Abstract
Aim
Calcitonin gene-related peptide (CGRP) plays a key role in migraine and is also known for its immunomodulatory properties. When released from neurons, CGRP can induce T-cell exhaustion markers, such as T-cell immunoglobulin and mucin-domain containing-3 (TIM3) and programmed cell death protein 1 (PD-1) on CD8+ T cells. It remains unclear whether this mechanism occurs in migraine and correlates with disease activity. This study aims to determine whether T-cell exhaustion markers could serve as migraine biomarkers.
Methods
Blood samples were collected from 63 migraine patients at the West German Headache Center and analysed using flow cytometry. The number of monthly headache days (MHD) was assessed retrospectively and correlated with the proportion of CD8+ T cells expressing TIM3 and PD-1. The data was adjusted for confounding factors, including age and sex.
Results
CD8+TIM3+ T cells correlated significantly with MHD (r = 0.313, 95% CI: 0.07–0.52, p = 0.012), while CD8+PD-1+ T cells showed a non-significant negative trend after Bonferroni correction (r = −0.254, 95% CI: −0.47 to −0.01, p = 0.044).
Conclusion
The potential association between T-cell exhaustion markers, such as TIM3, and migraine activity warrants further investigation. Although the clinical value of these biomarkers remains uncertain, future studies should consider including cellular immune components in their analyses.
This is a visual representation of the abstract.
Introduction
Calcitonin gene-related peptide (CGRP) has been shown to play a key role in the pathophysiology of migraine. Calcitonin gene-related peptide is released during a migraine attack 1 and is present at higher concentrations in chronic migraine. 2 Recent studies have demonstrated that neuronal CGRP directly influences T-cell differentiation via interaction with the RAMP3-CALCRL receptor. 3 Furthermore, CGRP released from pain fibres has been shown to drive the expression of T-cell exhaustion markers – including programmed cell death protein 1 (PD-1) and T-cell immunoglobulin and mucin-domain containing-3 (TIM-3) – on cytotoxic CD8+ T cells, an effect mediated through binding the CGRP receptor itself. 4 These findings indicate a neuron-driven modulation of the immune response.
Several studies have evaluated CGRP as a biomarker, with elevated levels reported in chronic migraine. 5 However, methodological limitations, including assay sensitivity, variability between ELISA kits, non-systemic analysis of different CGRP isotypes (alpha or beta), short CGRP half-life, and inter-individual variability, have led to inconsistent results.6,7 Thus, CGRP has not yet been established as a reliable diagnostic or prognostic biomarker.
Consequently, assessing cellular markers influenced by CGRP may be considered as a potential alternative, provided that these markers are more stable and easier to quantify than CGRP itself. Calcitonin gene-related peptide is released by the trigeminal nerve and, in concert with other neuropeptides, contributes to sterile neurogenic inflammation, leading to vasodilation, plasma extravasation and mast cell activation, processes thought to occur during migraine attacks. 8 However, as outlined above, CGRP exerts multiple effects on various cell types, including the modulation of T-cell differentiation and exhaustion.3,4 Nevertheless, its effect on T cells in the context of migraine remains insufficiently understood.
The present study focused on identifying potential CGRP-modulated markers in migraine. We therefore investigated whether T-cell exhaustion receptors on CD8+ T cells are associated with migraine activity using flow cytometry. Demonstrating such an association could enhance our understanding of immune modulation in migraine and help determine whether cellular components may serve as potential biomarkers in the future.
Methods
The study included patients from the West German Headache Center who met the ICHD-3 criteria 9 for either episodic or chronic migraine. The analysis was approved by the independent ethics committee of the University Hospital Essen (21-10133-BO). This study is reported in accordance with the STROBE guidelines for observational studies. 10 After written informed consent was obtained, blood samples were collected for the West German Biobank Essen (WBE). Sample availability depended on patient consent for biobank processing and on the operational feasibility of same-day peripheral blood mononuclear cell (PBMC) isolation. When possible, PBMCs were isolated and stored for later analysis.
All available WBE samples collected between October 2021 and June 2024 from patients diagnosed with migraine were used for the analysis. In cases with two time points, the most recent one was included. The PBMC were stained with Zombie Aqua™ and monoclonal human antibodies directed against CD3-FITC, CD4-PerCP-Cyanine5.5, CD8-BV785, CD56-BV421, PD-1-APC and TIM3-PE (all from BioLegend). Flow cytometry was conducted using a FACSCelesta (BD Biosciences), and downstream analysis was performed using FlowJo 10.10.0. See the Supplementary material for the detailed experimental protocol.
