Abstract
Introduction
Migraine is a common and disabling neurological disorder that requires effective acute treatment. However, access to optimal therapies varies significantly across healthcare systems worldwide. This systematic review assesses acute pharmacological treatments for migraine from a Mexican healthcare perspective, focusing on drug availability and proposing a practical treatment algorithm.
Methods
Following PRISMA guidelines, we systematically searched multiple databases from inception to November 2024. We included randomized controlled trials evaluating acute treatments for migraine in adults. Treatment efficacy data were synthesized narratively in accordance with the International Headache Society guidelines. Drug availability in Mexico was assessed through regulatory databases and clinical practice surveys.
Results
Thirty-six randomized controlled trials met inclusion criteria, evaluating triptans, nonsteroidal anti-inflammatory drugs (NSAIDs), calcitonin gene-related peptide antagonists, ditans, and combination therapies. Subcutaneous sumatriptan demonstrated high efficacy (83% pain relief at 2 hours), but it is limited in availability within the Mexican public healthcare system. Oral triptans (sumatriptan, zolmitriptan, eletriptan, and rizatriptan) are available; however, access varies between the public and private sectors. NSAIDs like ketorolac and ketoprofen show good efficacy and are widely accessible. Newer agents (rimegepant, lasmiditan) are available but expensive, limiting healthcare access.
Conclusion
Effective migraine treatment in Mexico necessitates a balance between evidence-based medicine and healthcare realities. While triptans remain first-line per international guidelines, NSAIDs offer practical alternatives given their accessibility. A structured approach considering drug availability, cost, and patient characteristics can optimize migraine care in Mexico and similar healthcare systems.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine is a prevalent and disabling neurological disorder that significantly impacts daily functioning, productivity, and quality of life. Characterized by recurrent headache attacks lasting 4 to 72 hours, migraine affects approximately 1 billion people worldwide, with higher prevalence among women. 1 In Mexico, population-based studies indicate a lifetime prevalence of 14.6% in women and 4.1% in men, with significant underdiagnosis noted across healthcare settings. 2
The healthcare utilization impact of migraine in Mexico is substantial and measurable. Emergency Department (ED) visits attributed to migraine represented 19% of all headache-related presentations from 2013 to 2022, with nearly 180,000 visits documented by the Mexican Ministry of Health. This burden disproportionately affects women, who comprise 78% of migraine-related emergency visits, reflecting the higher prevalence and severity of migraine in female patients. 3 The frequency of ED utilization suggests inadequate access to appropriate acute treatments in ambulatory settings, highlighting gaps in current treatment approaches.
The economic impact extends beyond direct healthcare costs to include productivity losses and reduced quality of life. While comprehensive economic burden studies specific to Mexico remain limited, the substantial ED utilization and high prevalence rates indicate significant financial consequences for both patients and the healthcare system. Lost productivity due to migraine-related disability affects not only individual patients but also broader economic productivity, particularly given the condition's peak prevalence during the prime working years.2,3
The International Headache Society (IHS) has recently updated its evidence-based guidelines for migraine pharmacological treatment, providing strong recommendations for acute management based on high-quality evidence using Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) methodology.4,5 These guidelines establish clear hierarchies of treatment.
Effectiveness is supported by subcutaneous sumatriptan, oral triptans, and specific Nonsteroidal anti-inflammatory drugs (NSAIDs), which receive strong recommendations based on evidence from randomized controlled trials.
However, implementing these guidelines requires careful consideration of local healthcare contexts, including variations in drug availability, cost constraints, healthcare infrastructure, and socioeconomic factors that influence access to treatment.
The Mexican healthcare system operates through three parallel components: employment-based social insurance schemes, public assistance services for the uninsured with financial protection, and the private sector, which includes providers, insurers, and manufacturers. 6 The system is divided into public and private sectors, with major public institutions including the Mexican Social Security Institute (IMSS), the Institute of Security and Social Services for State Workers, and Popular Health Insurance 6 and after IMSS-Bienestar, the largest social insurance institution, operates under a corporatist governance structure that reflects the political realities of the 1940s rather than modern needs. 6 The system faces significant challenges with segmentation, as nearly 14% of the population lacks financial protection, while others have multiple coverage types.6,7 Regarding financing efficiency, evidence suggests that out-of-pocket funding incentivizes more efficient hospital behavior, while public funding is more effective for complex, high-technology facilities. 8
Each sector maintains different formularies, access policies, and cost structures, creating complex variations in treatment availability that must be considered when developing practical treatment approaches.
This systematic review aims to provide a Mexican perspective on acute migraine treatments, synthesizing evidence from randomized controlled trials while emphasizing the practical implementation of these treatments within the Mexican healthcare system. Our research question addresses both clinical efficacy and healthcare delivery: What evidence exists for the use of acute pharmacological treatments for migraine, and how can this evidence be practically implemented within the constraints of the Mexican healthcare system? By integrating international evidence with local availability data, we propose a practical treatment algorithm that balances clinical efficacy with healthcare accessibility, addressing the gap between optimal evidence-based care and achievable clinical practice in the Mexican context.
Methods
Literature search and study selection
We conducted a comprehensive systematic review in accordance with the PRISMA guidelines. 9 This systematic review was not prospectively registered in PROSPERO, which represents a limitation of our study design that we acknowledge affects methodological transparency. Electronic databases, including MEDLINE, OVID, BVS, and Wiley Online Library, were searched from inception through November 2024 using a comprehensive search strategy developed by a medical reference librarian and peer-reviewed by a second librarian.
