Abstract
Introduction
The mechanism of action of non-invasive vagal nerve stimulation in the treatment of migraine is elusive. We studied its effect in a human model of pain, the nociceptive withdrawal reflex.
Methods
We enrolled 10 healthy subjects who underwent active non-invasive vagal nerve stimulation and sham treatment in a randomized, cross-over, sham-controlled study. Non-invasive vagal nerve stimulation was delivered with gammaCore®. The assessment of the nociceptive withdrawal reflex was performed at baseline (T0) and at 5 (T5) and 30 (T30) minutes after stimulation.
Results
Non-invasive vagal nerve stimulation significantly increased the reflex threshold to single stimulus at both T5 and T30 and the temporal summation threshold at T30. Sham treatment did not modify any parameters.
Discussion
These findings are consistent with a modulation of central descending pathways for pain control. An altered spinal and supraspinal control of pain has been described in primary headache, so this effect may partially explain the therapeutic effect of non-invasive vagal nerve stimulation.
Introduction
Non-invasive vagal nerve stimulation (nVNS) has become practical through a portable device (gammaCore®) which activates the vagus by transcutaneous electrical stimulation through the neck level with effects similar to those of implanted VNS (1). This observation was recently confirmed in both electrophysiology and neuro-imaging studies in humans. Cervical nVNS consistently elicited scalp somatosensory evoked potentials (SEPs) similar to those obtained with invasive techniques. The cortical SEP components disappeared when the device was placed over the sternocleidomastoid muscle, confirming that the observed response was related to the activation of vagal fibers (2,3). Using functional MRI, Frangos et al. demonstrated that auricular and cervical nVNS activates central pathways and nuclei consistent with vagal projections (4,5).
Increasing evidence suggests the efficacy of cervical nVNS as a potential treatment for migraine and cluster headache (6–10); nVNS was recently approved for the acute treatment of episodic cluster headache in the USA, and it is approved for the acute or preventive treatment of primary headache in the European Union.
The exact mechanisms of action of nVNS are not known. Pre-clinical evidence suggests that nVNS causes antinociception at spinal and supraspinal levels (11,12), whereas in human studies the variability of electrode positioning and stimulation parameters does not allow unequivocal interpretation of the findings (13,14).
The nociceptive withdrawal reflex (NWR) of the lower limb is a well-accepted neurophysiological tool for the assessment of spinal and supraspinal nociceptive pathways in healthy subjects and in pathological conditions (15). Data from the literature support the usefulness of the NWR study in primary headaches. The NWR threshold strongly correlates with the severity of migraine: It is reduced in chronic migraine, and it is normalized by the administration of preventive migraine drugs such as amitriptyline or serotonergic drugs (16). More recently, NWR detected a facilitation in the temporal summation process at the spinal level in an induced spontaneous-like model of migraine triggered by the oral intake of a nitric oxide donor (17). A facilitation of temporal summation was also observed using the NWR paradigm during the active phase of episodic cluster headache (18).
In the present study, we aimed at further investigating the antinociceptive effect of nVNS delivered with gammaCore in humans using the NWR paradigm in healthy subjects.
Materials and methods
Subjects
We enrolled 10 healthy subjects (five males, mean age 26.5 ± 2.2 years) in a randomized, cross-over, sham-controlled study. None of the subjects had a positive pathological anamnesis for neurological and/or pain disorders. All subjects had a negative personal and family history (first-degree relatives) for neurological disorders and in particular for migraine with or without aura and for cluster headache. None of the enrolled subjects had taken any drugs that are known to interfere with pain transmission in the 4 days prior to the neurophysiological sessions. Female subjects underwent the neurophysiological assessments outside the physiological menstrual window. Each subject underwent active nVNS and sham treatment in two separate sessions in a cross-over design, according to a computer-generated randomization list. The two sessions were performed 7 days apart. Five subjects received active nVNS stimulation first, whereas the other five subjects received the sham stimulation first. The study was approved by the local Ethics Committee and all subjects provided informed consent before enrolment. None of the subjects had previously experienced VNS stimulation, and they were not informed that the stimuli delivered in the cranial district could be expected to be more effective in modulating the pain reflexes under investigation.
Stimulation
nVNS was delivered with gammaCore (electroCore, LLC) which produces 5 kHz sine waves each lasting 200 µs, repeated once every 40 ms (25 Hz). When applied to the skin, the highest voltage produced is 24 V and the highest intensity is 60 mA.
