Abstract
Context
A variety of neuromodulatory approaches available today has broadened our therapeutic options significantly especially in drug refractory patients with chronic cluster headache and chronic migraine.
Overview
It is a dynamic field with a current trend to non-invasive transcutaneous stimulation approaches. However, sound studies providing evidence for the widespread use of these novel approaches are sparse. For invasive approaches, occipital nerve stimulation is now widely considered the treatment of first choice in chronic trigeminal autonomic cephalgias and – with limitations – chronic migraine. Although equally effective, deep brain stimulation is considered second-line treatment in cluster headache because of its potentially life-threatening side effects. Most recently, stimulation of the sphenopalatine ganglion has also been shown to effectively abort acute cluster headache attacks. Interesting other upcoming approaches include transcutaneous supraorbital nerve stimulation and transcutaneous vagal nerve stimulation.
Conclusion
Pearls and pitfalls of common invasive and non-invasive neuromodulatory approaches and open questions are summarised in this review along with recommendations for future studies.
Introduction
Chronic headaches affect up to 3% of the population. They lead to a significantly higher individual impairment than in patients with episodic subtypes (1,2) and put more strain on health care systems. In patients with chronic cluster headache, annual expenses amount up to €9073 per capita compared with €1819 for the episodic form (3). Despite a broad armamentarium of both abortive and prophylactic drugs, especially in patients with chronic headaches first- and second-line pharmacological therapies often fail and patients are regarded as drug refractory.
Available invasive and non-invasive neuromodulatory approaches.

DBS
The first case reporting efficacy of DBS of the posterior hypothalamus dates back to 2001 in a patient with medically intractable chronic cluster headache (7**). The treatment rationale was based on hormonal abnormalities as well as structural and functional neuroimaging studies pointing to the posterior hypothalamus as a pivotal structure in the pathogenesis of cluster headache (see Figure 1). Since then, data for more than 70 patients have been published for patients with trigeminal-autonomic cephalalgias, predominantly chronic cluster headache.
Implanted deep brain electrode into the right posterior hypothalamic regiom (arrow) in an axial magnetic resonance imaging (MRI) sequence.
For chronic cluster headache, the only randomised sham-controlled trial on DBS was published using a double-blind cross-over design in 11 patients (8**). The primary endpoint was defined as the difference of attack frequency per week for a period of one month of active (stimulator switched on) or no stimulation (stimulator switched off). Active stimulation did not result in a lower attack frequency during this period. However, in the subsequent 10-week open-label period, 55% of the patients experienced a reduction of attack frequency of 50% or more. The very brief experimental period of only one month and a wash-out phase of only one week are problematic. Furthermore, preset stimulation parameters instead of individual adjustments could have contributed to this negative finding for the experimental period while the response rate in the open-label phase is similar to those observed in uncontrolled studies and case series, which report response rates of ≥50% (defined as 50% or more reduction of attack frequency), overlooking follow-up periods of several years (9–12**,13). A long-term follow-up of the largest group of patients from Milan, Italy (n = 17) was recently published (14**). After a median follow-up of 8.7 years with a maximum of 12 years, six patients experienced sustained freedom from pain. In six more patients the chronic cluster headache reverted to an episodic pattern with prolonged remissions and a substantial improvement in 12 out of 17 patients (70%). In five of the six patients with substantial improvement, the stimulator was switched off after a median of 6.4 years with persistent benefit for a median of three years. Four patients with alternating attacks on both sides received bilateral stimulation but continued to suffer from daily attacks. In three patients a delayed loss of efficacy was observed after at least 12 months of substantial improvement. Adverse events included infections of the electrodes or the impulse generator in four patients (24%), electrode displacement in two patients (8%) and a non-symptomatic haemorrhage of the third ventricle as well as a seizure in one patient each (4%). One patient developed de novo contralateral attacks and five patients had received occipital nerve stimulation before without relevant benefit.
Hypothalamic DBS was also efficient in chronic paroxysmal hemicrania (15), short unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT syndrome) (16–18), and symptomatic trigeminal neuralgia due to multiple sclerosis but not in persistent idiopathic facial pain (19). Interestingly, acute hypothalamic DBS was insufficient to abort ongoing cluster headache attacks (20). Effective blinding is possible as interruption of DBS is generally not noted by the patient and relapses have been described (9,12**).
