Abstract
Background
Migraine attacks present a problem for patients who are unable to benefit from drug treatment. The Chordate System S020 delivers kinetic oscillation to the nasal mucosa, which is supposed to modulate the parasympathetic outflow involved in migraine pathophysiology. The objective of this clinical investigation was to evaluate the efficacy and safety of the Chordate System S020 in the treatment of acute migraine attacks.
Methods
In this randomized, placebo-controlled, double-blind, multicenter clinical investigation, participants were treated with the Chordate System S020 either in active or in placebo mode shortly after the onset of an acute, moderate to severe migraine attack. The primary endpoint was the proportion of participants whose headache pain improved to mild or no pain at 2 h after treatment start. The participants continued to report their response over the subsequent 48 h in a diary and were contacted by phone within 24 and 48 h after treatment. A final follow-up visit was performed 3 to 7 days after treatment.
Results
The study included 46 women and 5 men with a median age of 40.0 years. 42.3% of the participants who received active treatment (N = 26) as compared to 32.0% of participants who received placebo treatment (N = 25) met the primary endpoint of headache relief at 2 h (p > .05). The odds ratio between active and placebo of the pain-free rate was 0.54 (95% CI 0.04–5.43) at 2 h and 1.78 (95% CI 0.09–112) at 24 and 48 h. More participants in the active vs placebo group improved numerically in nausea (50% vs 36%) and photophobia (42.3% vs 24%) at 2 h. Treatment with the Chordate System S020 was safe and well tolerated.
Conclusions
Treatment with the Chordate System S020 had no statistically significant effect on acute migraine headache relief.
This is a visual representation of the abstract.
Background
Most migraine attacks require treatment. Pharmacologic treatment of migraine attacks includes analgesics such as acetaminophen or non-steroidal anti-inflammatory drugs, ergots and triptans. Triptans are serotonin 5-HT1B/1D-receptor agonists and inhibit the transmission of activated nociceptive trigeminal afferents trough possible effects on cranial vasoconstriction, and direct neuronal effects. Recently antagonists of the calcitonin gene-related peptide receptor, the gepants, and the 5-HT1F-receptor agonist lasmiditan were approved for the treatment of migraine attacks. In some patients, pharmacological treatment is not effective, not tolerated or contraindicated. Therefore, there is an unmet need for nonpharmacological options for the treatment of acute migraine attacks.
The Chordate System is a medical device system that is used to administer kinetic oscillation stimulation (K.O.S) in the nasal cavity. 1 The rationale for using K.O.S for the acute treatment of migraine is based on several aspects:
First, in many subjects experiencing migraine attacks, a number of cranial autonomic symptoms such as lacrimation and rhinorrhea frequently accompanies these. 2 This clinical observation suggests a parasympathetic involvement in migraine attacks. This assumption is supported by the fact that neuropeptides involved in parasympathetic communication, such as pituitary adenylate cyclase activating peptide (PACAP) and, to a lesser extent, vasointestinal peptide play a role in the pathophysiology of migraine.3,4 The experimental activation of these receptors triggers migraine-like attacks and—in the case of PACAP—scavenging the ligand with a monoclonal antibody has therapeutic potential. 5
Second, stimulation of the sphenopalatine ganglion, a key element of the cranial parasympathetic outflow, has been shown to modulate migraine attack susceptibility.6,7 In addition, as PACAP receptors are located on the parasympathetic and trigeminal system and in migraine, pain and cranial autonomic symptoms generally go hand in hand, the existence of a trigeminal-autonomic reflex has been postulated by many research groups. 8 Finally, stimulation with K.O.S induces intense unilateral lacrimation reflecting a modulation of the parasympathetic outflow without causing trigeminal activation as the stimulation is not painful.1,6,9
Based on these observations and the ability of K.O.S to modulate the neuroanatomical system, it was hypothesized, that by modulating parasympathetic outflow, K.O.S may be effective in reducing trigeminal neuronal activity thereby aborting acute migraine attacks.
