Abstract
Introduction
Cluster headache is an extremely debilitating and often difficult-to-treat headache disorder characterized by recurrent attacks of excruciating pain associated with cranial autonomic symptoms. Several invasive neuromodulation procedures have been evaluated in the past, but the combination of these procedures to maximize response has not been studied in groups of patients.
Materials and methods
In this single-center, retrospective, observational study, we present the long-term clinical evolution of seven patients suffering from refractory chronic cluster headache (CCH), treated with combined sphenopalatine ganglion (SPG) and greater occipital nerve (GON) invasive stimulation, ipsilateral to the side of the pain, between February 2010 and March 2021. The Visual Analogue Scale (VAS) and Headache Impact Test (HIT-6) were used to monitor the response.
Results
The mean follow-up time was 5.8 years ± 3.4 years (maximum 10.4 years). Six out of the seven patients experienced good-to-excellent initial pain relief and achieved almost complete remission of symptoms afterwards. Mean VAS and HIT-6 score values decreased significantly and steadily during most of the observation period. Electrode migration requiring reintervention occurred in two patients.
Conclusion
Synergistic invasive SPG-GON stimulation is a relatively safe and promising alternative for effective and long-lasting control of CCH. Further studies involving larger samples are warranted.
This is a visual representation of the abstract.
Introduction
Cluster headache is an extremely debilitating headache disorder characterized by recurrent attacks of excruciating pain associated with cranial autonomic symptoms. 1 In most patients, a clear circannual and circadian pattern of affection can be observed, 2 probably related to the involvement of the hypothalamus in the pathophysiology of this condition.3,4 Nonetheless, a few affected individuals exhibit the most disabling form of cluster headache, known as chronic cluster headache (CCH), in which there are no periods of absolute remission, or they are very brief. 1
The treatment of cluster headache remains challenging. In the episodic form, attacks often respond to oxygen, 5 triptans, 6 or ergotamine, 7 bridge therapy with sub-occipital injections of steroids combined with local anesthetics is generally effective for short-term relief, 8 and preventive treatment with verapamil or lithium is commonly beneficial. 9 Additionally, recent trials suggest that galcanezumab might be a viable alternative for some patients. 10 However, therapeutic options are less reliable in the chronic form, where patients are frequently refractory to conventional strategies. 11
In refractory chronic cluster headache patients, various peripheral and central invasive neuromodulation techniques and ablative procedures have been attempted, but the results remain inconclusive. Occipital nerve stimulation (ONS) has shown benefits, though its effects may take several months to manifest.12 In contrast, sphenopalatine ganglion (SPG) stimulation acts more rapidly and offers both preventive and on-demand abortive therapy. However, long-term efficacy remains a concern, as a previous study reported that only 61% of patients maintained a sustained therapeutic response after 24 months. 13 Reflecting these limitations, current guidelines assign no more than a level B recommendation to either approach as standalone therapy. Given these challenges, exploring new strategies that provide both rapid and sustained relief is essential.
To our knowledge, the effects of combined occipital nerve and sphenopalatine ganglion stimulation in cluster headache have not been studied in the past. The rationale behind this association relies on the convergence of fibers from both pathways into the dorsal longitudinal fasciculus and the trigemino-hypothalamic tract, which connect regions in the hypothalamus known to serve as targets for deep brain stimulation and structures in the brainstem that mediate head pain (Figure 1).14–16

Left: Fiber tractography presenting the dorsal longitudinal fasciculus/trigemino-hypothalamic tract in a healthy individual. Middle: Diagrammatic representation of the stimulating electrodes and their respective targets. Right: Anteroposterior skull x-ray depicting the placement of the sphenopalatine (indicated by the red arrow) and occipital (highlighted by the green arrow) stimulating electrodes.
Therefore, it seemed fair to hypothesize that combined ONS and SPGs could synergistically potentiate the modulatory effects indirectly exerted on the hypothalamus by each technique without substantially increasing the risk of complications to the level observed with deep brain stimulation.
In this study, we present the long-term clinical course of a series of patients suffering from refractory CCH (rCCH) who received combined invasive occipital and sphenopalatine ganglion neuromodulation.
Methods
This single-center, retrospective, observational study included seven patients (3F/4M) suffering from CCH, according to the diagnostic criteria of the current International Classification of Headache Disorders, 1 and considered refractory based on the Consensus Statement of the European Headache Federation. 11 Between February 2010 and March 2021, these patients underwent implantation of electrodes for SPG and greater occipital nerve (GON) stimulation ipsilateral to the side of the pain. Patients with uncontrolled hypertension, cerebrovascular disease, congestive heart failure, cancer, or severe psychiatric disorders were considered ineligible for the procedure.
