Abstract
Introduction
Anti-calcitonin gene-related peptide antibodies proved effective in the preventive treatment of chronic migraine. In this open label study, we aim to assess the effects of erenumab administration on neurophysiological and biomolecular profiles in a representative cohort of chronic migraine patients.
Methods
Forty patients with a history of chronic migraine for at least 12 months prior to enrollment, and previous failure of at least two different preventive therapies, were enrolled. After a 1-month observation period (T0), patients were treated with erenumab 70 mg s.c. (every 28 days) for a total of three administrations. At week 12, they returned for the end-of-protocol visit (T3). At T0 and T3, patients underwent recording of clinical features, recording of single stimulus (RTh), temporal summation (TST) thresholds of the nociceptive withdrawal reflex, venous blood sampling for miR-382-5p, and miR-34a-5p quantification.
Results
At T3, 31 patients (77.5%) qualified as 30% Responders (reduction in monthly migraine days by at least 30% in the last 4-week observation period). RTh (T0: 15.4 ± 8.1 mA, T3: 19.7 ± 8.2 mA) as well as TST (T0: 11.2 ± 5.8 mA, T3: 13.4 ± 5.0 mA) significantly increased at T3 in 30% Responders (p = 0.001 for both), while we did not observe significant changes in NON-responder patients. MiR-382-5p and miR-34a-5p levels were significantly lower after erenumab administration in the overall study population (p = 0.015, and p = 0.001, respectively), without significant differences between 30% Responder and NON-responder groups.
Conclusions
Different migraine phenotypes, characterized by different treatment susceptibility, may exist as suggested by the divergent behavior between neurophysiological and biomolecular findings in 30% Responder vs. NON-responder patients.
The study protocol was registered at clinicaltrials.gov (NCT04361721).
Introduction
Monoclonal antibodies (mABs) targeting calcitonin gene-related peptide (CGRP) proved effective in the preventive treatment of episodic and chronic migraine (1) as well as in difficult-to-treat patients such as those who had previously failed multiple prevention treatments (2) or those with associated medication overuse (MO) (3).
A characteristic dysfunction in chronic migraine (CM) is sensitization, occurring peripherally in the trigeminovascular system (4) but then spreading to the central nervous system, where it manifests with an increased neuronal excitability in multiple areas (5). Several neurophysiological studies have demonstrated the occurrence of central sensitization in the brain as well as at the spinal level in CM patients (6,7).
MicroRNAs (miRNAs) are involved in the generation and maintenance of chronic pain (8). Current evidence suggests that specific miRNAs may also play a role in migraine, thus representing possible biomarkers of the disease. MiR-34a-5p and miR-382-5p are implicated in the regulation of GABAergic signaling and in IL-10 gene expression, respectively. In a previous study, serum levels of miR-34a-5p and miR-382-5p were significantly elevated during migraine attacks. MiR-382-5p levels were also elevated during the pain-free period when compared to healthy controls (9). In addition, miR-34a-5p was also negatively modulated in the saliva of migraine adolescents after treatment with antimigraine drugs, thus suggesting a role for this MiRNA as a biological marker of treatment efficacy (10). Interestingly, pre-clinical data showed that CGRP stimulated in vitro the release of inflammatory cytokines in lymphocytes as well as IL-6 gene expression in macrophages through modulation of a specific miRNA (11). Altogether, these observations formed the rationale for exploring the possibility that miRNAs may represent a peripheral biological marker of the changes induced by the treatment with CGRP-targeting mABs in CM subjects.
The primary aim of this open label, hypothesis-generating study was the evaluation of the impact of erenumab treatment on neurophysiologically-recorded spinal sensitization in a representative cohort of CM patients who had previously failed at least two preventive treatments.
As the secondary outcome, we evaluated the effects of erenumab treatment on the expression levels of miR-34a-5p and miR-382-5p. Furthermore, we explored the possible role of neurophysiological and biomolecular parameters as potential predictors of treatment response.
Methods
Subjects
We enrolled 40 patients (age 50.8 ± 8.9 years, 25 females, age at migraine onset 14.8 ± 5.9 years) affected by CM, according to the ICHD-3 criteria (12) among those consecutively attending the Headache Centre at the IRCCS Mondino Foundation.
