a.m. and 2:00 a.m.) showed alterations in axial length and choroidal thickness rhythms, which could no longer follow a sinewave function with a 24 h period. This brief light exposure caused a transient stimulation in the ocular growth rate which may have subsequently resulted in myopic refractive error. 115 Interestingly, Sarfare et al. 116 revealed that evening bright light inhibits the effect of continuous hyperopic defocus and form deprivation while morning bright light has a greater inhibitory effect on transient ‘2 h’ hyperopic defocus. These findings suggest a peculiar interaction between the timing and duration of defocus and bright light exposure that the authors attribute to the duration and sign of the defocus signal in operation immediately following the bright light exposures. 116 The abolishment of light/dark cycles has also been studied in animal models and constant light has been shown to disrupt LIM and FDM in chickens.117,118 Corneal flattening was also observed in chickens reared under continuous bright light; however, no distinct observation was made with chickens reared in bright light with a diurnal pattern.119,120 This effect of continuous light on refractive error development and emmetropization appears to be unique to chickens, since rearing infant rhesus macaques in ambient constant light does not affect emmetropization. 88 This interspecies variation was attributed to difference between the avian and mammalian circadian systems. 111 In smaller mammals like mice, prolonged (18 h light/6 h dark) exposure to light does lead to a myopic shift, increased axial length and vitreous chamber depth (VCD), reduction in retinal Egr-1 mRNA transcript level, and decreased scleral fibre diameters in C57BL⁄6 in bred mice. 121
Temporal frequency of light
Emmetropization in chickens is dependent upon the temporal frequency of the light exposure: high temporal frequencies induce hyperopia and low temporal frequencies, myopia.122,123 Lan et al. 124 demonstrated that intermittent exposure to bright light at 15,000 lux for 1:1 and 7:7 min were more effective in controlling FDM when compared with continuous bright light exposure. A possible underlying mechanism for such findings could be that flickering light triggers the retinal ON and OFF pathways, thereby stimulating DA release. 125 Guinea pigs raised in 0.5 Hz flickering light (600 lux) for 12 h/day for 12 weeks presented a greater myopic shift in refraction and a larger increase in axial length ocular length compared with guinea pigs raised in 5 Hz flickering light (600 lux) or a control group which was raised in steady light (300 lux). 126 In another study, guinea pigs exposed to flickering light (505 nm, 600 lux, 0.5 Hz) for 12 h/day for 8 weeks showed a significant decrease in refraction and increase in axial length compared to animals exposed to 12 h/day of steady control light (600 lux). Furthermore, not only the levels of DA, but also of 3,4-dihydroxyphenylacetic acid (DOPAC) and homovanillic acid (HVA) which are primary and secondary metabolites of DA, respectively, were significantly increased in the flickering light group, with DA D1 and D2 receptors upregulated compared with the control. 127 Flickering rate and DA levels may hence play a role in myopia development in guinea pigs. 126 Apart from DA, Li et al. 128 found elevated concentrations of 5-hydroxytryptamine (5-HT) and 5-HT2A receptor expression in guinea pig groups raised under flickering light (600 lux, 0.5 Hz for 12 h/day for 8 weeks), while norepinephrine and epinephrine levels were reduced compared with control groups exposed to 300 lux of light for 12 h/day. C57BL/6 (B6) mice exposed to 6 weeks of flickering light (2 Hz: with 500ms of dark phase per second) for 12 h/day presented with a myopic shift (~ −9D) in refraction and increased axial length compared with the steady light control. 129
Spectral composition of light
The spectral composition of light has also been shown to play a key role in ocular growth and emmetropization. In chickens, exposure to red light (peak wavelength range: 615–641 nm) has been reported to induce myopia while rearing under ultraviolet light (UV) (peak wavelength: 375 nm) or blue light (peak wavelength range: 430–477 nm) induces hyperopia.130–132 Furthermore, ocular DA release and metabolism, as well as vitreal and retinal metabolomic profiles, were highly dependent upon the spectral composition of light.132,133 Among plausible explanations to this wavelength-dependent refractive error regulation, ocular longitudinal chromatic aberration (LCA), which leads to wavelength defocus and higher refraction of short-wavelength light compared with long-wavelength light by ocular optics, was supported by many authors.131,134,135 The hyperopic shift in response to short-wavelength blue light has also been reported in other, but not all, animal species such as Cichlid fish, 136 guinea pigs137–141 and some rhesus monkeys. 142 Comparatively, red light or eye-mounted red filters render tree shrew and rhesus monkey eyes hyperopic, while blue flickering light induces myopia and increases VCD.143–145 Interspecie differences in the spectral responses may to light not only to be due to protocol differences (e.g. duration of light exposure) but also to differences in retinal photoreceptor composition and sensitivity accross species.146,147