The search strategy employed detailed terms and Boolean operators to identify relevant trials: (“migraine” OR “headache”) AND (“acute treatment” OR “abortive treatment”) AND (“ketorolac” OR “sumatriptan” OR “ergotamine” OR “almotriptan” OR “prochlorperazine” OR “rizatriptan” OR “lasmiditan” OR “ditans” OR “NSAIDs” OR “ketoprofen” OR “ibuprofen” OR “ubrogepant” OR “CGRP antagonist” OR “zavegepant” OR “rimegepant” OR “lidocaine” OR “tramadol” OR “acetaminophen” OR “metoclopramide” OR “caffeine”) AND (“randomized controlled trial” OR “clinical trial” OR “placebo-controlled”). The search was validated by two co-authors and managed using Rayyan software to facilitate collaboration among reviewers.
We also searched clinical trial registries, government databases, conference proceedings, patient advocacy group websites, existing systematic reviews, and the websites of medical societies. These additional sources did not yield eligible studies beyond those identified through primary database searches.
The inclusion criteria comprised adults aged 18 years or older with a diagnosed migraine, as defined according to the International Classification of Headache Disorders (ICHD-3), 10 regardless of attack frequency, as acute treatment principles apply to all forms of migraine. We included randomized controlled trials of acute pharmacological therapy, specifically phase II or III studies comparing interventions to placebo or active comparators, with reported short-term efficacy outcomes, including pain relief or pain freedom at 2 hours. Only English or Spanish publications were included to ensure accurate interpretation of methodology and results.
Exclusion criteria eliminated nonrandomized studies, phase I trials, invasive treatments, studies focusing on nonmigraine headache disorders, and duplicate publications. Each study's migraine definition was accepted if it was compatible with the ICHD-3 criteria for migraine without aura or migraine with aura, recognizing that diagnostic criteria evolved during the timeframe of the included studies.
Drug availability assessment
Drug availability in Mexico was systematically assessed through multiple sources, including the Federal Commission for Protection Against Health Risks (COFEPRIS) regulatory databases, formularies from major public healthcare institutions (IMSS and ISSSTE – Instituto de Seguridad y Servicios Sociales de los Trabajadores del Estado (Institute for Social Security and Services for State Workers), and clinical practice surveys conducted among Mexican headache specialists. We defined availability categories as follows: wide availability denoted medications included in public healthcare formularies with broad geographic access and over-the-counter availability; partial availability indicated inclusion in some but not all public formularies, or medications available primarily through private pharmacies with geographic restrictions; limited availability described medications registered with COFEPRIS but not included in major public healthcare formularies, available primarily through private pharmacies, or requiring special authorization for coverage.
Risk of bias assessment
The risk of bias was assessed using version 2 of the Cochrane Risk of Bias tool for randomized trials (RoB 2), which evaluates five domains: randomization process, deviations from intended interventions, missing outcome data, outcome measurement, and selection of reported results. 11 Two independent reviewers (KV-J and IR-L) performed assessments, with a third reviewer resolving discrepancies. We acknowledge that studies published before 2010 employed less rigorous methodologies by current standards, and this methodological evolution was considered in our overall quality assessment, resulting in more conservative bias ratings for older studies (Figures 1 and 2).
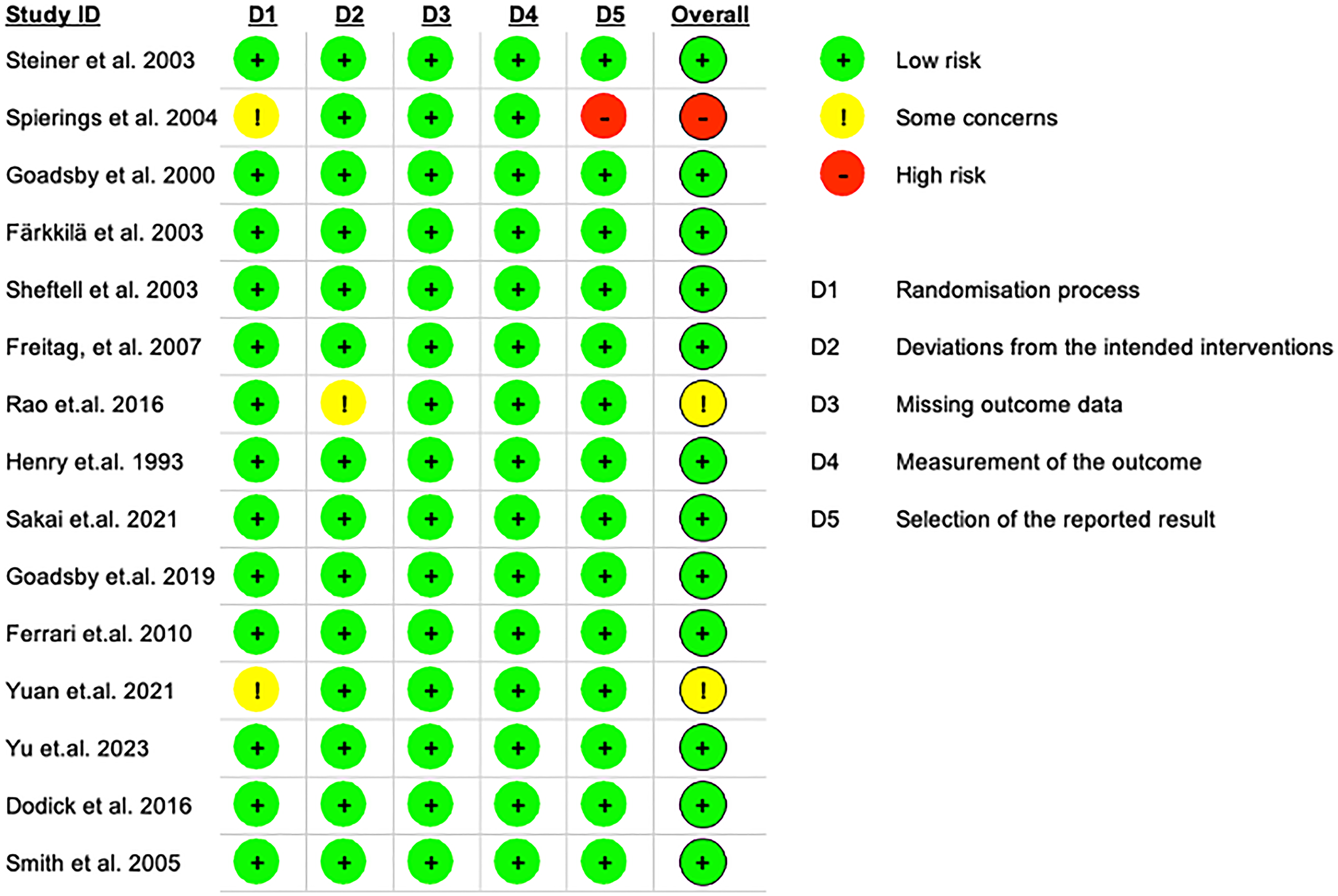
Risk of bias summary for the included randomized clinical trials (intention-to-treat analysis). This figure displays each study's risk of bias judgments (low, some concerns, or high) across the five domains of the RoB 2 tool, along with an overall judgment, focusing on the primary intention to treat analysis analysis population.