We adopted the stimulation parameters that have shown efficacy and safety in migraine (4). Briefly, the device was placed medially to the sternocleidomastoid muscle along the cervical branch of the vagus nerve, and the intensity of the stimulation was gradually increased until a mild but stable contraction of the ipsilateral orbicularis oris muscle was obtained. This same intensity was then used to deliver two 120-second stimulations, one on each side of the neck separated by 5 minutes.
Sham treatment was delivered bilaterally using gammaCore at the wrist level, 1 cm proximal to the carpal ligament along the median nerve. The intensity was gradually increased until a mild but stable contraction of the abductor pollicis brevis muscle was obtained. The treatment paradigm was identical to active nVNS. The selection of the wrist was based on the results of preparatory trials in healthy volunteers: Wrist stimulation consistently induced a motor response in the abductor pollicis brevis muscle similar to the motor response activated by nVNS in the ipsilateral orbicularis oris muscle.
Nociceptive withdrawal reflex measurements
The NWR was recorded from the right lower limb according to standardized procedure. All the examinations were conducted in the morning at 10:00 a.m. in a quiet room by the same blinded technician (MF). The subjects were seated with their ankle flexed at 90° and knee flexed at 130°. The electrical stimuli were delivered at the sural nerve behind the lateral malleolus with a pair of Ag/AgCl surface electrodes. The stimuli consisted of a train of five 1 ms squared pulses at 200 Hz. The electromyographic response (Synergy, Medelec, UK) was recorded from the capitis brevis of the right biceps femoris with a pair of Ag/AgCl surface electrodes. The evaluation of the reflex threshold was conducted with a staircase method, in which the intensity was gradually raised by 0.3 mA steps. The sweep parameters were: analysis time 300 ms, sensitivity 20 µV, filter bandpass 3–3000 Hz.
The reflex threshold of the NWR to a single stimulus (RT-SS) was the lowest intensity (mA) needed to elicit three consecutive stable muscular responses of at least 20 µV and 10 ms. The temporal summation threshold of the NWR (RT-TS) was evaluated as an indicator of spinal excitability using a train of five stimuli at a frequency of 2 Hz. RT-TS was the lowest intensity (mA) needed to elicit three consecutive stable muscular responses of at least 20 µV and 10 ms in the fourth and fifth sweeps. We also recorded the mean area under the curve (AUC – μV*ms) of the three muscular responses at RT-SS, the latency (ms) of the reflex at RT-SS, and the 0–10 points Visual Analogue Scale (VAS) at RT-SS.
For both active and sham treatment, NWR was recorded at baseline (T0) and at 5 (T5) and 30 (T30) minutes after the stimulation.
Statistical analysis
The sample size/power was determined using the Open Source Epidemiologic Statistics for Public Health (www.openepi.com). The primary outcome of the study was to assess the difference between active nVNS and sham treatment in RT-SS after 30 minutes. For the sample size/power calculation, we considered at least a 20% (standard deviation of 10%) difference in RT-SS between groups as clinically meaningful. This value is based on the data from the literature and on our previous experience in migraine and Parkinson’s disease (18,19). To calculate the sample size for a paired t-test (cross-over study), we used a level of significance of 0.02. With these parameters, the power of the study for 10 subjects was 0.9984.
For the statistical analysis, we used the Statistical Package for the Social Sciences (SPSS) for Windows, version 21.0. The Kolmogorov-Smirnov test confirmed a normal distribution of the data. Intra-group analysis was conducted with an ANOVA (analysis of variance) for repeated measures, followed by a post hoc Bonferroni’s correction for multiple comparisons. Differences between active nVNS and sham were analyzed with a t-test for paired samples. Data are presented as percentage variations from baseline, with the latter equaling 100%. The level of significance was set at 0.05 (corrected when necessary for multiple comparison) for all variables.
Results
Values of neurophysiological parameters of the nociceptive withdrawal reflex at baseline.

RT-SS: reflex threshold to single stimulus; AUC: area under the curve; VAS: Visual Analogue Scale; RT-TS: Reflex threshold to temporal summation.
Effect of active nVNS and sham on the neurophysiological parameters. Data in brackets represent the percent change from baseline, with baseline equaled to 100.
