Abstract
Introduction
Despite recent advances in migraine treatment there is a need for therapies with higher clinical efficacy and/or fewer side effects. Triptans (5-HT1B/1D/1F agonists) are essential in the present treatment regime and gepants (CGRP-receptor antagonists) are recognized as effective in acute migraine treatment. Triptans and gepants have different mechanisms of action and here we tested the hypothesis that a combination of these drugs (sumatriptan and olcegepant) would result in an additive effect.
Methods
Using the validated glyceryl trinitrate mouse model of migraine, we initially tested dose-response relationships of sumatriptan (0.1, 0.3, and 0.6 mg/kg IP) and olcegepant (0.25, 0.50, and 1.0 mg/kg IP) to find suitable high and low doses. Subsequently, we performed a combination study of the two drugs with a low and a high dose. All experiments were vehicle (placebo) controlled and blinded.
Results
Sumatriptan significantly reduced glyceryl trinitrate-induced allodynia (F(4,54) = 13.51, p < 0.0001) at all doses. Olcegepant also reduced glyceryl trinitrate-induced allodynia (F(4,53) = 16.11, p < 0.0001) with the two higher doses being significantly effective. Combining 0.50 mg/kg olcegepant with 0.1 or 0.6 mg/kg sumatriptan did not have any improved effect compared to either drug alone (p > 0.50 on all days) in our mouse model.
Conclusion
Combining olcegepant and sumatriptan did not have an additive effect compared to single-drug treatment in this study. Triptan-gepant combinations will therefore most likely not improve migraine treatment. Nevertheless, further studies are necessary, and combinations should also be examined in patients with migraine.
Introduction
Regardless of recent advances in migraine treatment, there is still a large unmet need in the clinic (1–5). Triptans are the most commonly used drugs, sumatriptan being the standard reference (6). Triptans are 5-HT1B/1D/1F agonists (7) inhibiting trigeminal calcitonin gene-related peptide (CGRP) release by hyperpolarizing nerve endings (8) and causing cranial vasoconstriction (9,10). Although beneficial in many patients, lack of effect and a relatively high incidence of side effects cause limitations (11–13). A meta-analysis of 53 high-quality, placebo-controlled randomized clinical trials showed an average sumatriptan (100 mg per oral) response rate of 59% (95% CI 57–61) for any headache response, 29% (95% CI 27–31) for pain freedom and 20% (95% CI 18–21) for sustained pain freedom (14), showing that sumatriptan often results in pain reduction and not pain freedom.
As an alternative to sumatriptan, small molecule CGRP receptor antagonists (gepants) have proven effective in the treatment of acute migraine attacks (15,16). This drug class does not show any of the cardiovascular side effects seen with triptans and has a prolonged action compared to triptans (10). The standard experimental gepant is olcegepant, originally developed by Boehringer Ingelheim and available for animal experiments (17). In late December 2019, the US Food and Drug Administration approved ubrogepant from Allergan for the acute treatment of migraine with and without aura (18,19). This approval followed the latest randomized trial where pain freedom at 2 hours was observed in 19.2% of participants in a 50 mg ubrogepant group (p = 0.002), 21.2% in the 100 mg group (p < 0.001) and 11.8% in the placebo group (20). Rimegepant, from Biohaven Pharmaceuticals, is expected during 2020 (21). The clinical efficacy of gepants has been in the same order or lower than that of the triptans (3,9,22). Thus, neither triptans nor gepants are optimally effective (14,20).
We investigated if combining sumatriptan and olcegepant has an additive effect compared to the individual compounds. An additive effect is likely in compounds with different mechanisms of action (23), as exemplified by the slightly higher efficacy of a sumatriptan–naproxen combination than either drug alone (24,25). Since neither triptans or gepants yield complete pain freedom, an additive effect might be possible. To test our hypothesis, we used the mouse glyceryl trinitrate (GTN) model of migraine as both sumatriptan and olcegepant reduce allodynia in this model (26,27). If increased effect could be shown, it would strongly support future human studies using combinations. On the other hand, if our hypothesis could not be confirmed, it would suggest that increase in clinical efficacy is unlikely in humans.
