Abstract
Background
This study is designed to evaluate whether cilostazol can trigger migraine attacks in men diagnosed with migraine without aura. Previous research has shown that cilostazol induces migraine in women, but its impact in men has not been established.
Methods
A total of 14 men and 15 women with migraine without aura will be recruited. Male participants will undergo a randomized, placebo-controlled crossover trial, whereas female participants will take part in an open-label study. For 12 h following administration, participants will be assessed for headache occurrence, related symptoms, and migraine-like episodes. The main outcomes include comparing the frequency of migraine-like attacks in men receiving cilostazol versus placebo and between cilostazol-treated men and women. Secondary outcomes include headache incidence, measurements of heart rate and mean arterial pressure and documentation of any adverse events.
Conclusion
The results are expected to enhance understanding of sex-specific mechanisms involved in migraine, which could inform more individualized diagnostic and therapeutic strategies.
This is a visual representation of the abstract.
Introduction
Migraine is a prevalent and disabling neurological disorder with a marked predominance in women after puberty. Despite this sex difference in prevalence and severity, the underlying biological mechanisms remain incompletely understood. 1 Among the various intracellular pathways implicated in migraine pathophysiology, cyclic adenosine monophosphate (cAMP) has emerged as a particularly relevant mediator. Cilostazol, a selective phosphodiesterase 3 (PDE3) inhibitor that elevates intracellular cAMP levels, has been shown to reliably provoke migraine attacks in individuals with a history of migraine.2–6 Prior studies have primarily involved women, leaving uncertainty about whether men exhibit a similar susceptibility to cilostazol-induced migraine (Table 1). The present study protocol was designed to address this knowledge gap by systematically evaluating the headache- and migraine-inducing effects of cilostazol in both men and women diagnosed with migraine without aura. To this end, we will conduct a randomized, placebo-controlled, double-blind crossover trial in men, alongside an open-label arm in women. This design enables the assessment of both within-subject effects of cilostazol compared to placebo in men and between-group comparisons of responses in men and women. The study aims to improve our understanding of sex-specific aspects of migraine mechanisms and contribute to the development of more individualized diagnostic and therapeutic approaches.
Prior studies evaluating the migraine-inducing effects of oral cilostazol (200 mg) in individuals with migraine without aura.

Methods
We will carry out a randomized, double-blind, placebo-controlled crossover trial to investigate the involvement of intracellular cAMP in triggering migraine attacks in 14 men diagnosed with migraine without aura (Figure 1). The study will be conducted at the trial unit of the department of Clinical Pharmacology, Pharmacy and Environmental Medicine (University of Southern Denmark), with experimental sessions expected to take place between October 2025 and June 2026. The protocol includes an initial screening session lasting approximately 1 hour, followed by two experimental sessions of around 2 hours each, resulting in a total participation time of about 5 hours. A washout interval of at least 7 days will separate the two experimental administrations. This approach closely follows the design of earlier provocation studies conducted in women with migraine without aura.2–6 Implementing blinding is critical to minimize bias and preserve the validity of the data, even when participants develop headache or migraine symptoms. This strategy will enable precise attribution of any observed effects to either cilostazol or placebo, ensuring the generation of reliable and objective evidence. In parallel, we will also conduct an open-label study enrolling 15 women with migraine without aura to examine the effects of cilostazol in migraine induction (Figure 2). As previous placebo-controlled trials and additional investigations have already demonstrated cilostazol's capacity to provoke migraine in women,2–6 repeating placebo-controlled procedures in this group would represent an unnecessary use of resources. The open-label design instead allows us to gather current comparative data in women without relying solely on historical controls.

Design of the study in men with migraine.