The spectral composition of light also has a prominent role in exerting protective effects against FDM and LIM. Torii et al. 148 suggested that exposure to violet light (VL:360–400 nm) can suppress myopia progression in chickens through the upregulation of the “myopia protective gene” EGR1. Similarly, blue and UV light exposure conferred a protective effect against myopia progression with a concomitant increase in the retinal DA levels. 132 However, the applicability of near UV and UV light to humans is limited due to the UV-blocking properties of the crystalline lens149,150 and the nonavailability of near UV receptors unlike in chickens and guinea pigs. In addition, in guinea pigs, short-wavelength blue light of 470 ± 5 nm with an intensity of 50 lux showed inhibition of LIM, while long-wavelength red light of 600 ± 5 nm with an overall luminance of 300 lux presented a myopic shift. The increased sensitivity to blue light by 0.35 log units compared with red light in guinea pigs may have contributed to this short-wavelength mitigation of eye growth. 139
The spectral tuning of refractive error development is also dependent upon the flicker frequency of light. For instance, blue light exposures are protective against myopic eye growth induced by low-frequency flickering light in chickens, while 8 weeks of flickering green light (5 Hz) at 800 lux was found to induce myopia and increase axial lengths in guinea pigs. 151 These findings suggest that high temporal frequencies may reduce the effects of wavelength defocus on ocular refraction, such as at low temporal frequencies, visual inputs are dominated by wavelength defocus signals, inducing hyperopic shifts at short wavelengths and myopic shifts at long wavelengths. While at high temporal frequencies, a myopic shift under blue light and a hyperopic shift under green light is a result of visual inputs being dominated by luminance signals and wavelength defocus signals being weakened. 152
Altogether, observational and experimental studies in humans and animal models suggest that exposure to high-intensity light, both in continuous or intermittent patterns, can slow the development of myopia. However, this impact of high-intensity light against myopia development in animal models is dependent on the means of myopia induction (i.e. more effective in FDM compared with LIM). Furthermore, today there is no clear consensus on a minimum or optimal light intensity to promote emmetropization and prevent or slow myopia development in humans. Such a threshold is variable in animal models, given differences in retinal circuitry and photoreceptoral composition. Conversely, a total of 40 min of outdoor time per day (i.e. a combination of exposure to high-intensity sunlight, increased spatial frequency, increased retinal focus, etc.) seems to be protective against myopia in humans; frequently, animal models for myopia, baring strong myopiagenic stimuli, require longer durations of high-intensity light per day to alleviate the development of this ocular condition. Although the spectral sensitivity to refractive error development in response to light has not yet been fully established, existing studies in animals (chicken and guinea pigs) and humans are in a fragile consensus that short-wavelength light may be protective against axial myopia development. Studies in NHP and tree shrews disagree with the latter statement. Finally, exposure to high-intensity and short-wavelength light needs to be timed carefully to avoid any potential disruptions to the circadian timing system of children and adolescents. Considering that all the parameters of light namely the intensity, duration, spectrum, pattern, and timing of light are synergetic, and given the scarcity of interventional clinical studies using light, tailored light-therapy strategies for myopia prevention are yet to be established.
Summary of experimental and observational studies in humans and animal models investigating the impact of light on myopia.

BEW, blue-enriched white light; CI, confidence interval; Clux, chicken lux; D, diopter; DL, day light; EL, experimental light; FDM, form deprivation myopia; FLM, flickering light–induced myopia; LCA, longitudinal chromatic aberration; LED, light emitting diodes; LIH, lens-induced hyperopia; LIM, lens-induced myopia; ML, middle-wavelength light; NDF, neutral density filter; NL, normal light; OR, odds ratio; SL, short-wavelength light; SW, soft standard white light; UV, ultra-violet; VL, violet light; WL, white light.
Physiological mechanisms mediating light-induced myopia prevention
Experimental research has been instrumental for elucidating the anatomo-physiological impact of light on ocular growth refractive error development. Although there are mixed opinions in the myopia research community on the involvement of light in the prevention of myopia, 162 the protective effect of high/higher intensity light against myopia cannot be ruled out, at least in experimental animal models of the condition and interventions in humans. 43 To date, however, the exact spectro-temporal characteristics of protective light regimens remain unclear. Understanding the underlying physiological and molecular mechanisms mediating light-induced myopia prevention is essential for data-driven successful translational interventions (Table 2).
Summary of studies investigating key signalling molecules linked to light-dependent ocular growth and refractive error development.