As shown in an H215O-positron-emission tomography (PET) study, hypothalamic DBS does not only affect the posterior hypothalamic region itself but also causes activation of the ipsilateral trigeminal system as well as widespread activations and deactivations, especially of structures belonging to the “pain matrix” (21**). In patients with chronic cluster headache receiving chronic DBS the ipsilateral cold pain and cold detection thresholds were increased in the first branch of the trigeminal nerve while short-time interruption of DBS had no effect on thermal thresholds (22). Microelectrode recordings of the stimulated region did not reveal a pattern specific for the posterior hypothalamic region (10,23,24). They could, however, provide some indirect information about the electrode position as neuronal discharge rates in the posterior hypothalamic region were significantly lower than in the medial thalamic region (23). In addition, the position of stimulation electrodes in the posterior hypothalamic region does not seem to be predictive of clinical efficacy as shown by an analysis in 10 patients receiving DBS for chronic cluster headache (25). This suggests that therapeutic effects are presumably not mediated by a direct stimulation of the hypothalamus itself but point to a specific effect of DBS in cluster headache with complex neuroplastic changes in various brain regions that exceed pure antinociception.
Somnographic studies revealed that circadian rhythmicity is not impaired by hypothalamic DBS (26), and clinically relevant effects on the autonomic nervous system are hardly apart from an increased sympatho-excitatory drive in the head-up-tilt test (26).
On the downside, some severe side effects have occurred: One patient experienced a lethal intracerebral haemorrhage, and one implantation had to be aborted as the patient perceived a panic attack with autonomic disturbances (12**). In another patient, a transient neurological deficit with temporary hemiparesis classified as reversible vasospasm of the Aa. thalamoperforantes occurred (27). Therefore, hypothalamic DBS should be restricted to departments with a stereotactic neurosurgery unit and ample experience in DBS implantations.
Other side effects comprise infection of electrodes or impulse generators, syncopes (although no relevant changes were found during autonomic testing apart from an increased sympathico-excitatory tone in the head-up-tilt test as shown by Vetrugno et al. (26)), diplopia and vertigo with higher amplitudes (8**,9) and changes in appetite and thirst.
In conclusion, hypothalamic DBS is an efficient approach causing complex changes in various brain regions involved in pain processing, but should not be the first choice in chronic trigeminal autonomic cephalgias because of its invasive nature with potentially lethal side effects.
Pearls
– High efficacy in various unblinded studies – Good long-term experience – Blinding feasible – Reasonable mechanism of action
Pitfalls
– Randomised controlled trial (RCT) negative for experimental period – Invasive with potentially lethal side effects, thus only second-line for patients who have failed or are not eligible for ONS and sphenopalatine ganglion stimulation (SPGS)
ONS
ONS in cluster headache and other trigeminal autonomic cephalgias
The efficacy of bilateral ONS (see Figure 2 as example) has been examined in a retrospective study for a mean follow-up period of 18 months. The response rate defined as a 50% reduction of attacks was 36% (five out of 14 patients) (28**,29). After encouraging results in a pilot study on eight subjects from a Belgian group (30**), a prospective long-term study (n = 14) was published on patients with unilateral ONS and a mean follow-up of 37 months (31**). A reduction of attack frequency of 90% or more was found in 11 patients (78%). Similar efficacy rates were found in a German sample (n = 10) with a follow-up of 18 months (32) and a prospective multicenter trial on bilateral ONS from France with a mean follow-up of 15 months: In the latter 10 out of 13 patients experienced a 50% or more reduction of attack frequency. Including further smaller case series more than 60 patients have been treated with ONS in chronic cluster headache with a favourable outcome: More than 50% of the patients responded (defined as ≥50% reduction of attack frequency). However, an RCT confirming these positive findings has never been conducted.
Bilateral occipital nerve stimulation on an anterior-posterior X-ray.
In other trigeminal autonomic headaches, patient-reported response rates of at least 80% in four out of six patients were found in hemicrania continua (28**). Out of seven patients treated with ONS in SUNCT and one patient with short-lasting unilateral neuralgiform headache attacks with cranial autonomic symptoms (SUNA), five reported an improvement of at least 50% (33).
Multiple pathophysiological mechanisms have been discussed for ONS. Peripheral nerve stimulation can exert direct effects on the nerve such as a frequency-dependent modulation of conduction velocity. Segmental effects as proposed by Melzack and Wall in the “gate control theory” are an additional mechanism (34). Afferent sensory A beta fibres block segmental transmission of nociceptive input from A delta and C fibres. As a peculiarity of the cranial region, occipital and trigeminal nociceptive afferent neurons converge to interneurons in the trigemino-cervical complex, which extends from the trigeminal nucleus caudalis to the upper cervical cord. This explains spreading of cervical pain to the frontal region or vice versa which can be seen frequently in primary headaches. Additionally, activation of ascending second-order neurons could modulate neuronal activity in regions such as the periaqueductal grey with a successive increase of descending antinociceptive pathways resulting in a net reduction of pain (see Bartsch et al. (35) for further information).
