Abstract
Background
Recently, amylin and its receptors were found in different structures involved in migraine pathophysiology. Here, we evaluate interictal concentrations of amylin and calcitonin gene-related peptide in peripheral blood as biomarkers for chronic migraine.
Methods
We prospectively recruited patients with episodic migraine, chronic migraine and healthy controls. Interictal amylin and calcitonin gene-related peptide levels were assessed in blood samples using enzyme linked immunosorbent assay.
Results
We assessed plasma samples from 58 patients with episodic migraine (mean age 37.71 ± 10.47, 87.9% female), 191 with chronic migraine (mean age 46.03 ± 11.93, 95% female), and on 68 healthy controls (mean age 43.58 ± 11.08 years, 86% female). Body mass index was 25.94 ± 4.53 kg/m2 for migraine patients and 25.13 ± 4.92 kg/m2 for healthy controls (
Conclusion
Interictal plasma amylin levels are higher in patients with chronic migraine and may serve as a diagnostic biomarker for chronic migraine.
Introduction
Chronic migraine (CM) is diagnosed when there is headache on at least 15 days per month for more than 3 months, with features of migraine on at least 8 days per month (1). CM affects 0.5% to 5% of the general population (2), imposing a substantial personal and societal burden on those affected (3). CM evolves from episodic migraine (EM), with an annual conversion rate of about 3% (4). Despite its considerable impact on daily activities and quality of life, CM remains underdiagnosed and undertreated (3,4). Early diagnosis of CM is a major challenge because of the extremely variable clinical phenotypes and thus identifying novel biomarkers will help improve CM diagnosis. In addition, it is likely that such biomarkers will shed further light on the mechanisms involved in the process of chronification.
Calcitonin gene-related peptide (CGRP) is a peptide that plays a key role in migraine (5). Although interictal CGRP levels have been previously studied in CM patients (6,7), the value of CGRP as a CM biomarker remains controversial. Recently, different clinical trials in patients with CM have shown that monoclonal antibodies (MAbs) targeting either CGRP or its receptor produce significant benefits (8). Yet, a subset of patients appears to be resistant to this therapy, suggesting the possibility of the existence of additional mediators that contribute to migraine.
Amylin (also known as islet amyloid polypeptide, IAPP) is a 37 amino acid peptide produced and secreted by β-cells in the pancreas, along with insulin (9,10). Amylin and CGRP are structurally related peptides that belong to the calcitonin peptide family and share some receptors and biological activities (10). Amylin and the amylin receptors are found in different structures involved in pain modulation in the context of migraine, including the trigemino-vascular system (11). Furthermore, a handful of preclinical studies suggested that amylin could have vasodilatory and pronociceptive actions (12,13). Therefore, it seems reasonable to hypothesise that amylin or its receptors may play a causative role in migraine.
Based on this hypothesis, this study set out to compare the interictal amylin and CGRP levels in peripheral blood between controls and patients with migraine, and to evaluate and compare the performance of CGRP and amylin in diagnosing CM in a large cohort of adults.
Patients and methods
Participants, settings, and clinical evaluations
We carried out this prospective, two-center, case-controlled study at two different tertiary outpatient headache clinics where adult patients with migraine were recruited, all between 18 and 65 years of age. The patients were diagnosed as having EM or CM for at least 12 months prior to enrolment, in accordance with the criteria of the International Classification of Headache Disorders, 3rd edition (ICHD-3) (1). The exclusion criteria were: a) severe systemic disease or disease that could alter the levels of neuropeptides, such as active infection, recent myocardial infarction or surgery (within the past 3 months), renal failure, severe liver disease, diabetes, inflammatory or neoplastic disease; b) immunosuppressed patients requiring treatment with steroids or immunosuppressants, HIV-infected patients, people with an addictive disorder to drugs or alcohol; c) patients who had recently started (in the 8 weeks prior to enrolment) a preventive migraine treatment, or who consumed opioids; d) pregnant or breastfeeding women; e) patients with a major psychiatric disorder or intellectual disability; and f) morbidly obese patients according to body mass index (BMI) criteria. Furthermore, patients who were on preventive medication should have been on a stable dose for at least 2 months prior to recruitment onto the study.
