Abstract
There is a multitude of scientific papers and guideline papers on the methodology of drug trials in migraine. Here, we try to condense this into a single paper and to make proposals for future consideration. Literature known by the authors and from reference lists of relevant publications was used for the history. Relevant literature for our proposals was searched on PubMed. The main headings in published guidelines, namely, Patient selection, Trial design, Evaluation of results and Statistics, have remained unchanged over the years. Most of the methodology has remained unchanged but the changes that have taken place are important. Chronic migraine has been studied separately from episodic migraine, children and adolescents distinguished from adults, and migraine without aura from migraine with aura. In trial design, the group comparison design has taken priority over the cross-over design, but the latter is suggested for investigator driven, comparative, dose finding and aura trials because of its superior power. There is a confusing number of possible primary end points: number of migraine attacks, number of migraine days, number of headache days and number of 50% responders in prophylactic trials, whereas two-hour pain free is agreed in acute trials. However, also 24- and 48-hour pain free have been suggested and headache relief is sometimes still used against recommendations. Most bothersome symptom has been requested as a co-primary end point by Food and Drug Administration (FDA). Our future suggestions are meant to provide food for thought for future committee work. We suggest that most bothersome symptom needs to be discussed with FDA as a co-primary end point in acute trials. It could also be discussed whether episodic- and chronic migraine need separate study. Migraine with- and without aura should be studied separately. Furthermore, two-hour pain free should be maintained as the primary end point but the use of stricter outcome parameters should be explored. In prophylactic trials, migraine days are recommended over migraine attacks and over 50% responders. For investigator-initiated trials, comparative trials and proof of concept trials by small companies, the cross-over design with its superior power is still recommended. Finally, the need to lump various guidelines into one major document should be considered. The methodology of drug trials in migraine has been worked out in detail. It is important that these guidelines be followed in all clinical trials. We highlight several issues that merit attention in the future.
Introduction
The methodology of drug trials in migraine is clinically important, intellectually challenging, complex and very interesting. Specialists caring for migraine patients should know something about this. At the most basic level, physicians caring for headache patients should be able to judge the methodology of the published literature. They should be able to make a personal judgment of the drugs that they prescribe. Many will participate in company-initiated drug trials and they should understand the principles of drug trial methodology to be valuable members of the team. Being just one out of many investigators is an important learning experience for young doctors and often the first way into migraine research. At a more senior level, there is the possibility of participating in publication committees, which requires a deeper understanding of the art.
The body of knowledge about drug trials in migraine is considerable. The scientific literature has moved from methodological papers by individual authors to guidelines authored by committees of the International Headache Society (IHS). Over the years, many editions of such guidelines have been published, each of considerable size and complexity, but also with a lot of repetition. It is therefore difficult to obtain a precise overview of the field. The intention of the present review is to present essentials of the methodology of drug trials in migraine in a single article. It is written by persons who first published several individual articles about the methodology of drug trials in migraine and thereafter (PTH) chaired three editions of the IHS guidelines. Hopefully, it will help readers understand the field without spending days studying the long and repetitious committee reports. As seasoned experts in the field, we also take the liberty of suggesting several questions for discussion by future IHS drug trial committees.
The first scientific articles
In the period 1968–1979, a few papers discussed the methodology of drug trials in migraine (1,2). It is interesting to see how primitive the reasoning in these papers seems today. But, at the time, there were very few drug trials available for analysis, no explicit diagnostic criteria for migraine were available and the double-blind design was not yet obligatory. The papers highlighted several problems but gave few answers that are valid today. A new era started with a paper published in the first volume of Cephalalgia (3). It focused on prophylactic treatments because there were no published trials of acute treatment that could support a methodological discussion. The paper laid out a solid ground upon which all subsequent papers and committee reports have been based. It made clear for the first time that only double-blind trials are valid and it listed a range of issues to be decided in the protocol of future prophylactic trials . They were grouped under four headings that have been used in all subsequent committee reports: Patient selection, Trial design, Evaluation of results and Statistics.