Inclusion criteria were: age 18–65 years; history of CM or CM + MO (according to the ICHD-3 criteria) for at least 12 months prior to enrollment; previous failure of at least two different pharmacological classes of preventive therapies.
Exclusion criteria were: pregnancy or breastfeeding; previous reported adverse reaction to latex; substance abuse; concomitant treatment with more than one preventive therapy; treatment with botulinum toxin in the last 3 months before enrolment.
A previous therapeutic failure was defined as: a) no reduction (<30%) in headache frequency after at least 6 weeks of treatment with an adequate dose, or b) the subject discontinued the treatment due to related adverse events or poor tolerability.
In our cohort, the principal comorbidities were: hypertension (10 subjects, 25.0%), anxiety and/or mild depression (19 subjects, 47.5%), disthyroidism (5 patients, 12.5%), and gastrointestinal complaints (11 subjects, 27.5%).
Clinical and demographic characteristics of our population are summarized in Table 1.
Baseline clinical and demographic data of study population.

CM: chronic migraine; CM+MO: chronic migraine associated to medication overuse; NSAIDs: nonsteroidal anti-inflammatory drugs; BoNT-A: botulinum toxin type A.
Note: p-value: t-test for independent samples for 30% Responder vs. 30% NON-responder. Data are presented as “mean ± standard deviation” or “absolute value (percentage)”.
Study procedures
This is an open-label study with a screening period followed by a 1-month baseline and a 3-month treatment period. The study was approved by the local Ethics Committee (P-20190105434).
All patients underwent a screening visit at the Headache Centre of the IRCCS Mondino Foundation during which they signed a written informed consent after a thorough description of the protocol by the investigator. Patients who fulfilled inclusion/exclusion criteria were enrolled in the study and started a baseline observation period for a month. This observation period aimed to prospectively confirm headache frequency as well as the diagnosis of CM. At the end of the baseline period (T0), patients completed the following procedures: recording of clinical and demographical features, vital signs evaluation, neurophysiological assessment, venous blood sampling for miRNAs quantification, and first injection of erenumab 70 mg s.c. The second injection of erenumab 70 mg was administered in hospital after 28 days (T1), while the third and last dose of erenumab 70 mg was self-administered at home by the patients themselves after an additional 28-day interval (T2). The last visit of the study (T3) was then planned 28 days after the last dose of erenumab. At T3, the patients underwent neurophysiological testing and venous blood sampling for miRNAs quantification. All the enrolled patients completed the study. Study procedures are summarized in Figure 1. The study protocol was registered at clinicaltrials.gov (NCT04361721).
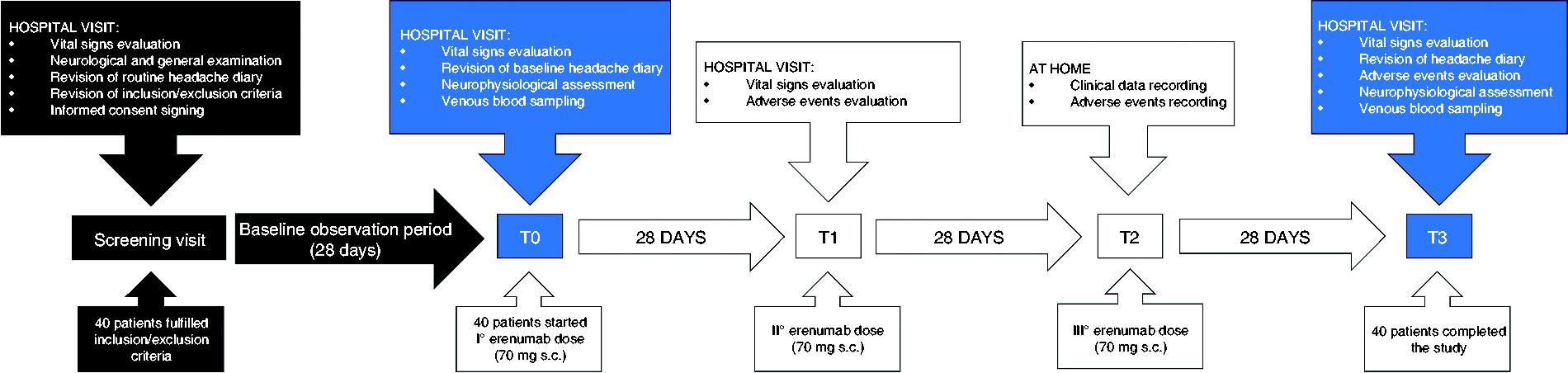
Flow-chart of the study procedures.
