Abstract
Aim
To evaluate the effectiveness and tolerability of non-invasive vagus nerve stimulation (nVNS) as acute or preventive treatment, or both, in a cohort of trigeminal autonomic cephalalgia (TAC) patients.
Methods
A service evaluation retrospectively included patients with TACs between January 2014 and February 2025 who had used, or currently use, nVNS. Data were collected from clinical letters. Data are presented as descriptive statistics analysis and non-parametric tests were performed.
Results
In total, 108 patients were included, 74 patients with cluster headache (CH), 10 with paroxysmal hemicrania, 15 with hemicrania continua, four with short-lasting unilateral neuralgiform headache with conjunctival injection and tearing (SUNCT), three with short-lasting unilateral neuralgiform with cranial autonomic symptoms (SUNA) and two with an undifferentiated TAC. Overall, 70 patients considered nVNS useful over a median time using nVNS of 47 (interquartile range = 18–66) months. The median time of use in patients who did not find nVNS useful was 7 (interquartile range = 4–12) months. Twenty-three patients reported an adverse event (AE), while no serious treatment-related AEs occurred. Fifty-nine patients withdrew from using the device, including 11 patients that initially reported nVNS as useful. All groups considered nVNS more useful as preventive, while cluster headache and SUNCT/SUNA patients also considered it useful as acute treatment.
Conclusions
Our findings complement previous evidence of the effectiveness and tolerability of nVNS in CH in addition to other forms of TACs. Interestingly, nVNS seems to be more effective as preventive rather than as acute treatment in our cohort.
This is a visual representation of the abstract.
Introduction
The trigeminal autonomic cephalalgias (TACs) are a group of primary headache disorders listed in the International Classification of Headache Disorders, 3rd edition that include cluster headache (CH), paroxysmal hemicrania (PH), hemicrania continua (HC), short-lasting unilateral neuralgiform headache with conjunctival injection and tearing (SUNCT) and short-lasting unilateral neuralgiform with cranial autonomic symptoms (SUNA) (1).
Non-invasive vagus nerve stimulation (nVNS) is considered an option for the treatment of primary headache disorders, especially for patients with debilitating and refractory conditions such as TACs. Open label use indicated that it would be useful for both the acute and preventive treatment of cluster headache (2). Two sham-controlled trials demonstrated significant and clinically meaningful effects of nVNS as acute treatment in episodic CH (ECH), but not chronic cluster headache (CCH) (3,4). An open label study against standard-of-care demonstrated the utility of nVNS for preventive treatment in chronic cluster headache (CCH) (5), which persisted over time (6). In all clinical trials, nVNS was considered a safe and well-tolerated treatment (2–6). Real-world data also suggests that nVNS is clinically beneficial for both acute and preventive treatment (7), and is a safe treatment for CH patients. The most commonly reported adverse events (AEs) are application site discomfort or local pain (8). Cost-effectiveness studies favor using nVNS as a treatment option, especially in CCH (9). For this reason, the National Institute for Health and Care Excellence (NICE) guidelines positioned nVNS as adjunctive therapy alongside standard treatment for CH patients (10).
However, there is a relative lack of data concerning less prevalent TACs such as PH, HC, SUNCT and SUNA. Two cases of HC who could not tolerate indomethacin, and who had previously responded to occipital nerve stimulation with a microstimulator (11), were reported to respond to nVNS (12). In an audit of the medical records of 15 patients with indomethacin-sensitive headache, seven of nine patients with HC reported a reduction in the severity of continuous pain and four of six patients with PH, reported benefit in attack duration, severity and frequency. These data suggest nVNS may be an alternative or adjunctive therapy in patients with PH or CH (13). A case-series of eight patients with chronic PH reported a potential use of nVNS as an alternative preventive treatment to indomethacin (14). An open-label, prospective clinical audit, that included four patients with HC and two with SUNA, showed a meaningful improvement in two of the HC patients, with a 73% and 80% reduction in the number of headache exacerbations per month as well as a 18% and 58% reduction in pain scores for these two patients (15). A more recent review concluded that nVNS is well tolerated and efficacious in patients with indomethacin-responsive headaches, such as HC and PH (16). Given interindividual variability and latency of response to preventive treatment with nVNS in primary headache disorders, it is recommended that nVNS should be trialled for at least three months to evaluate efficacy (16).