An [18F]2-fluoro-2-deoxy-D-glucose (18FDG)-PET study on patients with chronic cluster headache receiving ONS revealed that continuous stimulation resulted in a normalisation of regional hypermetabolism in structures belonging to the pain matrix and thus extensive central neuromodulatory changes (36).
Although device-related side effects are high (see below), ONS has displaced DBS as an invasive approach of first choice in trigeminal autonomic cephalgias because of its overall less invasive nature and similar results in terms of efficacy. As a downside, an RCT has never been performed in chronic cluster headache, first and foremost owing to blinding issues.
ONS in chronic migraine
Two case series in patients suffering from “transformed” migraine (which is defined as headache on more than 15 headache days per month in patients fulfilling the International Headache Society (IHS) criteria for episodic migraine) found a reduction of attack frequency or headache severity of more than 50% along with relevant improvements of migraine-associated disability in more than 85% of the participants. Despite these promising initial findings, larger studies have yielded ambiguous results. In the multicentre, randomised, sham-controlled occipital nerve stimulation for the treatment of intractable chronic migraine headache (ONSTIM) trial, 110 patients with refractory chronic migraine were enrolled. During the experimental phase of three months, 20 patients received adjustable stimulation, 16 preset stimulation and 17 in an ancillary group conservative treatment only. No change was observed in headache frequency and pain intensity. However, the responder rate was significantly higher in patients with adjustable stimulation (39%) compared to the preset (6%) and the control group (0%).
Another recently published large study of 157 patients with chronic migraine did not show any differences of the primary endpoint (defined as a ≥50% reduction in mean daily scores on a visual analogue scale (VAS)) after three months of stimulation between the stimulation (n = 105) and the sham (n = 52) group (37**). However, the number of participants reporting a 30% reduction of pain was significantly higher in the stimulation group along with a significant decrease in the number of headache days per month and migraine-related disability. A group of 20 patients not initially responding to test stimulation was excluded from the trial, which raises methodological concerns.
In the Precision Implantable Stimulator for Migraine (PRISM) study, which has been presented only in preliminary form (38), 125 patients with intractable episodic and chronic migraine were treated with either three months of bilateral active or sham-stimulation only if a preceding externalised test stimulation for five to 10 days was successful. The primary endpoint (reduction of headache days per month) was not met. Interestingly, a subgroup of patients with medication overuse (especially opioids) had a significantly worse outcome.
In summary, three larger RCTs have failed to show meaningful and conclusive improvements in their experimental period in this group of patients. However, the overall experience with ONS and the good results in the open-label period and in uncontrolled studies should be kept in mind. They do nevertheless strongly argue against an unreflected widespread use of this method in chronic migraine at present. Multidisciplinary integrated care should be considered instead, and it will be tempting to see if the increasing use of botulinumtoxin in chronic migraine will reduce the number of patients with drug-resistent chronic migraine.
ONS from a technical point of view
Although the long-term use seems to be effective and generally safe with follow-up periods of more than three years, high rates of device-related side effects including electrode migration, lead breakage, local infections and irritation from faulty lead insulation have been reported. Battery depletion is a problem inherent in all implantable devices without a rechargeable internal power supply – not only those used for ONS. Still, it was reported as the most common complication in all major studies affecting more than 60% of the patients leading to high revision rates (28**,29,30**,31**,37**,39–42**). Especially, patients using high stimulation intensities are more prone to frequent revisions. Although the rate of electrode lead migration was as low as 0% in some studies, it reached 100% in other studies (4**), illustrating the need for technical advances (see also Recommendations #6).
Stimulation-related side effects including an irritation within the receptive field of the occipital nerve, muscle contractions and local pain around the impulse generator are clinically less relevant (28**,29,30**, 31**,37**,39–42**). As paraesthesias are mandatory to achieve clinical efficacy in ONS, it is a matter of opinion whether to regard them as side effects generally or only in those patients who are reluctant to tolerate them. Thus, at present especially the technical implementation is far from optimum and needs improvements. As first devices with a rechargeable battery have been introduced, it will be interesting to see if revision rates can be lowered in the future.