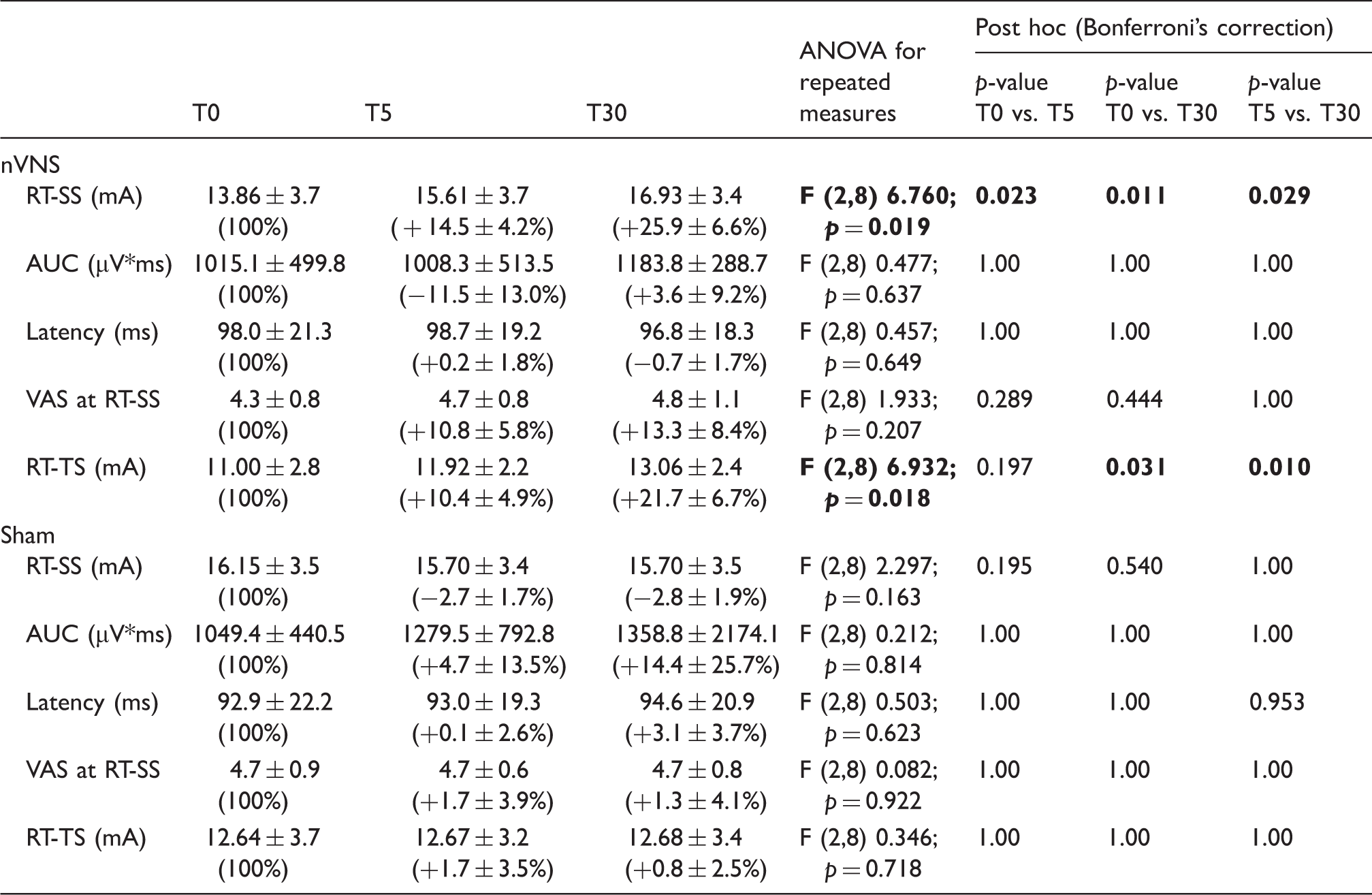
RT-SS: Reflex threshold to single stimulus; AUC: Area under the curve; VAS: Visual Analogue Scale; RT-TS: Reflex threshold to temporal summation. Note: Significant values are highlighted in bold.
Sham treatment did not modify RT-SS at either T5 (−2.67% ± 1.3%, p = 0.195) or T30 (−2.76% ± 1.9%, p = 0.540). Similarly, RT-TS was unaffected by sham at both time points (T5 +1.67% ± 3.5%, p = 1.00; T30 + 0.48% ± 2.5%, p = 1.00) (Table 2).