Nociceptive withdrawal reflex measurement (NWR)
The reflex was recorded at the lower limb according to a well validated procedure (13,14), by an expert technician (VG). The reflex was recorded between 09:00 AM and 11:00 AM. The subjects were tested in an interictal period, defined as: no headache in the previous 24 hours, or mild headache (≤3 on a 0 to 10 nociceptive rating scale) that does not fulfil ICHD-3 criteria for “migraine without aura” and did not require the intake of acute antimigraine drugs. The possible onset of headache, and the associated symptoms, in the 12 hours after the test were also recorded on the headache diary. During the neurophysiological procedure, patients were lying on their back in a comfortable position with their ankle flexed at 90° and knee flexed at 130°. The sural nerve was stimulated electrically behind the lateral malleolus with a pair of Ag/AgCl surface electrodes. The electrical stimulation was made of five consecutive squared pulses (1 ms, 200 Hz), randomly delivered every 60–120 seconds. The electromyographic sweep (Synergy, Medelec, UK) was recorded from the capitis brevis of the homolateral biceps femoris with a pair of Ag/AgCl surface electrodes. A staircase method was used for all the threshold evaluations, and the intensity was increased by 0.3 mA per step. The recording parameters were: analysis time 300 ms, sensitivity 20 mV, and filter bandpass 3 to 3000 Hz.
We evaluated:
The single stimulus reflex threshold of the NWR (RTh): the lowest intensity (mA) able to induce three consecutive stable muscular responses of at least 20 mV and 10 ms. The temporal summation threshold (TST), by means of a train of five electrical stimuli at a frequency of 2 Hz: the lowest intensity (mA) able to elicit three consecutive stable muscular responses of at least 20 mV and 10 ms in the fourth and fifth electromyographic sweeps. The subjective pain perception of RTh (VAS-RTh), and of the first (VAS-TST-1) and fifth (VAS-TST-5) stimulus of the TST on a 0–10 point visual analogue scale.
MicroRNAs expression
After overnight fasting, samples of venous blood (18 mL) were collected in sterile tubes from the cubital vein, between 8:00 AM and 11:00 AM. Subjects were tested outside a migraine attack, as previously defined.
The microRNAs expression was evaluated by real-time reverse transcription (RT) PCR in peripheral blood mononuclear cell (PBMCs). PBMCs were isolated immediately after blood collection. Briefly, blood samples were collected within ethylenediamine tetra-acetic acid (EDTA) containing tube and diluted in 1:1 ratio with phosphate buffer saline (PBS) (Sigma). Subsequently, diluted blood samples were slowly loaded into Ficoll separating solution (15 ml) (Sigma) and centrifuged at 800 g for 30 min at room temperature. PBMCs accumulated as the middle white monolayer were washed twice in sterile PBS at 300 g for 15 min. After washing, PBMCs pellets were resuspended in trizol and stored at −80° C until use. Total RNA (including all small non-coding RNAs) was extracted from pellets within 2 weeks from storage using the Direct-zol RNA Mini prep plus (Zymo Research) to perform microRNAs analysis. RNA quality was determined by an optical density (OD) 260/280 ratio ≥1.9 and OD 260/230 ratio ≥1.5 by using a NanoDrop Spectrophotometer (Nanodrop™ Thermo Fisher Scientific, Euroclone Milano). Synthesis of cDNA was performed by using MirXMirna First strand Synthesis (Takara-Diatech, Jesi-An Italy) and TB Green q-Rt PCR (Takara-Diatech, Jesi-An Italy) was used to determine expression levels of miR-34a-5p and miR-382-5p. The thermocycling conditions were: 95°C for 10 min, and 40 cycles of 15 s at 95°C, followed by 1 min at 60°C by a Light Cycler 480 Instrument RT-PCR Detection System (Roche, Milan, Italy). MicroRNAs expression was normalized with U6 (a type of small nuclear RNA), used as housekeeping gene. MicroRNAs levels were calculated according to 2−ΔΔCt = 2 − (ΔCt gene − ΔCt housekeeping gene) formula by using Ct (Cycle threshold) values (15).