Healthy controls (HC) consisted of healthy individuals, most of them employees from both university hospitals, matched with patients with migraine in terms of age, sex and BMI, without a personal or family history of migraine headaches and who complied with all the exclusion criteria. They were interviewed directly, and they did not have a personal history of chronic pain syndromes and were not taking any medication. After a screening visit, we invited eligible participants to complete a structured questionnaire. In patients with migraine, the type of migraine (with or without aura), the migraine frequency (episodic or chronic), the intensity of their headaches (measured as the average score on a visual analogue scale), the presence of allodynia, the duration of the disease (years) and other associated symptoms were registered. A complete medical record was available for all the participants, and their physical examination (with blood pressure readings), extensive blood tests (including fasting glucose) and neuroimaging data (where appropriate) were unremarkable in all cases. Basic anthropometric measurements were also obtained, including height, weight and BMI.
Ethics
The study received approval by the local research Ethics Committee of the Clinica Universidad de Navarra (approval number 109/2015), following the standards for medical research in humans recommended in the Helsinki Declaration. All participants provided their written informed consent prior to their enrolment on the study.
Data availability statement
The data that support the findings of this study are available from the corresponding authors on request
Laboratory tests
We extracted blood samples from all eligible participants in the non-dominant forearm after overnight fasting. The blood samples were obtained between 08:00 and 12:00 h. The blood (10 ml) was collected into BD P100 Vacutainer blood collection tubes coated with EDTA. Tubes were centrifuged within 1 h and no longer than 2 h after the extraction, in a pre-cooled centrifuge at 3.000
A researcher blind to the clinical data measured plasma amylin and CGRP levels using commercial ELISA kits and following the manufacturer’s recommendations. All determinations were performed in a single laboratory (Clínica Universidad de Navarra). One undiluted plasma aliquot, thawed one time, was used for each ELISA. For plasmatic amylin assessment, given the homology between the two peptides studied, an ELISA kit specific and sensitive for this peptide (EZHA-52K, Merck, Germany) was used, with CGRP-like immunoreactivity inferior to 1%, as reported by the manufacturer. The capture antibody recognises reduced human amylin and human amylin acid (deamidated amylin) but not the 1–20 fragment of amylin. All samples included in the study were tested in duplicate. The total amylin concentration was derived from a six-point standard curve generated with serially diluted standards of human amylin. Two quality control samples were included in each assay, for which the inter- and intra-assay coefficient of variability was 5.35% and 3.07%, respectively. For CGRP plasmatic assessment, a specific and sensitive ELISA kit was used (CGRP ELISA kit, A05481, Bertin Bioreagent, France). The amylin-like immunoreactivity of this ELISA kit was less than 0.01% according to manufacturer’s protocol. All samples were run in duplicate and the total CGRP concentration was derived from an eight-point standard curve generated with serial dilutions of recombinant CGRP standards. One quality control sample was included in each assay, which had an inter- and intra-assay coefficient of variability of 11.60% and 8.43%, respectively. All fluorescence values were measured on a spectrofluorimeter (PerkinElmer, EnVision 2104 multilabel reader, USA). Results were calculated using a standard curve, which was performed in all the plates, to determine the concentrations of both amylin and CGRP. Standard curves were constructed using the standard peptide included in the assays. These standard curves of six (amylin) or eight (CGRP) points in duplicate were lineal fixed with correlation coefficients higher than 0.95. After subtracting the blank values, the levels of absorbance were interpolated in its corresponding standard curve to obtain the reported concentration values of the peptides. All the values were in the linear range of standard curves. Moreover, each ELISA plate contained two quality control standards that were run in duplicate. These values were always between the parameters stated as correct in the manufacturer protocol.
Statistical analysis
Standard statistical analyses were performed using the MedCalc Statistical Software version 19.1.3 (MedCalc Software bv, Ostend, Belgium: https://www.medcalc.org 2019) and SPSS Statistics for Windows software (Version 26.0, Released 2019: IBM Corp., Armonk, NY). We estimated that a minimum sample size of 112 patients with migraine (56 with CM and 56 with EM) and 42 HC would be necessary to detect a mean amylin concentration difference between the study groups of 10 pg/mL, with a type I error of 0.05 and a power of 90%, assuming a SD of 15 pg/mL and a loss of 15% to follow-up.