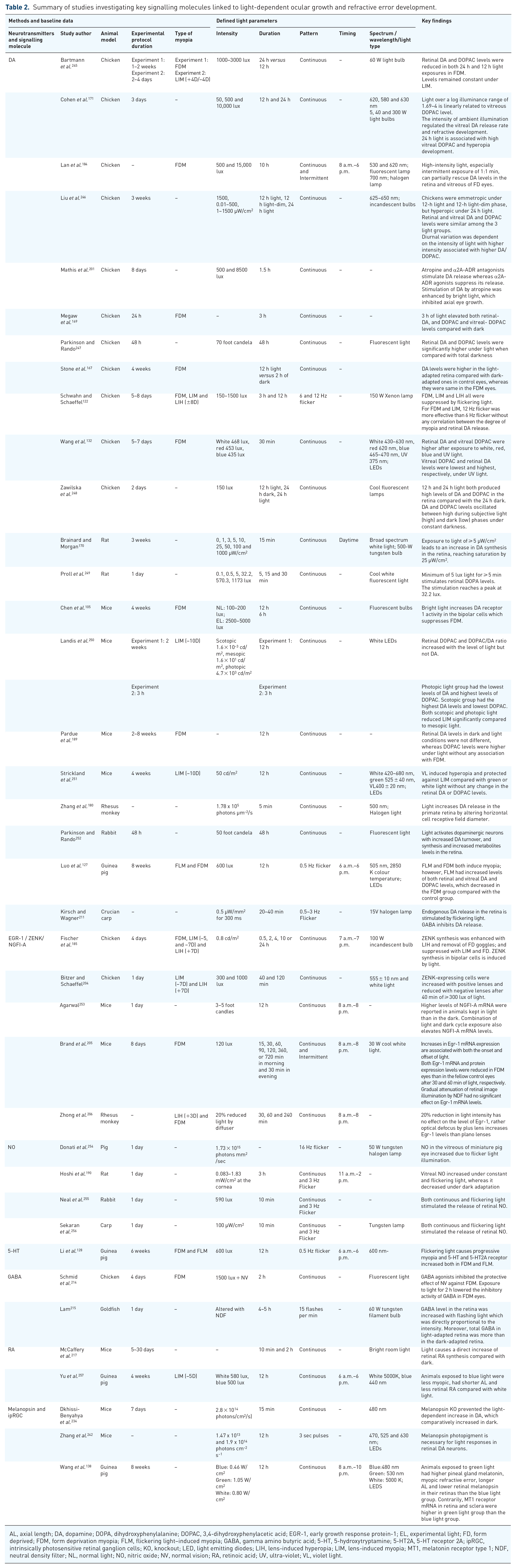
AL, axial length; DA, dopamine; DOPA, dihydroxyphenylalanine; DOPAC, 3,4-dihydroxyphenylacetic acid; EGR-1, early growth response protein-1; EL, experimental light; FD, form deprived; FDM, form deprivation myopia; FLM, flickering light–induced myopia; GABA, gamma amino butyric acid; 5-HT, 5-hydroxytryptamine; 5-HT2A, 5-HT receptor 2A; ipRGC, intrinsically photosensitive retinal ganglion cells; KO, knockout; LED, light emitting diodes; LIH, lens-induced hyperopia; LIM, lens-induced myopia; MT1, melatonin receptor type 1; NDF, neutral density filter; NL, normal light; NO, nitric oxide; NV, normal vision; RA, retinoic acid; UV, ultra-violet; VL, violet light.
Ocular pathways of myopia control
The sclera plays a vital role in determining the shape and size of the eye; consequently, it has long been of high interest for myopia intervention strategies with emphasis given to understanding the mechanism of pharmacological molecules on the scleral metabolism. 163 Nevertheless, subsequent studies have identified that the retina is the key signal regulator in the emmetropization process. Although there exist more convincing reports on the involvement of retinal signalling molecules which include DA and acetylcholine during emmetropization, the particular cell type and associated biochemical pathway involved are yet to be unveiled. 164 It is further hypothesized that these retinal molecules act through a cascade to communicate with retinal pigment epithelium (RPE) and choroid, which in turn releases a series of different molecules that regulate the scleral growth and remodelling 164 (Figure 2).