Materials and methods
Animals and housing
Male C57BL/6NTac mice were received at 7 weeks of age (Taconic, Denmark), body weights 21–31 g and left to acclimatize for 1 week prior to starting experiments. Mice were group-housed six per cage in eurostandard type III cages (42.5 × 27.6 × 15.3 cm, Tecniplast, Italy) with food (Altromin 1314F and sunflower/corn/peanut mix, Brogaarden, Denmark) and tap water ad libitum. Cages were floored with sawdust (Aspen chip, Tapvei, Estonia) and enriched with 2–3 red-tinted polycarbonate shelters and tunnels (Molytex, Denmark), play tunnel (size small, Scanbur, Denmark), nesting material (Happi-Mats and Enviro-Dri, Brogaarden, Denmark), biting sticks (Aspen Chew Bricks, Labodia, Switzerland), and autoclaved hemp rope (Fyns Kran Udstyr A/S, Denmark). The mice were housed in a light-, humidity-, and temperature-controlled environment (12:12 h light/dark cycle with lights on at 07.00 h, relative humidity 50 ± 10%, and 24°C). The animals were weighed on test days (every other day) and a total of 177 mice were used in the study. After the end of the test protocol, mice were anaesthetized by a mixed gas (70% CO2, Strandmøllen A/S, Denmark) and decapitated. All experiments were planned and carried out in accordance with the ARRIVE guidelines. Experiments were carried out between 08.00 and 15.00 hrs. Animal experiments were approved by the Danish Animal Experiments Inspectorate (ethical approval number 2017-15-0201-01358).
Compounds
Three different compounds were used in this study: The NO donor GTN, the 5-HT1B/1D/1F receptor agonist sumatriptan and the CGRP-receptor antagonist olcegepant HCl. GTN (7.89 mg/mL in 96% ethanol (EtOH), Cambrex Germany, distributed via the Capital Region Hospital Pharmacy, Denmark) was diluted in saline (Fresenius Kabi, Germany) to 1 mg/mL and administered to mice at 10 mg/kg (28,29). For the GTN vehicle control group, a corresponding amount of 96% EtOH was dissolved in saline (yielding a 12.2% EtOH solution). Sumatriptan (GSK Denmark, Imigran 12 mg/mL) was diluted in saline to three different dosages (0.1, 0.3, and 0.6 mg/kg), the highest dose being commonly applied (27,28,30). Olcegepant HCl (CAS 586368-06-1, MedChemExpress, NJ, USA) was dissolved in saline to three different dosages (0.25, 0.50, and 1.0 mg/kg), the highest dose already validated (27). All compounds were administered intraperitoneally (IP) in a volume of 10 mL/kg with 27G×3/4″ needles (Becton, Dickinson and Company Ltd., Ireland). For administration of both olcegepant and sumatriptan simultaneously, the concentration of injected compounds was doubled to limit the total injection volume to 10 mL/kg.
Mechanical sensitivity thresholds
For this study, we applied a validated mouse model of migraine (28,29,31). The model uses repeated injections of the known migraine trigger GTN (32,33) to sensitize mice (28,34). Chronic administration of GTN in mice has been shown to induce a continuous and additive general allodynia (26). As described by Pradhan and colleagues (2014), we measured allodynia in the mice via cutaneous mechanical sensitivity testing (28,29). Mice were left to acclimate for 45 min in individual, clear plexiglass cages (L9.5 × W9.5 × H13 cm) on a mesh floor (IITC Life Science, CA, USA) that allowed access to the paws. The plantar area of the left hind paw was stimulated with von Frey filaments (0.008 – 2.0 g (excluding 1.4 g), Ugo Basile, Italy) until a withdrawal of the paw occurred or for 2–3 s. Using the up-down method with four stimulations after the first breaking point (change in response), the response pattern and final filament was noted and a 50% withdrawal threshold was calculated as originally defined by Chaplan et al. (35) and optimized by application of exact inter-filament steps as described by Christensen et al. and calculated online at https://bioapps.shinyapps.io/von_frey_app/ (36). Prior to the first test day, mice were acclimated to the test chamber for 1 hour. Testing was performed every other day for 9 days, yielding a total of 5 test days (Figure 1) as described by Pradhan et al. (28).
