Abstract
Background
This study aims to assess the migraine-inducing effects of sildenafil in men with migraine without aura. While sildenafil has been shown to provoke migraine in women, its effect on men remains unknown.
Methods
The trial will enroll 12 men and 15 women, all diagnosed with migraine without aura. The men will participate in a randomized, placebo-controlled crossover trial and the women in an open-label study. Participants will be monitored for migraine attacks and associated symptoms for 12 h post-administration. The primary endpoints include the incidence of migraine attacks in sildenafil-treated men compared to placebo and between sildenafil-treated men and women. Secondary endpoints are headache incidence, heart rate, mean arterial pressure, and adverse events.
Conclusion
The findings will contribute to a better understanding of sex-related differences in migraine mechanisms, potentially leading to more tailored treatment approaches.
Introduction
Migraine exhibits a marked disparity between sexes, with women experiencing migraine at rates two to three times higher than men after puberty. Women's migraine attacks are not only more frequent but also tend to be longer and more debilitating, with reduced responsiveness to treatment compared to their male counterparts. 1 Despite the influence of various factors such as sex hormones, brain structure, genetic variations, life events, stress, and neuronal activity, the precise mechanisms underlying these sex-related differences in migraine remain elusive. Human provocation models, which involve experimentally inducing migraine attacks through specific triggers, offer valuable insights into these mechanisms. 2 One such trigger is sildenafil (Viagra®), a selective phosphodiesterase 5 (PDE5) inhibitor that raises intracellular levels of cyclic guanosine monophosphate (cGMP) by preventing its breakdown. Although sildenafil is primarily prescribed for erectile dysfunction, it has been shown to induce migraine attacks in individuals with migraine without aura. In previous studies, sildenafil triggered migraine in 83%–89% of study participants.3,4 In adults with migraine pretreated with a single subcutaneous injection of erenumab (140 mg), 10 out of 16 participants (63%) experienced a migraine attack after sildenafil administration. 5 However, these studies have predominantly involved women, leaving a gap in our understanding of sildenafil's effects in men with migraine without aura (Table 1). This trial seeks to fill this gap by conducting a randomized, double-blind, placebo-controlled crossover study to assess the migraine-inducing effects of sildenafil in men with migraine without aura. Additionally, the study will re-evaluate sildenafiĺs effects in women to explore potential sex-related differences. The results will yield new insights into the sex-specific mechanisms of migraine, potentially leading to more personalized and effective treatment strategies.
Prior studies evaluating the migraine-inducing effects of sildenafil in individuals with migraine without aura.

Methods
We will conduct a randomised, double-blind, placebo-controlled, crossover trial designed to evaluate the role of intracellular cGMP in the initiation of migraine in 12 men with migraine without aura (Figure 1). The study includes a screening visit (1 h) and two experimental days (about 2 h each), for a total duration of approximately 5 h. There will be a washout period of at least 7 days between the two provocations. The study design is similar to previous provocation studies conducted in women with migraine without aura.3,4 Blinding is essential to eliminate bias and ensure the integrity of our results, even if participants experience headache or migraine. By maintaining blinding, we will accurately attribute any observed effects to sildenafil or placebo, thus obtaining the most objective evidence possible. In accordance with previous provocation models of migraine, this methodological rigor is crucial for drawing valid conclusions about the headache-inducing properties of sildenafil. Moreover, we will conduct an open-label study to evaluate the role of intracellular cGMP in the initiation of migraine in 15 women with migraine without aura (Figure 2). The study includes a screening visit (1 h) and a single experimental day (about 2 h each), for a total duration of approximately 3 h. The role of sildenafil in inducing migraine in women has been confirmed through more than one placebo-controlled study.3,4 Therefore, replicating those studies is unnecessary. The open-label study is designed to collect comparative data in women with migraine avoiding reliance on historical cohorts.