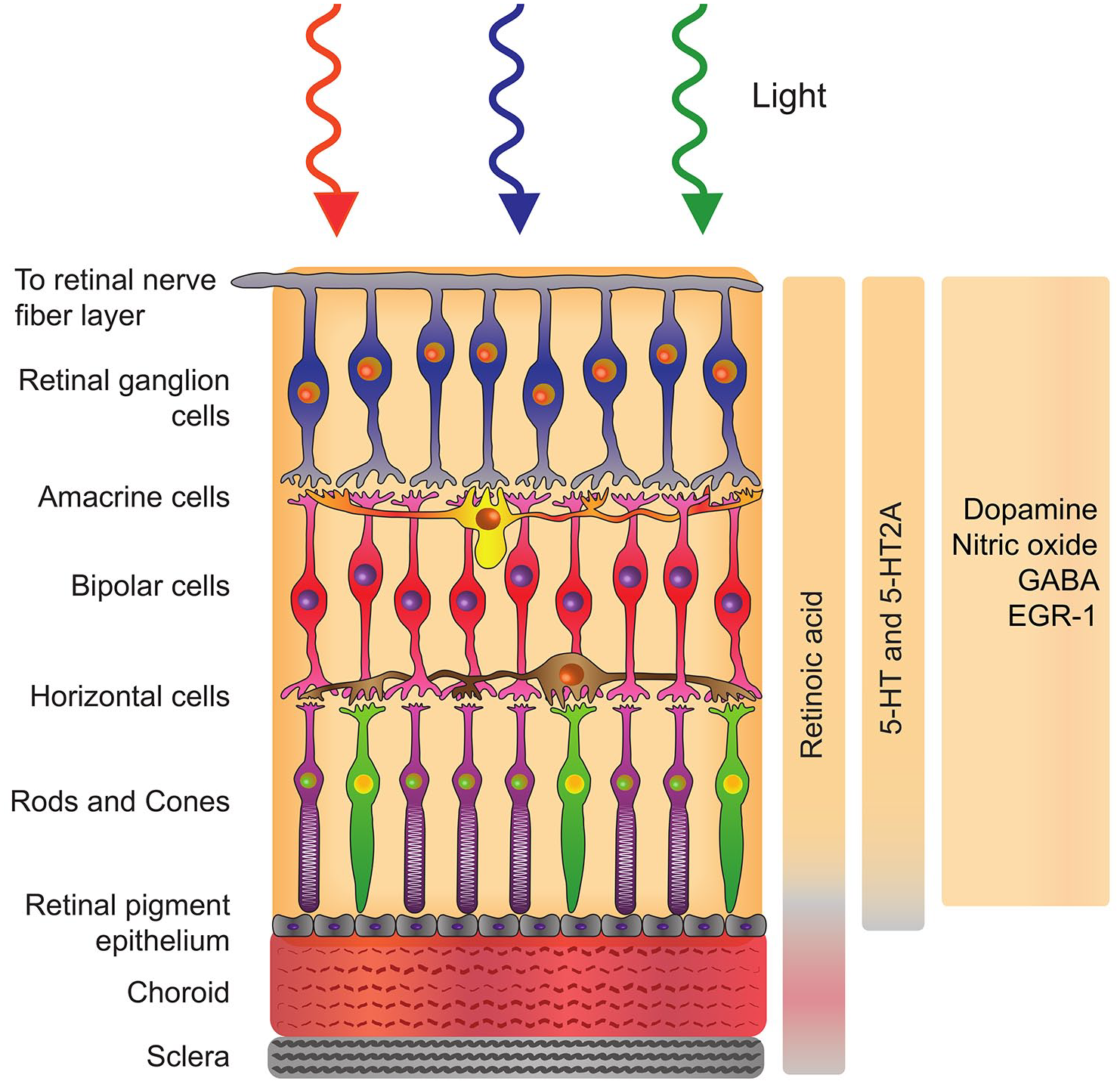
Schematic representation of the retina, retinal pigment epithelium, choroid, and sclera with corresponding molecules modulated by light stimulation.
Light, dopamine and refractive error regulation
DA, a neurotransmitter implicated in several physiological, cerebral and retinal functions, has been shown to be involved in the biochemical signalling cascade that controls emmetropization.165–167 In the retina, DA is released by amacrine cells and/or interplexiform cells, depending on the species. 168 A large body of evidence is available to support the hypothesis that DA is implicated in ocular growth 74 and a dose-response relationship has been established between DA synthesis and light intensity.169–171
Light-associated DA activity is mediated possibly through the D2 receptor pathway, thereby altering the signal that triggers axial elongation. Among DA receptor subtypes involved in the signaling, D2-like (D2 and D4) receptor plays a key role in FDM. 172 The ocular refractive development relies mainly on the balance between the activation of D1-like and D2-like receptors. Overactivation of D1-like receptor has been reported to induce hyperopia and vice versa. 173 DA can influence the development of myopia via changes in spatial tuning of cellular responses in the retina. Receptive fields are adjusted based on D1 and D2/D4 receptor activation via varied concentrations of DA. As light influences the release of DA, it is believed that different light conditions play a role in the spatial tuning of retinal cellular responses. 173 In the case of rod–cone gap junctions in bright light conditions, conductance is decreased as DA levels are high. The binding of DA to D2/D4 receptors reduces adenylate cyclase activity, leading to reduced cyclic AMP (cAMP) production and protein kinase A (PKA) inactivity.174,175 Rod–cone gap junctions are thus left uncoupled, resulting in reduced receptive field sizes and better visual acuity. Under low light conditions, DA release is low and rod–cone gap junctions are coupled in order for dim objects to be detected. 176 Apart from D2/D4 receptors, DA can also bind to D1 receptors. Horizontal cell gap junctions and AII amacrine cell gap junctions are regulated via this pathway.177,178 In high light levels, DA binds to D1 receptors and activates adenylate cyclase. Increased cAMP concentrations activate PKA, reducing conductance between horizontal cell-horizontal cell gap junctions. In AII amacrine-AII amacrine gap junctions, the production of protein phosphatase 2A (PP2A) is believed to reduce conductivity and coupling. 179 Horizontal cell coupling is involved in image contrast optimization 180 while AII amacrine cell coupling is involved in the summation of identical signals and removal of noise. 181 This control in rod–cone, horizontal cell, and AII amacrine gap conductance optimizes spatial tuning in the retina and allows for the production of high acuity images with good contrast in different light conditions. Light-regulated DA levels may thus control the development of myopia by modifying the attributes of perceived images.