Basic clinical data, including monthly headache days (MHD; three-month average), were obtained retrospectively from medical records. Migraine was classified as episodic or chronic based on MHD, with ≥15 days defining chronic migraine, even when monthly migraine days (MMD) were <8, as distinguishing migraine from headache days in clinical routine is challenging. Patients with known autoimmune diseases, secondary headache diseases, or uncertain diagnoses were excluded. Patients were permitted to use acute medication.
The correlation between MHD and the proportions of CD8+PD-1+ and CD8+TIM3+ T cells was assessed using Pearson's correlation coefficient, with Bonferroni correction for multiple testing (α = 0.025). Data was subsequently adjusted for age and sex using linear regression. Analyses and figures were generated using R (4.3.2) 11 and RStudio (2025.09.1).
Results
A total of 72 samples were collected. One patient was excluded due to idiopathic intracranial hypertension, two due to immune-mediated diseases, and four because of an uncertain diagnosis. One patient had two time points; the most recent was used. One sample failed flow cytometry quality control. The final analysis included 63 patients. Two patients with ≥15 MHD but <8 MMD were classified as having chronic migraine. Table 1 summarizes patient characteristics.
General characteristics of migraine patients.

Abbreviations: AMD = acute medication days; MHD = monthly headache days; MMD = monthly migraine days; NA = not available; SD = standard deviation.
The flow cytometry gating strategy is shown in Figure 1. The proportion of CD8+TIM3+ T cells was significantly correlated with MHD (r = 0.313, 95% CI: 0.07–0.52, p = 0.012; Figure 2). In contrast, CD8+PD-1+ T cells showed a negative trend that did not remain significant after Bonferroni correction (r = −0.254, 95% CI: −0.47 to −0.01, p = 0.044; Supplementary Figure S1). A linear regression model showed that the proportion of CD8+TIM3+ cells was significantly associated with MHD, while age and sex had no significant effect (see Supplementary Table S1).
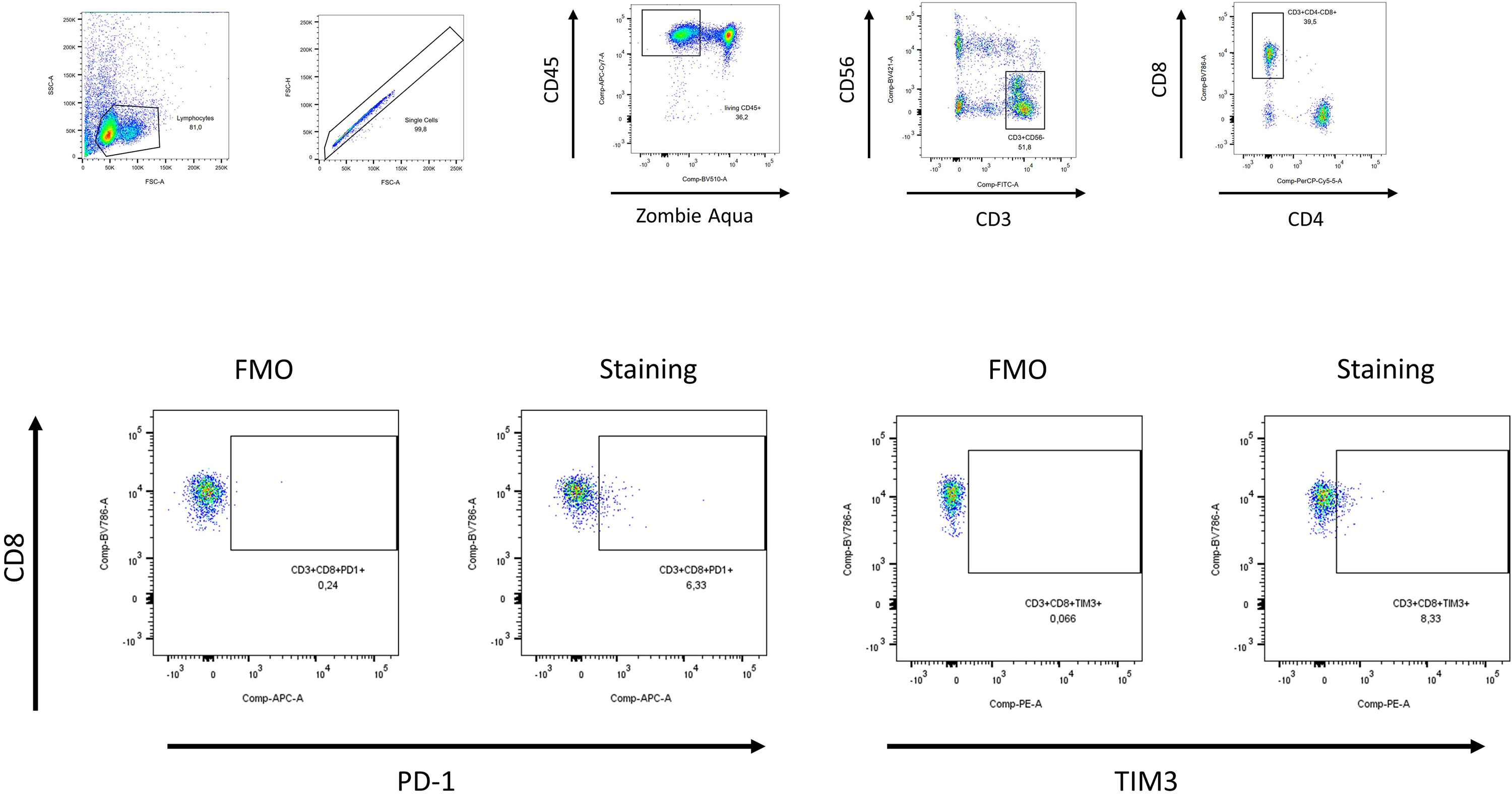
Gating strategy of flow cytometry. FMO = fluorescence minus one control; PD-1 = programmed cell death protein 1; TIM3 = T-cell immunoglobulin and mucin-domain containing-3.

Correlation between CD8+TIM3+ cells and MHD. Each data point represents an individual patient. The regression line is presented with the corresponding 95% confidence interval (grey-shaded area). MHD = monthly headache days; TIM3 = T-cell immunoglobulin and mucin-domain containing-3.
Discussion
In this study, we analysed the T-cell markers of PBMC samples from individuals with migraine, with a particular focus on the CGRP-related induction of the exhaustion markers PD-1 and TIM3. Our aim was to identify immunological alterations associated with migraine activity or chronification and to explore CGRP-influenced cellular biomarkers.