Early studies of ONS from a methodological point of view
Occipital neuralgia was the first indication for ONS, and Weiner and Reed reported a response rate of 100% (6**), leading to widespread dissemination of the method for various peripheral cranial nerves. A follow-up study questioned the diagnosis and highlights a basic prerequisite for studies introducing new treatments procedures: a proper diagnosis following accepted classifications (43,44). In two other studies reporting good efficacy (45,46), the inclusion criteria do not meet the IHS criteria for occipital neuralgia, raising the question as to which underlying headache entity was actually treated. It is noteworthy that occipital neuralgia as such is a very rare condition and mostly responds well to pharmacotherapy and occipital nerve blocks, which casts doubt on the reportedly high number of patients with drug refractory occipital neuralgia in these studies.
So what lessons are to be learned? An interdisciplinary approach should be mandatory for all centres: Experienced headache specialists should take care of patient selection and pre- and postimplant care. An experienced surgeon or interventionalist should be responsible for the implantation procedure itself. For cluster headache and migraine, criteria for drug-refractoriness have been published (47–49) in addition to the established diagnostic criteria (50), so we should adhere to them. Unfortunately, consensus criteria for further patient selection in invasive neuromodulation have been published only for cluster headache (51) internationally, although some national guidelines have modified them to cover ONS as well (52). Apart from these urgently needed recommendations on uniform and adequate patient selection, the methodology of studies varies substantially, calling for commonly accepted criteria to enhance comparability of studies and to avoid trials with a flawed design in the first place.
Pearls
– Large number of patients successfully treated in unblinded studies with good efficacy in trigeminal autonomic cephalgias and moderate efficacy in open-label studies/case series on patients with chronic migraine – As bilateral implantations are now standard in most centres, it is the method of choice for non-side-locked trigeminal autonomic cephalgias and primarily bilateral headaches – Implantation procedure moderately invasive
Pitfalls
– Ambiguous findings in RCTs on chronic migraine – No feasible sham paradigm – High rate of mostly device-related complications
SPGS
Ictal parasympathetic activation along with sympathetic hypoactivity is a central diagnostic feature of all trigeminal autonomic cephalgias. The preganglionic cranial parasympathetic fibres project from the superior salivatory nucleus to the sphenopalatine ganglion (SPG) in the pterygopalatine fossa, where they form the parasympathetic root and synapse to postganglionic fibres which supply the nasopharyngeal mucosa and the lacrimal and nasal glands and possibly the meningeal vessels and the meninges themselves. Simultaneously, a relevant nociceptive trigeminal activation mainly of V1 branches is observed during attacks, which can lead to further parasympathetic activation via the trigemino-parasympathetic reflex forming a centrally driven “vicious circle” during an attack. Several studies have targeted the parasympathetic branch by blocking the SPG, albeit with temporary relief only (53,54). Alternatively, lesions of the SPG have been effective in chronic cluster headache (55). As a non-destructive approach acute percutaneous SPGS with a removable electrode was examined in a small pilot study in cluster headache (n = five) and aborted 61% of the attacks within minutes (56). Similarly, SPGS in patients with acute migraine attacks (n = 10) aborted or substantially attenuated 50% of beginning attacks (57). Based on these findings, a multicentre randomised double-blind and sham-controlled trial (n = 32) has been conducted to examine the efficacy of acute stimulation in refractory chronic cluster headache with a new kind of implantable microstimulator (see Figure 3) which is powered and controlled transcutaneously by electromagnetic waves (58**). Twenty-eight patients who completed the experimental period reported a relevant pain relief in 67.1% of headache attacks treated with full stimulation compared to only 7.4% of attacks treated with sham and 7.3% with sub-perception threshold stimulation. Pain relief was defined as a decrease of headache intensity from ‘moderate’ or ‘severe’ to ‘none’ or ‘mild’ on the five-point scale 15 minutes after the beginning of stimulation.
Implanted sphenopalatine ganglion (SPG) neurostimulator. (a): anterior-posterior X-ray showing the electrode lead in the pterygopalatine fossa (1), the body of the neurostimulator with the microprocessor and the radio frequency-antenna (2) and the fixation plate (3). (b): Lateral view of implanted SPG stimulator.
Seven patients (25%) out of 28 achieved acute pain relief, 10 (36%) had a reduction of attack frequency of at least 50% from the beginning of stimulation until the end of the experimental period, and two (7%) experienced both. Side effects associated with the implantation technique were frequent and clinically relevant: Eighty-one per cent of the patients reported sensory disturbance after the implantation, mainly affecting maxillary nerve branches in 19 out of 32 patients (59.4%). While they subsided within an average of 97 days in the majority of patients (15 out of 19 patients, 78.9%), four out of 19 patients (21.1%) were still suffering from mild to moderate sensory disturbances when the manuscript was accepted. In contrast, the rate of device-related complications was comparably low.