Risk of bias summary for the included randomized clinical trials (per-protocol analysis). Like Figure 2, but for per-protocol analyses in the subset of trials where applicable. Differences between intention to treat analysis and per-protocol assessments are highlighted where relevant.
Data synthesis
Given the heterogeneity in study populations, interventions, outcome measures, and our primary focus on treatment availability rather than comparative efficacy, we performed a narrative synthesis rather than a quantitative meta-analysis. This approach enabled a comprehensive evaluation of treatment classes, incorporating healthcare system considerations essential for practical implementation guidance.
Results
Study characteristics
Our systematic search yielded 597 records after duplicate removal. Following title, abstract, and full-text screening, 36 randomized controlled trials published between 1990 and 2024 met the inclusion criteria (Figure 3). These studies evaluated acute treatments across four main therapeutic classes: triptans (n = 18 studies), NSAIDs (n = 6), CGRP antagonists (n = 7), and ditans (n = 3). Additionally, eight studies examined combination therapies (Table 1).
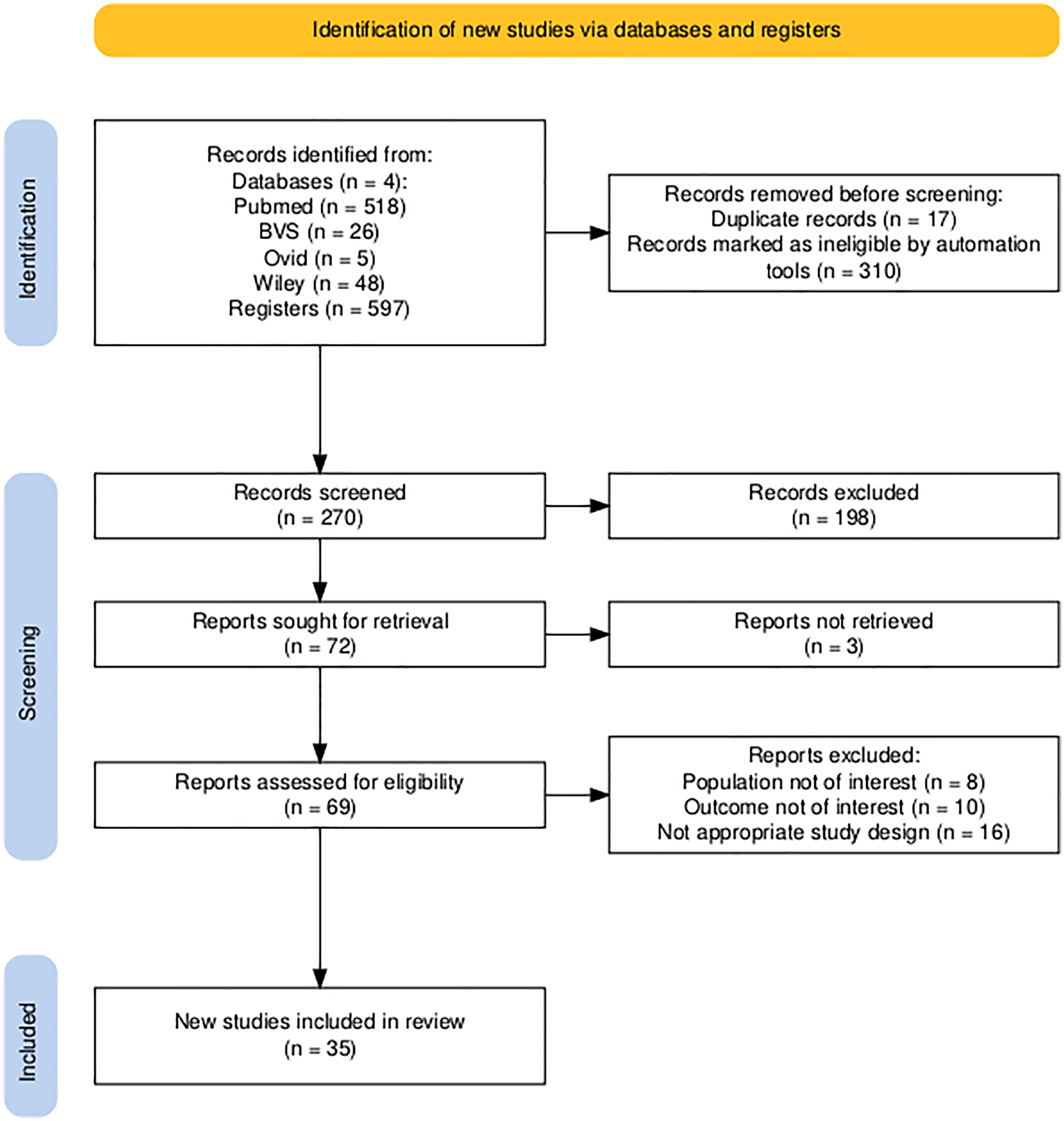
PRISMA flow diagram of study selection for the systematic review. This flowchart illustrates the number of records identified, screened, eligible, and included in the review, as well as the reasons for exclusion at each stage.
