Abstract
Current antimigraine drugs are believed, besides their direct vasoconstrictive effect, to inhibit calcitonin gene-related peptide (CGRP) release from trigeminal nerve endings during migraine.
Objective
The objective of this report is to establish a biomarker for the CGRP-interfering effect of antimigraine drugs.
Methods
We quantified the effect of sumatriptan on the trigeminal nerve-mediated rise in forehead dermal blood flow (DBF), induced by capsaicin application (0.6 mg/ml) and electrical stimulation (0.2–1.0 mA), in a randomised, double-blind, placebo-controlled, crossover study in healthy male (n = 11, age ± SD: 29 ± 8 years) and female (n = 11, 32 ± 7 years) individuals.
Results
DBF responses to capsaicin were attenuated by sumatriptan (ΔDBF, mean ± SEM: 82 ± 18 AU, p = 0.0002), but not by placebo (ΔDBF: 21 ± 12 AU, p = 0.1026).
Conclusion
We demonstrated that sumatriptan inhibits increases in DBF, induced by the release of, most likely, CGRP. Thus, our model may be used as a biomarker to establish the trigeminovascular effects of (potential) antimigraine drugs, such as CGRP receptor antagonists or antibodies directed against CGRP or its receptor.
Introduction
Understanding the mechanism of action of drugs helps to unravel underlying disease pathophysiology. Although the antimigraine drug sumatriptan, one of seven triptans currently available, was introduced about two decades ago, its precise action remains elusive. Besides direct vasoconstriction, mediated by the 5-HT1B receptor (1), triptans may also presynaptically inhibit release of the key neuropeptide in migraine, calcitonin gene-related peptide (CGRP) (1), most likely via 5–HT1D(/1F) receptors (1). Despite its success, 14%–30% of patients do not respond to subcutaneous sumatriptan (2). To understand the mechanism behind therapeutic nonresponse in migraine, a non-invasive biomarker, e.g. based on sumatriptan’s effect on trigeminal CGRP release, is needed. This is especially relevant now that antagonists and antibodies directed against CGRP or its receptor are being developed (3). Early trials indicate they are not effective in all patients, reinforcing the importance of investigating their trigeminovascular effects (3). To identify such a biomarker, we performed a randomised, double-blind, placebo-controlled, crossover study in healthy volunteers to investigate the blocking effect of sumatriptan, the most established acute antimigraine therapy (1), on the CGRP-mediated rise of capsaicin-induced dermal blood flow (DBF). We hypothesised that sumatriptan inhibits the capsaicin-induced DBF increases, which are most likely mediated by release of CGRP. To exclude physiological antagonism by direct vasoconstriction to sumatriptan, we used electrical stimulation (ES) of trigeminal afferents, increasing DBF partly via CGRP, but also via other mechanisms such as substance P and prostaglandin synthesis (4).
Methods
Standard protocol approvals, registrations and patient consents
The study protocol was reviewed and approved by the Ethics Committee of Erasmus MC, Rotterdam, and registered at the Netherlands Trial Register (ID: NTR4171). All participants gave written informed consent and the study was conducted in accordance with local laws, the ethical principles of the Declaration of Helsinki and the principles of Good Clinical Practice.
Design and procedures
Healthy non-smoking male and female individuals, aged 18–50 years (body mass index (BMI) 18–30 kg/m2), without history of cardiovascular disease or use of medication, were eligible. Females were using oral contraceptives and continued their use without the ‘stop week’ to avoid the confounding influence of varying steroid levels (5). The validated Leiden University Migraine Neuro Analysis (LUMINA) questionnaire (6) was used to exclude migraineurs.
Participants were investigated between September and November 2013 in a quiet, temperature-controlled room. They rested supine and did not speak. For all individuals, two visits were scheduled with a one-week washout in between and during the same time of day. Participants refrained from using vasoactive drugs (including nonsteroidal anti-inflammatory drugs (NSAIDs)) for >48 hours, and from consuming alcohol, caffeine-containing beverages and chocolate for >3 hours prior to the start of experiments. A light meal three hours before the start of the experiments was allowed.
Blood pressure and DBF responses to capsaicin (0.6 mg/ml) and ES (0.2–1.0 mA) were measured before and 30 minutes after either placebo or sumatriptan during the two research visits. Measurements before administration of sumatriptan or placebo were always performed on the right side of the forehead; measurements after administration of sumatriptan or placebo were performed on the left side of the forehead. As described in our method validation paper (4) DBF responses on the right and left side of the forehead are identical. Capsaicin solution and physiological saline for ES were placed in reservoirs specifically designed for this purpose (Perimed AB, Järfälla, Sweden). For electrical stimulation of the forehead skin, iontophoresis of physiological saline was applied. Capsaicin- and ES-induced dermal forehead vasodilatation, measured with a laser Doppler perfusion imager, is described in detail in our model validation paper (4).