Each group had many items, and a tentative answer was provided for them all. Many of these answers still stand today, whereasothers have been changed in subsequent committee work. Four years later, the same authors had gained experience with acute drug trials and were able to analyze the methodology of drug trials of acute migraine attacks (4). This was further expanded in a book chapter (5) The major sections on acute trials were the same as for prophylactic drug trials: Patient selection, Trial design, Evaluation of results and Statistics. But the questions in each section and the proposed solutions for acute trials often differed from prophylactic trials. For both acute and prophylactic trials, the need for better diagnostic criteria for migraine was obvious and explicit criteria were suggested, criteria that later were modified and included in the International Classification of Headache Disorders (ICHD)-1 (6). Much discussion and many solutions in these first modern era papers reflected that company budgets for trials were very small and the budgets for the scientists even smaller. A major task was therefore to optimize trial design so that as few patients as possible would be needed. This has later become obsolete in phase 3 trials because companies today are willing to include hundreds of patients in each group as a result of regulatory demands. For certain types of trials (comparative, dose finding and investigator-initiated trials), these considerations are still valid (see later). To what extent published trials had taken crucial methodological issues into account was analyzed (7). The results were not encouraging and called for much more attention to methodology in future trials.
IHS drug trial committee reports about acute drug treatment: First, second, third and fourth editions
The main features of these extensive committee guidelines are shown in Table 1. The table shows that most parameters have not changed over the years (8–11). However, the modest changes are important, and they can easily be identified in Table 1.
Design, selection of patients, trial design and evaluation of results of controlled trials of acute drug treatment of migraine attacks, as recommended in the four editions of the guidelines for drug treatment of migraine.
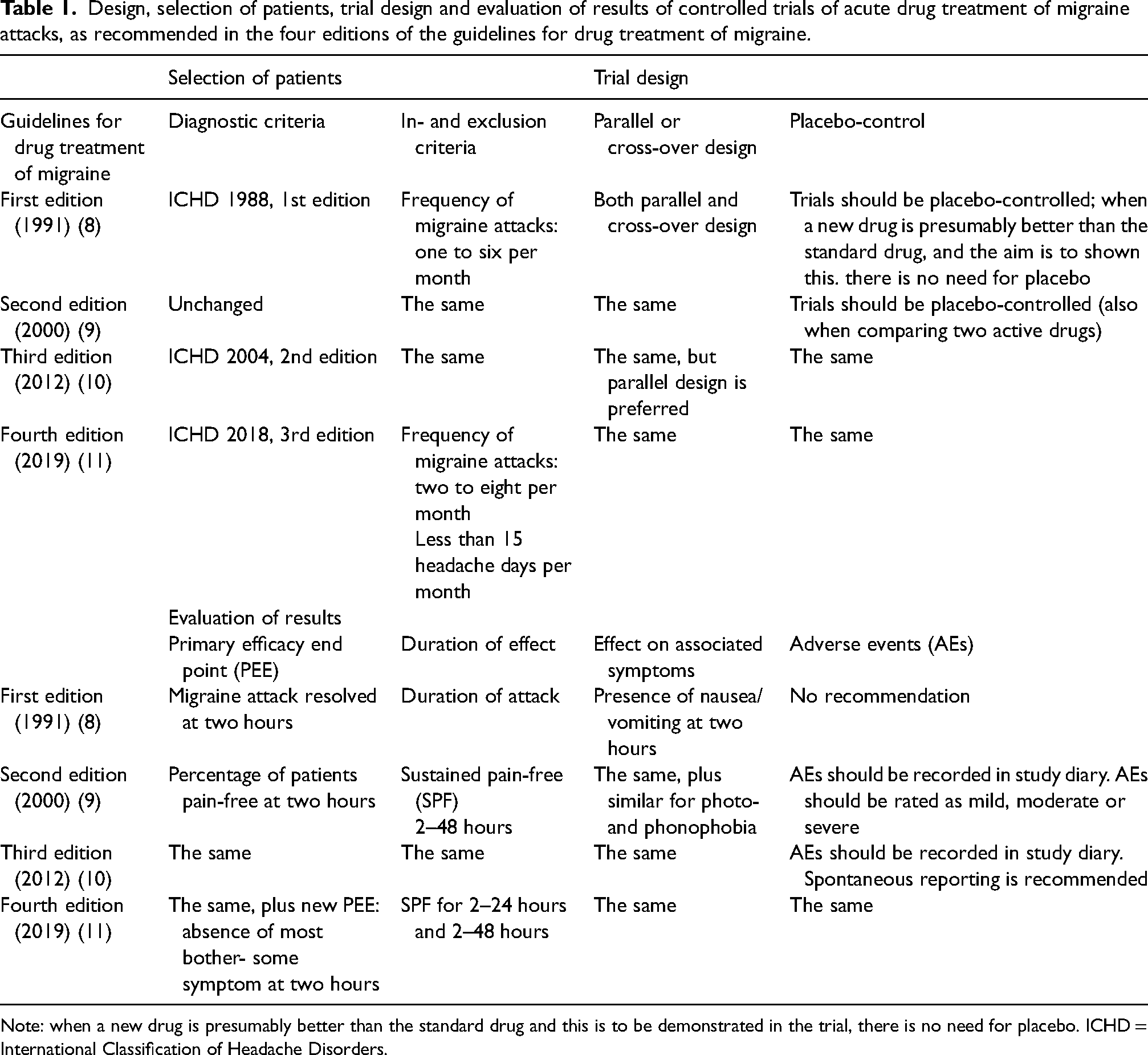
Note: when a new drug is presumably better than the standard drug and this is to be demonstrated in the trial, there is no need for placebo. ICHD = International Classification of Headache Disorders.
The most significant changes are “most bothersome symptom”, which was added in the Fourth Edition in 2019 because of a recommendation from the Food and Drug Administration (FDA), the reduction of headache relief to become a secondary outcome parameter and the increased focus on the recording of adverse events. Following FDA recommendations, the parallel group comparison design became preferred over the cross-over design.
IHS drug trial committee reports about preventive drug treatment: First, second, third and fourth editions