Descriptive statistics involved the use of the number (percentage), mean (SD), mean 95% confidence interval (95% CI), mean (SEM) or median 95% CI as appropriate. Missing data for continuous variables were allocated by means of a multiple imputation algorithm (14) and we examined the distribution of continuous variables with a D’Agostino-Pearson test. The Mann–Whitney U test was used to evaluate the continuous clinical and demographic parameters between migraineurs and HC, and between EM and CM. The one-way ANOVA test or the Kruskal-Wallis test were used to compare differences between groups (CM, EM, and controls).
A logistic regression analysis was performed to assess the association between the plasma amylin and CGRP concentrations and the risk of suffering migraine. As such, amylin and CGRP levels were grouped in quartiles. Receiver operating characteristics (ROC) curves were constructed to assess the sensitivity and specificity of amylin and CGRP to predict a diagnosis of migraine. The Areas Under the ROC curves (AUROCs) were compared by the method of DeLong et al. (15). The optimal point on the ROC curves that provided best discrimination was determined for amylin and CGRP. From the sensitivity and specificity determined from that optimal point, positive and negative predictive values, and positive and negative likelihood ratios were calculated.
Categorical variables were compared using a Chi-squared test and a Fisher’s exact test, as necessary. A
Results
Participants
This study was carried out on 191 patients with CM (mean age 46.02 ± 11.92 years; range 18–65 years), 58 patients with EM (mean age 37.70 ± 10.47 years; range 18–61 years) and 68 HC (mean age 43.58 ± 11.08 years; range 25–64 years). There were no significant differences in age, sex or BMI between patients with migraine and HC. Clinical characteristics of patients with migraine and HC are displayed in Table 1. There was a statistically significant difference in age between patients with CM and those with EM (
Comparison of the demographic and clinical data between patients with episodic and chronic migraine.

CM: chronic migraine; EM: episodic migraine; BMI: body mass index; HC: healthy controls; NA: Not applicable; Migraine: patients with EM and CM; VAS: visual analog scale.
aTwo-tailed independent samples Student’s t/Mann–Whitney U tests.
bChi-squared test/Fisher’s exact test.