Here, we evaluate the effectiveness and tolerability of nVNS as acute or preventive treatment, or both, in a cohort of TAC patients to add more information regarding rare forms of TACs.
Methods
We performed a service evaluation, which does not require Research Ethics Committee review in the UK (http://www.hra-decisiontools.org.uk/research). Patients included were seen in a tertiary headache center between January 2014 to February 2025, selecting the patients with a TAC diagnosis, who had used, or currently use, nVNS. We retrospectively collected data from structured letters documenting clinical encounters from trained headache clinicians. We included patients’ demographic features, such as sex and age, and clinical data regarding TACs subtype diagnosis and attack frequency, duration and intensity, the coexistence of other headache disorders fulfilling International Classification of Headache Disorders, 3rd edition (ICHD-3) criteria (1), psychiatric comorbidities, such as generalized anxiety disorder or major depressive disorder, number of previous preventives tried and number of concomitant preventives use, number of nVNS stimulations administered, nVNS use as an acute or as a preventive treatment, nVNS treatment response, period of time under treatment, AEs and reasons for withdrawal. Patients were instructed to apply the device to the neck in the pain side for two stimulations of two minutes each up to three times a day for preventive setting and tailored to individual needs for acute treatment. The treatment response with nVNS was considered clinically meaningful if the patient reported a decrease in the frequency, or duration, or both, of their attacks after a minimum of eight weeks of using nVNS. This decrease needed to be quantified and documented by the medical team in the follow-up letter after nVNS initiation, in terms of the frequency or duration of TAC attacks. Improvements reported in other concomitant primary headaches were excluded and clinical data from patients that could not clearly distinguish their TAC headache from other headache were excluded. Patient groups were organized based on different TAC diagnoses, and the patients with CH were divided into two subgroups: those with ECH and CCH. We excluded patients who initiated nVNS but for whom we did not have follow-up data.
Statistical analysis
The sample size was a convenience sample of those who had used nVNS. Descriptive analysis of categorical variables is presented in total number and percentages and continuous variables as mean (SD) or median (interquartile range (IQR)), as appropriate for their distribution. Non-parametric tests were used to do comparisons among groups and subgroups. For categorical variables, we used the chi-squared test and for continuous measures Mann–Whitney and Kruskal–Wallis tests, when the groups were independent, and the Wilcoxon signed rank test otherwise. Logistic binary regressions were performed to determine if the patient's characteristics or comorbidities influence nVNS treatment response and to study the influence of previous preventive failures and nVNS withdrawal. The results were presented as odds ratios (OR) with 95% confidence interval (CI). We conducted a simulation to estimate the theoretical power of the study to detect meaningful effects. The simulation indicated that the study may not have sufficient power to detect an effect of psychiatric comorbidities; however, for other confounders, the study had greater than 80% power to detect a statistically significant effect. p < 0.05 was considered statistically significant, and was adjusted for multiple comparisons using a Bonferroni correction. All statistical analyses were performed with IBM, version 29 (IBM Corp.).
Results
Population characteristics
We included 108 patients, 62 males (57%) and 46 females (43%) with a mean (SD) age of 53 (13) years. Seventy-four patients with CH were split between 27 ECH and 47 CCH, 10 with PH, 15 with HC, four with SUNCT, three with SUNA and two with an undifferentiated TAC (Figure 1).
