Abstract
Aim
Cluster headache (CH) is often underdiagnosed and improperly treated. We explored the characteristics of CH patients referred to our tertiary headache center and compared them to patients with migraine and tension-type headache (TTH).
Methods
This cross-sectional study was conducted at the Headache Center of the Charité – Universitätsmedizin Berlin from December 2015 to January 2023. We collected data on headache characteristics, comorbidities, prior treatments, and treatment recommendations from the doctor's letter generated after their first consultation. Patient-Reported Outcome Measures were assessed using the Headache Impact Test, the Depression, Anxiety and Stress Scale 21, and the Short Form 12. We compared characteristics of patients diagnosed with CH to those with migraine and TTH, matched for age, sex, and body mass index.
Results
From the overall cohort of 1468 patients (mean age 43 ± 14 years; 75% women), 56 patients (mean age 44 ± 9 years; 23% women) were diagnosed with CH. Among these 56 patients, 54% (n = 30) were diagnosed with chronic CH. Although 84% were correctly diagnosed before referral, 34% lacked first-line acute treatment, and 55% had not received guideline-recommended preventive medication. Compared to patients with migraine, patients with CH had a shorter disease duration from onset to referral (5.5 [7.3] years vs. 20.0 [21.0] years, p < 0.001). Smoking and illicit drug use were significantly more frequent among patients with CH (p < 0.001 and p < 0.05 for the comparison with migraine and TTH, respectively), but not the rate of manifest cardiovascular diseases. Patient-Reported Outcome Measures scores did not differ significantly.
Conclusion
Both acute and preventive treatment in patients with CH remains inadequate at primary and secondary care levels. The high prevalence of chronic CH suggests a high threshold for referral to specialized centers.
This is a visual representation of the abstract.
Introduction
Cluster headache (CH) is the most common form of trigeminal autonomic cephalalgias (TAC), characterized by recurrent attacks of unilateral severe headache accompanied by ipsilateral cranial autonomic symptoms.1,2 CH is frequently underdiagnosed, and patients often report a delay of several years between disease onset and diagnosis, also leading to a delay in adequate treatment. A study conducted in the United States found that 42% of patients experienced a delay of over five years before receiving an accurate diagnosis of CH. 3 The pain experienced during CH attacks is one of the most severe kinds of pain, leading to a significant negative impact on quality of life, mood, and employment status.4,5 This impact is notably greater in CH than in other primary headache disorders, and even more pronounced in patients with chronic CH (cCH) compared to episodic CH (eCH).6,7
Patients with CH report smoking and use of illicit drugs more often than the general population and the number of CH bouts and headache-related disability are associated with smoking intensity.8–10 Although to date the direction of causality remains controversial, it is undisputed that smoking and illicit drug use are risk factors for cardiovascular and/or respiratory disease, potentially contributing to the worsening of physical health in CH.11–13
Treatment options for CH have been recently summarized in the European Academy of Neurology (EAN) guidelines. 14 German national guidelines are consistent with these recommendations. 15 However, in Germany, only subcutaneous sumatriptan, intranasal zolmitriptan, and lithium are formally approved for the treatment of CH. Although high-flow oxygen and verapamil are not officially approved for this indication, they are commonly used in clinical practice as off-label therapies and reimbursed by statutory health insurance, based on decisions of the Federal Joint Committee (Gemeinsamer Bundesausschuss). Other off-label therapies, including monoclonal antibodies to calcitonin gene-related peptide, require a cost coverage request and approval by the health insurance provider. Lower-cost medications like topiramate are generally reimbursed without issue. Available treatments for CH lack specificity and may be limited by issues of tolerability or effectiveness. 16
Taken together, these factors contribute to a substantial disease burden and highlight the need for specialized headache care. However, the capacities of tertiary headache centers remain limited. 17 To date, no German study has specifically assessed patients with CH at the time of referral to a tertiary headache center in comparison to patients referred for other primary headache disorders. The aim of this study was (1) to assess the characteristics of patients with CH referred to our tertiary headache center, and (2) to compare those to patients with migraine and tension-type headache (TTH).