In this study, the Chordate System S020, a variant of Chordate System S011, was used to stimulate parasympathetic outflow through the nasal cavity. The Chordate System S011 has a CE market approval in the EU for the treatment of rhinitis. System S011 and System S020 are identical from a mechanical and an electrical point of view. The only difference is the software that controls the parameter settings. Exploratory treatments of patients with migraine attacks have demonstrated a reduction in pain intensity and attack frequency. A parallel group placebo-controlled study indicated that treatment with the Chordate System reduces headache with a rapid onset of action. 1
Based on the promising results the present clinical investigation was performed in a larger cohort of patients with migraine to further evaluate the effect of the Chordate System on pain and accompanying symptoms associated with an acute migraine attack.
Methods
Investigation design
The study was a prospective, interventional, randomized, placebo-controlled, double-blind, multicenter clinical investigation. One single migraine attack was treated with the Chordate System S020 in normal active mode or in placebo mode. After screening (up to 6 months before treatment), eligible participants were asked to return to the center at the onset (≤4.5 h) of their next moderate to severe migraine attack. Participants were randomized in a 1:1 ratio to receive active or placebo treatment. During the treatment and 2 h after initiation, changes in headache severity and symptoms were documented at the study center. The participants determined the time point of meaningful relief up to 4 h from treatment initiation and recorded their response over 48 hours on a diary card. A follow-up visit was scheduled 3 to 7 days after the treatment.
Investigational device
The investigational medical device was the Chordate System S020 consisting of a controller, a single use catheter with a balloon (A100) as well as a placebo module (CT100) from Chordate Medical AB, Stockholm, Sweden. The catheter was inserted into the participant's nasal cavity. For active treatment, the catheter balloon was inflated and oscillated for 15 minutes at 95 mbar pressure and 68 Hz frequency. For placebo treatment the catheter neither inflated nor oscillated but was connected to the controller. Treatment was applied to each nostril with less than 10 minutes between treatments. A placebo module was connected between the controller and catheter and allowed administration of treatment blinded to participants and investigators as it delivered active or placebo treatment according to the randomization code entered. A headband was used to keep the device in place. Treatment was administered by a doctor or nurse trained in the procedure.
Participants
Male and female individuals, 18 to 65 years of age with migraine, with or without aura according to the International Headache Society classification (International Classification of Headache Disorders 3 beta) were considered for participation in this study. The full list of inclusion and exclusion criteria is accessible as supplementary file (Supplemental Table S1).
Endpoints
The primary endpoint was the percentage of participants with headache relief (decrease in pain severity from moderate or severe at baseline to mild or no pain at 2 h post-treatment initiation without use of rescue medication). Secondary endpoints included the pain-free rate at 2 h (absence of pain without use of rescue medication); incidence of recurrence (return of moderate or severe pain in participants who were pain-free at 2 h) at 48 hours; sustained pain-free rate at 24 and 48 h; time to meaningful pain relief recorded by patients; change from baseline in nausea, photophobia, phonophobia, vomiting and disability at 2 h; participants global impression at 48 h (compared to the usual course of a migraine attack); and use of rescue medication within 48 h. We did not ask the participants whether they were aware of the treatment assignment.
Safety endpoints
Safety endpoints were the frequency, severity, safety of the device and outcome of all adverse events (AEs) until the follow-up visit.
Statistical analysis and sample size estimation
The sample size estimation was based on a likelihood ratio Chi-squared test (α = 0.05, 2-sided) with 86% power to detect a difference between active and placebo treatment in the primary endpoint. The sample size was planned with 100 participants, but enrollment was less than anticipated for several study centers. Based on a recent study with 36 participants showing positive results, 1 the sample size was reduced to 50 participants leading to a lower statistical power than originally planned.