We identified a significant association between TIM3 expression on CD8+ T cells and disease activity (MHD), suggesting a potential immunological consequence of ongoing CGRP release during migraine chronification initiated by trigeminal activation. Animal models support the relevance of immune checkpoint pathways in migraine: PD-1 and PD-L1 are expressed in trigeminal ganglia, and PD-1 inhibition increases CGRP expression, 12 indicating a role in neuronal–immune pain modulation. Neuron-induced T-cell exhaustion may therefore regulate immune responses and promote prolonged inflammation. While the association of TIM3 with disease activity is consistent with this hypothesis, the lack of significance after Bonferroni correction and the negative trend in PD-1 expression may point to distinct immunological mechanisms in migraine or reflect limitations of peripheral blood–based analyses.
Nevertheless, we found no correlation between PD-1 and TIM3 expression on CD4+ T cells and MHD (results not shown), suggesting that CD8+ T cells are more susceptible to T-cell exhaustion in this context. Furthermore, there is evidence that CD8+ T cells may have a protective role in the development of neuropathic pain. 13 It remains to be investigated whether this protective function also plays a role in migraine chronification and whether it is reduced due to T-cell exhaustion.
Elevated levels of CX3CL1, a marker of T-cell recruitment, have been reported in patients with migraine, indicating enhanced immune activation. 14 In chronic pain, clinical response to amitriptyline has been associated with changes in T-cell composition, including reduced CD8+ cells and increased regulatory T cells (Tregs). 15 In vitro, neuronal CGRP promotes T-cell differentiation towards a Treg phenotype, 16 while in episodic migraine Tregs appear less effective than in healthy controls. 17 Although evidence is still limited, these findings support a possible CGRP-modulated, immune-mediated mechanism in migraine, with CGRP-induced T-cell exhaustion potentially contributing to chronic migraine.
However, these considerations require further empirical validation. This preliminary study has several limitations, including heterogeneous treatment regimens, retrospective data collection, the small cohort size and the lack of a non-migraine control group, which should be considered when interpreting the findings. Furthermore, the cohort may not be representative of the general migraine population, as patients were recruited from a tertiary headache center. Additionally, a comparatively higher prevalence of migraine with aura was observed, which may limit the generalizability of the results. Moreover, samples could only be obtained from patients who consented to further processing in the biobank and on days when same-day PBMC isolation was logistically feasible. Altogether, these factors limit the generalizability of the results.