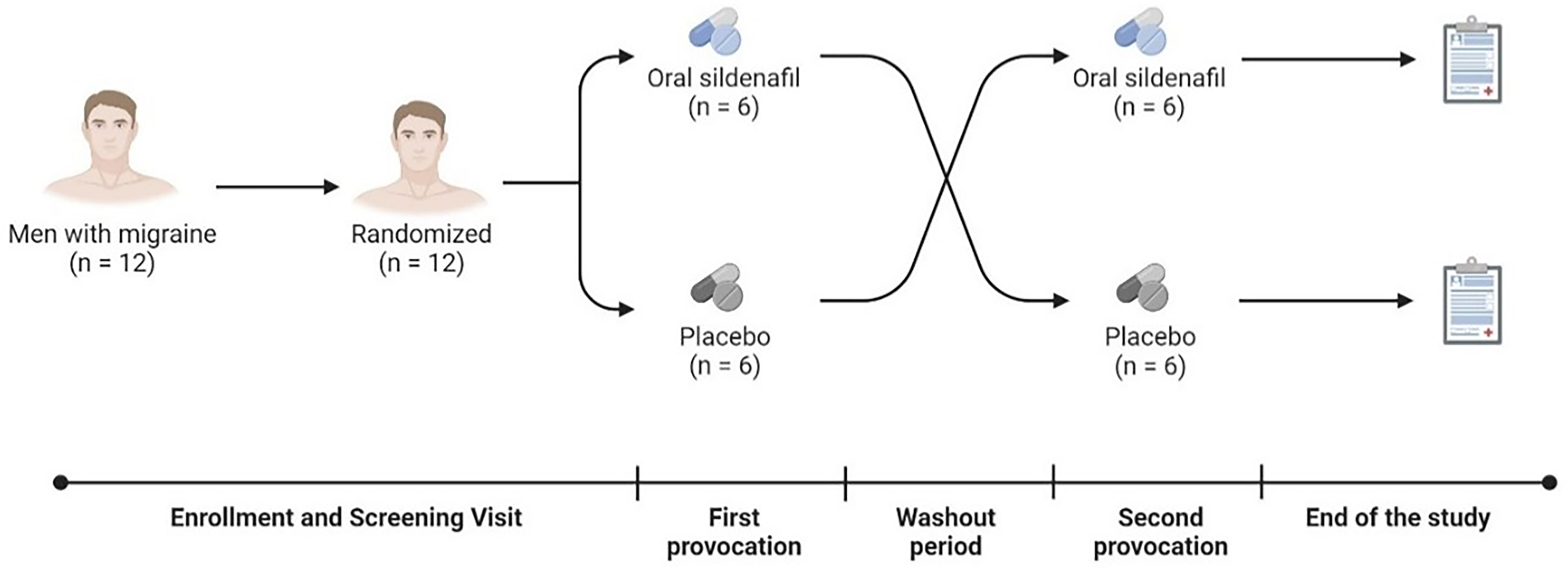
Design of the study in men with migraine.

Design of the study in women with migraine.
Ethics
Provoked migraine attacks are self-limiting, treatable, and do not cause lasting harm, making the model ethically acceptable and widely used for 30 years. 6 Participants will be invited to take an analgesic (ibuprofen or sumatriptan) in the presence of a mild headache, without compromising the objective of the study. The trial has been approved by the relevant ethical committee in accordance with the Declaration of Helsinki and all applicable local regulations. The trial is registered in the Clinical Trial Information System (CTIS) of the European Union under the number 2024-512014-17-02.
Study participants
Participants will be men and women aged 18–50 years, diagnosed with migraine without aura per the International Classification of Headache Disorders. 7 Patients will be recruited through the Department of Neurology at the Odense University Hospital, private clinics, general practitioners, recruitment websites and advertisements to patient organizations (Migræne Danmark, Danmarks Patientforening for Hovedpineramte and Migræne og Hovedpineforeningen). Eligible participants must have experienced 1–6 migraine attacks in the previous eight weeks and weight from 60 to 90 kg. Women are required to use safe contraception throughout the study. A urinary pregnancy test will be conducted during the screening visit and again immediately before the experimental day to confirm non-pregnancy status. Exclusion criteria include any other type of headache (except ≤3 days/month of tension-type headache), migraine less than 24 h before the experiment, recent consumption of coffee, cola, or alcohol (within 12 h), blood pressure abnormalities, recent use of analgesics (within 48 h), and certain medications (e.g., α-blockers, ritonavir, CYP3A4 inhibitors). Participants with cardiovascular or liver diseases, mental illness, substance abuse, smoking history, or other significant health issues are excluded, as are those with known allergies to sildenafil or participants who are either members of the clinical trial site staff or relatives of the site staff.