Characteristics of included randomized controlled trials evaluating acute migraine treatments.

RCT: randomized clinical trials; OR: odds ratio; ODT: orodispersable tablets; TEAE: treatment emergent event adverse; OTC: over‐the‐counter (analgesics); UTI: urinary tract infection.
The included studies enrolled a total of 27,537 participants, with individual study sizes ranging from 44 to 3005 patients. Most trials were conducted in outpatient settings, with smaller numbers involving evaluations in the ED or at home. The included studies predominantly enrolled patients with migraine without aura (approximately 70%), with the remainder having migraine with aura or both types. Mean age ranged from 35 to 45 years across studies, with 75% to 80% female participants reflecting the higher prevalence of migraine in women. Most studies excluded patients over 65 years, pregnant women, and those with cardiovascular comorbidities, which may limit generalizability to these populations. The risk of bias assessment revealed a generally low to moderate risk across studies (Figures 1 and 2), with older trials showing a higher risk due to the less rigorous methodological standards prevalent at the time of their conduct (Table 1).
Treatment efficacy by class
Our findings demonstrate strong concordance with recent IHS evidence-based guidelines, which provide robust recommendations based on high-quality evidence using GRADE methodology. 5 The efficacy data support clear treatment hierarchies while revealing significant implementation challenges within the Mexican healthcare context.
Triptans
Subcutaneous sumatriptan demonstrated the highest efficacy among all tested treatments, providing pain relief to up to 83% of patients within 2 hours, with about 50% experiencing complete pain freedom.12–26 This formulation is the most effective acute migraine treatment available, consistently outperforming other interventions across multiple trials. However, this highly effective formulation faces significant availability restrictions in Mexico's public healthcare system, creating a substantial treatment access gap that affects the majority of the population relying on public healthcare services, with varying availability in the private sector.
Among oral triptans available in Mexico, eletriptan showed robust dose-dependent efficacy. The 40 mg dose achieved pain relief at 2 hours in approximately 64% of patients, compared to 22% with the placebo, with pain freedom rates of 32% versus 6%, respectively. The 80 mg dose demonstrated even higher efficacy, with 74% achieving pain relief and 44% experiencing pain freedom, compared to placebo rates of 22% and 6%, respectively. Sustained relief at 24 hours was maintained in 44% to 47% of patients across both doses compared to only 11% with placebo.15–17
Zolmitriptan demonstrated consistent efficacy across the 5 to 10 mg dose range, with 61% to 75% of patients achieving pain relief at 2 hours, compared to 19% for the placebo. Pain freedom rates ranged from 43% to 47%, compared to approximately 1% with the placebo, and sustained relief at 24 hours was achieved in 71% to 79% of patients.10–12 Oral sumatriptan 100 mg yielded pain relief rates of 50% to 60%, while rizatriptan 10 mg provided pain relief in 69% to 70% of patients at 2 hours, with 30% achieving complete pain freedom.18–21
Oral formulations of sumatriptan, zolmitriptan, eletriptan, and rizatriptan are available through both public and private healthcare sectors in Mexico. However, cost considerations and formulary restrictions may limit access within public systems, creating disparities in treatment availability based on socioeconomic status and healthcare coverage type.
Ditans
Lasmiditan, representing the novel ditan class of 5-hidroxitriptamine 1F receptor or serotonin-1F receptor receptor agonists, demonstrated consistent efficacy across multiple dose levels.27–31 The 50 to 200 mg doses achieved pain relief at 2 hours in 59% to 61% of patients, with 30% to 40% experiencing pain freedom at higher doses. These findings support the IHS's strong recommendations for all lasmiditan doses based on high-quality evidence from well-designed randomized controlled trials.
While lasmiditan is available in Mexico, its high cost substantially limits accessibility in public healthcare settings, creating similar access disparities to those observed with CGRP antagonists (Table 2). The medication's novel mechanism of action offers particular advantages for patients with cardiovascular contraindications to triptans, but economic barriers prevent widespread utilization of this therapeutic option.
Clinical decision support: CGRP antagonists (gepants) versus ditans for acute migraine.

NSAID: nonsteroidal anti-inflammatory drugs; 5-HT1F: hidroxitriptamine 1F receptor or serotonin-1F receptor.
Nonsteroidal anti-inflammatory drugs
NSAIDs have shown strong efficacy, making them the most accessible treatment option throughout Mexico's diverse healthcare system.32–34 Intranasal ketorolac 30 mg achieved 72.5% pain relief at 2 hours, showing similar effectiveness to intranasal sumatriptan in direct comparisons. 22 Oral ketoprofen demonstrated dose-dependent results, with doses of 75 to 150 mg providing pain relief in 63% to 67% of patients, compared to about 30% with placebo. Intravenous ibuprofen 800 mg reached 74% pain relief; however, this form is mainly used in the emergency department due to its administration requirements.