Medication-related effects on receptor expression represent another important limitation. Although patients were permitted to use acute medication, incomplete documentation precluded its inclusion in the regression model; therefore, the potential influence of acute medication cannot be assessed. In addition, CGRP-targeting therapies (monoclonal antibodies or gepants) may modulate TIM3+ expression. While patients receiving CGRP monoclonal antibodies were included, but, to our knowledge, data on their effects on T-cell populations is lacking; therefore, a potential influence on the results cannot be excluded.
Moreover, reproducibility is essential; longitudinal analyses of disease activity are needed to confirm the correlation between TIM3 and MHD, considering potential changes in receptor expression across migraine attacks. Verifying the hypothesis that trigeminally released CGRP influences T-cell exhaustion markers will also necessitate additional in vitro and in vivo studies extending beyond flow cytometry. Furthermore, the correlation identified was relatively weak and variable (r = 0.313, 95% CI: 0.07–0.52), indicating that multiple other factors likely contribute to TIM3 expression.
Despite these limitations, these findings therefore suggest a promising direction for future research and highlight the value of incorporating cellular marker analyses into migraine biomarker studies.
Clinical implications
Our data suggests a potential association between the T-cell exhaustion marker TIM3 and migraine disease activity.
T-cell exhaustion may play a role in the pathophysiology of migraine or represent a secondary effect of the disease.
Further research using cellular markers is needed to confirm these findings and to explore the potential of TIM3 as a biomarker for migraine.
Supplemental Material
sj-docx-1-rep-10.1177_25158163261444000 - Supplemental material for Revealing the immune face: TIM3 as a window into T-cell immunity in migraine? – A retrospective cohort study
Supplemental material, sj-docx-1-rep-10.1177_25158163261444000 for Revealing the immune face: TIM3 as a window into T-cell immunity in migraine? – A retrospective cohort study by Armin Scheffler, Paolina Krois, Martha Kiljan, Christoph Kleinschnitz, Manfred Schedlowski and Dagny Holle-Lee in Cephalalgia Reports
Supplemental Material
sj-png-2-rep-10.1177_25158163261444000 - Supplemental material for Revealing the immune face: TIM3 as a window into T-cell immunity in migraine? – A retrospective cohort study
Supplemental material, sj-png-2-rep-10.1177_25158163261444000 for Revealing the immune face: TIM3 as a window into T-cell immunity in migraine? – A retrospective cohort study by Armin Scheffler, Paolina Krois, Martha Kiljan, Christoph Kleinschnitz, Manfred Schedlowski and Dagny Holle-Lee in Cephalalgia Reports
Footnotes
Acknowledgements
Human biological samples and related data were provided by the Westdeutsche Biobank Essen (WBE, UniversityHospital Essen, University of Duisburg-Essen, Essen, Germany; approval 21-WBE-123). Following Large Language Models were used to improve English quality: DeepL Wright, ChatGPT 5.2. The graphical abstract was prepared using Microsoft PowerPoint (Microsoft Corporation, Redmond, WA, USA) and is based on data generated with R and FlowJo, which are referenced in the manuscript.
Ethical considerations
The ethics committee of the University Hospital Essen approved the study (21-10133-BO).
Consent to participate
Written informed consent was obtained.
Consent for publication
The authors agree to publish with Cephalalgia Reports.
Author contributions
Armin Scheffler, Dagny Holle-Lee, Martha Kiljan and Manfred Schedlowski were responsible for study concept and design. Armin Scheffler and Paolina Krois were responsible for acquisition of data. Armin Scheffler, Martha Kiljan and Paolina Krois analyzed and quantified the data. Armin Scheffler drafted the manuscript and figures. Christoph Kleinschnitz provided editorial support for the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Armin Scheffler and this project was part of the UMEA Clinician Scientist programme, funded by Faculty of Medicine, University of Duisburg-Essen and German Research Fund (DFG).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dagny Holle-Lee has received scientific support and/or honoraria from Biogen, Novartis, Eli Lilly, Sanofi-Aventis, Teva, Allergan and Hormosan. Christoph Kleinschnitz has received honoraria, a consulting or advisory role to declare from Novartis and Teva. Armin Scheffler has received travel fees and/or honoraria from Teva and Novartis.
Data availability statement
Original data is available upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
