Abstract
Short-lasting unilateral neuralgiform headache with autonomic symptoms (SUNA) is a rare form of trigeminal autonomic cephalalgia. SUNA is frequently associated with a trigeminal neurovascular conflict and rarely occurs secondary to other intracranial pathology. We report a patient with SUNA that was associated with ipsilateral meningeal inflammation caused by idiopathic hypertrophic pachymeningitis (HP). During the 9-year follow-up, she experienced multiple episodes of SUNA, most of which occurred during exacerbations of the pachymeningitis. This is the third case of SUNA associated with HP reported in the literature. Based on this observation, we suggest that in patients presenting with SUNA, besides dedicated magnetic resonance imaging (MRI) of the trigeminal nerve, gadolinium-enhanced brain MRI should be performed to rule out HP.
Introduction
Short-lasting unilateral neuralgiform headache with autonomic symptoms (SUNA) is one of the most infrequent forms of trigeminal autonomic cephalalgia (TAC). Rarely, SUNA and other forms of TAC are secondary to intracranial lesions. 1,2 Here, we report a case of SUNA secondary to idiopathic hypertrophic pachymeningitis (HP).
Case
In 2008, a 22-year-old woman presented with right-sided attacks of severe unilateral stabbing headache with ipsilateral nasal congestion and miosis. The pain was located in the right temporal region and occurred almost exclusively at night. Single stabs lasted 2–20 s and initially tended to occur with such a high frequency that the pain was perceived in a sawtooth pattern. 3 The frequency of individual stabs varied, ranging from multiple stabs per minute to several stabs per day, and tended to decrease over time until these attacks spontaneously resolved after approximately 7 weeks.
Neurological examination and unenhanced brain magnetic resonance imaging (MRI) were unremarkable. Her symptoms met the International Headache Society diagnostic criteria of SUNA. 4 In 2011, she presented again with a similar headache episode, which now occurred throughout the day instead of predominantly at night, and diplopia, followed by subacute right-sided visual loss. On neuro-ophthalmological examination, visual acuity of her right eye was 1/300; there was no oculomotor, abducens or trochlear nerve palsy. Gadolinium-enhanced brain MRI including a three-dimensional (3D) T1 sequence showed marked meningeal hypertrophy and enhancement, most pronouncedly along the right cerebral hemisphere, with involvement of the meninges near the origin of the right optic nerve (Figure 1). Dedicated imaging of the trigeminal root revealed no neurovascular conflict. Cerebrospinal fluid (CSF) examination and extensive blood tests were unremarkable. She was suspected to suffer from an inflammatory pachymeningitis, possibly sarcoidosis, though no systemic signs were found. She was treated with methylprednisolone 1000 mg daily for 3 days, followed by oral prednisone 60 mg daily which was tapered over the course of several months. Her headache greatly improved within 3 days after initiation of methylprednisolone treatment. Follow-up MRI scans showed improvement of meningeal hypertrophy, though her visual acuity improved only slightly to 1/60. In 2012, she presented with a new, similar episode of SUNA, at which point she was referred to our tertiary clinic. There were no new neurological deficits, but the MRI revealed a slight increase in meningeal hypertrophy and enhancement. CSF examination was again unremarkable (opening pressure 15 cm H2O, leucocytes 1 × 106 cells/L, glucose 3.8 mmol/L, total protein 0.29 g/L, no oligoclonal immunoglobulin G (IgG) bands, IgG index 0.46). Exhaustive diagnostic tests for systemic and central nervous system inflammatory (including sarcoidosis), infectious and (para)neoplastic disorders were also unremarkable, including a whole-body 18-fluoro-deoxyglucose positron emission tomography scan which showed no abnormal uptake in lymph nodes, organs or other tissues and serum IgG4 concentration. A dural biopsy was not performed. Because no specific cause of the pachymeningitis could be found, she was diagnosed with idiopathic HP. She was again treated with methylprednisolone, followed by oral prednisone 60 mg daily, after which the meningeal hypertrophy and enhancement again improved; her SUNA also improved following treatment, though she did occasionally have additional SUNA episodes varying in duration from several days to 1 week without new neurological deficits. The prednisone was tapered over a prolonged course of 2 years. In 2014, approximately 6 months after the prednisone was discontinued, she experienced a new SUNA episode, this time with left temporal stabbing headache and a left-sided ptosis, diplopia with impaired left ocular elevation and mydriasis due to a partial oculomotor lesion with enhancement of this cranial nerve on contrast-enhanced MRI. She was treated with a third course of methylprednisolone after which her symptoms resolved within 72 h, followed by oral prednisone 50 mg daily and methotrexate 10 mg once per week. The prednisone was tapered over a course of 1 year. In 2016, she developed left-sided sudden deafness (20 dB hearing loss) without an increase in meningeal enhancement, nor enhancement of the vestibulocochlear nerve on 3D gadolinium-enhanced MRI, which resolved almost completely following treatment with 60 mg prednisone daily for 7 days (in accordance with the local sudden deafness protocol). In 2017, she again experienced an episode of SUNA with similar features which resolved spontaneously after several weeks. There were no new neurological deficits, no increase in meningeal enhancement on MRI and no new abnormalities on repeated audiometry and neuro-ophthalmological examinations. Regarding symptomatic treatment, paracetamol and non-steroidal anti-inflammatory drugs were completely ineffective. In 2012, lamotrigine 50 mg twice a day possibly had some effect in preventing SUNA attacks but was discontinued after 3 months because she preferred to use as little medication as possible. The patient provided written informed consent for the publication of this article.