When active nVNS was compared with sham, the percentage increase in RT-SS observed after active nVNS was significantly higher with respect to the increase observed after sham treatment at both T5 (p = 0.008) and T30 (p = 0.013). Accordingly, the percentage increase in RT-TS was significantly more pronounced following active nVNS than after sham at T30 (p = 0.013) (Figure 1).
Effect of nVNS and sham treatment on RT-SS and RT-TS. Data are expressed as percent change from baseline, with baseline equaled to 100. Δ = Active nVNS vs. Sham: p < 0.05. RT-SS: Reflex threshold to single stimulus; RT-TS: Reflex threshold to temporal summation.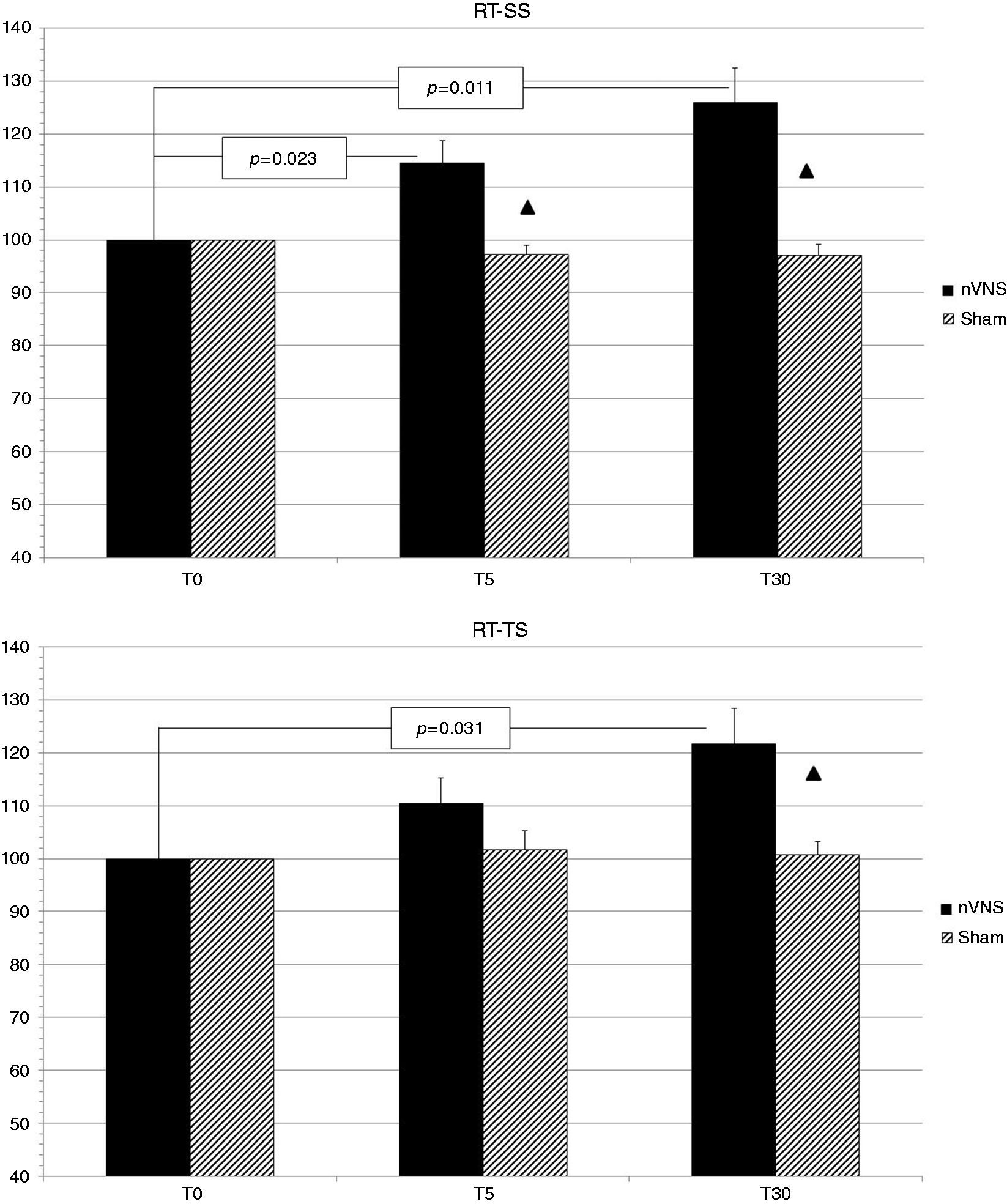
We did not find any significant changes in the other neurophysiological and psychophysical parameters after either active or sham treatment (Table 2).