Comparative analysis of CGRP and amylin levels between groups
Mean plasma levels of amylin were significantly higher in CM (47.10 ± 26.6 pg/mL) than in the EM (28.84 ± 18.8 pg/mL,
Comparison of the amylin and CGRP levels in HC and patients with CM and EM.

CGRP: calcitonin gene related peptide; CM: chronic migraine; EM: episodic migraine.
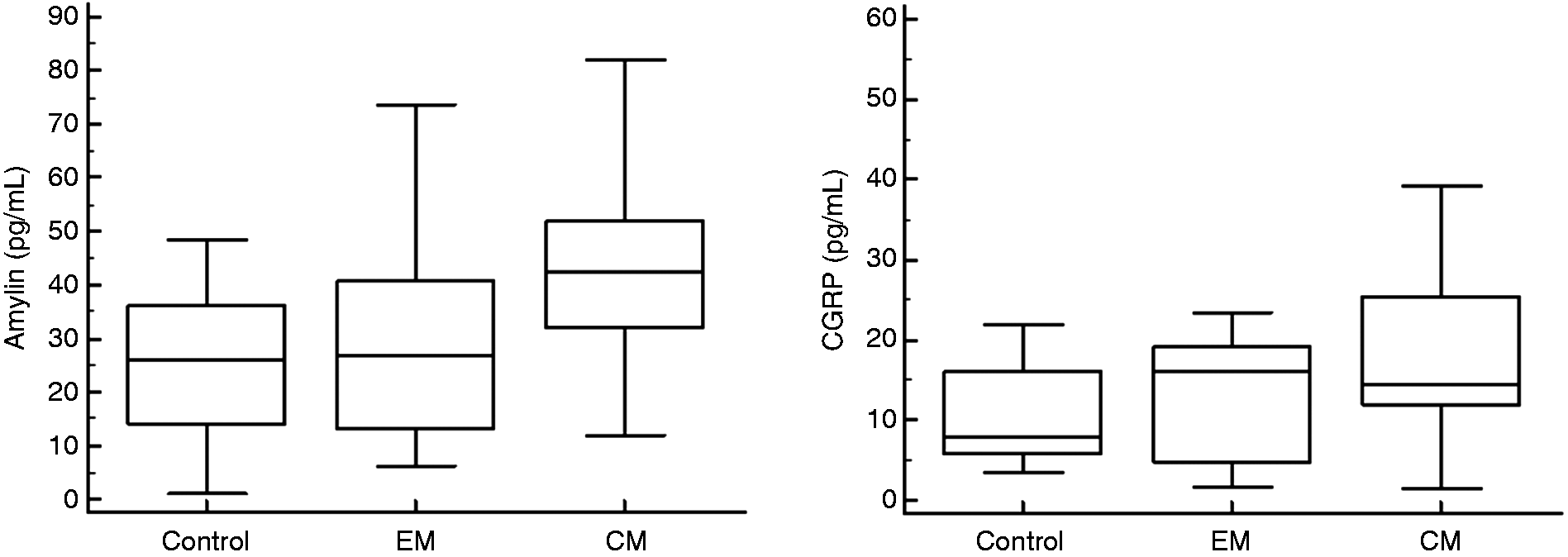
Plasma amylin and CGRP concentrations for HC, EM and CM.
The value of the plasma amylin and CGRP concentrations as a predictor for CM was assessed in CM patients. First, the participants were divided into four quartiles according to amylin levels. For each quartile increase in amylin, the odds of having CM, relative to the HC, increased by 2.90 (95% CI: 2.10–4.00,
Nonetheless, the plasmatic amylin concentrations showed a good correlation with the CGRP levels in patients with CM (r = 0.50;
Diagnostic performance of amylin and CGRP
In the ROC analyses, the AUC when comparing patients with migraine (EM and CM) and HC was 0.74 (95% CI: 0.69–0.79) for plasma amylin and 0.61 (95% CI: 0.53–0.69) for CGRP (Table 3).
Sensitivity, specificity, positive predictive value and negative predictive value of amylin and CGRP for the diagnosis of CM.

AUROC: area under the receiver operating characteristics; CI: confidence interval; CGRP: calcitonin gene related peptide; CM: chronic migraine; Migraine: patients with episodic and CM; NPV: negative predictive value; PPV: positive predictive value.
Using a cut-off of 39.68 pg/mL for the plasma amylin concentration (95% CI: 21.89–42.87), the sensitivity when differentiating migraine (episodic and chronic) from HC was 50.2% and the specificity was 88.2% (Figure 2). For CGRP, using a cut-off of 9.43 pg/mL (95% CI: 9.30–21.88), the sensitivity and specificity when differentiating migraine from HC was 72.3% and 60.3% respectively. The AUROC for the comparison between CM and HC was 0.80 for plasma amylin (95% CI: 0.75–0.85), and 0.63 for CGRP (95% CI: 0.56–0.68, Figure 3). Using a cut-off of 39.68 pg/mL for the plasma amylin concentration (95% CI: 29.69–45.30), the sensitivity and specificity to differentiate CM from HC was 57.6% and 88.2% respectively (Table 3, Figure 3). For CGRP, with thresholds of 9.43 pg/mL (95% CI: 9.30–21.88) the sensitivity was 74.4% and the specificity 60.3% to differentiate CM from HC.