The electrodes were implanted in a single surgery under general anesthesia. For the occipital nerve, a four-lead stimulating electrode (Medtronic Resume, Medtronic, Minneapolis, USA) was placed through a 2 cm midline incision at the C1 level, which was chosen for optimal access and secure anchorage. 17 For the sphenopalatine ganglion, an implantable 8- to 16-channel impulse generator (Medtronic Itrel3, Medtronic, Minneapolis, USA) was inserted via an infrazygomatic puncture, reaching the pterygoid fossa. 18 No preoperative tests were performed.
Over the next 72 h post-implantation, stimulation parameters were adjusted based on paresthesia thresholds. For the SPG lead, settings included an amplitude of 1–2 V, a pulse width of 60 μs, and a frequency of 140 Hz. For the GON lead, parameters were set at a pulse width of 350 μs, an amplitude of 4–9.5 V, and a frequency of 130 Hz.
Patients were followed every 3 months after surgery until the 90th month, and then every 5 months. The Visual Analogue Scale (VAS) and the Headache Impact Test (HIT-6) scale were used to monitor treatment response as primary and secondary outcome measures. In case of missing data (<2%), linear interpolation was applied. Data from subsequent visits were compared to baseline values using the last observation carried forward (LOCF) method and repeated measures ANOVA correcting for multiple comparisons with Dunnett's test. A p-value below 0.05 (adjusted for multiplicity) was considered statistically significant.
The study was conducted following the Declaration of Helsinki and approved by the Ethics Committee of the Cenit Foundation. Written informed consent was not required for this study due to specific legal and ethical guidelines outlined in Law 25.326, Article 11(d) and Article 28, and Ministry of Health Resolution 1490/07, Chapter 4, Section 3.
For the left panel in Figure 1, we used anonymized data from a healthy participant enrolled in a separate, ethically approved headache study (protocol NO-2024-00121794-UNC-CE#HNC, Ethics Committee of the School of Medicine, National University of Córdoba, Argentina), for which appropriate permission was granted. Data were processed with DSI Studio.19,20 A Diffusion Tensor Imaging scheme was used, with 32 diffusion sampling directions, a b-value of 1000 s/mm², an in-plane resolution of 2 mm, and a slice thickness of 2 mm. Regions of Interest were placed at Montreal Neurological Institute coordinates (–1, −46, −71) and (–2, −6, −11). Anisotropy and angular thresholds, as well as step size, were randomly selected within predefined ranges. Tracks shorter than 30 mm or longer than 200 mm were discarded.
In Argentina, compassionate-use surgical procedures such as combined SPG and GON stimulation may be authorized when conventional treatment options have failed. In all cases included in this study, the proposed intervention was individually evaluated and approved by the corresponding medical auditor based on the treating physician's rationale and documentation of treatment refractoriness.
Results
Baseline patients’ characteristics are summarized in Table 1. The mean follow-up time was 5.8 years ± 3.4 (maximum 10.4 years and minimum 1.3 years). Six out of the seven patients (86%) experienced good-to-excellent initial pain relief, defined as a ≥ 50% reduction in VAS scores compared to baseline, together with marked clinical improvement in attack severity and functional impact as assessed by the treating physicians. Almost complete remission of symptoms was achieved in most cases, defined as the complete absence of cluster attacks or only rare, very mild episodes without significant functional impact (Supplemental Figure 1). One patient stopped requiring stimulation 15 months after the placement procedure, and symptomatic remission was sustained until the 23rd month when the device had to be restarted. Another patient reported no perceived benefit from the therapy. The specific cause of this outcome remains unclear. However, it is worth noting that this perception may have been influenced by a generally negative or skeptical attitude, as the patient later requested a battery replacement when the device was depleted. To better illustrate the individual variability in treatment response, Supplemental Figure 1 presents the evolution of VAS and HIT-6 scores for each patient across the follow-up period. Regarding HIT-6 results, mean values decreased to the “Little or no impact” severity level already during the first control after surgery and remained close to the border with “Moderate impact” during subsequent visits (F3, 447 = 20.68, p = 0.003) (Figure 2). 21 Similar results were observed in the VAS, where a stable descent of 4 to 5 points on average was achieved (F4, 279 = 25.67, p = 0.001), from “very severe” to “moderate” pain (Figure 3). 22 Electrode migration requiring a reintervention occurred in two patients: one at 12 days and the other at 3 months after surgery. Regaining the initial benefits after reimplantation happened in one case. One patient passed away during the follow-up due to an unrelated cause (a traffic accident). Two patients reported pain at the site of the subclavicular pulse generator implantation. In one of them, a seroma developed and resolved spontaneously within one month, after which electrical stimulation was initiated without further issues. Another patient reported bothersome occipital paresthesia, which did not result in therapy discontinuation.