Discussion
Two 120-second nVNS stimulations delivered with the gammaCore produced a rapid increase in the neurophysiologically recorded pain threshold. The effect seems specific for nVNS because it was not observed with the innocuous heterosegmental somatosensory stimulation. The NWR paradigm is a powerful tool for the study of pain modulation that allows the reliable quantification of pain perception in different settings. In our series of young, healthy subjects, the broad stability of the reported VAS at different thresholds indicates that the data were accurately captured and represent a valid neurophysiological assessment.
The analgesic effect of VNS on nociception is not new because it was detected in different animal models, also in the trigeminal region, but its exact mechanisms remain elusive (20). Cervical nVNS activates vagal afferents that are connected to multiple structures involved in pain modulation and headache pathophysiology, i.e. the hypothalamus, dorsolateral prefrontal cortex, nucleus of tractus solitarius, trigeminal nucleus caudalis, spinal trigeminal nucleus, locus coeruleus, raphe nuclei, and periaqueductal grey (4,5,12). Additionally, VNS is known to increase serotonin levels in the dorsal raphe, a neurotransmitter that plays a primary role in descending pathways of pain control (21).
Previous data obtained with NWR demonstrated an altered supraspinal control of pain in migraine and cluster headache characterized by a dysfunction in the system affecting the “diffuse noxious inhibitory control” of pain (DNIC) and a facilitation of pain transmission (18,22,23). In particular, an altered functioning of the DNIC pathway appears to be crucial in the pathogenesis and in the progression of primary headaches (22). The anatomical and functional bases of the DNIC system have been widely studied. The structures involved are mainly represented by the hypothalamus (and its dopaminergic descending pathways), the periaqueductal grey, the subnucleus reticularis dorsalis, and the nucleus raphe magnus (18,24,25).
In our study the observed increase in RT-SS after nVNS, a parameter that correlates with the overall subjective pain perception, was consistent with a central modulation of the pain network through an inhibitory effect. The observed increase in the RT-TS 30 minutes after nVNS, which is more specific for spinal activation, is consistent with a modulation of the descending pathways for pain control and, in particular, with the inhibition of the wide dynamic range neurons at spinal and trigeminal levels (24).
On the basis of the present findings and available evidence, we speculate that nVNS exerts an antinociceptive effect via the activation of structures involved in DNIC that act both at supraspinal and spinal levels in a top-down manner. Considering that none of the subjects in our study reported any pain during cervical nVNS, we are inclined to rule out a direct activation of DNIC by nVNS. It seems more likely that nVNS and DNIC act on shared central and descending pathways that are relevant in the pathophysiology of primary headaches, such as the hypothalamus, raphe nuclei, and periaqueductal grey as described above (23,24). The modulation of the DNIC system may represent only one of the mechanisms of action involved in the therapeutic effect of nVNS in migraine and cluster headache. Alternative possibilities include a direct modulation of the hypothalamus and its dopaminergic descending pathways (primarily for cluster headache) and/or of the trigeminal nucleus caudalis and spinal trigeminal nucleus (primarily for migraine) (11,12,26).
Our study had several limitations of note. Despite the calculated power of the study, the small sample size does not allow the drawing of definitive conclusions, and the present findings need to be confirmed in a larger population. In addition, NWR can be modulated by different conditions. In our procedure, we aimed to control multiple variables, for example, circadian fluctuations, menstrual cycle (in women), drug intake, and the subject’s comfort. The level of attention of the subject during the electrophysiological evaluation is crucial, however, and we can’t rule this out as a possible confounding variable.
Future studies will hopefully provide more precise details on the structures, mechanisms, and mediators involved in physiological and pathological effects of nVNS.
Footnotes
Key findings
nVNS produces a rapid-onset inhibitory effect on the NWR in healthy subjects.
nVNS acts both on the RT-SS and on the RT-TS components of the NWR.
The analgesic effect of nVNS is likely to effect pain facilitation mechanisms, maybe via the activation of structure involved in the diffuse noxious inhibitory control (DNIC).
In this study in healthy subjects, nVNS produced a significant increase in pain thresholds. This mechanistic effect could contribute to the clinical efficacy reported in the treatment of primary headache.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