The primary endpoint and pain-free rate at 2, 24, and 48 h were analyzed using a logistic regression model including treatment and baseline migraine pain intensity as explanatory variables (2-sided, α = 0.05). A Kaplan–Meier analysis was done for time to meaningful relief and any use of rescue medication (in addition to the use of rescue medication at specific time points). Nausea, photophobia, phonophobia, vomiting, disability, and participant's global impression on a 7-point scale were analyzed descriptively. The safety analysis was based on the safety analysis set (SAF, all participants who were treated with the investigational medical device at least once). All efficacy analyses were based on the modified full analysis set (mFAS, all participants from the SAF with an acute migraine attack of moderate to severe pain intensity at the time of treatment who did not use acute migraine medication within the 48 h prior to onset of the acute migraine attack and had at least one post-baseline efficacy evaluation).
Results
Participant disposition and baseline characteristics
Of 116 screened participants, 51 were included in the SAF and mFAS. One participant (placebo) was randomized by mistake but not treated (Figure 1). More women (90.2%) than men participated in the study, and most were white (96.1%). The number of participants diagnosed with “migraine with aura” was higher in the placebo group (36%) compared with the active treatment group (19.2%), otherwise demographics and baseline characteristics were comparable between the treatment groups (Table 1).

Participant disposition. N = number of participants.
Participant demographics and baseline characteristics (SAF).

BMI: body-mass-index; max: maximum; min: minimum; N: number of participants in data set; n: number of participants; SAF: safety analysis set; SD: standard deviation.
Primary endpoint
Numerically more participants in the active (11 participants, 42.3%) than in the placebo group (8 participants, 32.0%) experienced headache relief at 2 h (Table 2). This difference was not statistically significant (p > .05).
Headache relief at 2 h (primary endpoint) and pain-free rate at 2, 24, and 48 h post-initiation of treatment (mFAS).

Endpoints were analyzed using a logistic regression model including treatment and baseline migraine pain intensity as explanatory variables.
CI: confidence interval; n: number of participants; mFAS: modified full analysis set; OR: odds ratio.
Odds ratio between active and placebo treatment.
2-Sided, α = 0.05.
Secondary endpoints
Two participants in the active and three participants in the placebo group were pain-free at 2 hours. None of these five participants reported a headache recurrence at 48 hours.