Participants were injected subcutaneously with sumatriptan (6 mg; a gift from GlaxoSmithKline, Zeist, the Netherlands) or placebo (saline) at the lateral side of the upper thigh. Randomisation, allocation and blinding were coordinated by the Erasmus MC pharmacy. ‘Insulin syringes’ with placebo or sumatriptan were prepared and labelled on the experiment day in a room inaccessible to the researchers. With a questionnaire at the end of each experiment, side effects were registered.
Statistical analysis
Sample size was based on detection of a 25% decrease in capsaicin-induced DBF after sumatriptan (4), at 5% significance (two-tailed) with 80% power. Baseline and maximal DBF (Emax) response, expressed in arbitrary units (AU), to capsaicin and ES, was calculated before and after sumatriptan or placebo. Differences in DBF responses to capsaicin (our primary endpoint) and ES during sumatriptan and placebo were calculated for each participant. Responses to sumatriptan and placebo were compared within participants using Student’s paired t-test. Current response curves of the ES sequence (0.2 mA–1.0 mA) were analysed with repeated-measures analysis of variance (ANOVA). A p < 0.05 was considered to indicate significance.
Results
Twenty-two healthy volunteers (11 males) participated (Figure 1 and Table 1). Since results were similar between genders, data were pooled.
Recruitment flow diagram. Demographics of the study population. BMI: body mass index; BP: blood pressure measured recumbent; HR: heart rate. Mean ± (SD).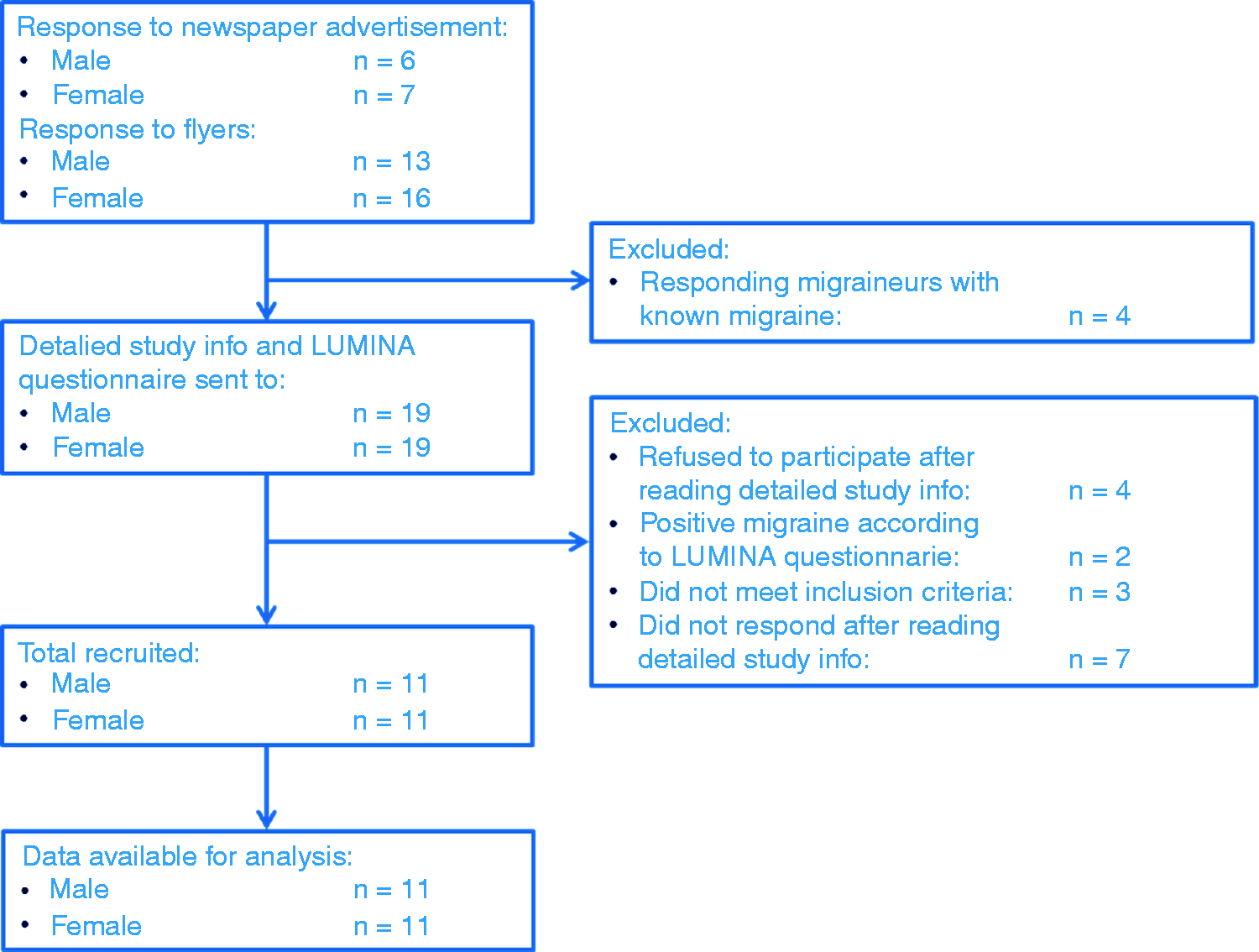

Systolic and diastolic blood pressure significantly increased after sumatriptan, while diastolic blood pressure slightly increased after placebo (Supplementary Table 1).