Retinal DA levels are decreased in chicken eyes subjected to form deprivation, 182 but return to normal levels upon cessation of the latter. 183 Interestingly, Schwahn and Schaeffel 122 were able to prevent FDM but not able to retain DA content and release in the retina with the use of 12 Hz at a duty cycle of 4% flickering lights. The change in retinal DA levels for different durations of flicker lights, however, did not correlate with the level of inhibition of myopia. Conversely, ocular DA levels are also dependent upon the spectral content of light with a trend towards higher retinal DA release under UV light compared with white light. 132 The protective effect of bright light against myopia was also found to be dismissed when chickens wearing diffusers were injected with spiperone, a DA antagonist, before exposure to bright light, 40 indicating DA’s role in the light-driven myopia-control pathway. In chickens that were form deprived, exposure to 15,000 lux of light resulted in partial rescue of retinal DA levels, while it did not alter the levels of ZENK, an immediate early gene in the amacrine cells involved in the regulation of ocular axial length of.184,185 Furthermore, retinal DA may modulate choroidal thickening and a subsequent reduction in axial length by triggering other neuromodulators such as nitric oxide (NO). 186
According to Hartline, 187 luminance-associated visual information is decoded in the brain through parallel pathways, namely, increments (ON) and decrements (OFF). Low luminance and optical blur which are considered as key risk factors of myopia, trigger the ON luminance pathway and thereby lower the release of retinal DA. 188 The role of ON and OFF pathways in refractive error development is extrapolated using a genetically modified mouse model wherein the nob mice were subjected to FDM. The results from the study exhibit that myopia development in mouse eye is stimulated primarily due to defect in ON pathway in addition to the low DA level and blurred vision imposed by form deprivation which is evident from the loss of visual function along the ON pathway. 189
Other neurotransmitters and signaling molecules involved in light-mediated refractive error regulation
Nitric oxide
NO is a neurotransmitter that is involved in the regulation of retinal responses. Vitreous concentrations of NO are dependent on ambient light conditions 190 and may play a role in the protection against developmental myopia. In a study conducted by Carr and Stell, 191 intravitreal injection of NO synthase substrate L-arginine (L-arg) or NO donor sodium nitroprusside was able to dose-dependently inhibit the development of myopic refraction and axial elongation. In addition, NO may also play a role in the regulation of choroidal thickness, as intravitreal injection of NO synthase inhibitor NG-nitro-L-arginine methyl ester rapidly and transiently inhibited choroidal thickening and promoted choroidal thinning in chicken eyes recovering from FDM and in eyes mounted with +15D lenses. 192
Similar to DA, NO affects horizontal cell gap junction conductance and coupling. In the presence of bright light, NO is released in amacrine cells and activates guanylate cyclase after diffusion into retinal neurons. This results in an increase in cyclic guanosine monophosphate (cGMP) levels and the activation of cGMP-dependent protein kinase. Phosphorylation or dephosphorylation of connexin 35 in chickens and connexin 36 in mammals present in the retinal gap junctions alters horizontal and amacrine cell gap junction conductivity via NO, which in turn stimulates retinal cell uncoupling.193–195 Subsequently, this increases the overall optokinetic contrast sensitivity in chicken eyes, especially at high spatial frequencies. 196 NO may thus contribute to the prevention of myopia development by modulating the receptive field sizes and spatial contrast sensitivity.197,198
Atropine
Although low-dose atropine eye drops are used to prevent or slow myopia development in children, 198 the underlying mechanisms of atropine action remain poorly understood. Atropine is a muscarinic antagonist that also acts as a potential alpha 2-adrenergic receptor (α2A-ADR) antagonist. In chickens, that lack muscarinic receptors in the ciliary muscles, atropine can still reduce experimental myopia development, 199 and both atropine and other α2A-ADR antagonists have been shown to stimulate DA release by activating the tyrosine hydroxylase immunoreactive amacrine cells.200,201 Conversely, α2A-ADR agonists strongly suppress the release of DA.200,201 Interestingly, the actions of atropine and bright light of 8500 lux were recently reported as additive, increasing DA release in the vitreous of chickens that received an intravitreal atropine injection and were exposed to bright light for 1.5 h, compared with chickens that received the same treatment but were exposed to 1.5 h of standard light of 500 lux. 201
EGR1 (ZENK)
Early growth response protein-1 (EGR-1) or ZENK is a protein encoded by the EGR-1 gene. Lower levels of EGR-1 or ZENK have been associated with increased axial elongation and vice versa. 202 EGR-1 is considered as a well-established and documented protective gene for myopia.203–205 Upregulation of ZENK is associated with inhibition of ocular elongation linked with hyperopic defocus and the recovery from form deprivation. The modulatory expression of ZENK is clearly evident in amacrine cells containing glucagon. In chickens, EGR-1 suppresses ocular axial elongation and when EGR-1 is knocked down in mice, the eye exhibited distinct axial elongation. 202 It was found that 30 min of exposure to visual stimuli following form deprivation can regulate ocular growth by modulating the expression of ZENK. 185