Although the number of subjects in the experimental period was relatively low and attack frequency was not a primary outcome, SPGS could exert both acute and preventive effects. The underlying pathophysiological rationale is plausible und device-related complications seem to be significantly less problematic than in ONS. On the downside, sensory impairment seems to be a major complication of the implantation procedure that needs further attention.
Judging from the recently published data, the overall complication rate does not exceed that of ONS. Yet, it seems that they tend to decrease over time unlike in ONS. As a further limitation, no long-term safety data are available for SPGS at present.
Lastly, SPGS allows us to study the (patho-)physiology of the cranial parasympathetic system in vivo with the fascinating possibility to provoke attacks by low-frequency stimulation in patients with cluster headache and treat them with high-frequency SPGS as recently shown by a Danish group (59).
Pearls
– Good acute efficacy as shown in RCT – Putative preventive efficacy – Reasonable mechanism of action – Low rate of device-related side effects
Pitfalls
– Relatively low responder rate in experimental period – High rate of mostly transient complications related to implantation procedure – Blinding problematic – Limited to unilateral headaches without history of side-shift
High cervical spinal cord stimulation (hcSCS)
High cervical stimulation of the dorsal column was studied prospectively in a small sample of refractory chronic cluster headache patients (n = seven) with a mean follow-up of 23 months. Continuous stimulation (except for one patient with intermittent use) led to an impressive and immediate reduction of mean attack frequency from 6.0 to 1.4 with a responder rate of 86%. However, dislocation of electrodes, rapid depletion of batteries and lead breakage occurred in five out of seven patients with frequent revisions. As in other unilateral approaches, two patients reported a side-shift of attacks. Although the underlying concept is intriguing and hcSCS is probably more effective than ONS, the high rate of complications strongly argues against its clinical use.
Pearl
– Good efficacy
Pitfalls
– Very high revision rate, technical shortcomings – Low number of patients – Blinding problematic
Vagal nerve stimulation (VNS)
Good response to VNS with a reduction of attack frequency was observed in three out of four patients who received VNS for intractable epilepsy and suffered from comorbid episodic migraine (60); for a review see Jenkins and Tepper (61). These high response rates of more than 50% have been corroborated by anecdotal reports on further patients with migraine (62–64), chronic daily headache (64), chronic cluster headache (63,65) and chronic tension-type headache (66) only – partly with concomitant refractory epilepsy. Recent experimental data in rats show that neuronal firing in the spinal trigeminal nucleus in response to electrical stimulation of the dura is reduced under continuous stimulation of the left cervical vagal nerve (67). Most side effects are generally mild, but cough, dyspnea, vocal cord paresis, and hoarseness can be incapacitating. As the electrodes are wrapped around the nerve during implantation, this can cause permanent adherence to the nerve by formation of scar tissue and makes removal or replacement of electrodes problematic or even impractical. VNS should therefore be limited to the minuscule group of patients with both intractable headache and co-morbid epilepsy. Since VNS has been approved in the United States to treat drug-resistant major depression, VNS could also find a place in drug-resistant chronic migraine and/or chronic daily headache when they are associated with major depression.
Transcutaneous VNS (tVNS) could overcome these limitations. Recently, two devices have been licensed as medical devices in Europe with promising results on experimental pain (68) but limited benefits in two small case series in patients with primary headaches which have been presented in preliminary form (69,70). Ongoing studies in patients with migraine and cluster headache will clarify their therapeutic potential.
Pearls
– High response rate in various headache syndromes – tVNS: non-invasive and easy to use
Pitfalls
– Anectdotal reports only (partly on patients with both epilepsy and primary headaches), no RCTs – Invasive VNS potentially irreversible
Supraorbital nerve stimulation (SNS)
Anectodal reports propose efficacy of SNS alone in trigeminal pain involving V1 (71,72) and in combination with ONS (73). Results of a larger randomised controlled multicentre trial on the efficacy of transcutaneous supraorbital nerve stimulation (tSNS) in 67 patients with episodic migraine suggest efficacy of intermittent stimulation of 20 minutes per day for three months with a significant decrease of 2.06 headache days per month after three months compared to sham stimulation with only 0.32 days (74). The 50% responder rate was significantly greater in the verum (38.1%) than in the sham group (12.1%), and the number of monthly migraine attacks (p = 0.044), headache days (p = 0.041) and acute anti-migraine drug intake (p = 0.007) were significantly reduced in the verum but not in the sham group. There were no adverse events. Despite the lack of a true placebo condition and the relatively moderate reduction of headache days per month, tSNS represents a promising approach.