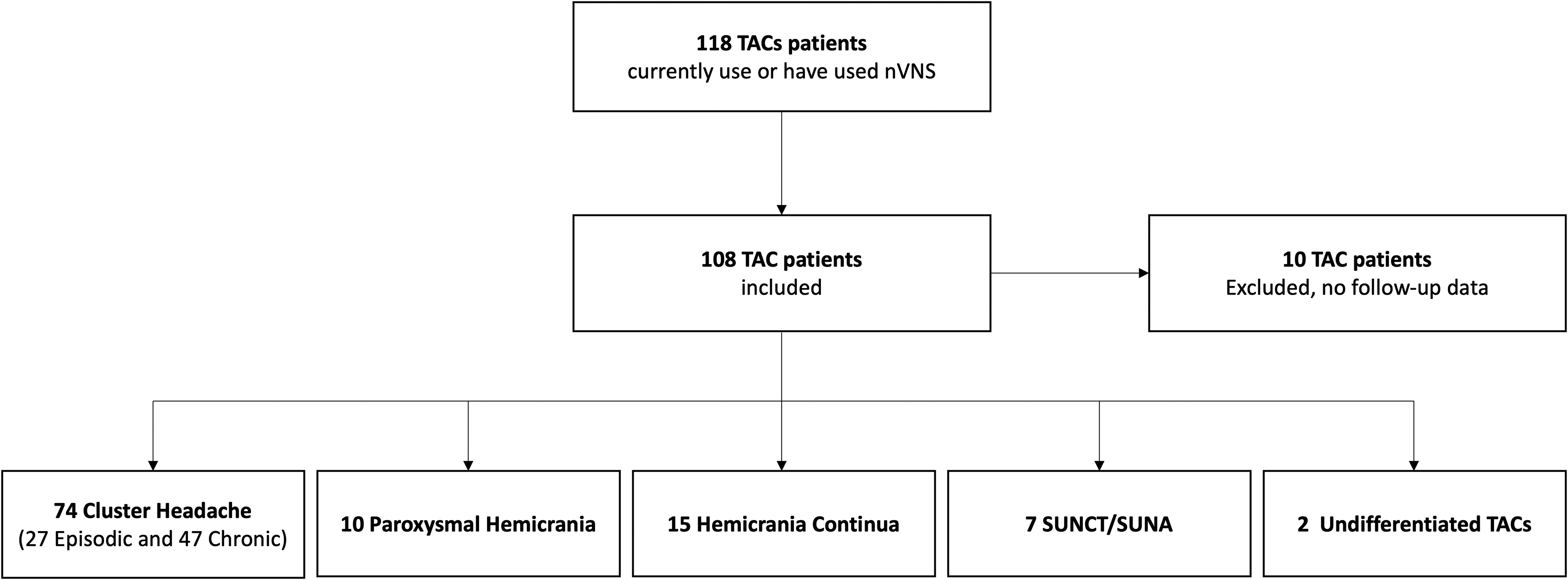
Population characteristics.
The median number of previous preventive treatment failures was 5 (IQR = 3−5) and the median number of concomitant preventives was one (IQR = 0−1). Sixty-eight (63%) patients had previously tried preventive treatment with verapamil, 64 (59%) previously had a greater occipital nerve block, 47 (44%) patients tried indomethacin, 38 (35%) patients tried melatonin, 29 (27%) patients tried steroids, 18 (17%) patients tried lithium, 11 (10%) patients tried dihydroergotamine infusion, eight (7%) patients tried lamotrigine and two (2%) patients had tried galcanezumab.
In Table 1 we summarize the demographic and clinical data by TAC diagnosis groups before the initiation of the neuromodulation treatment.
Data summarized by the different TACs diagnoses at baseline.

AE: adverse event; CH: cluster headache; HC: hemicrania continua; IQR: interquartile range; N: number; nVNS: non-invasive vagus nerve stimulation; PH: paroxysmal hemicrania; SUNA: short-lasting unilateral neuralgiform with cranial autonomic symptoms; SUNCT: short-lasting unilateral neuralgiform headache with conjunctival injection and tearing; TAC, trigeminal autonomic cephalalgia; UND: undifferentiated.
The Wilcoxon signed rank test were used to evaluate the difference in attacks frequency and duration after nVNS treatment (significance at: *p < 0.05; **p < 0.01; *** p < 0.001).
Kruskal–Wallis test.
Fisher's exact test.
Comorbid diagnoses
In the overall study population, 62 patients (57%) had another concomitant ICHD-3 headache diagnosis, 60 patients (96%) were diagnosed with migraine, one patient (2%) with hypnic headache and one with trigeminal neuralgia (2%) Regarding the coexistence of another primary headache and TAC groups: 41 (55%) cluster patients also had a diagnosis of migraine and one patient (1%) hypnic headache. Six patients (60%) with PH had migraine and one patient (10%) had a previous history of trigeminal neuralgia. Five (33%) HC patients, all seven SUNCT/SUNA patients and one patient (50%) with an undifferentiated TACs also had a migraine diagnosis. There was no difference in the presence of comorbid primary headache between the TACs (χ2 = 19.557, d.f. = 12, n = 108, P = 0.076).