Statistical analysis
Regarding sample size calculation, as primary outcome we considered the TST modification at T3 when compared to T0 in the overall study population. Based on previous assessments, we considered meaningful a 2.0 ± 4.0 mA between groups difference (7,14). The calculation for a dependent-samples test (confidence interval (two-sided): 95.0%; power: 80%; expected correlation (ρ): 0.5) suggested a minimum sample size of 32 patients.
The Statistical Package for the Social Sciences (SPSS), version 21.0 (Windows), was used for all the computations. A normal distribution of our data was confirmed at baseline as well as at T3 by the evaluation of “skewness” and “excess kurtosis”, and by calculation of z-tests for both.
Continuous variables are presented as mean ± standard deviation, while categorical data will be presented as number (percentage). MiRNAs levels are expressed as mean ± standard error of the mean, to allow comparison with previous published data (9).
Responder patients were defined by a reduction in migraine days by at least 30% in the last 4-week observation period (30% Responder), according to the IHS Guidelines (16). We also performed a sub-group analysis where we considered patients with a reduction in migraine days by at least 50% in the last 4-week observation period (50% Responders).
Patients that did not reach the 30% reduction in monthly migraine days for the primary analysis, or the 50% reduction in monthly migraine days for the sub-group analysis, were defined as 30% NON-responder or 50% NON-responder, respectively.
The analysis was performed with an ANOVA test for repeated measures with factor TIME (two levels: T0, T3), and, for neurophysiological and biomolecular data, factor GROUP (two levels: 30% Responder, 30% NON-responder). When the TIMExGROUP interaction yielded a statistically significant output, a post-hoc analysis was performed and corrected for multiple comparisons according to a Bonferroni’s correction. Post-hoc analysis was performed with a one-way ANOVA for repeated measures to evaluate the effect of factor TIME separately in 30% Responder and 30% NON-responder groups.
To exclude a significant influence of clinical and demographic features on neurophysiological and biomolecular evaluations, we performed separate analyses with an ANOVA for repeated measures with factor TIME, and factors: gender (male vs. female), medication-overuse (presence vs. absence), aura (presence vs. absence), and mild anxiety and/or mild depression (presence vs. absence).
Between-groups association of variables was tested with a t-test for independent samples, while statistical association among categorical variables was tested with Pearson χ2 test or Fisher’s exact test. In the sub-group analysis (50% Responder vs. 50% NON-responder), a further Bonferroni’s correction was applied to control for multiple testing.
The level of significance was always set at α = 0.05, always corrected for multiple comparisons if appropriate.
Results
Clinical effectiveness of erenumab
Erenumab produced a significant reduction in the number of migraine days per month over time (T0: 21.6 ± 6.1, and T3: 9.6 ± 8.6; p = 0.001). At T3, the percentage of 30% Responders was 77.5% (31 out of 40 patients), with an average percent reduction of migraine days per month of 56.6 ± 33.0.
At T0, 30% Responders did not differ from 30% NON-responders as regards clinical or demographic variables (Table 1).
In line with these results, the headache days per month, and the days of acute drug intake per month were significantly reduced by erenumab administration (p = 0.001 for both).
During the 12-week follow-up, a total of 17 patients (42.5%) complained of at least one adverse event. The reported adverse events were: constipation (25.0%), rash and pruritus (5.6%), menstrual irregularities (11.2%), and asthenia (11.2%).
Neurophysiological evaluation at baseline, and modifications after erenumab administration
The baseline parameters of the nociceptive withdrawal reflex are presented in Table 2.