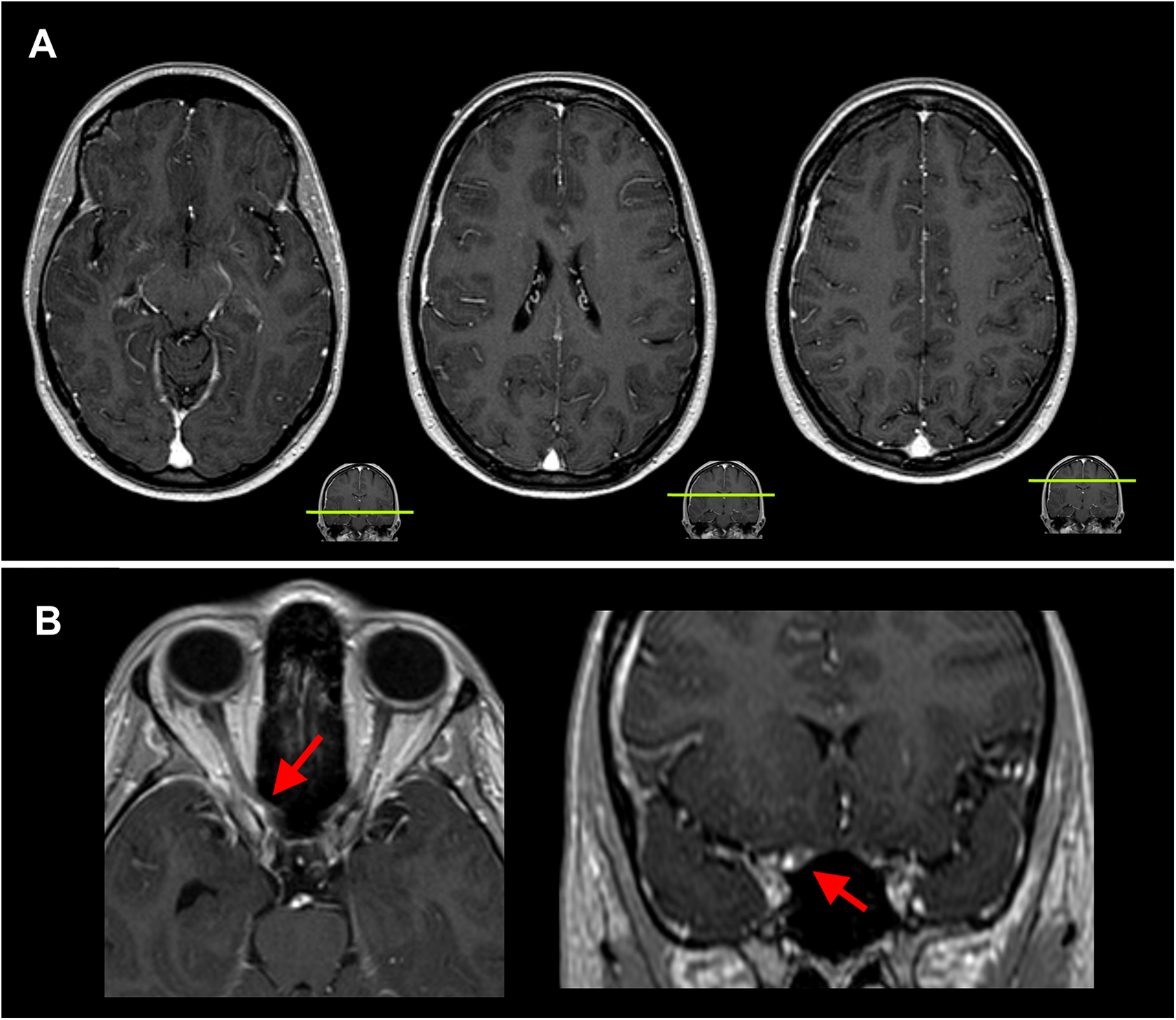
Brain MRI showing meningeal hypertrophy and enhancement. Representative slices of the gadolinium-enhanced T1 sequence are shown. (a) Hypertrophy and enhancement of meninges along the right convexity. (b) The arrow points to a focal hypertrophic meningeal nodule that compresses the right optic nerve just prior to entering the orbit.