Pearls
– Positive outcome on attack frequency in RCT on tSNS – Non-invasive (tSNS) – Few adverse effects (tSNS)
Pitfalls
– Blinding problematic – Only little improvement in terms of the absolute number of headache days in RCT
Other non-invasive approaches
All transcutaneous neuromodulatory techniques have the advantage of being non-invasive, incurring a very low risk of device- and procedure-related side effects. In addition, they are generally cheaper (both in terms of device-related costs and costs of the implant procedure). Despite these major advantages, the body of evidence supporting their widespread use is unfortunately even smaller than for most invasive approaches. While implanted devices in general allow both intermittent stimulation to abort attacks and continuous stimulation with potentially better prophylactic efficacy, the use of non-invasive devices is mostly limited to intermittent stimulation. One other major drawback is the lack of true blinding in most approaches and the lack of RCTs.
Transcutaneous electric nerve stimulation (TENS)
A relatively old Cochrane review from 2004 concluded that TENS was effective only in patients with migraine and tension-type headache in combination with other electrical stimulation paradigms. In addition, it was effective in patients with tension-type headache if combined with physical therapy (75). A more recent trial compared thrice weekly low frequency-high intensity TENS sessions in chronic tension-type headache (n = 138) to 50 mg imipramine daily over a period of three months. In both groups VAS scores decreased significantly with significantly higher benefit in the imipramine group (76). Despite an impressive sample size, a true placebo condition was missing and the VAS score as primary outcome is debatable.
Transcutaneous direct current stimulation (tDCS)
tDCS is an established non-invasive technique allowing cathodal (i.e. inhibitory) or anodal (i.e. excitatory) cortex stimulation. Efficacy has been reported especially for chronic pain conditions in small pilot studies (77,78). However, recent meta analyses found that current evidence is insufficient to promote clinical use in painful conditions (79,80). Likewise, a small sham-controlled study could not corroborate a prophylactic effect on attack frequency of cathodal tDCS of the occipital cortex (81). Although anodal tDCS of the motor cortex was found to reduce pain intensity and length of migraine episodes in another small study in patients with chronic migraine (82), several shortcomings limit the clinical utility. Information on headache frequency as a recommended primary outcome criterion in prophylactic studies is missing (83), the sample size of both the stimulation and the sham group (n = eight, respectively, n = five) is critically low and the use of 2 mA stimulation intensity raises questions about effective blinding (84).
Transcranial magnetic stimulation (TMS)
Results for the prophylactic efficacy of repetitive TMS (rTMS) in chronic migraine are controversial (85,86). A consensus on the brain region to target and the stimulation parameters (including stimulation frequency and intensity, but also the choice of magnetic coil) is missing, which makes it problematic to recommend rTMS at present. In addition, the technically demanding setup prevents widespread use at the patients’ homes. Single-pulse TMS (spTMS) over the occipital cortex has been studied in patients with migraine with aura in a large trial (n = 201) with a portable device (87). spTMS aborted acute attacks more effectively within the following two hours than sham treatment if spTMS was applied in the first hour after the aura started. On the downside, there was no real sham paradigm, triptans were more effective and the device was studied only in attacks with aura. Thus, its efficacy in the remaining 70%–90% of migraine patients (those without aura) has yet to be shown.
Conclusion
Selected pitfalls of invasive neuromodulation.

CCH: chronic cluster headache; CM: chronic migraine; DBS: deep brain stimulation of posterior hypothalamus; hcSCS: high cervical spinal cord stimulation; ONS: occipital nerve stimulation; RCTs: randomised controlled trials; SNS: supraorbital nerve stimulation; SPGS: sphenopalatine ganglion stimulation; VNS: vagal nerve stimulation.
Efficacy of invasive neuromodulatory approaches.