Baseline DBF decreased after sumatriptan (ΔDBF: 11 ± 4 AU, p = 0.0126), but not after placebo. The DBF response to capsaicin decreased after sumatriptan (ΔDBF: 82 ± 18 AU, p = 0.0002) but not after placebo (ΔDBF: 21 ± 12 AU, p = 0.1026, Figure 2(a)). ΔDBF sumatriptan was different from ΔDBF placebo (p = 0.0036). A sub-analysis showed that in 5/22 volunteers the DBF response to capsaicin did not decrease after sumatriptan. The DBF response to ES was not affected by either sumatriptan or placebo (Figure 2(b)).
Forehead dermal blood flow (DBF) responses to capsaicin and electrical stimulation (ES). (a) Maximal DBF response to capsaicin before (grey bars, pre-) and after (black bars, post-) placebo and sumatriptan. (b) Maximal DBF responses to ES with an increasing stimulation current before placebo (grey triangle), after placebo (black circle), before sumatriptan (grey diamond) and after sumatriptan (black square). Data are presented as mean ± SEM. *Significant decrease in DBF response to capsaicin after sumatriptan.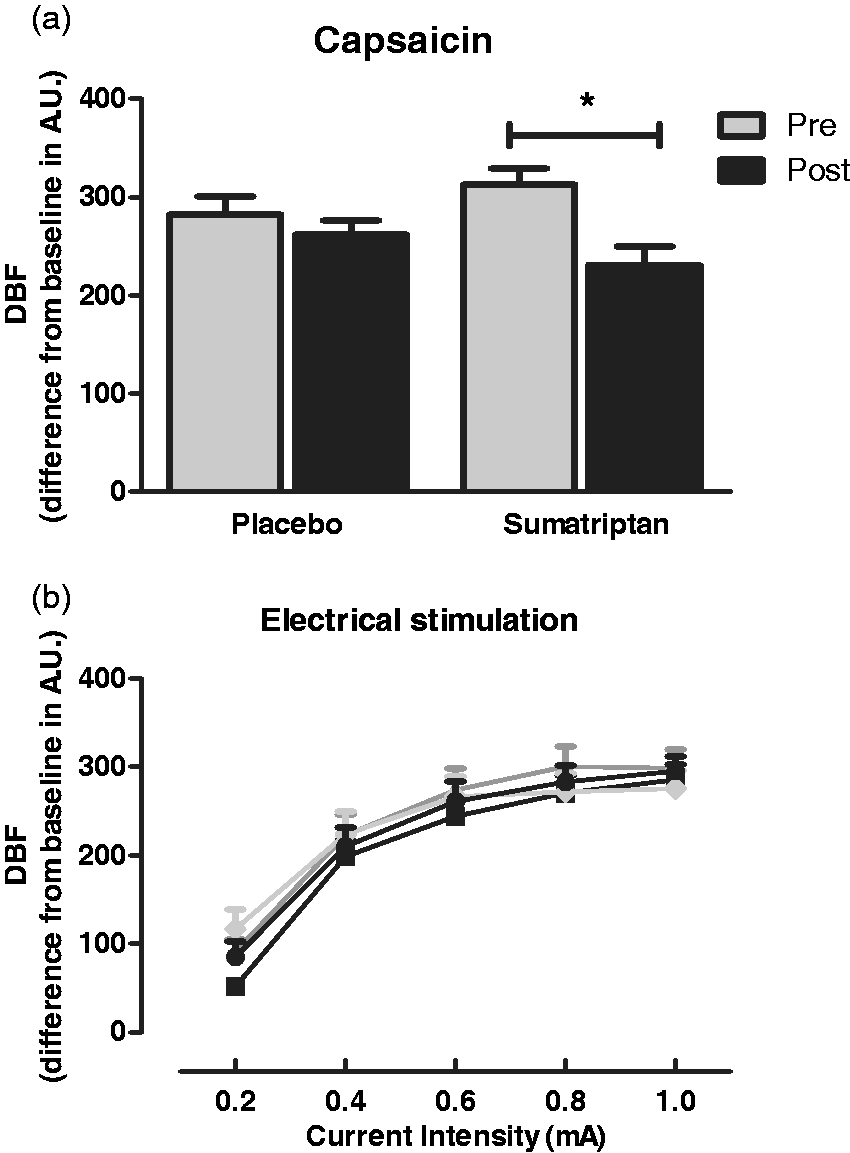
Pain at the injection site was the most reported side effect after sumatriptan (15/22) compared to placebo (6/22), followed by a general heavy pressing feeling (14/22 after sumatriptan versus 1/22 after placebo). Eight of 22 volunteers reported stiffening of the neck muscles after sumatriptan; this was not reported after placebo. Anxiety was reported by 4/22 volunteers after sumatriptan and by none after placebo.