The main features of these extensive committee guidelines are shown in Table 2. The table shows that most parameters have not changed over the years with subsequent editions (8–10,12). However, the modest changes are important, and they can easily be identified in Table 2.
Design, selection of patients, trial design and evaluation of results of controlled trials of preventive drug treatment of migraine attacks, as recommended in the four editions of the guidelines for drug treatment of migraine.
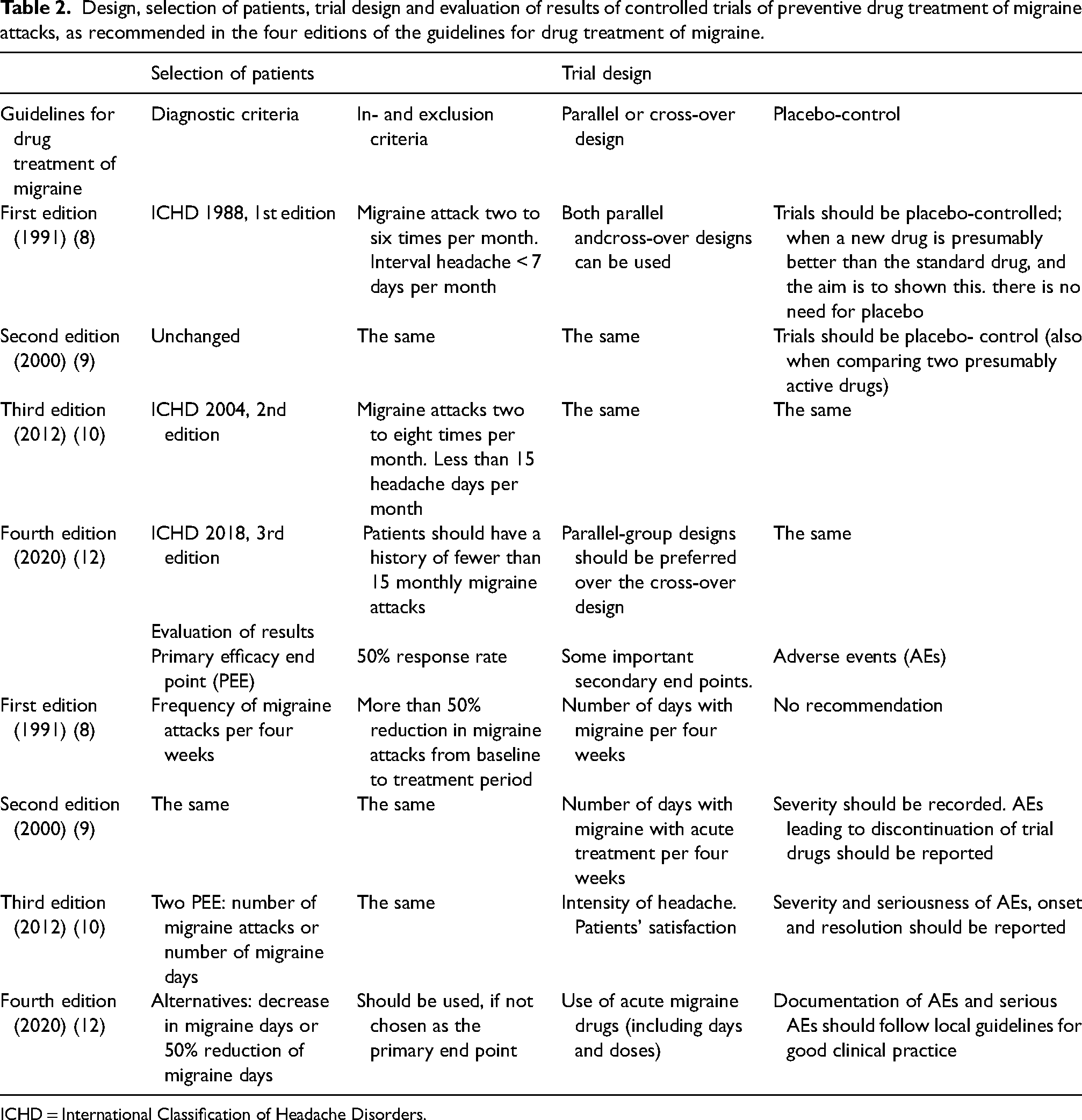
ICHD = International Classification of Headache Disorders.
The most important changes were inclusion of attack frequency up to eight per month but less than 15 days of headache to distinguish episodic and chronic migraine, the gradual shift to only recommend the parallel group comparison design, the increasing focus on adverse events and the acceptance of three possibilities for main outcome parameter in the 4th Edition: number of migraine attacks, number of migraine days and 50% responder rate. The ones not selected for main parameter should be reported as secondary outcome parameter.
Migraine subtypes
Chronic migraine
Chronic migraine has been the topic of two committee guidelines in the belief that it posed special problems in the study of prophylactic medications, whereas it has been assumed that attack treatment (acute treatment) would not show such differences (13,14) (Table 3)
Design, selection of patients, trial design and evaluation of results in randomized, controlled trials of preventive drug treatment of chronic migraine, recommended in the two editions of the guidelines for preventive drug treatment of chronic migraine.