At 24 and 48 h, two participants in the active and one participant in the placebo group reported sustained pain freedom without use of rescue medication (Table 2). The median time to meaningful pain relief recorded by patients was shorter with active than placebo treatment (Kaplan–Meier analysis, Figure 2). A higher proportion of participants reported an improvement in nausea and photophobia in the active group (50% and 42%, respectively) than in the placebo group (36% and 24%, respectively) at 2 h (Table 3). The proportions of participants reporting an improvement in phonophobia and vomiting did not differ between groups. There was no apparent difference between active and placebo groups in the presence of symptoms associated with migraine or the rating of migraine-related disabilities at 24 or 48 h (data not shown).
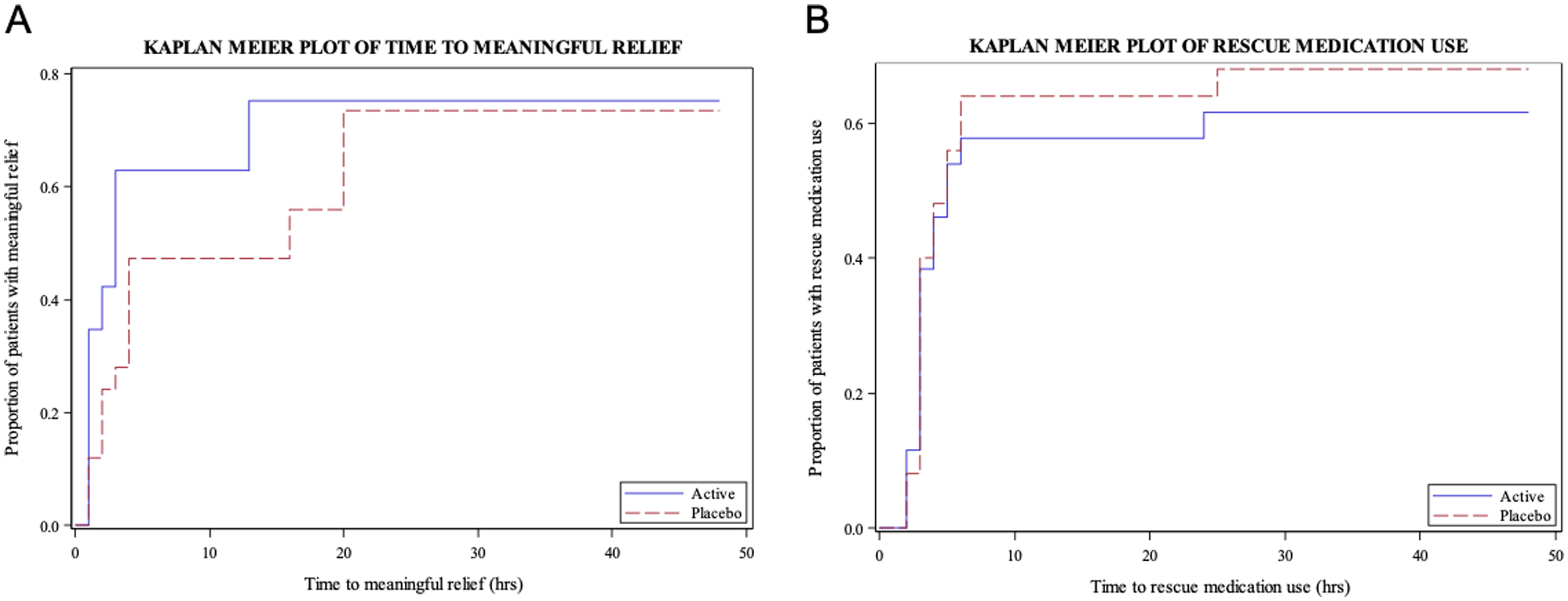
Kaplan–Meier analyses of time to meaningful pain relief (A) and time to rescue medication use (B), mFAS. Participants without any meaningful relief were censored at 48 h, or, in case rescue medication was taken, at the time of first use of rescue medication (A) hrs = hours, mFAS = modified full analysis set.
Change from baseline in migraine-associated symptoms at 2 h post-initiation of treatment (mFAS).

Data show number (%) of participants affected.
mFAS: modified full analysis set; N: number of participants.
62% of participants in the active and 76% in the placebo group had a better global impression at 48 h, while 8% in both groups had a worse impression.
Most participants in both groups had used rescue medication at 48 h (Figure 2), although the proportion of participants was slightly lower in the active group (61.5%) than in the placebo group (68.0%). Two participants (7.7%) in the active group and none in the placebo group took rescue medication at 2 hours.
Safety endpoints
Overall, 11 treatment-emergent AEs (TEAEs) were reported in six participants (Table 4). No serious AEs were reported. Three severe TEAEs were reported by one participant in the active group (nasal congestion and thirst) and one participant in the placebo group (fatigue). Four events occurring in two participants in the active group were assessed as related to treatment. The most frequent events were nasal congestion (3 participants in the active group) as well as nasal discomfort, dizziness, paresthesia, and mucosal inflammation, each occurring in one participant in the active and placebo group.
Overview of adverse events (SAF).

Percentages are based on the number of participants within each group.
AE: adverse event; N: number of participants in data set; n: number of participants; SAE: serious adverse event; TEAE: treatment-emergent adverse event.