ROC-curve analysis for amylin and CGRP, comparing with migraine patients (episodic and chronic) and HC. (a) AUC for amylin 0.74 (95% CI 0.69–0.79,

ROC curve analysis for amylin and CGRP, comparing the CM group with HC. (a) AUC for amylin 0.808 (95% CI 0.755–0.854,
The same methodology was used to discriminate between CM and EM, and the AUROC curve for plasma amylin was 0.74 (95% CI: 0.68–0.79) and 0.53 (95% CI: 0.47–0.59) for CGRP. Using a cut-off of 34.22 pg/mL for plasma amylin (95% CI: 20.39–42.49), the sensitivity was 69.1% to differentiate CM from EM and the specificity was 72.4%. For a plasma CGRP concentration of 15.21 pg/mL (95% CI: 5.71–16.71), the sensitivity was 70.16% and the specificity 58.62% to differentiate CM from EM. Finally, we did not observe that the use of plasma CGRP and amylin together improved the diagnostic accuracy to differentiate CM from HC compared with plasma amylin concentration alone.
Discussion
In the present work, we described an increase in the interictal plasma levels of CGRP and amylin in patients with CM relative to healthy participants without a history of headache. More importantly, we found that plasma amylin concentrations showed a better diagnostic performance than CGRP in differentiating CM from HC in adult patients. We also observed that, in patients with CM, plasma amylin, but not CGRP concentrations, were significantly increased when compared with those patients with EM. Although the value of interictal plasma amylin for CM diagnosis is limited due to insufficient sensitivity, our findings suggest that increased plasma amylin levels may be implicated in migraine chronification, opening new possibilities for the development of preventive therapies that target amylin receptors.
The increased interictal plasma CGRP concentrations detected in individuals with migraine are consistent with those identified in previous studies (6,16,17), but not in all of these (7). Such discrepancies may be due to technical issues or even the clinical characteristics of the populations included in each study (6,7). It was previously found that CGRP levels were significantly higher in women with CM and a history of migraine with aura attacks than in those who had never experienced an aura (6). However, we did not find any difference between the plasma CGRP or amylin concentrations in either men or women, nor in migraine sufferers with or without aura. Moreover, clinical variables such as age, analgesic overuse, depression and the use of preventive treatment did not appear to influence the plasma CGRP or amylin levels. In addition, CGRP and amylin levels were not associated with the appearance of allodynia, in accordance with recent clinical (7) and experimental observations (18).
It is largely unknown how the increase in amylin influences migraine. Amylin, CGRP and their receptors are expressed throughout the trigemino-vascular system (11,19) and may be expressed in sensory neurons (20,21). Previous studies suggest that high CGRP levels could reflect persistent activation and subsequent sensitisation of the trigemino-vascular system (6), and may indicate a mechanism driving the progression of EM to CM due to a shift from CGRP involved activity-dependent to activity-independent central sensitisation (22). Elevated interictal amylin may function somewhat similarly to elevated CGRP. The activation of the trigemino-vascular system could be induced by amylin either through a direct effect on the trigeminal ganglion or an interaction with CGRP, as AMY1 receptors can be activated by CGRP (9,23). On the other hand, amylin may have some actions in the CNS. Although amylin expression data is not available for the human brain (11), evidence from animal studies shows limited amylin expression in the CNS (10,11). Amylin and the amylin receptors are present in the hypothalamus (24,25), the brainstem (24,26) and the circumventricular organs (11,24), structures that are involved in the pathophysiology of migraine (27). Taking into account that the proportion of amylin that crosses the blood-brain barrier is very low (11), central actions of amylin may occur predominantly outside the blood-brain-barrier, mainly through the circumventricular organs (10,11). Recently, it was demonstrated that prolonged amylin administration in a rat chronic neuropathic pain model of spared nerve injury worsened cold allodynia, suggesting a pronociceptive role, which the authors presume may result from supraspinal inputs including the aforementioned circumventricular organs (28). We argue that amylin may contribute to migraine chronification predominantly via sensitising trigeminal neurons, but actions on circumventricular organs, and the hypothalamic and brainstem neurons may also occur.
No previous studies have assessed plasma amylin levels in patients with migraine or other pain conditions. However, amylin has been studied extensively in patients with obesity, a modifiable risk factor for migraine progression (4,29). Interestingly, the levels of amylin are elevated in patients with obesity (30), and experimental data suggest that amylin may contribute to the activation and sensitisation of the trigemino-vascular system observed in obesity (31). In this study, patients with morbid obesity were excluded and no relationship between amylin and BMI was found.