Methods and materials
Study design and participants
This work is part of a cross-sectional survey study conducted at the Headache Center, Charité – Universitätsmedizin Berlin, Germany. The design has been described in detail in a prior publication. 18 Briefly, from December 2015 to January 2023, all patients visiting our headache center for the first time were invited to voluntarily complete an anamnesis questionnaire prior to their first appointment. This paper questionnaire included prespecified questions on (i) symptoms, disease duration, prior neuroimaging (cranial computer tomography [cCT] or cranial magnetic resonance imaging [cMRI]), and prior pharmacological and nonpharmacological treatments (structured as a table to fill in medication name, dose, and efficacy), (ii) substance use and comorbidities (structured as a checklist of predefined options with a possibility to enter details in a free-text field) as well as (iii) patient-reported outcome measures (PROMs) including the (a) Headache Impact Test-6 (HIT-6), 19 (b) Depression, Anxiety and Stress Scale 21 (DASS-21), 20 and (c) Short Form-12 (SF-12). 21
During the consultation at the headache center, the responsible physician made a headache diagnosis based on the patient's medical history and clinical examination and documented the diagnosis, prior treatments, comorbidities, and recommended treatments in a doctor's letter. These physicians were either board-certified neurologists with advanced training in headache medicine or neurology residents working under the close supervision of a certified headache specialist. Only those patients who completed the questionnaire and had available documentation from the consultation were included in the analysis. We did not collect data on the diagnoses or characteristics of patients who declined participation. In Germany, a referral from either primary or secondary care is a prerequisite for accessing tertiary care services, except for patients with private insurance who may present with a self-referral. We recorded the type of referral as a referral from the primary level of care (general practitioner), secondary level of care (neurologist, pain specialist), or self-referral.
For this analysis, we included all patients with a diagnosis of CH or probable CH according to the ICHD-3 criteria. We excluded patients who had CH and a second concomitant headache disorder. From the database of all patients referred for their first consultation at our center, we retrieved in a 1:1 ratio patients with a diagnosis of migraine and patients with a diagnosis of TTH, respectively, matched to the patients with CH for age, sex, and body mass index (BMI). Patients had to have a diagnosis of isolated migraine or TTH without a second concomitant headache disorder. To ensure comparability between groups, we employed nearest-neighbor matching without replacement, manually selecting one patient with migraine and one with TTH for each patient with CH, based on closest values for age (±2 years), sex, and BMI (±1 kg/m²).
Prior and recommended treatments
We assessed the use of prior acute and preventive treatments as well as treatment recommendations from our center according to the guidelines of the EAN. 14 Nonguideline medications were grouped collectively as “other.” Concerning treatment recommendations, several treatments could be recommended at the same time (e.g., recommendation to try oxygen and a triptan during different attacks). Information on prior treatments and comorbidities was drawn from both the anamnesis questionnaire and the doctor's letters. When available, questionnaire data were used as the primary source, but if information was missing or incomplete, the physician's documentation was used to supplement the data. There were no contradictions between sources, so no predefined hierarchy or decision algorithm was necessary. Recommended treatments were derived exclusively from the doctor's letters.
Substance use and comorbidities
Information on substance use was retrieved from the anamnesis questionnaires. Patients were asked to classify their (1) smoking habits as “never,” “past smoking,” “occasional,” or “current daily,” (2) alcohol consumption as “never,” “rarely,” “occasionally,” or “regular,” and (3) use of illicit drugs as “never,” “rarely,” “occasionally,” or “regular.” In line with German law during the study period (2015–2023), illicit drugs were defined as substances prohibited under the German Narcotics Act (Betäubungsmittelgesetz), including the groups of cannabinoids (cannabis), stimulants (e.g., cocaine, amphetamines), dissociatives (ketamine) and hallucinogens (lysergic acid diethylamide, psilocybin). Patients could report both frequency and substance type via structured and free-text fields. However, we did not collect information on whether the use was recreational or related to headache self-medication.
Information on comorbidities was retrieved from patient questionnaires and cross-checked with the doctor's letter written during the patient's initial consultation at our center. Cardiovascular diseases included arterial hypertension, heart disease, and/or stroke. Respiratory disease was defined as chronic disease that affects the lungs and other parts of the respiratory system. For psychiatric diseases, we recorded diagnoses of anxiety and/or depression. Sleep disorder was recorded as present or absent, without assessment of specific diagnoses such as obstructive sleep apnea syndrome.