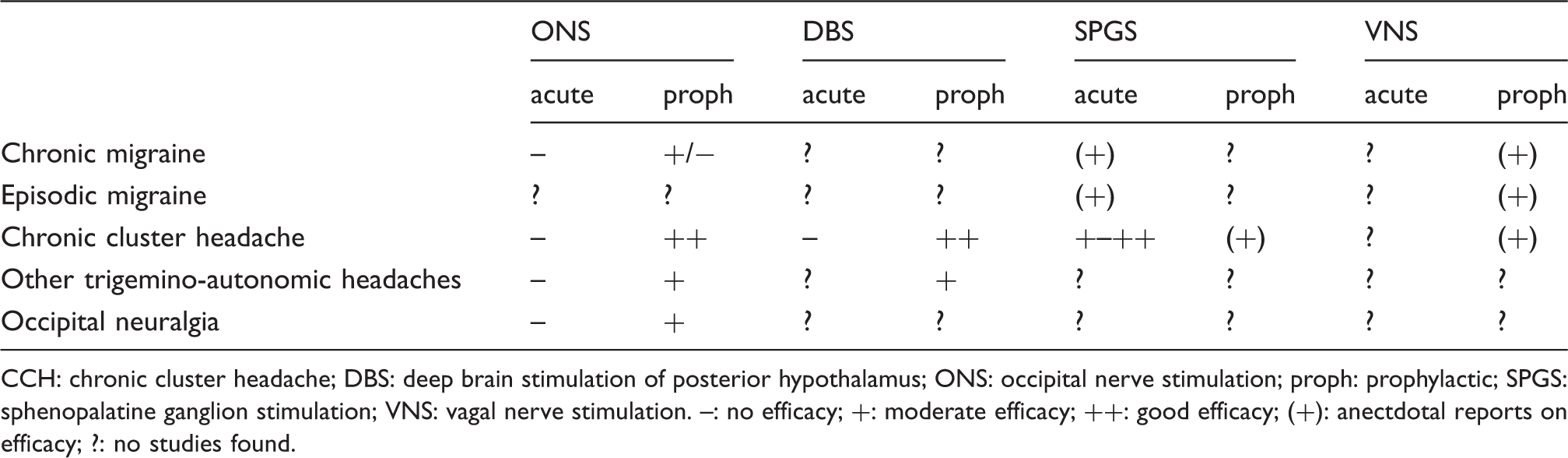
CCH: chronic cluster headache; DBS: deep brain stimulation of posterior hypothalamus; ONS: occipital nerve stimulation; proph: prophylactic; SPGS: sphenopalatine ganglion stimulation; VNS: vagal nerve stimulation. –: no efficacy; +: moderate efficacy; ++: good efficacy; (+): anectdotal reports on efficacy; ?: no studies found.
Open questions for the future
A number of open questions arise from what we currently know about invasive and non-invasive neuromodulation:
The emerging and rapidly growing field of non-invasive devices calls for adequately powered and thoroughly designed randomised sham controlled multicenter studies to prove their therapeutic efficacy. These studies should furthermore help to find their role relative to invasive strategies and should clarify whether they should always be tried before moving on to implantable devices. The phenomenon of “side-shift” seems to be ubiquitous in unilateral neurostimulation of strictly one-sided headache and occurs in up to 36% of the patients (31**). Even in SPGS, contralateral cluster headache attacks partly reoccurred in patients with a history of it regardless of ipsilateral stimulation efficacy (personal communication Anthony Caparso, Autonomic Technologies Inc., 2013). However, it is important to distinguish between a de novo side-shift in patients who have never had contralateral attacks (9,30**,31**) as found in studies with side-shift as an exclusion criterion and those who report reoccurrence or increased frequency of contralateral attacks known to the patient (28,88). Therefore, it is not only necessary to understand the underlying pathophysiological phenomena but also to identify individual risk factors to help patients choose the right approach. Should we consider unilateral approaches such as SPGS in patients with cluster headache who report frequent side-shifts or rather opt for bilateral stimulation such as ONS in these cases? Little is known about individual predictors of efficacy before implantation. Paraesthesia in the receptive field of peripheral nerves is mandatory to achieve clinical efficacy in peripheral nerve stimulation while medication overuse seems to be a negative predictor of efficacy, at least in ONS (43). In contrast, the response to occipital nerve blocks is generally not predictive of good response in ONS (29,30**,42**,89). Given the potential side effects and the relevant costs incurred, identification of further and more individualised markers could be helpful for the affected individual as well as from a socio-economic point of view. Especially, neurophysiological markers could be helpful here. They could help in choosing the approach with the highest likelihood of success in the individual patient to advance towards an individualised treatment. Is “the bigger always the better” when combining different invasive approaches such as SNS and ONS? The combined stimulation of the supraorbital and the occipital nerve effectively reduced pain in seven out of eight patients with chronic migraine for at least 50% (73). As in combined preventive treatment with two or more drugs, potentially higher rates of adverse events have to be weighed against potentially increased efficacy. However, until a larger group of patients can be overlooked, combined techniques should be restricted to controlled trials. The development of “tolerance” to stimulation has been suggested in deep brain stimulation (14**) and can be observed in ONS studies as well. The phenomenon of vanishing therapeutic efficacy over time with the necessity to adjust stimulation parameters deserves further attention, although it is certainly challenging to attribute decreasing therapeutic efficacy to singular factors and separate it from the natural course of the underlying headache disorder. In some studies an externalised test stimulation was performed for as little as five up to 28 days with a positive response as a prerequisite for an internalised permanent stimulation. This could exclude patients with delayed response as reported in the literature. Is it justifiable to provide patients with episodic and medically intractable primary headache disorders (such as patients with cluster headache who suffer from long bouts) with implantable neuromodulatory devices (90) or should this group be limited to non-invasive approaches with proven efficacy? Should patients with drug-refractory stable and long-lasting secondary headache disorders be eligible for neuromodulatory