Direct evidence for a causative role of CGRP in migraine comes from clinical studies in which exogenous intravenous infusion of CGRP to migraine patients can trigger a migraine-like headache (32). To date, no infusion of amylin has been performed in patients with migraine to establish whether amylin may cause headache, but some information about the effect of amylin can be obtained from the effects of the amylin analog pramlimtide, indicated for patients with diabetes (33,34). The most frequent adverse effect of pramlintide in clinical trials was nausea (10,33,34), a common and disabling symptom of migraine, and headache was also reported in some studies (34). Although the exact mechanism for the adverse effects of pramlintide is presently unknown, the occurrence of nausea is thought to be mainly mediated by stimulation of specific amylin receptors in the circumventricular area postrema (10,34) through the activation of the nucleus of the solitary tract, which is also activated in migraine patients who experience nausea (35).
Four MAbs targeting the CGRP ligand or its receptor are currently available for migraine prevention (8). Erenumab has high affinity for the CGRP receptor and is reported not to significantly block other human calcitonin family receptors, including amylin receptors, although detailed characterisation of specific amylin receptors has not yet been reported (36). On the other hand, MAbs directed against the ligand may prevent CGRP from binding to amylin receptors (10,19,23). However, the available data on the safety and efficacy of the different MAbs are similar and do not allow us to establish that the additional action on the amylin receptors is relevant from the clinical point of view (8,19). Future studies should elucidate the importance of amylin receptors in migraine pathophysiology.
The strengths of our study are that the sample collection, and the CGRP and amylin measurements, were performed on a large number of patients attending specialist headache units and under strict levels of quality control. Also, migraine participants were in good physical health, as we employed strict exclusion criteria to eliminate possible effects of most potential confounders. Possible limitations include the fact that our patients were recruited from a selected headache clinic population. While we believe our study to be representative of the population as a whole, future studies could eliminate this concern by addition of a primary care center and a newspaper/radio recruitment arm to that of headache specialty practice. Additionally, the sample size between patients with CM and the other two groups was different. Nevertheless, sample size calculation was performed before starting the study, and both HC and EM groups included a sufficient number of subjects. Moreover, the changes in plasma amylin were analysed in cross-sectional data and using only one assay for each peptide. Furthermore, it is important to understand that amylin and CGRP measurements may be influenced by multiple factors including patients’ comorbidity, pre-analytical methodology and the assay kit used. Finally, another possible limitation is that most of our migraine patients (80%) were receiving preventive treatments, which could have influenced the levels of CGRP and amylin. However, for ethical reasons, migraine prophylaxis was not discontinued.
The data presented here demonstrate, for the first time, that interictal plasma amylin levels were elevated in peripheral blood in patients with CM. Further studies will be needed to clarify the pathophysiological relevance of this increase in amylin concentrations in relation to migraine chronification, as well as to establish the value of amylin as a biomarker of CM in routine clinical practice and the potential of amylin receptors as novel therapeutic target for migraine prevention.
Article highlights
Interictal plasma amylin levels are elevated in peripheral blood in patients with CM. Plasma amylin concentrations showed a better diagnostic performance than CGRP in differentiating CM from HC in adult patients. Our findings suggest that amylin is involved in migraine chronification, opening new possibilities for the development of preventive therapies that target amylin receptors.
Footnotes
Acknowledgements
We wish to particularly acknowledge the patients for their willingness to participate in this study and to the Biobank of the University of Navarra for its collaboration. We want to thank the “Fundación Fuentes Dutor” for its support to the research performed at the Center for Applied Medical Research (CIMA) of the University of Navarra.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Spanish Research Network on Cerebrovascular Diseases RETICS-INVICTUS plus, grant/award RD12/0014/0009; Center for Applied Medical Research (CIMA), University of Navarra; Spanish Ministry of Economy and Competitiveness–Instituto de Salud Carlos III, grant/award PI15/01578.
The storage and use of human biological samples was in part supported by the Biobank of the Foundation for Applied Medical Research (CIMA, University of Navarra, Pamplona, Spain).