The widespread availability of NSAIDs across both public and private healthcare sectors, combined with their inclusion in public healthcare formularies and relatively low cost, makes them particularly valuable for Mexican patients. Oral ketorolac, ketoprofen, and ibuprofen are readily accessible across all healthcare settings, providing essential treatment options when triptans are unavailable, contraindicated, or financially inaccessible.
CGRP antagonists
The CGRP antagonist class showed promising efficacy based on high-quality evidence from recent randomized controlled trials.35–39 Rimegepant 75 mg achieved headache relief at 2 hours in 58.1% of patients, compared to 37% with placebo. Additionally, 42.6% of patients maintained sustained relief at 48 hours, compared to 25% with placebo. Pain freedom at 2 hours occurred in approximately 20% of patients, compared to 12% with placebo, demonstrating clinically meaningful benefits beyond placebo effects.
Intranasal zavegepant provided pain relief at 2 hours in 61.2% of patients compared to 41% with placebo, with 44.5% experiencing sustained relief at 48 hours. Pain freedom rates ranged from 15% to 20% versus 5% to 10% with the placebo. 34 Ubrogepant achieved pain relief at 2 hours in 62.57% of patients compared to 48.2% with placebo, with 36.7% maintaining sustained relief at 24 hours.38–40
Despite demonstrated efficacy supported by robust clinical trial evidence, access to CGRP antagonists in Mexico remains severely limited. Only rimegepant has become available in the Mexican market, but its high cost restricts accessibility primarily to private healthcare settings and patients with substantial financial resources (Table 2). Zavegepant and ubrogepant are not yet available, further limiting treatment options for patients who might benefit from this therapeutic class.
Combination treatments
Several combination approaches demonstrated superior efficacy compared to monotherapy, though implementation faces practical constraints.41–46 Sumatriptan 100 mg, combined with promethazine 25 mg, achieved pain relief at 2 hours in 62.2% of patients, compared to 37.2% with sumatriptan alone, with 85% maintaining sustained relief at 48 hours. 40 The fixed-dose combination of sumatriptan 85 mg with naproxen sodium 500 mg showed 65% pain relief, compared to 28% with placebo. Additionally, 48% of patients achieved sustained relief at 24 hours, compared to 18% with placebo.43–45
While combination therapies offer enhanced efficacy, their implementation in Mexico faces challenges, including higher costs, limited availability of fixed-dose combinations, and the need for patients or providers to coordinate multiple medications when fixed combinations are unavailable.
Alignment with international guidelines
Our findings demonstrate strong concordance with recent IHS evidence-based recommendations developed using GRADE methodology and quantitative meta-analysis. 4 Among treatments with strong IHS recommendations that are available in Mexico, oral triptans, including sumatriptan, zolmitriptan, eletriptan, and rizatriptan, demonstrate consistent efficacy supported by high-quality evidence. NSAIDs such as ibuprofen, diclofenac, and acetylsalicylic acid are widely accessible and show proven effectiveness. Lasmiditan and rimegepant, despite strong evidence supporting their use, face significant cost barriers that limit their practical implementation.
Treatments with strong IHS recommendations that have limited availability in Mexico include subcutaneous and intranasal sumatriptan formulations. In contrast, ubrogepant and zavegepant are not yet available in the Mexican market. This alignment validates our systematic approach while highlighting critical accessibility gaps that require attention in healthcare policy and potential interventions to improve treatment access across socioeconomic strata.
Mexican clinical treatment algorithm
Based on our systematic analysis of efficacy evidence and Mexican healthcare realities, we developed a practical treatment algorithm that integrates IHS recommendations with local healthcare constraints. The algorithm provides structured decision-making guidance that considers attack severity, drug availability, cost constraints, patient characteristics, and healthcare setting, enabling providers to optimize treatment selection within available resources while maintaining alignment with evidence-based principles (Figure 4).

This algorithm illustrates how to treat acute headaches at home (or at work), in the doctor's office, and in the ED. While we recommend different treatment options, resource availability and individual assessment will determine the most suitable and effective therapeutic approach.
Initial assessment begins with migraine diagnosis confirmation using ICHD-3 criteria, followed by attack severity evaluation. Attack severity assessment employs standardized criteria where mild attacks involve pain intensity 1 to 3 on a numerical rating scale (0–10) with minimal functional impairment, moderate attacks encompass pain intensity 4 to 6 with some functional impairment but the ability to continue activities with difficulty, and severe attacks involve pain intensity 7 to 10 with significant functional impairment requiring cessation of everyday activities.
For mild-to-moderate attacks, oral NSAIDs represent the first-line approach, with ketorolac 30 mg, ketoprofen 100 mg, or ibuprofen 400 to 600 mg providing effective treatment with wide accessibility. Combination analgesics may serve as alternatives when affordable and available, though care must be taken to avoid medications containing butalbital or excessive caffeine that may promote medication overuse patterns.
Moderate-to-severe attacks are optimally treated with oral triptans when accessible and cost-effective, including sumatriptan 100 mg, zolmitriptan 2.5 to 5 mg, or eletriptan 40 mg. When triptans are unavailable due to cost or formulary constraints, high-dose NSAIDs provide reasonable alternatives, though with somewhat reduced efficacy compared to triptan therapy.
ED management of severe attacks may employ intravenous ketorolac 30 mg or subcutaneous sumatriptan when available. Refractory cases benefit from combination approaches, including intravenous antiemetics such as metoclopramide or domperidone with analgesics, or greater occipital nerve blocks with lidocaine and corticosteroids for selected patients.46–51 Status migrainosus requires intravenous corticosteroids, magnesium sulfate, dihydroergotamine, antiseizure medications, and anesthetic agents in addition to standard acute treatments, with hospitalization considered for prolonged, severe presentations.