In terms of psychiatric comorbidities, 11 patients (10%) had at least one psychiatric comorbidity, based on documentation of previous psychiatric evaluation, including eight patients (7%) with depression and four patients (4%) with anxiety. That represented five (6.8%) cluster patients with depression and two (3%) with anxiety, one patient (10%) with PH and depression, two HC patients (13%) with anxiety and one HC (7%) and one SUNCT/SUNA patient (14%) with depression.
nVNS treatment use
In patients included, treatment use was analyzed at clinical follow-up after a minimum of 8 weeks under nVNS and the median nVNS time treatment in our population was 26 (IQR = 5–59) months. Overall, 70 (65%) patients considered nVNS useful during a median time using the device of 47 (IQR = 66–18) months. The minimum period of use was two months and the maximum period of use was 157 months. The timing of assessing nVNS usefulness varied, with a range of three to six months. The majority (59 patients (84%)) found this treatment useful as a preventive treatment and 33 patients (47%) also found it useful as acute treatment. The median number of stimulations a day for preventive treatment was 6 (IQR = 6–9). The number of stimulations per day was not associated with treatment response (U = 902, z = 0.177, p = 0.859).
Overall, the patients who did not consider nVNS useful had a higher median number of previous preventive failures (U = 1020; z = −2.009; p = 0.012). The baseline number of attacks a day (U = 978, z = −1.049, p = 0.294) and attack duration (U = 1253, z = 1.790, p = 0.074) did not influence the nVNS response.
A logistic regression was performed to analyze whether patient characteristics (age and sex) and comorbidities (coexisting migraine or psychiatric comorbidities) could influence the nVNS treatment response. The Hosmer–Lemeshow test was used as a goodness of fit measure and it confirmed that the model fitted our observed data (χ2 = 3.012, d.f. = 8, n = 70, p = 0.934). None of the four predictors influenced nVNS response (see supplementary material, Table S1). In addition, due to the higher proportion of concomitant migraine, we compared the nVNS treatment response in patients with and without concomitant migraine; there was no difference (χ2 = 0.203, d.f. = 1, n = 108, p = 0.689).
In all groups of TAC patients, nVNS was generally considered more useful as a preventive rather than acute treatment (Table 1). Nevertheless, SUNCT/SUNA patients also found it useful as an acute treatment in 50%. Of the patients that found nVNS useful, 70% had a reduction of more than 50% in attack frequency, with 23% of the patients having a greater than 50% in attack duration. No difference was noted between patients with and without migraine in terms of the number of the patients that achieve a reduction in attack frequency superior to 50% (χ2 = 0.859, d.f. = 1, n = 48, p = 0.428).
nVNS tolerability
Twenty-three patients (22%) reported at least one AE, the description of each AE reported is presented in Figure 2. The most common side effects were self-limiting neck discomfort and facial pain ipsilateral to the site of stimulation. No serious treatment-related AEs had been recorded in clinical letters associated with the use of nVNS. The percentage of AEs by TACs groups is shown in Table 1. A chi-squared test of independence was performed to examine the relationship between difference TACs groups and AEs and a significant relation was noticed (χ2 = 14.464, d.f. = 4, n = 107, p = 0.006), meaning that the rate of AEs is different between the different TACs groups. We proceeded to perform a between groups analysis (see supplementary material, Table S2) and concluded that the rate of AEs in HC patients was significantly higher than in ECH and CCH patients, 53%, 7% and 17%, respectively (χ2 = 11.212, d.f. = 1, n = 42, p = 0.002 and χ2 = 7.831, d.f. = 1, n = 62, p = 0.014) and also between ECH and PH patients, 7% and 40%, respectively (χ2 = 5.705, d.f. = 1, n = 37, p = 0.035).

The description of each adverse event reported is presented.
nVNS withdrawal
Fifty-nine (55%) patients withdrew from using nVNS, including 11 patients (19%) who had considered it initially useful, over a median time using nVNS of 7 (IQR = 4–12) months. In 35 patients (59%) lack of efficacy was the reason for withdrawal, six patients (10%) did not tolerate the device and seven patients (12%) withdrew due to other reasons, such as denial of individual funding requests, company supply issues or other institutional issues. These seven patients that withdrew nVNS because of other reasons were excluded from this analysis. Overall, the patients that withdrew nVNS had a higher median number of previous preventive failures (U = 1594, z = 2.393, p = 0.017) but no association was found with age (U = 1256, z = 0.045, p = 0.964), number of attacks a day (U = 1312, z = 1.578, p = 0.115) or attack duration at baseline (U = 880, z = −0.915, p = 0.360).