Baseline neurophysiological and biochemical data of study population.

RTh: single stimulus reflex threshold; VAS-RTh: subjective pain perception at RTh; TST: temporal summation threshold; VAS-TST-1: subjective pain perception of the first stimulus of TST; VAS-TST-5: subjective pain perception of the fifth stimulus of TST; RQ: relative quantification, RQ = 2−ΔΔCt = 2 − (ΔCt gene − ΔCt housekeeping gene); Ct: cycle threshold as defined in methods section; p-value: t-test for independent samples for 30% Responder vs. 30% NON-responder.
Note: Neurophysiological parameters are presented as “mean ± standard deviation”. MicroRNAs expressions are presented as “mean ± standard error of the mean”.
At the end of the 12-week treatment period with erenumab, a significant interaction TIMExGROUP was described for both RTh and TST (p = 0.001 and p = 0.005, respectively).
At the post-hoc analysis, 30% Responders showed a significant increase in RTh (T0: 15.4 ± 8.1, T3: 19.7 ± 8.2; p = 0.001) as well as in TST (T0: 11.2 ± 5.8, T3: 13.4 ± 5.0; p = 0.001) at T3 (Figure 2). At variance, we did not observe any significant change in RTh and TST in the 30% NON-responders (p = 0.277, and p = 0.449 respectively).

Changes in neurophysiological parameters after erenumab administration. Study population: n = 40; 30% Responders: n = 31; 30% NON-responders: n = 9.
We did not find significant effects of factor TIME and GROUP as well as their interaction for the subjective perception of pain, as measured by VAS-RTh, VAS-TST-1, and VAS-TST-5.
The neurophysiological changes were not influenced by gender, concomitant diagnosis of medication-overuse or migraine with aura, presence of mild anxiety and/or mild depression.
Seventeen patients (42.5%) at T0 and 12 patients (30%) at T3 developed a migraine attack the day after the procedures. This distribution was neither statistically significant between T0 and T3 (p = 0.302), nor between 30% Responder and 30% NON-responder groups (p = 0.664).
Biomolecular analysis of study population at baseline, and modifications after erenumab administration
The baseline levels of miR-382-5p and miR-34a-5p are presented in Table 2.
We did not find significant differences between 30% Responder and 30% NON-responder groups as regards baseline levels of miR-382-5p (10.0 ± 2.0, and 10.2 ± 2.7 respectively, p = 0.973), or miR-34a-5p (18.7 ± 3.5, and 15.7 ± 4.3 respectively, p = 0.661).
After erenumab treatment, the level of both miR-382-5p and miR-34a-5p were significantly lower in the overall study population (p = 0.015 for miR-382-5p, and p = 0.001 for miR-34a-5p). After erenumab treatment, we did not find significant differences between 30% Responder and 30% NON-responder groups, as demonstrated by the non-significant effects of factor GROUP or interaction TIMExGROUP for both miR-382-5p (p = 0.688, and p = 0.596, respectively) and miR-34a-5p (p = 0–820, and p = 0–366, respectively) (Figure 3).

Changes in miRNAs expression after erenumab administration. Study population: n = 40; 30% Responders: n = 31; 30% NON-responders: n = 9.
The biomolecular changes were not influenced by gender, concomitant diagnosis of medication-overuse or migraine with aura, presence of mild anxiety and/or mild depression.
Subgroup analysis for 50% Responder patients
In our population, 27 (67.5%) subjects qualified as 50% Responders. No additional information could be derived from this analysis as regards clinical, demographic and biochemical data. At variance, we noted an interesting pattern as regards neurophysiological parameters. Similarly to the 30% Responder group, at T3 the 50% Responders experienced a significant increase in both RTh (T0: 14.3 ± 6.9, T3: 18.6 ± 7.1; p = 0.001) and TST (T0: 10.3 ± 4.7, T3: 12.6 ± 3.9; p = 0.001), while these thresholds were not significantly modified in 50% NON-responder group. In addition, 50% Responders presented a significantly lower RTh at baseline (14.3 ± 6.9 mA) when compared to 50% NON-responders (20.3 ± 8.4 mA) (p = 0.028) (Figure 4). TST was comparable between these groups at baseline (p = 0.066).