Discussion
Our major finding is the inhibition of capsaicin-induced vasodilatory DBF responses after sumatriptan but not after placebo, confirming our hypothesis that sumatriptan inhibits the release of, most likely, CGRP, which is the key neuropeptide in migraine. Blood pressure changes (1) and side effects were as expected. While side effects may have reduced the blinding efficiency in some participants during recording, a potential limitation of our study, it is important to highlight that data analysis was performed completely blinded. Although our study was performed in individuals without migraine, the proportion (5/22, i.e. 23%) who did not display inhibition of capsaicin-induced DBF increases after sumatriptan is in the same range as the number of non-responders to subcutaneous sumatriptan (2). We did not investigate whether clinical non-responders to subcutaneous sumatriptan do correlate to the non-responders in our model because the design and proper execution of such a trial is complicated due to the seven different triptans that are now available as well as the heterogeneity in the definition of an adequate response to triptans among different studies (7).
The decrease in baseline DBF after sumatriptan indicates direct vasoconstriction, in agreement with several preclinical (8) and clinical (magnetic resonance angiography, MRA) studies (9,10). Nevertheless, not all MRA studies are consistent. A study with provoked migraine in patients demonstrated sumatriptan-induced constriction of the middle meningeal but not the middle cerebral artery, while sumatriptan constricted both arteries in healthy volunteers (9). In spontaneous migraine attacks, sumatriptan constricted extracranial arteries and the cavernous portion of the internal carotid artery (10). The constriction of the cranial vasculature in these studies did not uniformly coincide with the resolution of pain, suggesting involvement of additional mechanisms in the efficacy of triptans. Indeed, several studies suggest that part of the action of triptans is mediated via inhibition of neuropeptide release. For example, elevated serum levels of CGRP were normalised after sumatriptan (11). As DBF responses to capsaicin are almost entirely inhibited by CGRP receptor blockade (12), the inhibition of capsaicin-induced DBF responses by sumatriptan in our experiments indicates that triptans may inhibit CGRP release. The 26% decrease in DBF response after sumatriptan in our study may seem modest, but is similar to the 33% decrease in jugular CGRP in migraineurs using subcutaneous sumatriptan as reported earlier (11).
Hypothetically, the decrease of capsaicin-induced DBF could have been due to physiological antagonism (direct vasoconstrictor effect of sumatriptan), as described above. However, the responses to ES should then have been affected by sumatriptan as well, which was not the case (Figure 2(b)). Thus, we conclude that the inhibition of capsaicin-induced DBF after sumatriptan is due to its effects on neuropeptide release, most likely of CGRP.
In conclusion, trigeminovascular effects of potential antimigraine drugs, such as antagonists or antibodies directed against CGRP or its receptor, can be investigated with our human model.
Article highlights
Sumatriptan inhibits increases in dermal blood flow induced by the release of calcitonin gene-related peptide (CGRP). We have established a research tool to investigate the trigeminovascular effects of (potential) antimigraine drugs, such as CGRP receptor antagonists or antibodies directed against CGRP or its receptor.
Footnotes
Acknowledgements
Sumatriptan was a generous gift from GlaxoSmithKline, Zeist, the Netherlands. We would like to acknowledge E. Jäger, Dr S. Labruijere and Dr S. Lankhorst for excellent assistance.
The Dutch Brain Foundation and the NWO had no role in the design or conduct of the study.
Part of this paper has been presented as an abstract at the European Migraine Trust Meeting (Ibrahimi et al. J Headache Pain 2014; 15 (Suppl 1): G16).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grants from the Dutch Brain Foundation (project number 2012(1)-66) and the Netherlands Organization for Scientific Research (NWO) (VIDI 917.11.349 to AMVDB and VIDI 917.11.319 to GMT).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