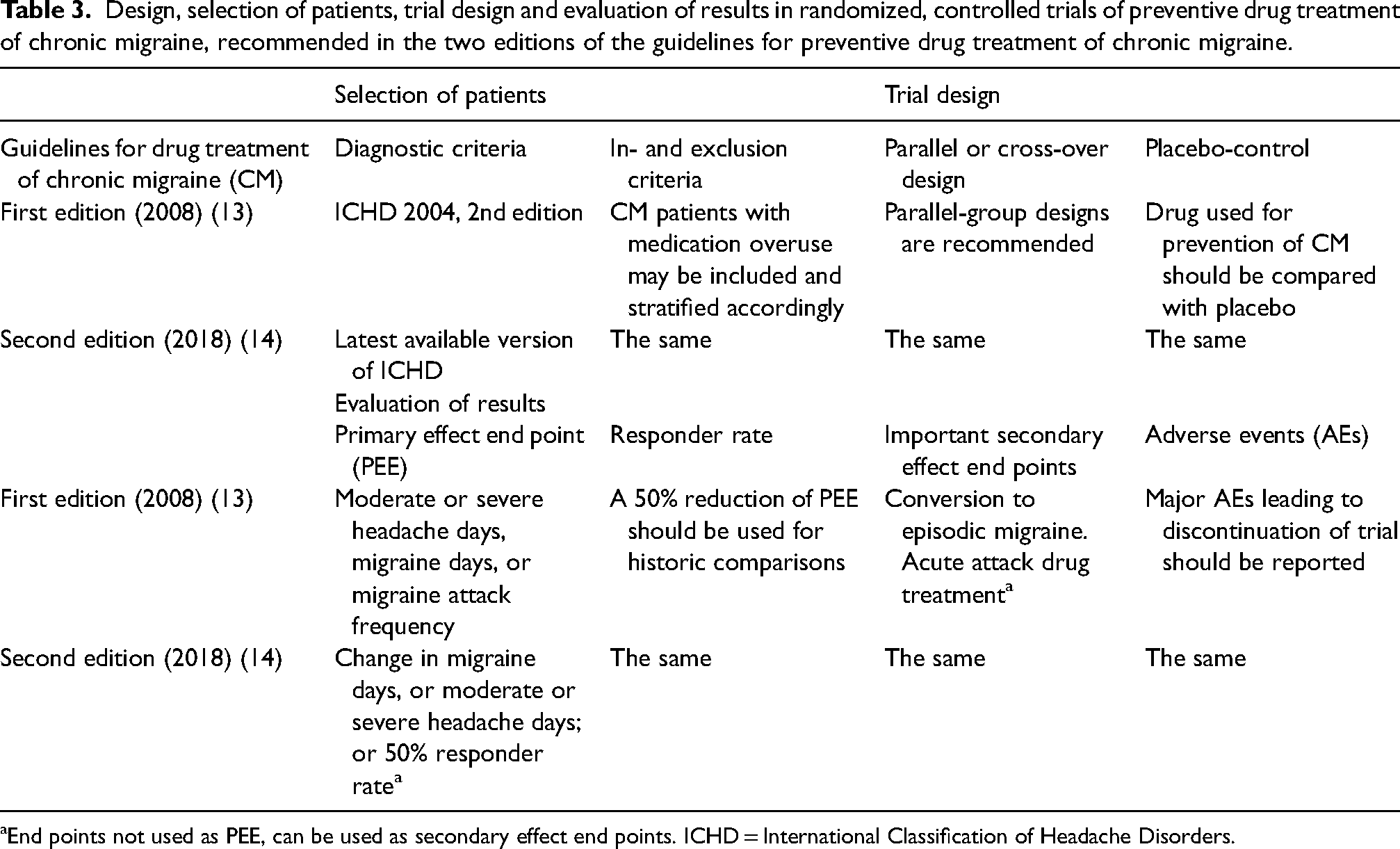
End points not used as PEE, can be used as secondary effect end points. ICHD = International Classification of Headache Disorders.
The main differences from previous recommendations for migraine in general are that medication overuse became allowed but should be recorded, that moderate or severe headache days could be a main outcome parameter and that conversion to episodic migraine should be a secondary outcome parameter.
Children and adolescents
Migraine in children and adolescents has been the topic of one committee guideline and an additional update version in the belief that they pose special problems in the study of preventive medications, whereas it has been assumed that attack treatment (acute treatment) would not show such differences (15,16) Table 4.
Design, selection of patients, trial design and evaluation of results, of controlled trials of preventive drug treatment of migraine in children and adolescents.