Mean and 95% confidence interval Visual Analogue Pain Scale scores evolution throughout the observation period. The asterisks denote statistically significant differences with respect to the baseline.
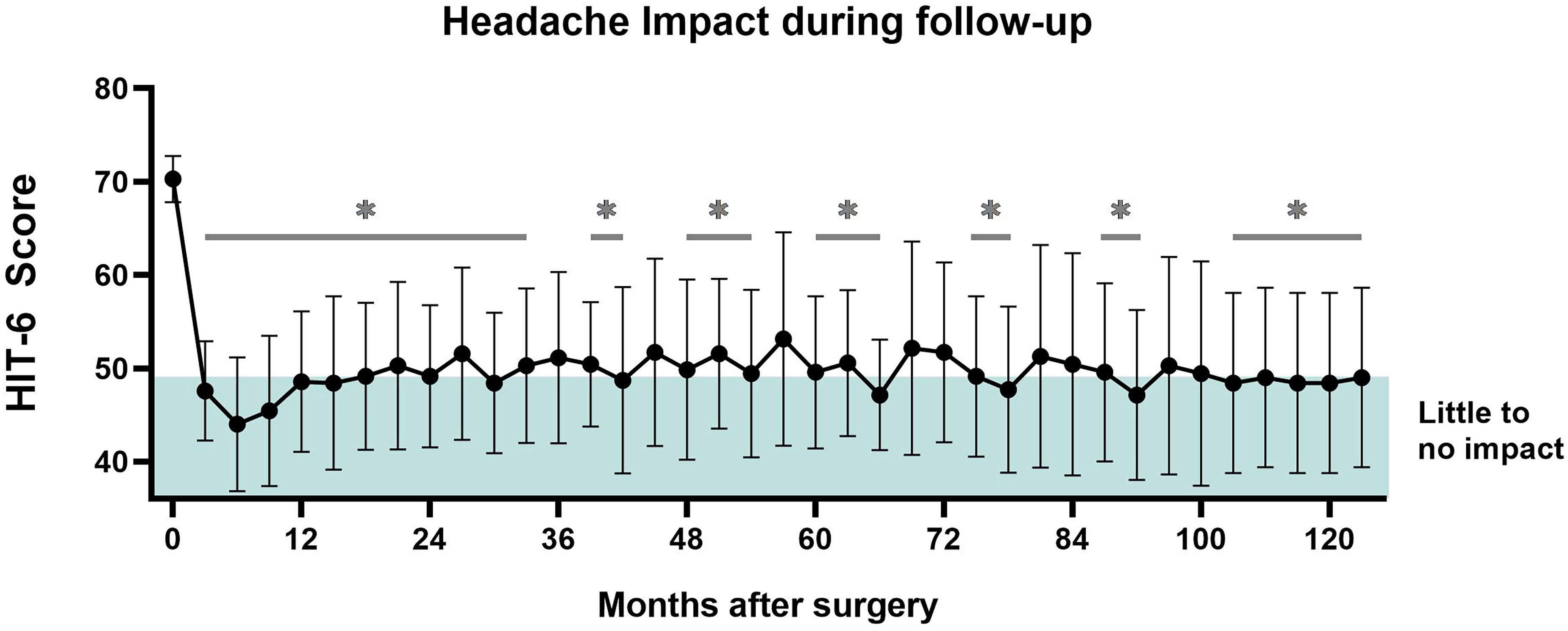
Mean and 95% confidence interval Headache Impact Test-6 scores evolution throughout the observation period. The asterisks denote statistically significant differences with respect to the baseline.
Summary of baseline data, prior treatment history, and comorbid conditions in patients undergoing combined sphenopalatine ganglion and greater occipital nerve stimulation.

HIT-6: Headache Impact Test; VAS: Visual Analogue Scale.
Discussion
Here we report, for the first time, that combined invasive SPG and GON neurostimulation significantly and enduringly improves CCH symptoms in a series of refractory patients. Moreover, the number and severity of complications presented indicate that this approach is relatively safe compared to others of its kind, resulting in a convenient risk-benefit profile. These findings are discussed in detail below.
Hypothalamic involvement in cluster headache has been robustly established.3,4 The clinical features of this condition initially raised suspicion, which was later supported by well-designed neuroimaging studies. Subsequently, deep brain stimulation (DBS) has been attempted as a therapeutic strategy in a limited number of highly refractory patients, targeting various regions in the hypothalamus and midbrain. While uncontrolled studies mostly showed positive results, 23 the only randomized clinical trial conducted thus far yielded negative findings. 24 One potential explanation for contrasting results could be differences in electrode placement, 25 as this technique modulates relatively restricted areas. Treatment efficacy is likely influenced by the direct or indirect modulation of a broader subset of structures. 26 DBS also carries significant risks, with numerous serious adverse events reported, including death due to intracerebral hemorrhage in one patient. 27 As an alternative, a combined approach like the one we present could offer advantages. By indirectly modulating hypothalamic function and nearby structures through a less invasive peripheral method, multiple hypothalamic and midbrain areas can be simultaneously reached via the widespread connections of the stimulated pathways. This approach maintains a substantially safer risk profile compared to DBS.