Patient-reported outcome measures
(a) Headache Impact Test-6
The impact of headache on daily life was measured using the validated German version of the HIT-6. 19 The HIT-6 is a self-administered questionnaire designed to evaluate the impact of headaches over a short period. It consists of six items rated on a five-point Likert scale, measuring headache frequency, pain severity, and the impact on daily activities, social participation, and concentration, as well as the emotional burden. We report rates of little or some impact (grades I and II) versus substantial or severe impact (grades III and IV).
(b) Depression Anxiety Stress Scale 21
Psychological distress was assessed using the DASS-21. 20 The DASS-21 is a self-administered questionnaire to measure the presence and severity of depression, anxiety, and stress symptoms. It comprises three subscales, each with 7 items, for a total of 21 items rated on a four-point Likert scale ranging from 0 to 3. Symptoms are categorized as mild, moderate, severe, or extremely severe based on established cutoff values. The German version of the DASS-21 has demonstrated strong reliability and validity. 22 We indicate rates of no/mild/moderate versus severe/extremely severe symptoms of depression, anxiety, and stress.
(c) Short Form-12
Health-related quality of life was assessed with the SF-12. 21 The SF-12 is a self-administered questionnaire designed to assess quality of life across eight domains. Each item is rated on a Likert scale. The SF-12 generates two composite scores: the Physical Component Summary (PCS) and the Mental Component Summary (MCS), reflecting overall physical and mental health status, respectively. The German version of the SF-12 has been validated, demonstrating good reliability and validity. 23 We indicate rates of a high physical and mental impact defined as a score ≤40 points, respectively.
Study objectives
The primary objective for this study was to characterize patients with CH at the time of the first consultation in our center including the description of (i) demographic data, the duration of disease before referral, and headache characteristics, (ii) prior and recommended treatments, (iii) substance use and comorbidities, and (iv) headache impact, psychological distress as well as health-related quality of life the by means of the abovementioned PROMs. Exploratory objectives were the comparisons of these characteristics to patients with migraine and TTH, and of PROMs between patients with cCH and eCH, and within eCH patients in-bout versus out-bout at their first consultation.
Statistical analyses
Statistical analyses were performed with SPSS 27.0 (IBM SPSS Statistics©, Armonk, NY, USA). Missing values are indicated for each analysis. Categorical variables are reported as absolute numbers (n) and percentages (%), numerical variables as mean value ± standard deviation in case of normal distribution, and median [IQR] in case of non-normal distribution. Differences in continuous variables were determined using the Student t-test or Mann–Whitney U test. Categorical variables were compared between two groups (CH vs. migraine and CH vs. TTH) by Pearson’s chi-squared test or the Fisher exact test. Due to the explorative character of the comparative analyses, no adjustments for multiple testing were made. Effect sizes were calculated for group comparisons where applicable: Cohen's d for independent t-tests, r (derived from Z as r = Z/√N) for Mann–Whitney U tests, and φ for Pearson's chi-square tests. For contingency tables larger than 2 × 2, Cramér's V was reported instead of φ.
Ethical standards
The study was approved by the ethics committee of the Charité – Universitätsmedizin Berlin (EA4/246/23). Due to the retrospective nature of this study, written informed consent from enrolled patients was not required under local regulations. This study is reported in accordance with the “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) statement for cohort studies. 24
Data accessibility
Anonymized data may be shared at the request of any qualified investigator for purposes of replicating procedures and results.
Results
Demographical data and headache characteristics
The selection process for the overall study is detailed and illustrated in Figure 1, adapted from the main publication. 18 Of 1468 patients presenting for the first time at our tertiary headache center (43 ± 14 years; 75% women), 56 (44 ± 9 years; 23% women) were diagnosed with CH. Six other patients were diagnosed with CH and concomitant migraine and were not included in this analysis. Thirty-three patients fulfilled all diagnostic criteria for CH, 23 had probable CH.

Flowchart for patient selection. Adapted from Overeem et al.18 Licensed under CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/). Changes were made to the original figure to adapt the “eligibility and included” section, providing subgroup details for patients with cluster headache (CH), migraine, and tension-type headache (TTH) relevant to this subanalysis.