Special clinical considerations guide treatment modifications in specific patient populations. Patients with cardiovascular risk factors should avoid triptans, with treatment relying on NSAIDs or CGRP antagonists when available and affordable. Frequent attackers experiencing more than two episodes per month require preventive therapy consideration and acute treatment limitation to fewer than 10 days per month to prevent medication overuse headache development. Cost-constrained healthcare environments benefit from generic NSAID prioritization as first-line treatment, reserving triptans for severe attacks or NSAID treatment failures.
Private healthcare settings allow consideration of newer agents, such as rimegepant or lasmiditan, for patients with triptan contraindications. Combination therapies may be offered when clinically indicated and available.
Discussion
Integration of evidence with healthcare realities
This systematic review demonstrates the complex challenge of implementing evidence-based migraine care within the constraints of real-world healthcare systems. Our analysis reveals strong concordance between international evidence and treatment efficacy, while simultaneously highlighting substantial gaps between optimal care and achievable practice in the Mexican healthcare context. The IHS guidelines provide robust recommendations based on high-quality evidence; however, practical implementation requires careful adaptation to local healthcare infrastructure, economic constraints, and patterns of medication availability.47–52
In Mexico, the direct and indirect costs of migraine in 2022 accounted for 1.15% of the country's GDP, equivalent to $14.77 billion. Direct effects contributed 0.295% of GDP, while indirect effects added 0.293% of GDP. 53
The evidence clearly supports subcutaneous sumatriptan as an effective treatment for acute migraine, achieving superior pain relief and freedom from pain rates compared to all other interventions. However, the severe limitations in accessing this formulation within Mexico's public healthcare system create a significant treatment gap affecting the majority of migraine patients who depend on public healthcare services. This disparity illustrates the broader challenge facing healthcare systems worldwide: bridging the gap between evidence-based optimal care and equitable, accessible treatment delivery.
Oral triptans represent a crucial compromise between efficacy and accessibility, offering substantial benefits over placebo while maintaining broader availability compared to subcutaneous formulations. The consistent efficacy demonstrated across sumatriptan, zolmitriptan, eletriptan, and rizatriptan supports their role as first-line treatments for moderate to severe attacks when accessible. However, cost barriers within public healthcare systems continue to limit their utilization, forcing many patients to rely on less optimal but more accessible alternatives.
Healthcare system implications and access disparities
The implementation of evidence-based migraine treatment in Mexico reveals profound disparities between public and private healthcare sectors that extend beyond simple medication availability to encompass broader questions of healthcare equity and access. 54 Public sector challenges include the limited inclusion of triptans in IMSS and ISSSTE formularies, substantial cost barriers for newer therapeutic agents, and an urgent need for policy interventions to improve access to generic medications and expand formularies. These limitations create systematic disadvantages for patients who cannot afford private healthcare or the costs of supplemental medicines (Table 3).
Availability of drugs for acute migraine management in the private and the public health systems in Mexico.
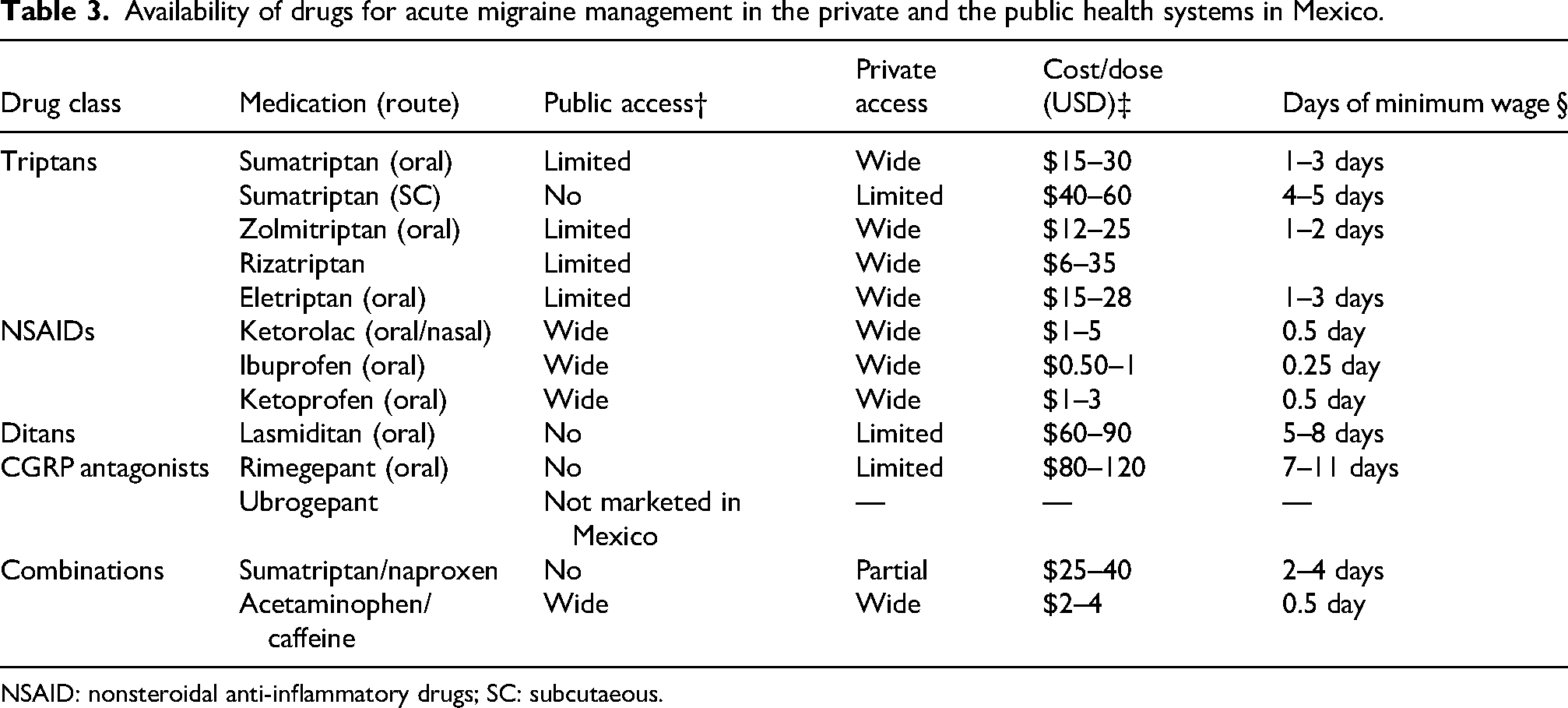
NSAID: nonsteroidal anti-inflammatory drugs; SC: subcutaeous.