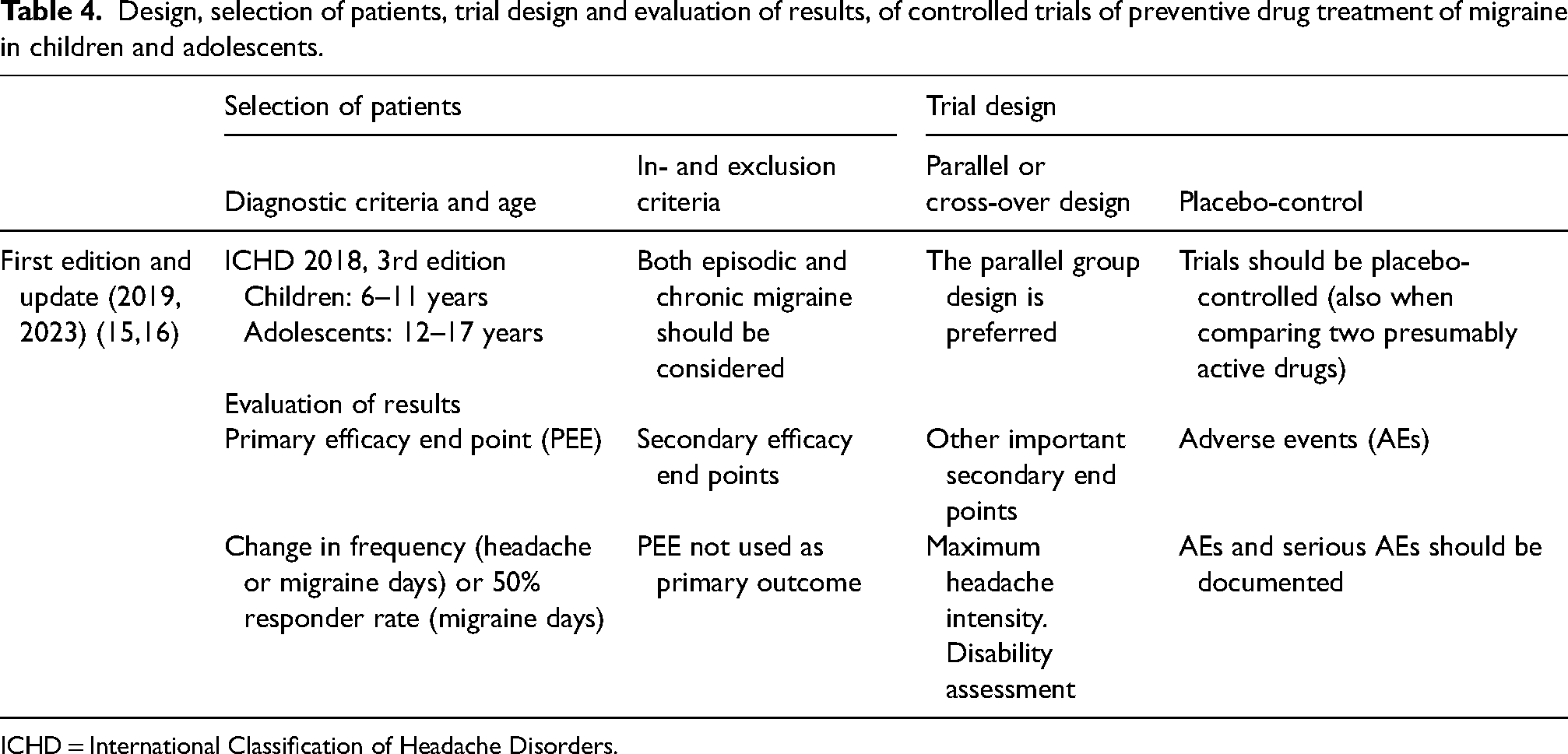
ICHD = International Classification of Headache Disorders.