The intensity of light exposure was reported to be positively correlated with the ZENK expression in chicken retinal amacrine cells; however, this effect is not related to the duration of light exposure. 185 Albeit, Ashby et al. 203 reported bidirectional response of Egr-1 mRNA levels with a 50% decrease and >200% elevation in Egr-1 mRNA levels in lens-induced (−5D) myopic eyes of guinea pigs during the induction (day 7) and the recovery periods, respectively.
ZENK-responsive bipolar cells are usually the cone ON-bipolar cells; hence, the bipolar cells are seen to have ZENK induction as a function of light intensity. 206 Furthermore, EGR-1 mRNA transcript levels are also dependent on the spectral content of light; chickens reared under VL for 7 days (12 h light/dark cycle) showed upregulation of EGR-1 in chorioretinal tissues, compared with blue light exposure. In addition, the eyes exposed to VL were significantly less myopic compared with those exposed to fluorescent light. 148
5-HT and 5-HT2A receptor
Serotonin [i.e. 5-hydroxytryptamine (5-HT)] is a neurotransmitter synthesized in central nervous system. Lens-induced myopic eyes of Guinea pigs have significantly higher levels of 5-HT and 5-HT2A receptor. 207 Constant square-wave 0.5 Hz flickering light can induce myopia of progressive nature in guinea pigs. 128 The 5-HT and 5-HT2A receptors were found to increase in both myopia due to flickering light and form deprivation. This indicates that 5-HT is possibly involved in the induction of myopia and it acts by binding to 5-HT2A receptor. 128 The 5-HT2A receptor expression was found to be increased and the concentrations of norepinephrine and epinephrine were decreased in guinea pigs’ eyes following both the exposure to flickering light and form deprivation. As hypothesized by Li et al. 128 by binding to 5-HT2A receptor, 5-HT may strengthen scleral remodelling and influence ocular axial growth.
Gamma aminobutyric acid
Gamma aminobutyric acid (GABA) is an inhibitory neurotransmitter in the retina and brain. There are three types of GABA receptors, the GABA(A) receptors facilitate the feedback between horizontal cells and cones, GABA(B) receptors regulate intracellular messengers and neuronal function, and GABA(C) receptors are involved in mediating GABAergic synaptic functions in the outer and inner retinas. Eye growth and refractive development in chickens is regulated by these 3 receptors of GABA.208,209 GABA(C) antagonists were found to be most effective at preventing LIM, although other receptors can also prevent myopia. 210 GABA(A) and GABA(C) agonists decreases DA release in the retina, whereas GABA antagonists increase DA release.209,211 Moreover, amacrine cells release GABA molecules which bind to fast-acting ionotropic receptors in the retina.212,213 In FDM, the DA and GABAergic neurotransmitter pathways interact. Exposure to fluorescent lights (1500 lux) for 2 h lowers the inhibitory activity of GABA in form-deprived eyes. 214 This protective effect of bright light against FDM while overcoming the effects of GABA agonists involves an increase in the D2 DA receptor activity. 214 In contrary, goldfish retina demonstrated an increase in GABA level directly proportional to flashing light intensity. 215
Retinoic acid
Retinoic acid (RA) is a lipid-soluble metabolite derived from retinol or vitamin A, which acts as a regulator of growth, differentiation, and development of several cell types, including epithelial and neuronal cells. RA also acts as a neuromodulator that sends information regarding the illumination to the outer plexiform layer of retina. 216 McCaffery et al. 217 first reported the light-mediated increase in RA synthesis in both retina and RPE samples of mice exposed to bright room light for 10 min. This increase was also directly proportional to the age of the mice, wherein the older mice reported increased RA release in response to light. Similarly, the impact of 20 min of bright room light retina and RPE samples revealed a strong RA activity when compared with samples kept in darkness. 218 Dirks et al. 218 also noted that RA synthesis is light-dependent and DA-independent in the carp eye indicating that these two modulatory systems are not inter-dependent but act in parallel. Apart from this, studies in various animal models like chickens,219–221 guinea pigs222,223 and marmosets 224 revealed that changes in RA synthesis are species-dependent, where myopia induction led to a decreased RA level in chickens, and increased RA levels in guinea pigs and marmosets.222,224,225 Choroidal RA biosynthesis is regulated exclusively by retinaldehyde dehydrogenase 2 (RALDH2) 226 and fundal tissue aldehyde dehydrogenase-2 (ADH2). 219 In the sclera, RA and glycosaminoglycan (GAG) levels observe an inverse relationship; with increasing RA levels, the GAG levels decreases and vice versa.220,224,225 The mechanism of action of RA might be through the remodelling of scleral extracellular matrix 224 or the modulation of cell coupling.227,228
Melanopsin and intrinsically photosensitive retinal ganglion cells