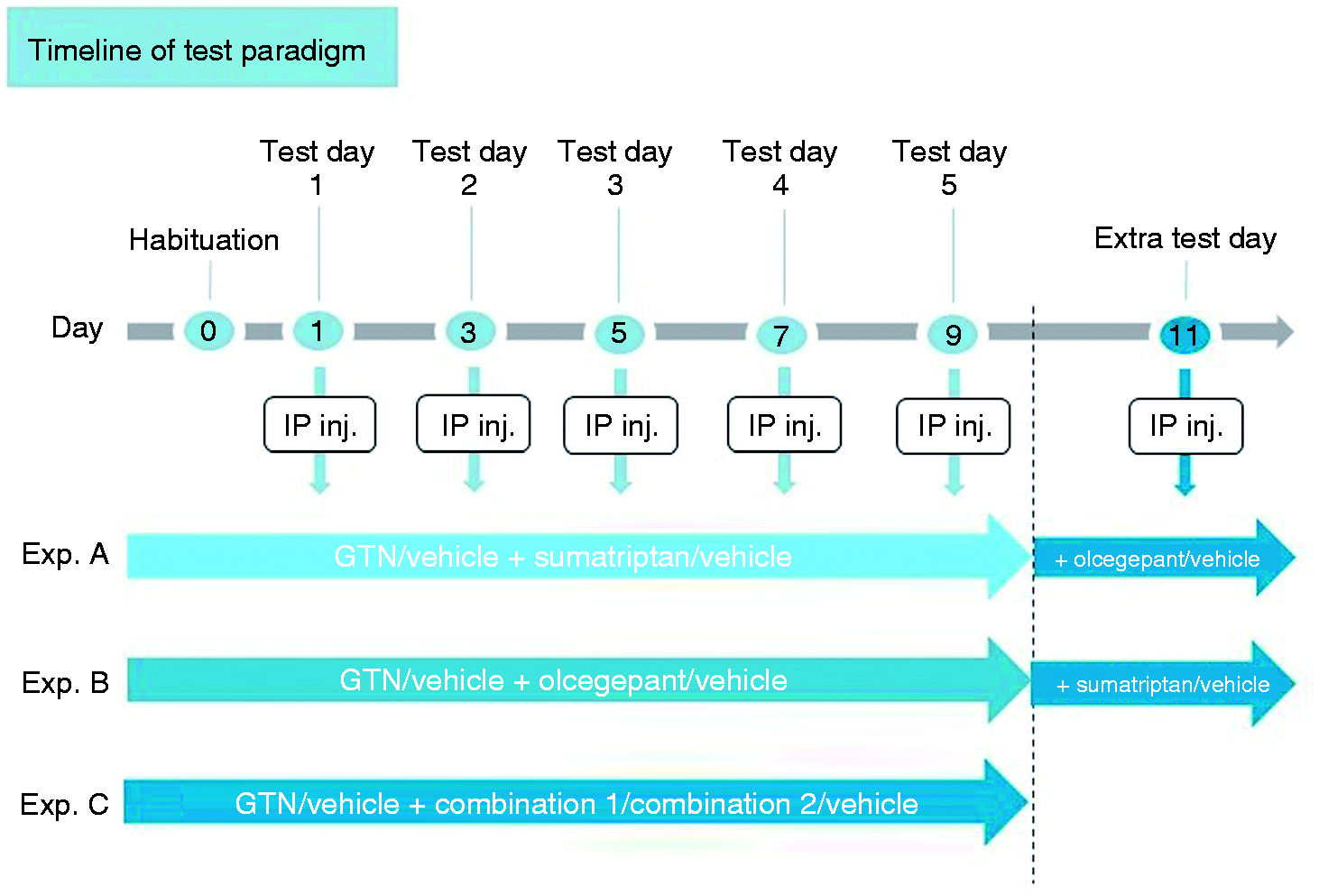
Experimental timeline. Following 1 day of habituation (day 0), five tests were done every other day over the course of 9 days. After the regular paradigm, an extra test day (day 11) was added in experiment A and B to examine acute add-on treatment after repeated treatment.
Experimental protocols
Three experiments were performed using the GTN mouse model described above. Firstly, two separate dose-response experiments were performed with either sumatriptan or olcegepant in three different doses, the highest being the ones currently used in our group (27). For both experiments, a GTN control group and a GTN vehicle control group were included. Baseline thresholds were measured in the mice before IP administration of GTN. Mice received sumatriptan or olcegepant injections 1 h and 15 min after GTN injections, when they were put in the testing chambers for habituation prior to testing sensitivity 2 h post GTN injection. The same protocol and timing were used for the third experiment examining potential additive effects through combination of olcegepant in a fixed dose with either a low or high dose sumatriptan (doses found in the previous dose-response experiments, combinations termed 1 and 2, respectively). The same two control groups as in the dose-response experiments were included as well as an olcegepant control group (the fixed dose) and a sumatriptan control group (high dose) for references. Study setup is summarized in Table 1 and Figure 1. The experimenter was blinded to treatments.