Study treatments
The treatment utilized in this study consists of 1 tablet (100 mg) of sildenafil “Orion”. Men with migraine will receive a capsule containing a tablet of sildenafil “Orion” that will be indistinguishable from placebo (glucose monohydrate). Women with migraine will receive a tablet of sildenafil “Orion” in an unblind manner. Supply, labeling and packaging of the study medicines will be performed by Glostrup Apoteket for this purpose. The dosage of sildenafil is fixed for all subjects and is identical to that previously used to investigate the headache-inducing properties of sildenafil in women with migraine without aura.3,4
Screening
All study participants will be given oral and written information, and they will give their written consent according to the Declarations of Helsinki prior to any study procedures. At the Screening Visit, a review of medical history will be performed to confirm subject eligibility, including a reassessment of the migraine diagnosis. A complete physical examination per standard of care, including a neurological exam, will be conducted by a medical examiner trained in headache disorders. Investigators will also check for any findings that would constitute grounds for study exclusion. All participants are informed that they, at any time throughout the study, can withdraw their consent without justification and it will not affect future treatment.
Randomization
Randomization will occur in men with migraine at the beginning of the first experimental day, after the eligibility assessment for study procedures. The randomization will be conducted according to a computer-generated schedule produced by staff not involved in experimental procedures and concealed from the research team. The randomization sequence will be masked in sequentially numbered, opaque sealed envelopes for each participant. Eligible subjects will be randomized to receive either sildenafil or placebo first and then they will be crossed over to receive the opposite intervention. Envelopes will be stored at a secure location and will be available to the research team only for emergency unblinding. Subjects and investigators will be blinded to the randomization assignment and double blinding will be maintained until the end of trial.
Study procedures
Consistent with previous provocation studies, headache diaries will be used to record headache occurrence, characteristics, and accompanying symptoms until 12 h post-administration. Heart rate, blood pressure, and adverse events will also be recorded during the study. Headache intensity will be measured with a numerical rating scale (NRS) from 0–10, where 0 indicates no pain and 10 represents the maximum pain imaginable. Criteria for defining a migraine-like attack in a human provocation study have been published previously. 8 Provoked migraine attacks must fulfil at least one of two categories. Category one describes headache with at least two of the following clinical features: unilateral location, pulsating quality, moderate to severe pain intensity, and aggravation or avoidance of routine physical activity. Additionally, headache must be accompanied by at least one of the following symptoms: nausea, vomiting, or photophobia and phonophobia. Category two describes headache that mimics the patient's usual migraine attacks and is treated with a rescue medication. Participants will be required to remain in bed during the 90-min monitoring period after the experimental administration. After 90 min, they may return home if they feel ready. Before leaving, each participant will receive a headache questionnaire to record headache characteristics, accompanying symptoms, use of analgesics, and any unexpected side effects for up to 12 h post-administration. The information from the questionnaire will be collected the day after through a phone call conducted by an investigator blinded for the treatment/placebo.
Concomitant medications
Abortive treatments for migraine are permitted during the study, with the exception of the 48 h preceding the experimental days. A single tablet of ibuprofen (400 mg) or sumatriptan (100 mg) will be offered to participants in case they complain of headaches during the first 90 min after the experimental administration. If the participant develops headache or migraine-like symptoms after leaving our Department, he/she is allowed to take the analgesics he/she normally assumes at home. Concomitant chronic medications including preventive treatments for migraine and excluding α-blockers, ritonavir, and drugs inhibiting the CYP3A4 enzyme, are permitted if the doses remain stable throughout the study. To qualify as “stable”, participants must not have altered any medication dosages within 14 days prior to the study entry (Screening Visit) and must maintain consistent dosages throughout the entire study period.
Endpoints
The primary endpoints are: (1) the difference in the incidence of migraine-like attacks between sildenafil- and placebo-treated men with migraine without aura and (2) the difference in the incidence of migraine-like attacks between sildenafil-treated men and women with migraine without aura. The secondary endpoint are (1) the difference in the incidence of headache between sildenafil- and placebo-treated men with migraine without aura, (2) the difference in heart rate and mean arterial pressure (MAP) until 90 min post-administration between sildenafil- and placebo-treated men with migraine without aura and (3) the difference in the incidence of adverse events during the entire observational period (until 12 h post-administration) between sildenafil- and placebo-treated men with migraine without aura.