Melanopsin is an atypical photopigment expressed in ganglion cells, rendering them intrinsically photosensitive.229,230 These intrinsically photosensitive retinal ganglion cells (ipRGCs) complement the visual photoreceptors and convey photo transduced signals to nonvisual centres in the brain, including the suprachiasmatic nucleus governing most circadian rhythmic expressions in the body (e.g. sleep, alertness, melatonin secretion at night) (for review, see Najjar and Zeitzer 231 ). Melanopsin is predominantly sensitive to bright blue light (~480nm), a wavelength reported to induce hyperopia and reverse experimental myopia in some animal models130–133 and the increase in ocular DA levels upon bright light exposure could potentially be due to the stimulation of melanopsin and the synaptic and functional connection between the ipRGCs and dopaminergic amacrine cells.232,233 In addition, melanopsin knockout mice display a decline in ocular DA levels, 234 and preliminary findings highlight a direct, yet unclear, role of melanopsin in refractive error development. 235 In humans, some authors attributed a reduction in sleep quality observed in highly myopic children to a decreased ipRGC function in myopic eyes, in addition to high demands at school and distress over poor vision. 236 Nevertheless, studies investigating the pupillary light reflex reported no alterations in the response in mild and moderate myopic participants and no associations between refractive error and the ipRGC inputs to the pupil control pathway.237,238
The retinal clock plays an essential role in adapting retinal physiology and visual function to the light/dark changes and holds with its outputs (e.g. melatonin, DA) a major role in the regulation of eye growth and refractive error development in birds and mammals. 239 A additional, potential pathway for photic ocular growth control involves the phase shifting aptitudes of the retinal clock by light 240 through the potential contribution of ipRGCs, neuropsin (OPN5), 241 rods and/or middle-wavelength (MW) cones and excitatory influences upon dopaminergic amacrine cells.242–244
The development of light-therapy strategies for myopia
While increasing exposure to outdoor light levels can successfully be implemented through national outdoor programmes, to prevent the onset of myopia and slow myopia progression in progressing myopes, implementation remains suboptimal in some circumstances. 258 On the other hand, the optimization of architectural lighting or development of light-therapy devices requires a holistic understanding of the benefits and side effects of light characteristics (intensity, timing, pattern and spectrum) on ocular growth and neurophysiology (e.g. circadian rhythms, sleep and alertness). Yet, some studies have shown promising results by either increasing light intensity indoors in school-based interventions 43 or adopting a daily light-therapy approach. 259 According to Hua et al. 43 increasing the ambient light levels to 558 lux at the desk and 440 lux at the blackboard in classrooms can reduce the percentage of new myopia onset. Concomitantly, a pilot study carried out in China has shown that both students and teachers can adapt to a bright classroom having a light intensity between 1,330 and 4,060 lux. 260 These findings suggest that moderately high intensities of light indoors (e.g. classrooms) could yield sufficient protection against myopia in children. 42 In accordance with the latter statement, yet adopting a more individualized light-therapy approach using light delivery glasses, Read et al. 259 showed that exposure to ~500 lux for 30 min in the morning for 1 week increased in choroidal thickness in young adults. Although Read et al. did not investigate the impact of light therapy on refractive error development per se, these results may be promising given the association between choroidal thickness and refractive error development. The spectral modulation of light reaching the retina can also offer promising therapeutics for myopia. A recent study by Ofuji et al. 261 have shown that wearing VL-transmitting glasses for 2 years, and engaging in outdoor activity for 2 h can reduce ocular axial length and increase choroidal thickness in a child with high myopia. As reported by Torii et al., 148 an increased expression of EGR1 gene may have led to this protective effect of VL- transmitting glasses against myopia. In addition, a DA increase in response to the UV light could also be postulated based on data available from the form-deprived chicken model. 132 Neuropsin (OPN5), is a UV-sensitive and bistable (λmax: 380 nm and 470nm) photopigment that is ubiquitously expressed in mammalian ganglion cells.244,262 In a recent study, it was reported that VL stimulation prevented myopia in mice and identified OPN5-expressing retinal ganglion cells as a key for emmetropization in this animal model. 241 On the other side of the spectrum, a randomized controlled trial (ClinicalTrials.gov Identifier: NCT04073238) is ongoing at Zhongshan Ophthalmic Center to test the efficacy of low-level red light therapy (LLLT) to control the progression of myopia, the authors hypothesize that effect of LLLT is potentially through inhibiting scleral hypoxia and thereby improving the choroidal blood perfusion. In a similar study, slowing of myopia progression was shown among children treated with low-level laser therapy for 6 months, probably by inhibiting the NO synthesis and inflammatory cytokines thereby decreasing the severity of oxidative stress.263,264 The effects of the spectral composition of light have to be corroborated carefully with its intensity, pattern, timing, and its effects on circadian system when considering light therapy as a potential treatment for myopia in children.