Neurophysiological parameters at baseline in the 50% Responder group.
Discussion
From a clinical point of view, our data are in line with the results published by Tepper et al. in 2017 (17). In particular, we confirm a significant reduction of migraine days (56.6 ± 33.0% reduction from baseline, p = 0.001), of headache days and of use of symptomatic drugs during the last 4 weeks of our 12-week treatment period. The percentage of patients who were 30% Responders at T3 was 77.5%, slightly higher than the percentage reported by Tepper et al., a difference that is likely ascribable to our open-label design (17).
Regarding the neurophysiological results, we confirmed our hypothesis of an improvement in central spinal sensitization after three monthly administrations of erenumab in the 30% Responder group, witnessed by a significant increase in the RTh and in the TST at T3 in this group. No change was instead observed in the neurophysiological parameters of 30% NON-responders. This suggests that the observed improvement in spinal sensitization is likely related to the clinical outcome; that is, the reduction of migraine recurrence, rather than to a direct effect of erenumab. In this frame, it is worth noting that nociceptive withdrawal reflex is mainly mediated by WDR neurons at spinal level, and this is particularly true for the temporal summation threshold, a direct measure of the wind-up phenomena (18). The elegant study conducted by Melo-Carillo et al. in an animal model of migraine pain showed that an anti-CGRP mAb inhibited the activation and the central sensitization of the trigeminovascular system after experimentally induced cortical spreading depression. This effect is primarily mediated by high-threshold (HT) neurons, without a significant contribution by the wide-dynamic range (WDR) neurons (19). We believe that the evaluation of the NWR represents a strength of the present study, as it is a sensitive and validated tool to objectively and quantitatively record central sensitization at the spinal level. Moreover, the relation between the NWR and migraine has been widely studied in the past, providing solid foundations on which to build our hypothesis. In an experimental migraine model, the NWR showed that subjects with a positive provocation test developed signs of central sensitization, proving sensitive enough to differentiate between subjects with low-frequency and high-frequency migraine sub-types (14,20). Moreover, NWR recording allowed to demonstrate an altered supra-spinal pain control in CM, namely a defective diffuse noxious inhibitory control system (21). Finally, it was demonstrated that in patients with CM associated to MO headache, central sensitization improved after the successful application of a detoxification protocol, along with an acute modulation of the endocannabinoid system metabolism (7).
The finding regarding the reduced RTh at baseline in the 50% Responders has to be interpreted with caution, due to its preliminary nature, and because it was demonstrated only in a secondary analysis, but it is nonetheless very interesting because it prompts the possibility to use neurophysiological parameters for the prediction of drug response. For the above reasons, it is not possible at this time to provide further analysis (sensitivity/specificity analysis, or ROC curve), but we plan to proceed with the enrollment to further expand our knowledge on the relevance of this topic.
At variance from the neurophysiological parameters, the expression of mir-382-5p and mir-34a-5p was significantly reduced after erenumab treatment, regardless of the clinical outcome. This finding suggests a direct modulation of erenumab treatment on the pathways that are implicated in the expression of miR-34a-5p and miR-382-5p. MiRNAs are able to regulate gene expression and also to mediate epigenetic modifications in neuropathic pain and migraine (8,10,22). CGRP can promote the release of pro-inflammatory cytokines, such as TNF-α, IL-1β, IL-6, and anti-inflammatory cytokines, such as IL-10, thus contributing to the modulation of pain threshold and to trigeminal nerve fibers sensitization (23,24). MiR-382-5p is implicated in IL-10 gene expression and therefore its reduction is in keeping with erenumab-mediated inhibition of CGRP pro-inflammatory activity (9). mir-34a-5p negatively modulates the GABAergic signaling and its peripheral expression decreases in serum and saliva of young migraine patients under drug treatment (10). Thus, the reduced expression of mir-34a-5p during erenumab treatment may reflect a more active GABAergic transmission from the parabrachial nucleus to amygdala, a pathway that plays a role in chronic pain modulation (25).