Discussion
We describe a rare case of SUNA that is associated with idiopathic HP, with a close temporal relationship between the onset of pain and inflammation and side concordance between the unilateral pain and the location of the most pronounced inflammation. One prior case report described two cases of SUNA associated with idiopathic HP, which makes the present case the third reported in the literature. 2 We provide a unique follow-up of 9 years, whereas the two previous cases were followed up for less than a year. From the 9 years of follow-up in our case, we can conclude with a high level of certainty that HP is the correct diagnosis, as the clinical course fits idiopathic HP and has provided no arguments for an alternative diagnosis. However, it should be noted that idiopathic HP is a diagnosis per exclusionem (a comprehensive summary of the differential diagnosis is provided elsewhere) 5 and probably represents a spectrum of currently unknown immune diseases. For example, IgG4-related HP was first described in 2009 and before then, such patients would have been classified as idiopathic. 6 We cannot rule out IgG4-related HP in the present case, as a meningeal biopsy (which is the gold standard) was not performed, but this seems less likely based on the lack of involvement of other organs systems after 9 years of follow-up and the lack of elevation of serum IgG4. Another advantage of having a 9-year follow-up is that our case shows that a new SUNA episode can be a warning sign of new meningeal inflammation, as was the case in several episodes with new neurological deficits, but that a new SUNA episode can also occur without new neurological deficits and without new visible signs of inflammation on MRI, as was the case in 2017.
SUNA is idiopathic in the majority of cases, but several reports have described isolated cases of SUNA secondary to an epidermoid cyst at the cerebellopontine angle, 7 multiple sclerosis with extensive lesions in the brain stem and middle cerebellar peduncle, 3 vertebral artery dissection with infarction of the medulla oblongata, 8 post-traumatic lesions 9 and a SCN9A mutation. 10 Short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT), a condition that is closely related to SUNA (it has been suggested that SUNCT and SUNA are clinical phenotypes of the same syndrome 11 ), has been described secondary to pituitary lesions 3 and a range of posterior fossa lesions. 1 Furthermore, a literature review of 222 cases of SUNA/SUNCT reported vascular compression of the trigeminal nerve in 16.9% of cases, 11 and it was argued that this might be an underestimation because many reports did not clarify whether detailed images of the trigeminal nerve root were made. In the current case, dedicated imaging of the trigeminal root revealed no neurovascular conflict. The involvement of the trigeminal nerve in many of these cases of secondary SUNCT/SUNA fits the current notion that the trigeminal–autonomic reflex (activation of the trigeminocervical complex resulting in cranial autonomic outflow) plays an important role in the pathophysiology of trigeminal autonomic cephalalgias. 12 In the current case of SUNA secondary to HP, compression of the trigeminal nerve due to inflamed meninges seems the most likely cause, though this could not be demonstrated on repeated high-resolution gadolinium-enhanced 3D T1 sequences, which might be due to limited sensitivity of MRI for detecting small and localized foci of inflammation. Alternatively, it might be hypothesized that nociceptive input in the ophthalmic division of the trigeminal nerve from the inflamed meninges activates the trigeminal–autonomic reflex in this case. Of note, we found no cases in the literature that suggest a reversed causality, that is, meningeal inflammation with cranial nerve deficit following SUNA or SUNCT. An important lesson that can be learned from this case is that in patients with SUNA, an underlying (idiopathic) pachymeningitis can initially be missed when performing brain MRI without contrast-enhanced sequences. Based on this observation and the previously described cases of SUNA secondary to intracranial lesions or vascular compression of the trigeminal nerve, we would suggest performing contrast-enhanced brain MRI and dedicated MR angiography of the trigeminal nerve in all patients with SUNA.
Clinical implications
We report a rare case of SUNA associated with ipsilateral idiopathic HP.
During the 9 years of follow-up, recurrent episodes of SUNA were associated with exacerbations of meningeal inflammation in most but not all the episodes.
In patients with SUNA, performing MRI is mandatory and should include a gadolinium-enhanced sequence to rule out HP.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