Discussion
Previous exploratory treatments with the Chordate System in patients with migraine have indicated a reduction in pain intensity and attack frequency. 1 Based on the results, the present investigation was initiated to evaluate impact of the novel technique in patients with acute migraine attacks. The treatment setup with 15 min stimulation in each cavity (at 68 Hz and 95 mbar) was perhaps not sufficient to produce an effect on headache intensity and secondary endpoints in 26 patients with acute migraine attacks compared with the 25 patients receiving placebo. Eleven patients of the 26 in the active group showed headache relief at 2 h and 8 out of 25 in the placebo group. The patients without pain relief had taken rescue medication at 24 h. Due to the small sample size and the small difference in the proportion of subjects between the treatment groups that experienced headache relief, the effects on the secondary endpoints were not significant. In addition, we observed a high response rate to sham stimulation. The sham response rate, however, was within the range of what has been observed in other trials. It is highly unlikely that the high sham response would have been induced by the sham intervention as the deflated catheter was introduced (which is not painful) but neither inflation nor oscillation took place. In conclusion, the results of this study must be considered neutral. We did not ask the participants whether they were aware of the treatment assignment and therefore could not validate blinding. From a safety perspective, the treatment with the Chordate System S020 was generally well tolerated.
Evidence that kinetic oscillation stimulation triggers parasympathetic is derived from the fact that the stimulation induces cranial autonomic symptoms. Patients do not describe the procedure as painful; hence one can deduce that in these patients no nociceptive trigeminal activation occurs. This is supported by an fMRI study which showed in patients that do not experience the procedure as painful, no hypothalamic activation. 6 Nevertheless, we cannot exclude that in a few patients some degree of pain/trigeminal activation may have occurred in the active treatment group.
Positive aspects of the study are that this method appears safe when used in a migraine attack and has minimal side effects, which is in line with a larger study when used in migraine prevention. 10 If shown to be effective in a future larger trial, the technique could be applied in pregnant women who have a limited choice of medications for the treatment of migraine attacks. Shortcomings of the study are the small sample size and the choice of headache relief instead of pain-free after 2 h as the primary endpoint.
After the termination of this study, a second study was performed to investigate the use of kinetic oscillation stimulation for the preventive treatment of chronic migraine in 132 patients. 10 The primary endpoint showed a significantly larger reduction of MHD with active treatment (−3.5 days, n = 67) compared with sham treatment (−1.2 days, n = 65) (p = .0132). This nonpharmacological nature of the treatment suggests that K.O.S could be a valuable addition to the current therapeutic options for the management of chronic migraine.
Conclusion
In conclusion, treatment with the Chordate System S020 had no statistically significant effect on acute migraine headache relief. In addition, we observed no statistically significant impact on migraine features such as pain-free rate, sustained pain-free rate, time to meaningful relief, migraine-associated symptoms, migraine-derived disabilities, or global impression. Treatment with the Chordate System S020 was generally well tolerated.
Highlights
The Chordate System S020 delivers kinetic oscillation to the nasal mucosa, which is supposed to modulate the parasympathetic outflow
Treatment with the Chordate System S020 had no statistically significant effect on acute migraine headache relief.
In addition, we observed no statistically significant impact on migraine features such as pain-free rate, sustained pain-free rate, time to meaningful relief, migraine-associated symptoms, migraine-derived disabilities, or global impression.
Treatment with the Chordate System S020 was generally well tolerated and the most frequent side effects were symptoms associated with migraine
Abbreviations
adverse event
kinetic oscillation stimulation
modified full analysis set
pituitary adenylate cyclase activating peptide
safety analysis set
treatment-emergent adverse event
Supplemental Material
sj-docx-1-rep-10.1177_25158163251377699 - Supplemental material for Efficacy and safety of kinetic oscillation stimulation in the nasal cavity for the treatment of acute migraine attacks: A randomized, placebo-controlled, double-blind investigation
Supplemental material, sj-docx-1-rep-10.1177_25158163251377699 for Efficacy and safety of kinetic oscillation stimulation in the nasal cavity for the treatment of acute migraine attacks: A randomized, placebo-controlled, double-blind investigation by Hans-Christoph Diener, Uwe Reuter, Andreas Straube, Arne May, Holger Kaube, Charly Gaul, Tobias Freilinger, Larissa Wirtz, Catrina Milazzo and Jan Hoffmann in Cephalalgia Reports
Footnotes
Author contributions
Hans-Christoph Diener and Jan Hoffmann wrote the first draft of the paper. All authors contributed by revising and editing of the manuscript. All authors approved the final version.