In children and adolescents, parental observations are accepted. A six-month history is minimum for inclusion and recruitment should cover the whole calendar year in order to avoid influence of the well-known dip in attack frequency during summer holyday. Age at recruitment should be 6–17 years and the 2023 paper points out that all these ages should be studied together. A secondary analysis could subdivide into two age groups. Contraceptive measures are very important in the adolescent group but not always thought about. The many special childhood syndromes are not mentioned but should be excluded if they are suspected of influencing the treatment result or contraindicate use of the trial medication. Consent of the children is not discussed but must be obtained in addition to consent from the parents. Body weight will vary considerably according to age. It is suggested to stratify according to this, but there are no specific recommendations. The 2023 paper (16) importantly specifies how migraine days are counted taking not only typical, but also atypical attacks into account. Headache days are an alternative that may be preferred. It is also pointed out that there are no documented differences between chronic migraine (CM) and episodic migraine (EM) in children and adolescents other than attack frequency and that they should be studied together. A possible sub-analysis could look at differences post hoc, but this requires relatively large controlled trials. There is more emphasis on disability measurement in this than in previous guideline documents.
Migraine with aura
Differences between migraine with aura (MA) and migraine without aura (MO) have only been briefly discussed in previous guideline papers. They have largely focused on MO and assumed that MA can be included under the same guidelines. However, the evidence for a difference between MA and MO has increased in recent years. Apart from the changes in cerebral blood flow suggesting cortical spreading depression in MA and not in MO (17), differences in genetics (18) and differences in menstrual and pregnancy relations (19) have also been demonstrated. It is of major importance that the response to triptans is smaller in MA than in MO (20) and that tonabersat was effective in reducing auras but not in a material dominated by MO (21,22). In the following discussion, we draw heavily on a previous paper specifically discussing aspects of drug trial methodology in MA (23).
In acute trials, the primary outcomes should be either reduction of aura duration or freedom from pain 2 h after dosing. Long lasting aura is important in the first case, but the study is very difficult. Only very fast acting drugs may have a chance to shorten the aura. Inhalation of amyl nitrite has been tried (unpublished data by the K. Jensen) but it caused terrible headaches and was only tried a few times. It seems unlikely that it will ever be possible to shorten the aura. In studies aiming at relieving the headache, a consistent and severe pain phase is important. Dosing should normally be at the onset of aura hoping to prevent the headache but triptans do not work if given before the headache (24). Therefore, trials giving the drug at the onset of pain may also be needed. Other headaches are not a reason for exclusion. They are easily distinguished because of the characteristic symptomatology of the aura.
In previous preventive trials, patients with both MO and MA attacks have usually been selected for a high frequency of attacks of any kind, which means a high frequency of MO attacks, but, in trials of MA, the frequency of MA attacks should obviously be a priority. Patients should be included whether they have a headache following the aura or not and only migraine with typical aura should be included. Patients with brain stem aura are extremely rare and the diagnosis is disputed, whereas patients with hemiplegic aura differ in genetics and in response to migraine provoking agents (25). It is very important to use a calendar of the type suggested previously so that each attack can be diagnosed as MA or MO (24). Patients should have a relatively high frequency of auras, but such patients are rare. We suggest as a minimum one aura per month. MO or other headaches should not be an exclusion criterion as they can easily be distinguished with an appropriate diary. Attack frequency in MA fluctuates and a run-in period should not be used for in- or exclusion. Patients should be included based on their estimate of average attack frequency over the preceding three months. The cross-over design is preferable to obtain enough power because eligible patients with a sufficiently high attack frequency are rare. A group comparison design needs a huge number of centers and will be very costly. The primary outcome should be number of auras and a possible co-primary outcome is the frequency of headaches following an aura.
Preventive trials for only one type of migraine are relatively rare. Thus, in the 2nd and 3rd editions of the textbook: The Headaches, the distribution of 90 preventive randomized, controlled trials (RCTs) according to MO and MA were: 79 RCTs (88%) with MO and MA combined; three RCTs (3%) with MA, and eight RCTs (9%) with MO (26,27).
It should only be a question of time before regulatory authorities demand separate studies in MA and MO.