approaches? Interestingly, for both SPGS and tSNS prophylactic efficacy has been suggested from intermittent stimulation, which raises questions about the underlying potential pathophysiological principle. However, these observations will have to be confirmed by additional studies. Implantable neuromodulatory devices especially offer the unique chance to advance our concepts of headache pathophysiology by studying changes during an attack in vivo. As already shown for patients with cluster headache receiving hypothalamic DBS, areas previously confined to neuroimaging and transcranial neurophysiological approaches are now directly accessible even during acute headache attacks. The same holds true for SPGS. Future studies will hopefully teach us more about the interplay of the peripheral cranial nerves with the central nervous system and the cranial autonomic system. Shall we treat non-refractory primary headaches such as frequent episodic migraine with non-invasive neuromodulatory devices rather than with first-line drugs given that their efficacy has been proven?
Recommendations for future studies
The comparability of studies needs to be improved. Consensus criteria for primary and secondary outcomes with a uniform definition of response for studies on acute and prophylactic efficacy of neuromodulatory approaches are urgently needed along with further specifications such as the minimal duration of stimulation adjustment treatment periods and consensus criteria for pharmacological refractoriness. Is a reduction of headache days per month of at least 30% acceptable as suggested by Silberstein and colleagues (37**), or is it too permissive and should we better continue adhering to the recommendations defined for prophylactic drug treatment (i.e. 50%) (83)?
The quality of studies needs improvement. Although even larger case series have been published especially for ONS and DBS, the number of RCTs is negligible. Studies should be adequately powered and meet all currently required criteria for RCTs. No valid conclusions can be drawn from vague inclusion criteria, first and foremost the headache diagnosis itself, which should adhere to the current IHS classification (50). As patients with concomitant medication overuse have worse outcomes than those without overuse in some studies, it is uncertain if they should be excluded from neurostimulation studies. Results from patients with concomitant medication-overuse headache should at least be analysed separately to answer this question. The downside of an increasing dissemination of neuromodulatory approaches could lead to a loss of relevant knowledge. Therefore, patients with invasive neuromodulation should be treated only within studies or at least be followed with the help of nationwide patient registries. As the induction of paraesthesia is regarded as mandatory for peripheral nerve stimulation, effective blinding paradigms remain an unresolved issue. As complications especially in ONS are high (see McGreevy et al. (91) for a recent review), the manufacturers are called on to further improve their devices (such as enhancing the durability of electrodes, leads and battery life or further developing rechargeable microstimulators) and advance implantation techniques including neurosurgical training. As shown by a first retrospective calculation for hypothalamic DBS in chronic cluster headache (92), neuromodulatory approaches can be cost effective despite relatively high initial expenses for the device itself and the implantation procedure. With healthcare budgets tightening both non-invasive and invasive neuromodulatory approaches will have to prove their cost efficiency, which should be considered when designing future studies. Lastly, the success of any neuromodulatory device critically depends on well-orchestrated teamwork involving an experienced surgeon or interventional pain specialist and an experienced headache expert who selects refractory patients after a proper diagnosis according to the International Classification of Headache Disorders-II, recommends the approach most suitable for the individual patient and takes responsibility for post-implant care, which is often the most demanding and time-consuming part. Instead of numerous small sites with low case numbers, specialised centres should be installed guaranteeing sufficient expertise.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
TPJ has received honoraria for lectures and consulting from Autonomic Technologies Inc and MSD Germany.
ML has nothing to declare.