The private healthcare sector offers substantially broader treatment options, including access to newer agents for appropriate patients and opportunities for combination therapies when clinically indicated. This disparity creates a concerning two-tiered treatment system where patient outcomes depend significantly on economic status rather than clinical need alone. The ethical implications of this disparity require attention from healthcare policymakers and professional organizations to develop strategies for improving treatment equity across socioeconomic strata.
Geographic disparities further complicate treatment delivery, with urban areas typically providing better access to specialist care and newer medications. In contrast, rural areas face additional constraints, including limited availability of neurologists and restricted formularies. These geographic inequalities necessitate differentiated treatment approaches, emphasizing the critical importance of training primary care physicians in evidence-based migraine management, particularly in regions with limited access to specialists.
Evidence-based treatment selection and clinical decision making
Our systematic analysis supports a structured approach to treatment selection that balances clinical evidence with practical constraints. The evidence hierarchy clearly demonstrates superior efficacy for migraine-specific treatments over nonspecific analgesics, supporting the IHS's emphasis on triptans as first-line therapy for moderate-to-severe attacks. However, the Mexican healthcare context requires a more nuanced approach that considers NSAIDs as practical first-line options given their superior accessibility and proven efficacy, particularly for mild-to-moderate attacks.
The emergence of newer therapeutic classes, including CGRP antagonists and ditans, provides important alternatives for patients with triptan contraindications or inadequate responses. The high-quality evidence supporting lasmiditan across multiple dose levels and rimegepant's demonstrated efficacy represent significant advances in migraine pharmacotherapy. However, their high costs create substantial barriers to widespread implementation, limiting their benefits primarily to patients with access to private healthcare or exceptional financial resources (Table 3).
Combination therapies offer enhanced efficacy compared to monotherapy approaches, with robust evidence supporting the use of sumatriptan-naproxen combinations and adjunctive antiemetics. However, implementation challenges include higher costs, limited availability of fixed-dose combinations, and the complexity of coordinating multiple medications when fixed combinations are unavailable. Healthcare providers must weigh these implementation challenges against the potential benefits of enhanced efficacy when considering combination approaches.
Inappropriate treatment avoidance and provider education
Effective migraine management involves avoiding specific treatments, even if they are used in some cases: (1) “Opioids”: Should not be used due to inferior efficacy compared to migraine-specific therapies (30–40% pain relief versus 60–80% for triptans).55,56 Additionally, there is an increased risk of medication overuse headache (hazard ratios of 2.5–3.0), 57 dependency risks—especially in Mexico 58 —and strong recommendations against their use from international guidelines. 4 The “Butalbital-containing Analgesics” should be avoided because of the high risk of medication overuse headache (up to 50% in chronic cases), 59 as well as concerns about sedation, dependency, and lack of migraine-specific efficacy.5,60 “Tramadol” is more common in Mexico; its risk-benefit profile is poor, and it should only be used if triptans and NSAIDs are ineffective or contraindicated. 61
Enhancing provider education on these guidelines is vital to improving patient outcomes and minimizing dependency risks.
Economic and policy implications
The economic burden of migraine in Mexico, evidenced by nearly 180,000 ED visits over the past decade, underscores the urgent need for improved acute treatment access and healthcare policy reform. The substantial healthcare utilization associated with inadequate migraine management suggests that cost-effectiveness analyses should guide formulary decisions, as providing access to more expensive but highly effective treatments may reduce overall healthcare costs through decreased emergency utilization and improved productivity outcomes.
Policy recommendations emerging from our analysis include several actionable strategies for improving migraine care across Mexico. Expanding public healthcare formularies to include at least one oral triptan option would provide substantial benefits for patients with moderate-to-severe attacks who currently lack access to migraine-specific treatments. Implementing cost-sharing programs for newer therapeutic agents in appropriate patients could bridge the gap between optimal care and economic constraints. Developing regional treatment protocols that account for variations in geographic access would help standardize care while acknowledging local resource limitations.
Healthcare policies should also promote the availability of generic medications through regulatory streamlining and support continuing medical education programs focused on evidence-based migraine management. Professional organizations and healthcare institutions should collaborate to develop standardized treatment protocols that can be implemented across different healthcare settings while maintaining flexibility for local adaptation.
Implementation strategies and clinical practice integration
The successful integration of evidence-based migraine treatment into Mexican clinical practice requires a comprehensive approach addressing provider education, patient empowerment, and healthcare system optimization. Healthcare providers require systematic training in recognizing migraine patterns, selecting appropriate acute treatments, and implementing early intervention strategies to improve outcomes while reducing healthcare utilization and associated costs.
Provider education programs should emphasize the importance of early treatment initiation, as delayed intervention typically results in reduced efficacy and increased duration of disability. The concept of treating attacks when pain is still mild should be promoted, as early intervention with appropriate medications can prevent progression to severe, disabling headache phases that require more intensive interventions and result in greater functional impairment.