We compared previous preventive failures and nVNS withdrawal using a logistic regression with proportional odds. The Hosmer–Lemeshow test result indicated that the model was a good fit to the observed data (χ2 = 11.945, d.f. = 8, n = 52, p = 0.154). Of the nine predictors, two previous preventive failures were significant: melatonin and lithium (see supplementary material, Table S3). Previous melatonin (OR = 0.244, 95% CI = 0.086–0.692, p = 0.008) or lithium failure (OR = 0.131, 95% CI = 0.024–0.727, p = 0.020) was associated with an increased likelihood of nVNS withdrawal.
Comparison between episodic vs chronic cluster subgroup patients
In the CH group, 74 patients were included, 27 (36%) patients with ECH and 47 (64%) with CCH diagnosis. The mean (SD) age for ECH patients was 55 (15) years and for CCH was 51 (13) years. The male-to-female ratio was higher in ECH patients (12.5:1) compared to CCH patients (1.5:1). There were no differences between the prevalence of coexisting primary headache disorders or psychiatric comorbidities between the ECH and CCH patients on nVNS. There were no differences between ECH and CCH patients with regard to usefulness of nVNS as acute (30% vs. 38%; p = 0.248) or preventive treatment (56% vs. 64%; p = 0.454) or adverse effects (7% vs. 17%; p = 0.309) from nVNS. The ECH and CCH groups had similar percentage of patients with a reduction of more than 50% in attack frequency (85% vs. 73%, p = 0.471). However, withdrawal was greater in CCH patients when compared to ECH (χ2 = 4.723, d.f. = 1, n = 74, p = 0.030).
Discussion
The data supports the efficacy, safety and tolerability of nVNS for the preventive treatment of TACs. As expected, the majority of the patients included were CCH patients (17), as this is the dominant TAC. Despite the predominance of CH in males, the frequency of female patients was higher in the PH, HC and SUNCT/SUNA group of patients. In the CH group of patients, the male: female ratio was higher in ECH than in CCH patients. Previous studies have shown a decrease in the male: female ratio in CCH after the age of 40 years (18). A recent Swedish cohort of patients showed that female patients are generally more severely affected by CH: more are diagnosed with the CCH subtype, report longer bouts and use prophylactic treatment more often (19).
Interestingly, the majority of the patients in our cohort had a coexisting migraine diagnosis, however, our study was not designed to evaluate the effect of nVNS in either the acute or preventive treatment of migraine. Nevertheless, we are aware that the prevalence of concomitant headache diagnoses in our population was remarkably high, and that this fact could impact the generalizability of the current study's results. However, in our cohort the presence of migraine did not affect the nVNS response or withdrawal.
According to lifetime prevalence studies, depression rates in CH patients range from 4.6% to 56.6% and anxiety rates from 4.6% to 24%; however, in our cohort, the presence of anxiety and depression were not that high, especially in a refractory population with a higher number of previous preventive treatment failures (20). When comparing TAC groups, the HC and SUNCT/SUNA patients presented with a higher frequency of concomitant anxiety or depression diagnosis.
In our cohort of cluster patients, nVNS was found to be more beneficial as a preventive, rather than as acute treatment. We observed a more significant reduction in the frequency of attacks compared to the duration of attacks. Contrary to the results of the ACT1 and ACT2 trials (3,4), our results indicated that nVNS had a clinically meaningful effect for both acute treatment of ECH and CCH (30% and 38%, respectively). One reason for the discrepancy between our findings and those studies may be related to their primary outcome measure, which focused on the number of patients achieving “responder status” within 15 minutes. In our analysis, we evaluated the overall response without a specific time cutoff. Patients with chronic TACs who experience background pain may find it more challenging to achieve pain relief within that timeframe. Furthermore, in the ACT2 study, when they assessed the proportion of CCH subjects who achieved responder status for 50% of treated attacks at 15 minutes, rather than a pain-free outcome, the percentage increased from 5% to 29%.