Overview of experimental groups and drug administration (IP) in the three experiments. A total of 177 male micewere used.

*Mice from this group died during the 9-day protocol.
**A mouse was transferred from vehicle to glyceryl trinitrate (GTN) control group by accident after the first day.
+Acute add-on treatment after the regular paradigm; repeated sumatriptan-treated animals received 1.0 mg/kg olcegepant and repeated olcegepant-treated animals received 0.6 mg/kg sumatriptan.
Acute add-on treatment in mice receiving repeated treatment
After the regular paradigm with 5 test days, the animals were tested one more day (day 11, see Figure 1) to examine whether a rescue with another drug was possible. At this point, mice had received repeated injections of either sumatriptan or olcegepant in various doses. Mice receiving the lowest and middle dose of drug A (sumatriptan or olcegepant) were administered a high dose of drug B (olcegepant or sumatriptan) and the same test protocol for mechanical sensitivity measured by allodynic response was followed (Table 1).
Data evaluation and statistical analyses
Mice were randomized within each home cage to avoid potential cage effects and when possible, adjusted to have each treatment group represented in each cage. Group sizes were based on previous data with this particular model (27,28) and experience showing that sample sizes ranging from n = 10–12 per group ensure that the study is not underpowered. This is highly important: using a model with a relatively small effect window and with the experimenter completely blinded, which consequently leads to more variation. Statistical analyses were done in GraphPad Prism 8 (Graph Pad Software Inc., CA, USA). Parametric parameters were evaluated, and normality was assessed by Q-Q plots and the Shapiro-Wilk test. Data transformation by square root (SQRT) was used in order to achieve normally distributed data with equal variances. A two-way repeated measures ANOVA (mixed effects analysis when values were missing) was used with Dunnett’s test for multiple comparisons (when comparing to the GTN control group) or Tukey’s multiple comparisons test (when comparing reference groups to combination groups). In the additive effect test, areas under the curve (AUCs) were calculated for each animal and normalized to the mean AUC for the vehicle animals (animals not participating in every test day were excluded to not represent artificially low AUCs). A one-way ANOVA with Dunnett’s test for multiple comparison was done for all groups and for comparing the two combination groups with either of the single treatment groups. Significance level was set at 95% with p ≤ 0.05 considered statistically significant. All data are shown as means ± standard errors of the mean (SEMs).
Results
All three doses of sumatriptan reduced acute allodynia to a varying degree
To determine the proper dose of sumatriptan to be used in the additive experiment, a dose-response experiment with sumatriptan was initially performed. Acute responses to GTN provocation and sumatriptan treatment are shown in Figure 2(a). Overall, mixed-effect analysis indicated a significant effect of treatment groups (F(4,54) = 13.51, p < 0.0001). Post hoc analysis showed that no groups were different at baseline on the first day. Two hours post administration of GTN, a significant allodynia was seen compared to the vehicle group on every test day (p < 0.0001 and p = 0.0004 on day 3). The lowest dose of sumatriptan, 0.1 mg/kg, showed significant inhibition of the allodynia on days 7 and 9. The middle dose of sumatriptan, 0.3 mg/kg, significantly reduced the allodynic response to GTN on all test days. The high dose of sumatriptan, 0.6 mg/kg, had a significant inhibitory effect on GTN provocation on test days 1, 3, and 5 but no significant effect thereafter. The low (0.1 mg/kg) and high (0.6 mg/kg) doses of sumatriptan were chosen for further studies, as they both seemed to be effective without resulting in complete inhibition of the GTN-induced allodynia.

The inhibitory effect of (a) sumatriptan or (b) olcegepant on glyceryl trinitrate (GTN)-induced allodynia. Following baseline measurement, 50% withdrawal thresholds were measured 2 h after GTN (10 mg/kg, IP) or vehicle administration on 5 test days (1, 3, 5, 7, and 9). Subsequently (a) sumatriptan (0.1, 0.3, or 0.6 mg/kg IP) or vehicle and (b) olcegepant (0.25, 0.50, or 1.0 mg/kg IP) or vehicle were administered on the same days. The vehicle group was significantly different from the GTN group on all days (p < 0.0001 and (a) p = 0.0004 on day 3, and (b) p = 0.0002 on day 7, #). Raw data were square root transformed (SQRT) and are represented as means ± SEMs.