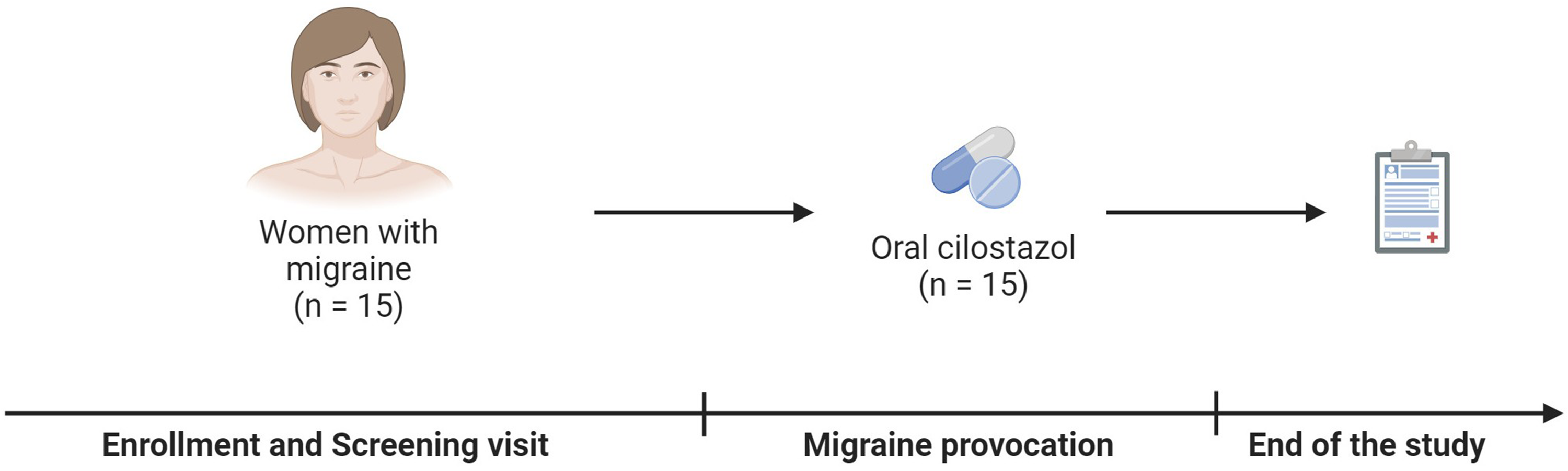
Design of the study in women with migraine.
Ethics
Migraine attacks induced in this experimental setting are self-limited, responsive to treatment, and do not result in any permanent damage, which has made this model ethically acceptable and extensively employed over the past decades. 7 Participants will be advised to take an analgesic, such as ibuprofen or sumatriptan, even if only mild headache symptoms occur, without affecting the primary aims of the study. Participants will receive a small financial compensation as well as reimbursement for travel expenses. The protocol has received approval from the appropriate ethics committee, in compliance with the Declaration of Helsinki and all relevant local regulations. The study has been registered in the European Union Clinical Trial Information System (CTIS) under the identifier 2025-521520-29-00.
Study participants
Eligible participants will be men and women between 18 and 65 years of age who meet the diagnostic criteria for migraine without aura as defined by the International Classification of Headache Disorders. 8 Although no formal cut-off for migraine frequency was specified in the study protocol, recruitment will primarily target individuals with episodic migraine and relatively low attack frequency, consistent with previous provocation studies. Recruitment will occur through the Department of Neurology at Odense University Hospital, private neurology practices, general practitioners, online recruitment platforms, and advertisements distributed via patient organizations, including Migræne Danmark, Danmarks Patientforening for Hovedpineramte, and Migræne og Hovedpineforeningen. To qualify, individuals must have a body weight ranging from 60 to 100 kilograms. Female participants will be required to maintain effective contraception for the duration of the study. A urine pregnancy test will be administered both at the screening visit and immediately prior to the experimental session to ensure non-pregnancy. Exclusion criteria encompass the presence of any additional headache disorder. The only exception is infrequent tension-type headache (≤3 days per month), which is permitted. Further exclusion criteria include onset of headache within 24 h before the experimental visit, recent intake of coffee, cola, or alcohol within the last 8 h, abnormal blood pressure readings, or the use of analgesics in the previous 48 h. Use of certain medications, such as anticoagulants, antiaggregants and drugs that inhibit CYP3A4 and CYP2C19 activity will also preclude participation. Additional exclusions include cardiovascular disease, cardiac arrhythmia, unstable angina pectoris, impaired liver function and/or kidney function, a recent history of myocardial infarction and/or major cardiac surgery, psychiatric conditions, substance misuse, history of smoking or other clinically significant health concerns. Individuals with known hypersensitivity to cilostazol, as well as those employed by the clinical trial site or their immediate relatives, will likewise be excluded. Inclusion and exclusion criteria of the study are reported in the Supplementary Material.