Consent to participate
The study was approved by the Independent Ethics Committee of the Medical Faculty of the University Duisburg-Essen in agreement with the local ethics committees at all participating institutions (![]() ). Written informed consent was obtained from all study participants. The study was conducted in accordance with appropriate local laws and regulations, the International Standards Organization guideline 14155 and the Declaration of Helsinki. The trial was registered on clinicaltrials.gov (NCT02185703).
). Written informed consent was obtained from all study participants. The study was conducted in accordance with appropriate local laws and regulations, the International Standards Organization guideline 14155 and the Declaration of Helsinki. The trial was registered on clinicaltrials.gov (NCT02185703).
Data availability statement
The data supporting the findings from the current study are available from the corresponding author on reasonable request considering restrictions of participant confidentiality.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hans-Christoph Diener received honoraria for oral presentations from: Betapharm, Lundbeck, Orion and WebMD. The German Research Council (DFG) supports headache research by Hans-Christoph Diener. Hans-Christoph Diener serves on the editorial boards of Cephalalgia, Lancet Neurology and Drugs.
Uwe Reuter received no personal honoraria. Institutional fees for consulting and lectures within the past three years were received from Abbvie, Novartis, Lundbeck, Perfood, Pfizer and TEVA. Financial support for research by Novartis and BMBF. Uwe Reuter is associate editor of JHP.
Andreas Straube received honoraria as speaker and/or consultant for Novartis, Lundbeck, Betapharm and TEVA. Financial support for his research by Novartis, Innovation Fund, German Research Association (DFG) and University of Munich (LMU).
Charly Gaul received honoraria for consulting and lectures within the past three years from Abbvie, Lilly, Novartis Pharma, Hormosan Pharma, Sanofi-Aventis, Lundbeck, Perfood, Vectura Fertin Pharma, Chordate, Pfizer, Dr Reddys, Merz, Reckitt-Benckiser and TEVA. His research is supported by a grant from the German Research Foundation (DFG). He does not hold any stocks in pharmaceutical companies.
Arne May received honoraria as speaker and/or consultant for Novartis, Ipsen, Tiefenbacher, Betapharm and TEVA.
Tobias Freilinger received honoraria as speaker and/or consultant for TEVA Pharmaceuticals, Novartis, Lundbeck, Lilly, UCB and Bayer. Research support from German Research Association (DFG) and University of Tuebingen.
Caterina Milazzo and Larissa Wirtz are employees of FGK Clinical Research GmbH
Jan Hoffmann is currently a full-time employee of H. Lundbeck A/S. The clinical trial was finalized prior to his employment at H. Lundbeck A/S. Before being employed at H. Lundbeck A/S he received honoraria for consulting activities and/or serving on advisory boards and/or for giving lectures/presentations from AbbVie, Allergan, Autonomic Technologies Inc., Cannovex BV, Chordate Medical AB, MD-Horizonte, Eli Lilly, Hormosan Pharma, H. Lundbeck A/S, Novartis, Pfizer, Sanofi and Teva. He holds stock options from Chordate Medical AB. He received personal fees for Medico-Legal work as well as from NEJM Journal Watch, Oxford University Press, Quintessence Publishing, Sage Publishing and Springer Healthcare. He also received research grants from Bristol Myers Squibb, International Headache Society (IHS), National Institute of Health and Care Research (NIHR), Medical Research Council (MRC), and the Migraine Trust. Jan Hoffman serves as Associate Editor Frontiers in Pain Research, as well as for The Journal of Headache and Pain. Previously he served as Associate Editor for Cephalalgia, Cephalalgia Reports, and for the Journal of Oral & Facial Pain and Headache. He served as an elected member of the Board of Trustees of the International Headache Society as well as a Council Member and Treasurer of the British Association for the Study of Headache. The remaining authors have no conflict.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by Chordate Medical AB.
Trial registration
The trial was registered on clinicaltrials.gov (NCT02185703) on 09-Jul-2014.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