Sample size
According to a previous study conducted in 12 women with episodic migraine without aura, we assumed that 65% of the male participants would develop migraine after sildenafil, and 5% after placebo. 3 Significance level was set at 5%, while the risk of type 2 error was set to 20% with 80% power. According to the asymptotic approximation for McNemaŕs test, we calculated a sample size of 12 male participants. 9 We will additionally enroll 15 women with migraine without aura in an open-label study to generate comparative data without the need of replicating previously placebo-controlled studies.3,4
Missing data
We aim to include 12 completers for the placebo-controlled trial and 15 participants for the open-label design. Participants who do not complete the study will be excluded from the final analysis. Based on similar studies, the anticipated drop-out rate is minimal, and missing data is expected to have a negligible impact on both primary and secondary endpoint analyses. 2 As such, no imputation methods for missing data will be applied in this study.
Statistical analysis
Baseline demographics, vital signs (blood pressure and heart rate), headache characteristics (pain localization, pain intensity, pain quality, and aggravation by neck movements) and associated symptoms (photophobia, phonophobia, nausea and vomiting) will be summarized by mean and standard deviation or median and interquartile range for continuous variables, or with number and percentages for categorical variables. According to the normality of the data assessed using the Shapiro-Wilk test, continuous variables will be summarized using mean and standard deviation for normally distributed data or median and interquartile range for non-normally distributed data. The primary endpoints are binary and will be analyzed using the McNemaŕs test. The secondary endpoints evaluating the incidence of headache and adverse events will be analyzed using the McNemaŕs test. The secondary endpoint evaluating the difference in heart rate and MAP between groups will be evaluated by calculating the area under the curve (AUC). According to the trapezium rule, 10 AUC is calculated from baseline until 90 min post-administration to obtain summary measures and to analyze differences between sildenafil and placebo. AUC data will be compared between groups using a paired two-way t-test or the Wilcoxon matched-pairs signed rank test, depending on the normal distribution of the data. P-values are reported as two-tailed with a level of significance of 5%. Statistical analysis and graphs will be performed using Prism 8.3.0 (GraphPad).
Conclusion
Migraine is a common neurological disorder that predominantly affects women, with pain resulting from the activation and sensitization of trigeminal sensory fibers. 11 Hormones such as estrogen and prolactin, which are more prevalent in women, sensitize trigeminal neurons, suggesting female-specific mechanisms in migraine. 12 However, the understanding of migraine mechanisms in men remains limited. Women with migraine are particularly sensitive to drugs like sildenafil, which elevate intracellular cGMP levels in neurons.3–5 We hypothesize that men are less affected by sildenafil due to the reduced influence of hormones like estrogen and prolactin, leading to fewer migraine attacks under similar conditions. This difference could reflect a distinct pathophysiology in men, which might have future therapeutic implications. Sildenafil is well-tolerated at a dose of 100 mg in healthy men, 13 so it is unlikely that the study participants will experience relevant side-effects. In single-dose studies conducted in healthy volunteers, doses up to 800 mg were associated with adverse reactions similar to those seen at lower doses. 14 Sildenafil has been previously administered in pregnant women, premenopausal women with a major depression disorder and women with heart failure and a reduced ejection fraction.15–17 The migraine research community has recently highlighted the need to investigate migraine mechanisms in men, as they are underrepresented in clinical studies. 18 This gap not only limits the generalizability of findings but also leaves men underserved in terms of tailored migraine treatments. Addressing these sex differences is critical for advancing our understanding of migraine and ensuring effective therapies for both men and women.
Clinical implications
The study could reveal sex-specific differences in sildenafil's migraine-inducing effects, leading to more individualized migraine treatments for men and women.
By clarifying the role of cGMP in men with migraine, the study will inform the development of more precise, mechanism-based treatments.
Footnotes
Acknowledgments
None.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The preparation and submission of the study protocol to the relevant authorities was supported by the Oda og Hans Svenningsens Fond (grant number V30).
Ethics
The trial has been approved by the relevant ethical committee in accordance with the Declaration of Helsinki and all applicable local regulations.