0.50 and 1.0 mg/kg olcegepant had significant inhibitory effect on GTN-induced allodynia
Next, a similar dose finding experiment was performed to determine the proper dose of olcegepant to be used when examining additive effect. Acute responses to GTN provocation and olcegepant treatment are shown in Figure 2(b). Overall, mixed-effect analysis showed a significant effect of the treatment groups (F(4,53) = 16.11, p < 0.0001). Subsequent post hoc analysis showed that no groups were different at baseline on day 1, and 2 hours after administration GTN induced a significant allodynic response compared to vehicle throughout the protocol (p < 0.0001 and p = 0.0002 on day 7). Olcegepant 0.25 mg/kg did not significantly inhibit GTN provocation on any days. Olcegepant 0.50 mg/kg and 1.0 mg/kg had significant effects on GTN provocation, although the effect size decreased during the paradigm. The dose 0.50 mg/kg was chosen for further additive studies, as it was effective without completely inhibiting the allodynia induced by GTN.
No additive effect of the olcegepant-sumatriptan combination
For the additive study, four control groups and two combination groups were chosen based on results shown in Figures 2(a) and 2(b). Control groups were GTN (positive control), vehicle (negative control), olcegepant 0.50 mg/kg, and sumatriptan 0.6 mg/kg (reference controls). The two combination groups were olcegepant 0.50 mg/kg + sumatriptan 0.1 mg/kg (combination 1) and olcegepant 0.50 mg/kg + sumatriptan 0.6 mg/kg (combination 2). Overall, mixed-effect analysis indicated a significant effect of both reference and combination groups (F(5,55) = 7.95, p < 0.0001). Subsequent post hoc analyses showed that no groups were different at baseline on day 1. Two hours after administration, GTN induced a significant allodynia compared to vehicle on every test day (p < 0.0001). For better resolution, reference groups and combination groups have been split into two separate figures plotted with the same control groups (Figure 3(a) and 3(b), respectively). Olcegepant 0.50 mg/kg and sumatriptan 0.6 mg/kg significantly attenuated the GTN-induced allodynia on days 1 and 3 and again the effect size decreased from here (Figure 3(a)). Both combination 1 and 2 significantly attenuated the GTN-induced allodynia on days 1 and 9. On days 3 and 7, only combination 1 had a significant effect and on day 5 both were trending (p = 0.09 and p = 0.08 for combinations 1 and 2, respectively, see Figure 3(b)). However, when comparing the two combination groups to either the olcegepant or sumatriptan reference group, no significant difference was found on any days (p > 0.5 on all days). These data strongly suggest that the combination treatments did not have a greater effect on the GTN-induced allodynia compared to the separate treatments.

Glyceryl trinitrate (GTN)-induced allodynia treated with repeated injections. Fifty percent withdrawal thresholds measured 2 h after GTN (10 mg/kg, IP) or vehicle administration followed by injections of either single or combination treatments or vehicle on 5 test days (1, 3, 5, 7, and 9). (a) Reference groups olcegepant 0.50 mg/kg and sumatriptan 0.6 mg/kg compared to control groups GTN and vehicle. Vehicle group was significantly different from GTN group on all days (p < 0.0001, #). (b) Combination groups 1 and 2 compared to control group GTN and vehicle. Please note that control groups are the same on both graph 3(a) and 3(b). Vehicle group was significantly different from GTN group on all days (p < 0.0001, #). Raw data was square root transformed and are represented as means ± SEMs.
Single and combination treatments yield an overall treatment effect of 73–81%
In order to better visualize the overall effects of the treatments and compare the combination therapies to single-drug treatments, AUCs for each animal were calculated and means were normalized to the mean vehicle AUC (Figure 4). All groups were significantly different from the GTN group (F(5,53) = 9.02, p < 0.0001): vehicle, p < 0.0001), olcegepant 0.50 mg/kg, p = 0.01; sumatriptan 0.6 mg/kg, p = 0.008; combination 1, p = 0.0009; and combination 2, p = 0.02. However, no difference was found between the two reference groups and the two combination groups, as all four of them yielded an effect size between 73–81%, showing no improved effect.