Study treatments
The intervention in this trial involves administering cilostazol “Aliud” at a dose of 200 mg. Male participants with migraine will receive two capsules containing either cilostazol (a single tablet of 100 mg) or placebo (lactose monohydrate), designed to be identical in appearance to maintain blinding. Female participants will take two cilostazol tablets openly, without blinding. Preparation, labeling, and packaging of all study medications will be carried out by Region Hovedstadens Apotek. The cilostazol dosage is standardized across all participants and matches the regimen previously employed to assess cilostazol's migraine-inducing effects in women with migraine without aura.2–6
Screening
Before any study-related activities take place, all participants will receive both verbal and written information about the study and will be asked to provide written informed consent, in accordance with the Declaration of Helsinki. During the screening visit, a medical investigator will review each participant's medical history to confirm eligibility, including a verification of the migraine diagnosis. A comprehensive physical examination, including a neurological assessment, will be performed by a clinician experienced in headache disorders. Investigators will also evaluate any findings that may constitute reasons for exclusion. Participants will be reminded that they are free to withdraw consent at any point during the study without providing a reason, and that doing so will not impact their future medical care.
Randomization
Randomization for male participants will take place on the first experimental day, once eligibility has been confirmed. The allocation sequence will be computer-generated by staff independent of study procedures and concealed from the research team. Sequentially numbered, opaque, sealed envelopes will hold the randomization assignments for each participant. Eligible men will be randomly assigned to receive either cilostazol or placebo in the first session, followed by the alternate treatment in the second session. The envelopes will be securely stored and accessible only for emergency unblinding. Both participants and investigators will remain blinded to treatment allocation, and the double-blind conditions will be maintained throughout the study period.
Study procedures
As in prior provocation studies, participants will complete headache diaries to document headache onset, characteristics, and associated symptoms for 12 h following administration. Measurements of heart rate, blood pressure, and any adverse events will be systematically recorded. Headache intensity will be rated using a numerical rating scale (NRS) from 0 to 10, where 0 represents no pain and 10 indicates the worst pain imaginable. The definition of a migraine-like attack follows published criteria 9 : attacks must meet at least one of two categories. The first category requires headache with at least two of these features: unilateral location, pulsating quality, moderate to severe intensity, or worsening with routine activity. In addition, at least one symptom such as nausea, vomiting, or sensitivity to light and sound should be documented. The second category describes headaches that resemble the participant's typical migraine attacks and lead to the use of rescue medication. After cilostazol administration, participants will remain resting in bed or seated in a chair during a 90-min observation period. If they feel well enough, they may go home afterward. Before discharge, they will receive a headache questionnaire to record pain characteristics, symptoms, use of analgesics, and any side effects during the subsequent 12 h. The following day, an investigator blinded to the treatment allocation will collect this information by telephone or email. A detailed overview of all assessments performed at each study visit is provided in the Supplementary Material.
Concomitant medications
The use of acute migraine treatments is allowed throughout the study, except during the 48 h preceding each experimental session. Should participants experience headache within the initial 90 min after administration, they will be offered a single dose of ibuprofen (400 mg) or sumatriptan (100 mg). If headache or migraine-like symptoms occur after participants have left the department, they may take their usual home medications as needed. Concomitant chronic therapies, including preventive migraine treatments, will be permitted as long as they do not involve anticoagulants, antiaggregants and drugs that inhibit CYP3A4 and CYP2C19 activity. To be considered stable, any concomitant treatments must not have undergone dosage adjustments within 14 days prior to the screening visit and must remain unchanged throughout the entire duration of the study.
Endpoints
The primary endpoints are (1) the difference in the incidence of migraine-like attacks between men receiving cilostazol and those receiving placebo, and (2) the difference in the incidence of migraine-like attacks between cilostazol-treated men and cilostazol-treated women with migraine without aura. Secondary endpoints include (1) the difference in headache incidence between cilostazol- and placebo-treated men, (2) changes in heart rate and mean arterial pressure (MAP) up to 90 min after administration between cilostazol and placebo conditions in men, and (3) the difference in the incidence of adverse events recorded during the 12-h observation period following administration between cilostazol- and placebo-treated men.
Sample size
Drawing on prior research involving women with episodic migraine without aura, we estimated that approximately 72% of male participants would experience a migraine attack following cilostazol administration, compared to 10% after placebo.2–6 We set the significance level at 5% and accepted a type II error rate of 20%, yielding 80% statistical power. Using the asymptotic approximation for McNemar's test, the required sample size was calculated to be 14 men. 10 Additionally, we plan to recruit 15 women with migraine without aura in an open-label design to collect comparative data without repeating previously conducted trials.2–6
Missing data
Our objective is to include 14 men completing the placebo-controlled crossover study and 15 women completing the open-label arm. Participants who fail to complete all study procedures will be excluded from the final analysis set. Based on dropout rates observed in similar protocols, we expect minimal attrition, with negligible impact on primary and secondary endpoint analyses.2–6 Therefore, no imputation techniques for handling missing data will be applied.