Suggestions for the future
Some issues in current guidelines are worth further discussion. It is perhaps advisable in future not to split such guidelines up into many separate publications. In time, there will also be guidelines for drug trials in several other types of headaches, and the result of continuing the present policy will be a very large number of guidelines. One for each headache disorder seems to be a reasonable compromise. Our present analysis has shown that for each new guideline there is only a small amount of information that differs from other guidelines. We propose in future to deal with acute and prophylactic treatment, episodic and chronic migraine, migraine with- and without aura, children and adults together. Thus, we propose one comprehensive document developed by the main standing committee under which working parties should deal with the special issues in short sections that should discuss only what is different from the guidelines in the main document. The ICHD has used this format with considerable success. Such a comprehensive document from the IHS should only be published at relatively long intervals, but parts of it could be updated at different intervals on behalf of the committee as single papers in Cephalalgia. Many drug trials have separately studied CM and EM (28). Our extensive literature search has shown that drugs have the same effect in CM and EM with the possible exception of botulinum toxin. Conversely, we found no drug that is effective in EM and not in CM when appropriately tested. This is in accordance with pathophysiological and genetic knowledge showing that there is no fundamental difference between the two. As pointed out by the IHS committee on drug trials in pediatric migraine, it is only the frequency of attacks that is different. Patients quite often spontaneously or because of preventive treatment change from CM to EM or from EM to CM (29). Finally, the diagnostic criteria for CM have been criticized and revised criteria suggested to use only attack frequency to distinguish CM from EM (30,31). The wisdom of generally requesting trials separately in CM and EM should therefore be questioned in discussion with regulatory authorities. One reason for separate trials is the frequent presence of medication-overuse headaches (MOH) in CM but not in EM. Thus, in mixed materials there would usually be too few MOH patients for a reasonably powerful sub-analysis of this group. The main reason for the distinction is, however, to reserve expensive treatments for CM, but that should be done by therapeutic guidelines rather than by regulatory demands to drug trials.
There are real differences between MA and MO in pathophysiology, genetics, cerebral blood flow and response to drugs, as discussed above. For these reasons, it seems important in future to strengthen guidelines with regard to MA and to recommend drug trials in MA and MO separately.
When two presumably active acute or preventive treatments for migraine are compared, a placebo control should be included (11,12). In recent comparative, preventive RCTs without a placebo-control, the 50% reduction in migraine days was 62% after galcanezumab and 61% after rimgepant (32) There is, however, no scientific proof of this without a placebo-control, because, in a review of the placebo effect for a >50% reduction in migraine days (33), there were two preventive RCTs with greater than 60% reduction of migraine days on placebo. Even in comparative RCTs showing a difference between two active drugs, a placebo control should be included for assay sensitivity (12). This is also the case for acute RCTs in migraine (11).
Adverse events (AE) are particularly important in the prophylaxis of migraine because most drugs have similar efficacy. In the first and only RCT using AE as a primary outcome, subcutaneous erenumab (n = 389) and oral topiramate (n = 388) were compared for migraine prevention (34) The primary end point was medication discontinuation because of adverse events during the double-blind phase. In the erenumab treated group 11% discontinued medication because of AEs compared to 39% in topiramate treated group (p < 0.001). Thus, AE was a good end point which should be used more in future comparative drug trials.
It may be considered if an even tougher outcome parameter than pain free at two hours should be primary. For example, 24 or 48 hours of freedom from pain or even requesting such response to be consistent. This is what patients really want and it would probably make it easier to distinguish between active treatments.
In prophylactic trials, migraine days should be preferred over migraine attacks. The latter is difficult to use in chronic migraine, whereas the former can be used for all migraine patients. Moderate or severe headache days should be abandoned as a primary outcome parameter.
Investigator driven trials, comparative trials and phase 2 trials of small compagnies should receive more attention in future guidelines.
Conclusions
The excellent guidelines from the IHS have greatly improved drug trials in migraine. Despite this major achievement, there are still issues in need of further attention
The methodology of drug trials in migraine has been described in multiple publications and is fragmented This paper has synthesized this information It is an easy inroad to understanding the principles of drug trials
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