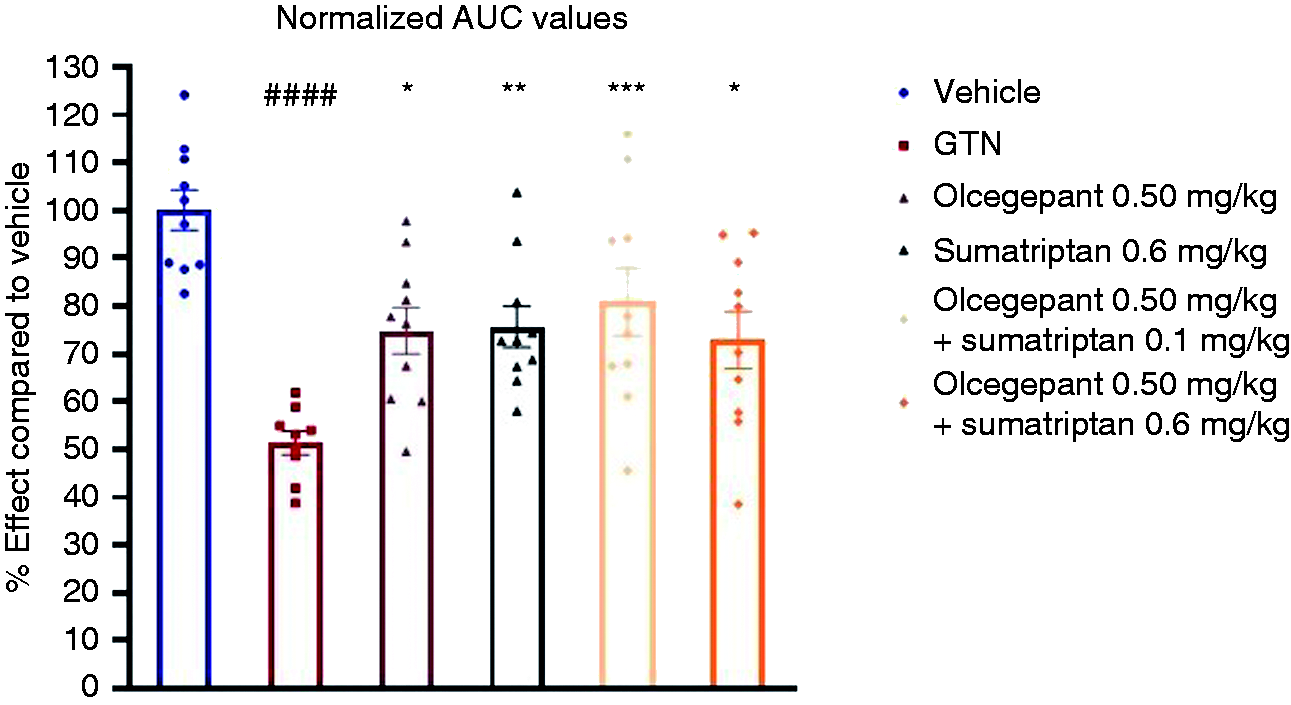
Overall AUC of each treatment based on means of individual subject AUC. Values were normalized to mean vehicle AUC and are shown in % effect. All treatment groups were significantly different from the glyceryl trinitrate (GTN) control group. The GTN group was significantly different from the vehicle group. The two reference groups (olcegepant 0.50 mg/kg and sumatriptan 0.6 mg/kg) were not different from the two combination groups. Overall withdrawal threshold of the GTN group was 51.4% compared to the vehicle group. Olcegepant 0.50 mg/kg was 74.9%, sumatriptan 0.6 mg/kg was 75.7%, combination 1 was 80.9% and combination 2 was 72.9%. AUCs are represented with individual values and means ± SEMs.
Combination treatment after end of paradigm was ineffective
It was clear from our dose-response experiments that the anti-nociceptive effect of sumatriptan and olcegepant gradually decreased as the repeated test paradigm proceeded. After the regular 9-day paradigm in the GTN model, we therefore added an extra test day with add-on treatment for groups receiving low or middle dose of either sumatriptan or olcegepant. This was to investigate whether a rescue of the decreased anti-nociceptive effect was possible by combining with an alternative drug.
Acute response to 1.0 mg/kg olcegepant added in the repeated sumatriptan study is shown in Figure 5(a). Overall, mixed-effect analysis suggested a significant effect of treatment groups (F(4,106) = 8.59, p < 0.0001). Post hoc analyses showed continued allodynia in the GTN group compared to vehicle on the same day (p = 0.0012). Adding 1.0 mg/kg olcegepant to 0.1 mg/kg sumatriptan did not have any significant effect on day 11. Adding 1.0 mg/kg olcegepant to 0.3 mg/kg sumatriptan resulted in significant inhibition of the GTN-induced allodynia on day 11 but no more than on day 9 when mice were treated only with sumatriptan. Mice receiving 0.6 mg/kg sumatriptan continued to be no different from the GTN control group.