The remaining TAC patients in our study cohort found nVNS useful, the majority as a preventive treatment rather than an acute treatment. This may relate to issues of device use during the attack or also be related to the duration of the attacks. Patients with shorter attacks may not be able to benefit from this treatment, although half of the patients with SUNCT/SUNA did find it useful. However, our results demonstrated that the number of attacks a day and the attack duration did not influence the nVNS response, suggesting that attack duration is perhaps not a reason for acute use of nVNS being found to be generally less useful than preventive use in our cohort. In terms of nVNS as a preventive treatment, the response seems not to be dependent on the number of stimulations, highlighting the importance of individual adjustments of parameters for each patient (21). In agreement with previous evidence reported in the literature, the nVNS therapeutic dose needed could also vary by individuals and be age dependent (16).
As previously reported in a meta-analysis (22), our results are in concordance with a good tolerability of nVNS. Remarkably, of all patients who withdrew using the device, only 10% did so because they did not tolerate it. Interestingly, when comparing TACs groups, HC patients reported AEs more often than CH patients. This may be related to sample size, or to the underlying biology of hemicrania continua. This difference was also noted in PH compared to ECH patients, but with a weaker association.
Despite almost 70% of patients finding nVNS useful, 55% of our population withdrew its use and of those patients, 20% found it initially useful. Patients with a higher number of previous preventive failures are more likely not to find nVNS useful and to withdraw, especially if they had previously tried melatonin or lithium. The data support using nVNS as an earlier option.
Limitations
Our study reports data on nVNS in TAC patients from a single tertiary headache centre, and it is important to consider how other centres may find the device's use. Moreover, our sample was dominated by cluster headache, with only a small contribution from other TACs. This reflects the demographics of TACs, and the broader issue that UK NHS funding is specifically directed at cluster headache. Given that the data was collected retrospectively, it was not possible to evaluate the effectiveness of nVNS more objectively, as the quantification of attack frequency, duration and intensity after nVNS initiation were not reported in a systematic way in all of the follow-up clinical letters. These limitations inform how we may collect information going forward. The patients were also not evaluated for the usefulness of the treatment at the same interval, which also could have influenced some of our results. To avoid a responder bias, patients who did not consider nVNS useful were also included in the treatment duration analysis.
In conclusion, our findings complement previous evidence of the effectiveness and tolerability of nVNS in CH. In addition, information regarding the other forms of TAC is added to what is a small overall public literature. Further prospective studies should cover the effectiveness and tolerability, particularly with PH, HC, SUNCT and SUNA patients.
Clinical implications
nVNS is a well-tolerated treatment in TACs in general. nVNS is useful for both preventive and acute attack treatment in cluster headache. nVNS should be considered as an earlier option of preventive treatment in all TACs.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251370339 - Supplemental material for Neuromodulation in trigeminal autonomic cephalalgias: 11-year experience of non-invasive vagus nerve stimulation
Supplemental material, sj-docx-1-cep-10.1177_03331024251370339 for Neuromodulation in trigeminal autonomic cephalalgias: 11-year experience of non-invasive vagus nerve stimulation by Catarina S Fernandes, Usman Ashraf and Peter J Goadsby in Cephalalgia
Footnotes
Acknowledgments
Stefania Maniataki and Fiona Greenwood, Specialist Headache Nurses, King's College Hospital, London, assisted in identifying relevant patient data.
Ethical statement
Author contributions
Catarina S Fernandes: Conceptualization; data curation; formal analysis; methodology; writing – original draft; writing – review and editing. Usman Ashraf: Data curation; writing - review and editing. Peter J Goadsby: Conceptualization; data curation; formal analysis; methodology; supervision; validation; writing – review and editing.
Funding
The work is supported, in part, by the NIHR South London and the Maudsley Biomedical Research Centre (NIHR203318).
Declaration of conflicting interests
Catarina S Fernandes: No conflict. Usman Ashraf: UA reports in the last 12 months receiving a fee from the Massachusetts Medical Society. Peter J Goadsby: Unrelated to this work, PJG reports, over the last 36 months, personal fees for consulting from Aeon Biopharma, Abbvie, Aurene, CoolTech LLC, Dr Reddy's, Eli-Lilly and Company, Epalex, Kallyope, Linpharma, Lundbeck, Pfizer, PureTech Health LLC, Satsuma, Shiratronics, Teva Pharmaceuticals, and Vial, and personal fees for advice through Gerson Lehrman Group, Guidepoint, SAI Med Partners, Vector Metric, and fees for educational materials from CME Outfitters and WebMD, and publishing royalties or fees from Massachusetts Medical Society, Oxford University Press, UptoDate and Wolters Kluwer.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