Andersen et al. (2016) demonstrated that, among more than 30 different microRNAs, miR-382-5p and miR-34a-5p were significantly elevated in the serum of episodic migraine patients when compared to healthy controls (9). Moreover, the two miRNAs exhibit different behaviors: miR-34a-5p was elevated only during migraine attacks, while miR-382-5p was found to be increased also during the migraine-free period. A direct comparison to our results is not warranted, because here we enrolled a cohort of CM patients, we lack a healthy control group, and miRNAs expression was tested in different specimens (serum vs. PBMCs). Even with the discussed limitations, the baseline increases of miR-382-5p and miR-34a-5p expression (10-fold increase, and 18-fold increase, respectively) appear to be more marked when compared to the results by Andersen et al. Moreover, the levels of miR-382-5p and miR-34a-5p after erenumab treatment (4-fold increase, and 3.5-fold increase, respectively) are descriptively comparable to the results of the Danish group. Taken together, these observations may suggest that these miRNAs are related to migraine severity and disease activity, and may thus provide a rationale for future studies.
The divergent behavior between neurophysiological and biomolecular findings may suggest that different migraine phenotypes, with different treatment susceptibility, may exist among the chronic migraine spectrum. In particular, we described a subset of patients in which, although erenumab administration exerts a biological effect as demonstrated by a significant reduction of miRNAs levels, this was neither coupled with an improvement of clinical outcomes nor with changes in spinal sensitization.
The basic idea that different migraine phenotypes could exhibit different clinical responses to the anti-CGRP mAbs is not new. Indeed, Christensen et al. in 2018 demonstrated that patients with good clinical response to erenumab had a high susceptibility to CGRP administration (CGRP phenotype), namely an elevated migraine induction rate, and a more severe and long-lasting induced headache (26).
Some limitations must be acknowledged in the interpretation of our results. Firstly, the study design lacks a placebo group for a blind comparison or a healthy control group. This choice was made not only to improve the study feasibility, but also because, due to the lack of preliminary data, we first needed to probe the actual existence of a biological effect of erenumab administration on either neurophysiological or biochemical parameters. Another limitation is represented by the fact that the 30% NON-responder group was slightly smaller than expected due to the unexpectedly higher clinical efficacy of erenumab treatment. It is therefore possible that we missed potentially significant differences between groups due to an insufficient power of the study. Finally, we aimed to evaluate our patients during an interictal phase and tested them after a 24-hour pain free-interval; however, we could not control for the occurrence of a headache after the testing procedures and obviously the chance to develop such a headache was theoretically higher for NON-responders. Indeed 42.5% of subjects developed a migraine attack the day after the procedures at T0 and 30% at T3; these percentages however did not significantly differ between Responder and NON-responder groups.
Our study advances our knowledge on the possible biological effects of the new anti-CGRP mAbs therapies and may represent a stepping stone for designing larger studies powered to confirm our data in adequate samples and to evaluate the possible role of neurophysiological parameters as predictors of treatment response.
Key findings
Erenumab administration in chronic migraine patients improved central spinal sensitization only in patients with a reduction in monthly migraine days of at least 30%. Independently from clinical outcome, miRNAs levels were reduced in chronic migraine patients after erenumab administration. Responder patients to erenumab treatment were characterized by divergent neurophysiological and biomolecular phenotypes.
Footnotes
Acknowledgements
The authors thank the Research Nurse Team of the Headache Science Centre of the IRCCS Mondino Foundation for their precious assistance in all of the activities.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CT received honoraria for the participation to advisory boards or for oral presentations from Allergan, ElectroCore, Eli-Lilly, Novartis, and Teva. CT has no ownership interest and does not own stocks of any pharmaceutical company. CT serves as Chief Section Editor of Frontiers in Neurology – Section, Headache Medicine and Facial Pain, and on the editorial board of The Journal of Headache and Pain. GS received honoraria for the participation to advisory boards or for oral presentations from Eli-Lilly and Novartis. The remaining authors have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Research Grant from the Italian Ministry of Health to IRCCS Mondino Foundation (Ricerca Corrente 2017-2019).