Acute add-on treatment of drug B on glyceryl trinitrate (GTN)-induced allodynia treated with repeated injections of drug A. Fifty percent withdrawal thresholds were measured 2 h after GTN (10 mg/kg, IP) or vehicle and subsequent single (drug A) or combination (drug A + B) administration. (a) 0.1 mg/kg and 0.3 mg/kg sumatriptan-treated groups acutely received 1.0 mg/kg olcegepant while 0.6 mg/kg sumatriptan was kept as control. (b) 0.25 mg/kg and 0.50 mg/kg olcegepant-treated groups acutely received 0.6 mg/kg sumatriptan while 1.0 mg/kg olcegepant was kept as control. Please note that day 9 represents the same values as shown in Figure 2. Raw data were square root transformed (SQRT) and are represented as means + SEMs.
Acute response to 0.6 mg/kg sumatriptan added in the olcegepant dose-response study is shown in Figure 5(b). Overall, mixed-effect analysis suggested a significant effect of treatment groups (F(4,80) = 19.16, p < 0.0001). Post hoc analysis showed continued allodynia in the GTN group compared to vehicle on the same day (p < 0.0001 on day 11). Adding 0.6 mg/kg sumatriptan to 0.25 mg/kg olcegepant did not significantly attenuate the GTN-induced allodynia on day 11, albeit a small effect was seen (p = 0.1). Adding 0.6 mg/kg sumatriptan to 0.50 mg/kg olcegepant significantly attenuated the GTN-induced allodynia on day 11 but not more than on day 9, when mice were treated with olcegepant only. Mice receiving 1.0 mg/kg olcegepant continued to be no different from the GTN control group. Collectively, these data suggest adding either sumatriptan or olcegepant after a regular paradigm could not repress the decreasing effect of the drugs and improve the inhibition of the GTN-induced allodynia.
Discussion
In this validated mouse model of migraine, there is no increased effect of combining olcegepant and sumatriptan either at high or at low doses compared to single drug treatments. Both olcegepant and sumatriptan reduced GTN-induced allodynia when given alone but had no additive effect when combined. Moreover, no dose of either drug or combinations yielded complete inhibition of the GTN-induced allodynia as previously seen with the CGRP antibody ALD405 (27).
The GTN mouse model of migraine is valid and translational
The GTN mouse model is well-validated in terms of back-predictive validity (26–30) and direct validity to its human counterpart (32,37). GTN causes a general hyperexcitation of central nociceptive circuits that subsequently contributes to a central sensitization of the nociceptive system. This central sensitization of the nociceptive system is directly evident in both rodent models and migraine patients as cutaneous allodynia (27,28,38). Previous studies have shown that intrathecal and intracerebroventricular administration of sumatriptan, olcegepant, and a CGRP antibody do no inhibit GTN-induced allodynia (28,39). This indicates that downstream GTN-induced mechanisms are indeed peripheral despite a possibly central initiation. We measured GTN-induced allodynia in the hind paw of the mouse, which in previous studies was more accurate but otherwise gave similar results as testing in the periorbital region ((40–42), unpublished data by the authors). The model responds to sumatriptan and olcegepant (27,28), drugs specific to migraine and cluster headache and without general analgesic activity (43–45). Using cutaneous allodynia as a surrogate marker for the activation of the nociceptive system and central sensitization is a validated way to examine the effects of migraine-specific treatments and to further understand basic mechanisms involved in migraine (28). However, an animal model has its limitations (please see below).
Possible shared pathway of sumatriptan and olcegepant?
We hypothesized that having two migraine-specific drugs with different mechanisms of action would result in an additive effect. Sumatriptan has four proposed mechanisms of action: i) inhibition of transduction in the trigeminal nucleus caudalis (46–48), ii) vasoconstriction and inhibition of vasodilation during migraine attacks (49), iii) inhibition of trigeminal afferent nerve activity (50) and iv) inhibition of CGRP release via 5-HT1D/1F receptor activation (46,51,52). However, sumatriptan does not antagonize already established CGRP-induced effects as seen in a human study by Falkenberg et al. (53). As mentioned in the introduction, neither triptans nor gepants yield complete pain freedom in all patients. Taken together, we therefore hypothesized that established CGRP-induced effects could have been treated by adding olcegepant, a CGRP-receptor antagonist, even when using a high or established dose of sumatriptan. Yet we did not observe any additive effect in our study when we combined sumatriptan and olcegepant (see Figure 3(b)). Sumatriptan inhibits CGRP-release and olcegepant blocks the effect of CGRP, which could represent a common pathway. The mechanisms of the drugs may also collide further down the migraine pain cascade. Thus, activation of the 5-HT1B/1D/1F receptors, via G-protein, inhibits the function of cyclic AMP (cAMP). Olcegepant blocks the effect of CGRP which normally, via G-protein, stimulates the function of cAMP. In other words, a shared effect of triptans and of olcegepant is that both reduce the amount of cAMP. Our results suggest that this common pathway is important and may explain the lack of additive effect in our model and possibly also in human migraine.