Patient education represents another critical component of optimal migraine management, extending beyond simple medication instructions. Patients need a comprehensive understanding of their condition, recognition of early warning signs and trigger patterns, and knowledge of when and how to use acute treatments most effectively. Self-management strategies, including trigger identification and lifestyle modifications, should complement pharmacological interventions to provide holistic care approaches that address both acute and preventive aspects of migraine management.
Study limitations and research priorities
Our systematic review has several significant limitations that should be acknowledged when interpreting results and planning future research directions. Regional healthcare access variations within Mexico are not fully captured in our analysis, and comprehensive cost-effectiveness data were not available for all treatments. Patient preference data remain limited, and long-term outcomes beyond 48 hours were not extensively evaluated in the included studies. Most trials excluded patients over 65 years of age, pregnant women, and those with significant cardiovascular or cerebrovascular comorbidities, which may have limited the generalizability of the findings to these essential patient populations.
The lack of prospective registration represents a methodological limitation that affects transparency and reduces confidence in our approach. Future systematic reviews should include prospective registration to enhance methodological rigor and reduce potential bias in study selection and analysis approaches.
Future research priorities should focus on developing region-specific treatment guidelines that account for differences in urban versus rural access, conducting comprehensive cost-effectiveness analyses of various treatment strategies within the Mexican healthcare context, and evaluating the real-world effectiveness of proposed treatment algorithms through implementation studies. Long-term outcome studies examining the impact of improved access to acute treatment on overall healthcare utilization, productivity, and quality of life provide valuable data for informed healthcare policy decision. 62
Contribution to Mexican migraine care
This work represents the first systematic integration of international migraine treatment guidelines with the realities and constraints of Mexican healthcare. While individual Mexican medical societies have provided treatment recommendations, no comprehensive evidence-based algorithm specifically addressing drug availability and healthcare system constraints has been previously developed for the Mexican context. Our approach provides a framework that can be adapted for use in other healthcare systems facing similar challenges in implementing evidence-based care within resource-constrained environments.
The treatment algorithm we propose provides practical guidance that aligns with international best practices while acknowledging local limitations. This balanced approach provides clinicians with evidence-based tools for optimizing patient care within available resources, potentially improving outcomes for the substantial population of Mexican migraine patients who currently lack access to optimal treatments.
Conclusion
Effective acute migraine treatment in Mexico requires an approach that combines evidence-based medicine with the realities of the healthcare system and socioeconomic challenges. Our systematic review reveals that, although triptans remain the gold standard according to IHS guidelines, the Mexican healthcare context necessitates a more comprehensive treatment strategy that addresses accessibility, cost, regional differences in healthcare delivery, and clinical effectiveness. The evidence indicates that migraine-specific treatments are generally more effective than nonspecific analgesics for most patients experiencing moderate-to-severe attacks.
Clinical implications
Evidence-based treatment algorithm adapted for Mexican healthcare: This study provides the first systematic integration of IHS guidelines with Mexican drug availability and healthcare realities, offering clinicians a practical decision-making framework that considers both efficacy and accessibility.
NSAIDs emerge as practical first-line options: While triptans remain the gold standard per international guidelines, NSAIDs (ketorolac, ketoprofen, and ibuprofen) offer effective and widely accessible alternatives for Mexican patients, particularly in public healthcare settings where triptan access is limited.
Significant treatment access gaps have been identified: Subcutaneous and intranasal sumatriptan formulations, among the most effective acute treatments, have limited availability in Mexico's public healthcare system, highlighting critical gaps that require policy attention and formulary expansion.
Newer agents show promise but face cost barriers: CGRP antagonists (rimegepant) and ditans (lasmiditan) demonstrate strong efficacy with favorable safety profiles, but their high cost limits accessibility in public healthcare, creating disparities in treatment options between private and public sectors.
Setting-specific treatment strategies optimize care. The proposed algorithm provides tailored approaches for home, office, and ED settings, enabling healthcare providers to maximize treatment effectiveness based on available resources and clinical context, while maintaining alignment with international best practices.
Supplemental Material
sj-docx-1-rep-10.1177_25158163251399923 - Supplemental material for Acute pharmacological treatments for migraine: A systematic review with Mexican healthcare perspective
Supplemental material, sj-docx-1-rep-10.1177_25158163251399923 for Acute pharmacological treatments for migraine: A systematic review with Mexican healthcare perspective by Maria-Karina Velez-Jimenez, Adriana Patricia Martínez-Mayorga, Ildefonso Rodriguez-Leyva, Marisol Jannet Figueroa Medina, Maria Teresa Reyes-Alvarez, Juan Carlos Pérez-García, Rubén Darío Vargas-García, Daniel San-Juan, Miguel Angel Morales-Morales, Emilio García-Gómez, Mauricio Pierdant-Perez, Carlos Trenado and Marco Antonio Martínez-Gurrola in Cephalalgia Reports
Footnotes
Acknowledgements
This work was supported by Pfizer Mexico. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article, or the decision to submit it for publication.
ORCID iDs
Author contributions
MV was involved in conceptualization, data curation, and writing—original draft; APM and IR in conceptualization, supervision, and writing—review & editing; MJF in investigation and data curation; MTR in resources and data collection; JCP and EG in investigation and data collection; RDV in data validation and visualization; DS in supervision, writing—review & editing, and project administration; MAM in investigation, project administration, and data collection; MP in methodology and statistics; CT in methodology, data interpretation, and statistics; and MAM in resources, data validation, and visualization.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Pfizer Mexico (grant number NA).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
All data supporting the findings of this review are derived from previously published studies, which are cited in the manuscript. Any extracted or summarized data used in the preparation of this review are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