With the availability of inexpensive lighting systems and electronics nowadays, many nonmedical devices ranging from side lamps to wearables are on the market, with the claim of alleviating the myopia epidemic. Unfortunately, many of such devices overstate their claims and are sometimes not data-driven. Today, to optimize indoor light-therapy strategies for myopia we would suggest the following pathway:
I. Elucidate interspecies differences in the light-driven emmetropization process;
II. Recognize the most suitable animal model(s) for studying the impact of light on emmetropization. This model(s) should closely mimic human ocular physiology and refractive error development;
III. Pinpoint anti-myopiagenic light parameters in the selected animal model and test the synergetic anti-myopiagenic aptitudes of light parameters. For example, test dynamic lighting and tailor the spectral composition and timing of moderate light intensities (indoor) across the day;
IV. Establish reliable short-term biomarkers allowing a fast and reliable, evaluation of the impact of light on refractive error development in humans. Considered one of the earliest observable ocular changes during the development of refractive errors and ocular growth, short-term changes in choroid thickness may represent a reliable biomarker for the signalling cascade that results in longer term changes in ocular growth in response to light. While myopiagenic stimuli (e.g. accommodation, hyperopic defocus) are often associated with a transient thinning of the choroid as compared with anti-myopiagenic stimuli (e.g. anticholinergic agent, myopic defocus) which are associated with a transient choroidal thickening;
V. Evaluate (1) the safety and (2) efficacy of these light parameters in humans by investigating short-term changes in biomarkers;
VI Confirm the preventive efficacy of these light parameters in a randomized longitudinal clinical trial;
VII. Develop data-driven light-therapy strategies/devices for myopia control in children.
Conclusion
In this narrative review, we presented the current knowledge on light-driven modulation of ocular growth and emmetropization based on studies in human and animal models. In addition, we also highlighted potential neurobiological mechanisms involved in the protective effect of light on myopia onset and suggested a potential pathway for the translational development of noninvasive light-therapy strategies for myopia prevention in children. Overall, available data from humans and experimental animal models suggest that high-intensity light even in discontinuous patterns is capable of preventing myopia onset. These findings support the need for well-devised outdoor programmes in children, especially in countries where myopia is prevalent. Nevertheless, less intense light levels (~500 lux) delivered in classrooms or using light delivery wearables may also be protective against myopia. Similarly, the spectro-temporal tuning of such moderate light levels has shown promise for myopia control, especially in animal models. However, the development of tailored light-therapy strategies for myopia control in humans remains challenging given the gaps in understanding the synergetic impact of light parameters within other environmental features. In addition, while working on this review, our team noticed a lack of standardized reporting of experimental light characteristics between studies. The standardization of light reporting through a reporting guideline could allow for a better comparison of findings and protocols between studies, and enable more effective meta-analyses.
During this COVID-19 pandemic, toddlers, children and teenagers alike are exposed to unprecedented amounts of indoor time, sparking concerns over an ever more severe myopia boom.265–268 Notwithstanding these peculiar circumstances, today there is a need for consensus on optimal, feasible and noninvasive light interventions for myopia prevention in children be it through increased time outdoors or adapted architectural lighting or light-therapy devices.
Footnotes
Author contributions
Manuscript writing: ARM, CL, SB, LWYS. Critical revisions: VAB, SSM, DM, RPN. Study design, conceptualization and funding acquirement: RPN.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by A∗STAR-JANSSEN World without disease (A∗JWWD) (18/4/91/00/099), and by Singapore Government through the Industry Alignment Fund – Industry Collaboration Project Grant (I1901E0038) and Johnson and Johnson Vision.