Clinical relevance
Our study is relevant to clinicians who now have the option to combine marketed triptans and gepants. Despite being validated and translational, the mouse model is of course not identical to migraine. Therefore, combination therapy should still be studied in other preclinical models to add evidence to whether human clinical trials should be pursued. However, we suggest that this particular combination should not be given the highest priority. Based on our data, we do not believe patients will benefit from the specific combination and it should not be used before additional preclinical as well as proper clinical trials show a positive effect. In future, it would be relevant to examine whether the effect of treatment could be increased by combining a centrally acting drug with a peripherally acting drug. Such combinations are already used in other, very different, aspects of pain such as in post-surgical pain management (54–56) and chronic pain (57). We suggest testing the combination of a gepant and lasmiditan, a selective 5-HT1F agonist of the ditan-class that crosses the blood-brain-barrier (58). The combination of an NSAID and a gepant should also be studied and is more likely to show benefit than the combination of triptans and gepants and may show a bigger effect than the slight additive effect of triptans and NSAIDs (24,25).
Based on our study, we could not rescue a decreasing anti-nociceptive effect by acutely adding either olcegepant or sumatriptan to already established paradigms (Figures 5(a) and 5(b)). Yet, it is important to note our results are based on acute treatments and do no examine the effects of prophylactic treatment. Important questions regarding prophylactic treatments need to be addressed in future studies. Antibodies targeting either CGRP or the CGRP receptor are currently being used prophylactically. Possible rescue or additivity with acute agents like sumatriptan is relevant to examine, especially in the periods with low levels of antibodies in the body before the next injection. This may also be relevant with prophylactic gepants like atogepant (19).
Strengths and limitations of the study
Major strengths of the present study were that our mouse model is well validated and that drug doses were chosen based on our own dose-response studies. The applied mouse model is excellent at differentiating between no, medium and high effect, but cannot easily distinguish small differences. That would require extremely large group sizes, which would not be feasible. Additionally, using these protocols we have not measured drug concentrations in the animals and the risk of having reached Emax with the higher doses of the drugs is therefore present. Additional pharmacodynamic experiments would be necessary to examine this. A further limitation is the use of only male mice. Previous experiments with the model have shown female mice to be slightly more sensitive and more variable in their response but they have not observed any sex differences in drug responses (28). Our conclusion would likely not have changed, had we had both sexes represented. Moreover, an overall limitation in our model is the use of a singular endpoint (cutaneous allodynia as a nociceptive marker) to measure a complicated drug response and disorder. To confirm lack of additive effect, studies with other measuring endpoints would be fitting. Lastly, a general limitation is the use of a rodent model to research a complex, human disorder. The degree to which the results can be translated into human migraine is uncertain. It should be noted that species differences in 5-HT1 receptor subtypes have been described including agonist-induced inhibition of trigeminal CGRP release (59). While the inhibition in rodents seem to be through 5-HT1B activation, 5-HT1D receptors are activated in humans (60). Thus, it has been suggested that the rodent 5-HT1B receptor is complemental to the 5-HT1D subtype in other species including humans (61). Despite these limitations, animal models have led to numerous translational successes and changes in pain research and treatment (62).
Conclusion
There is still a need for migraine treatments with higher clinical efficacy. As sumatriptan and olcegepant target different receptors, it was a straightforward option to combine them. However, no additive effect was seen in this animal model which makes clinical studies less attractive until additional preclinical studies have been made.
Article highlights
Both olcegepant and sumatriptan significantly attenuated GTN-induced allodynia in a validated mouse model of migraine. Combining olcegepant and sumatriptan did not have an additive effect compared to single-drug treatment in mice. Triptan-gepant combinations should not be used in the clinic unless relevant trials in patients, against expectations, show efficacy.
Footnotes
Author contributions
CE, SLC and DMK designed the study. CE performed all experiments and data analysis. The manuscript was written by CE with support from SLC, DMK, and JO. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was provided by the Candys Foundation.