Regarding peripheral neurostimulation techniques for CCH, both SPG and GON have shown positive results in controlled trials (although without a sham for GON).28,29 However, ≥ 50% responder rates (in terms of attack frequency) were only 36% for SPG and 44.6% for GON in those studies, suggesting that there might be room for improvement with the use of each of these two techniques as standalone treatments. Indeed, our results indicate that a combination of both approaches might increase the percentage of responders, a finding that would need to be corroborated in a proper randomized, sham-controlled trial. Nonetheless, given the prolonged period of observation that we recorded, it appears safe to say that positive results observed with this combined approach are stable over time.
To date, three case reports have disclosed favorable outcomes following combined stimulation in rCCH patients. One case combined non-invasive vagus nerve stimulation with DBS, 30 another combined GON stimulation with supraorbital nerve stimulation, 31 and a third—similar to our study—reported combined SPG and occipital nerve stimulation (ONS), 32 published shortly after we presented our preliminary results. 33 Although no studies have directly evaluated the association of SPG and GON stimulation, a sequential approach for the surgical treatment of cluster headache involving both these targets has been presented in the past. 34 In that series, patients subsequently underwent radiofrequency ablation of the SPG, bilateral stimulation of the occipital nerves, and finally, deep brain stimulation of the ipsilateral posterior hypothalamus if the preceding procedure was ineffective. 34 We consider that our simultaneous implantation of both electrodes in a single surgical procedure may offer advantages over this sequential protocol. By reducing periprocedural and anesthesia-related risks, shortening recovery time, and minimizing hospital stays, this approach could enhance patient safety and overall outcomes. Additionally, these benefits translate into significant cost savings by reducing both direct and indirect healthcare expenses.
Our study has strengths and limitations worth considering. First, as previously mentioned, this is the first case series of CCH patients treated with combined SPG and GON stimulation that we are aware of and, aside from isolated case reports,31,35 a pioneer in reporting results from combined neurostimulation in this condition. The long-lasting observational period is possibly a major positive feature of our analysis as it limits bias secondary to fluctuations in disease severity, which may affect clinical trials. 36 Regarding limitations, although comparable to other series and not trivial considering the epidemiology of this disease, the sample size is a drawback that hampers drawing firm conclusions from our study. Additionally, even though we evaluated headache severity and impact, the number of attacks, which is customarily quantified in this sort of analysis, was not registered. This decision was made because patients were mostly unwilling to collaborate by providing calendar-based registries, and because of its nature, recall bias could largely affect this variable. In future studies, refining stimulation parameters and comparing different types of electrodes could be valuable. Patients should be closely followed during the first months after surgery due to the risk of lead migration.
Conclusions
Synergistic invasive SPG-GON stimulation appears to be a relatively safe and promising alternative for effective and long-lasting control of CCH. Further studies involving larger samples are warranted.
Article highlights
Innovative approach: First study evaluating combined SPG and GON stimulation in a group of refractory CCH patients.
Sustained relief: Six out of seven patients experienced significant pain reduction, with lasting symptom remission.
Long-term outcomes: Mean follow-up of 5.8 years, confirming the potential of this neuromodulation strategy for CCH.
Supplemental Material
sj-docx-1-rep-10.1177_25158163251355554 - Supplemental material for Long-term effectiveness of combined unilateral sphenopalatine and occipital nerve stimulation in patients with refractory chronic cluster headache
Supplemental material, sj-docx-1-rep-10.1177_25158163251355554 for Long-term effectiveness of combined unilateral sphenopalatine and occipital nerve stimulation in patients with refractory chronic cluster headache by Juan Carlos M Andreani, Osvaldo Bruera, Marco Lisicki, Diego Bashkansky and Fabián Piedimonte in Cephalalgia Reports
Footnotes
Acknowledgements
Use of artificial intelligence
ChatGPT was used to improve the readability and language of the article. All authors carefully reviewed and edited the result and approved the final version of this article.
Authors’ contributions
All authors contributed significantly to the conception, design, execution, or interpretation of the study. Each author has reviewed and approved the final version of the manuscript and takes full responsibility for the content.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted following the Declaration of Helsinki and approved by the Ethics Committee of the Cenit Foundation. Written informed consent was not required for this study due to specific legal and ethical guidelines.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