Statistical analysis
Baseline characteristics such as demographics, vital signs (blood pressure and heart rate), headache features (location, intensity, quality, aggravation by neck movement), and associated symptoms (photophobia, phonophobia, nausea, vomiting) will be presented as means and standard deviations or medians and interquartile ranges for continuous variables, and as counts and percentages for categorical variables. Normality of distribution will be assessed with the Shapiro–Wilk test. Continuous variables with normal distribution will be summarized as means and standard deviations, while non-normally distributed data will be presented as medians and interquartile ranges.
The primary binary endpoints will be analyzed using McNemar's test. Similarly, secondary binary outcomes such as the incidence of headache and adverse events will be assessed with McNemar's test. For the secondary endpoint examining heart rate and MAP, the area under the curve (AUC) will be calculated from baseline to 90 min post-dosing, applying the trapezium rule. 11 AUC values will be compared between cilostazol and placebo using either a paired t-test or the Wilcoxon matched-pairs signed-rank test, depending on the data distribution. All p-values will be reported as two-tailed, with statistical significance set at 5%.
Conclusion
Migraine is a highly prevalent neurological disorder that affects women disproportionately, partly due to hormonal influences such as estrogen and prolactin, which are known to sensitize trigeminal neurons and contribute to female-specific mechanisms of migraine. 12 In contrast, the biological underpinnings of migraine in men remain comparatively unexplored. Prior research has demonstrated that women with migraine exhibit pronounced sensitivity to agents like cilostazol, which elevate intracellular cAMP levels in both vascular smooth muscle cells and neurons.13–15 Immunohistochemical studies have shown that PDE3 and calcitonin gene-related peptide (CGRP) are co-localized within the trigeminal ganglion, suggesting that PDE3 inhibition may modulate CGRP-dependent signaling pathways. 16 We hypothesize that the lower susceptibility of men to cilostazol-induced migraine is partly due to the reduced influence of hormones such as estrogen and prolactin, resulting in fewer migraine attacks under comparable experimental conditions. This observation may point to fundamental differences in migraine pathophysiology between sexes, with potential implications for developing more personalized treatment approaches. The importance of studying these sex-related differences has been underscored by the migraine research community, which has identified the underrepresentation of men in clinical studies as a critical limitation. 17 This imbalance restricts the generalizability of current findings and leaves men underserved by evidence-based therapies. Addressing these gaps is essential to advance our understanding of migraine mechanisms and to ensure that treatment strategies are effective across sexes. Together with ongoing research, including a provocation trial investigating the role of sildenafil in men with migraine, 18 this study will contribute valuable insights into potential sex-specific differences in cAMP- and cGMP-mediated pathways, ultimately informing the development of targeted therapies for all individuals living with migraine.
Clinical implications
This research may uncover sex-related variations in the migraine-provoking effects of cilostazol, paving the way for more personalized treatment strategies tailored to men and women.
By elucidating the contribution of cAMP signaling in men affected by migraine, the study will support the development of targeted, mechanism-driven therapeutic approaches.
Supplemental Material
sj-docx-1-rep-10.1177_25158163251388611 - Supplemental material for Migraine-inducing effects of cilostazol in men with migraine without aura: Protocol for a randomized, placebo-controlled crossover trial
Supplemental material, sj-docx-1-rep-10.1177_25158163251388611 for Migraine-inducing effects of cilostazol in men with migraine without aura: Protocol for a randomized, placebo-controlled crossover trial by Sarmad Al-Wakeel, Haya Sanjikdar, Ali Al-Tamimi and Lanfranco Pellesi in Cephalalgia Reports
Footnotes
Acknowledgments
None.
Ethical considerations
The protocol has received approval from the appropriate ethics committee, in compliance with the Declaration of Helsinki and all relevant local regulations.
Consent to participate
Not applicable.
Consent for publishing
Yes.
Author contributions
LP conceived and supervised the preparation of study protocol. SAW, HS, and AAT contributed to the development of the study protocol. All authors reviewed and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received funding from Oda og Hans Svenningsens Fond, which did not participate in the conduct of the study.
Declaration of conflicting interests
Lanfranco Pellesi serves on the editorial boards of The Journal of Headache and Pain, BMC Neurology, European Journal of Medical Research, Pain Research and Management, and Brain Conflux, and is Editor-in-Training at Clinical and Translational Science. The rest of the authors declare they have no conflict of interest.
Data availability statement
Not applicable.
Open practices
Not applicable.
Trial registration
The trial is registered in the Clinical Trial Information System (CTIS) of the European Union under the number 2025-521520-29-00.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