As previously reported, 18 47 of these 56 patients (84%) had already received the diagnosis at primary or secondary levels of care, one patient had been diagnosed with TTH, and the other eight patients had not received a headache diagnosis prior to referral. About half of the patients (n = 29, 52%) were referred from the secondary level of care, while the remainder were referred from the primary level (n = 13, 23%) or self-referred (n = 14, 25%). The proportion of patients who received a correct diagnosis of CH prior to referral was similar across all referral types: 85% (11/13) for primary care, 83% (24/29) for secondary care, and 86% (12/14) for self-referral (Cramér's V = 0.035, p = 0.967). Neuroimaging had been performed prior to referral in 37 patients (66%), corresponding to cCT in 8 patients (14%) and cMRI in 29 patients (52%).
Patient characteristics are displayed in Table 1. The median disease duration from onset to referral was 5.5 [7.3] years. cCH was diagnosed in 30 patients (54%). Of 26 patients diagnosed with eCH, 10 patients (38%) were in-bout at the time of their first consultation. Median weekly attack frequency during bouts was 21 [14], and median bout duration in patients was 6.5 [13.3] weeks.
Demographical data and headache characteristics.

Values are given as mean (± standard deviation) or median [IQR] for continuous data and as n (%) for categorical data. p Value from Pearson Chi² test for categorical variables, Student t-test, or Mann-Whitney U test for continuous variables. Effect sizes are reported as φ for Pearson's chi-square tests with 2 × 2 tables, Cramér's V for larger contingency tables, Cohen's d for Student t-tests, and r (derived from Z) for Mann–Whitney U tests. Bold values denote statistical significance at the p < 0.05 level.
CH: cluster headache; PLC: primary levels of care; SLC: secondary levels of care; TTH: tension-type headache.
p for comparison between CH and migraine.
p for comparison between CH and TTH.
Neuroimaging including cranial computer tomography and cranial magnetic resonance imaging.
Missing data for n (%) patients: 7 (13).
In patients with migraine, the rate of chronic cases was numerically lower, but not statistically significant (φ = 0.13, p = 0.256). The median disease duration between onset and referral was significantly higher (Z = −5.953, r = 0.56, p < 0.001).
In patients with TTH, the percentage of patients with a chronic form was significantly higher (φ = 0.33, p < 0.001), while the median disease duration between onset and referral was similar.
Prior treatment attempts and recommended treatments
For acute treatment, patients with CH reported a median number of 2 [2] prior treatment attempts. Three (5%) patients had not tried any acute medication before. The most commonly tried acute treatment was oxygen in 31 (55%) patients, followed by other treatments not recommended by current guidelines (Table 3). Those included ibuprofen, metamizole, diclofenac, indomethacin, and opioids. Subcutaneous or intranasal sumatriptan and intranasal zolmitriptan had been tried less frequently (n = 12, 21%, n = 9, 14%, and n = 17, 30%, respectively). The most frequently recommended acute treatments during the first visit at our center were intranasal zolmitriptan (n = 40, 71%), oxygen (n = 39, 70%), and subcutaneous sumatriptan (n = 31, 55%).
At the time of referral, patients reported having tried 1 [2] preventive treatments. Twenty-five (45%) patients had not tried any preventive medication before. Verapamil had been prescribed to 19 (34%) patients (Table 3). The second most commonly prescribed treatment was the group of “other,” nonguideline-recommended treatments in 15 (27%) patients. Those included lamotrigine, gabapentin, carbamazepine, zonisamide, flunarizine, amitriptyline, indomethacin, opipramol, clomipramine, duloxetine, and mirtazapine. Short-term preventive treatment with oral corticosteroids had been prescribed to 14 (25%) patients. The rate of patients having tried topiramate was higher than for lithium (n = 9, 16% vs. n = 2, 4%).
The most frequently recommended preventive treatment at the first visit in our tertiary headache center was verapamil in 38 (68%) patients, followed by oral corticosteroids in 19 (33.9%) and “other” treatments in 6 (10.7%) patients. The latter corresponded to a recommendation of indomethacin for one patient to exclude paroxysmal hemicrania, gabapentin for four patients, and erenumab for one patient. Topiramate was recommended more frequently than lithium (n = 6, 11% vs. n = 3, 5%). For six patients (11%), no preventive medication recommendations were documented, as they had been out-bout for several months. A reevaluation of preventive treatment was planned for their next consultation.
Invasive and noninvasive procedures had been tried by only two patients—one with noninvasive vagus nerve stimulation and one with a greater occipital nerve block—and were recommended to three patients (Table 2).
Type of prior, current, and recommended treatment attempts.
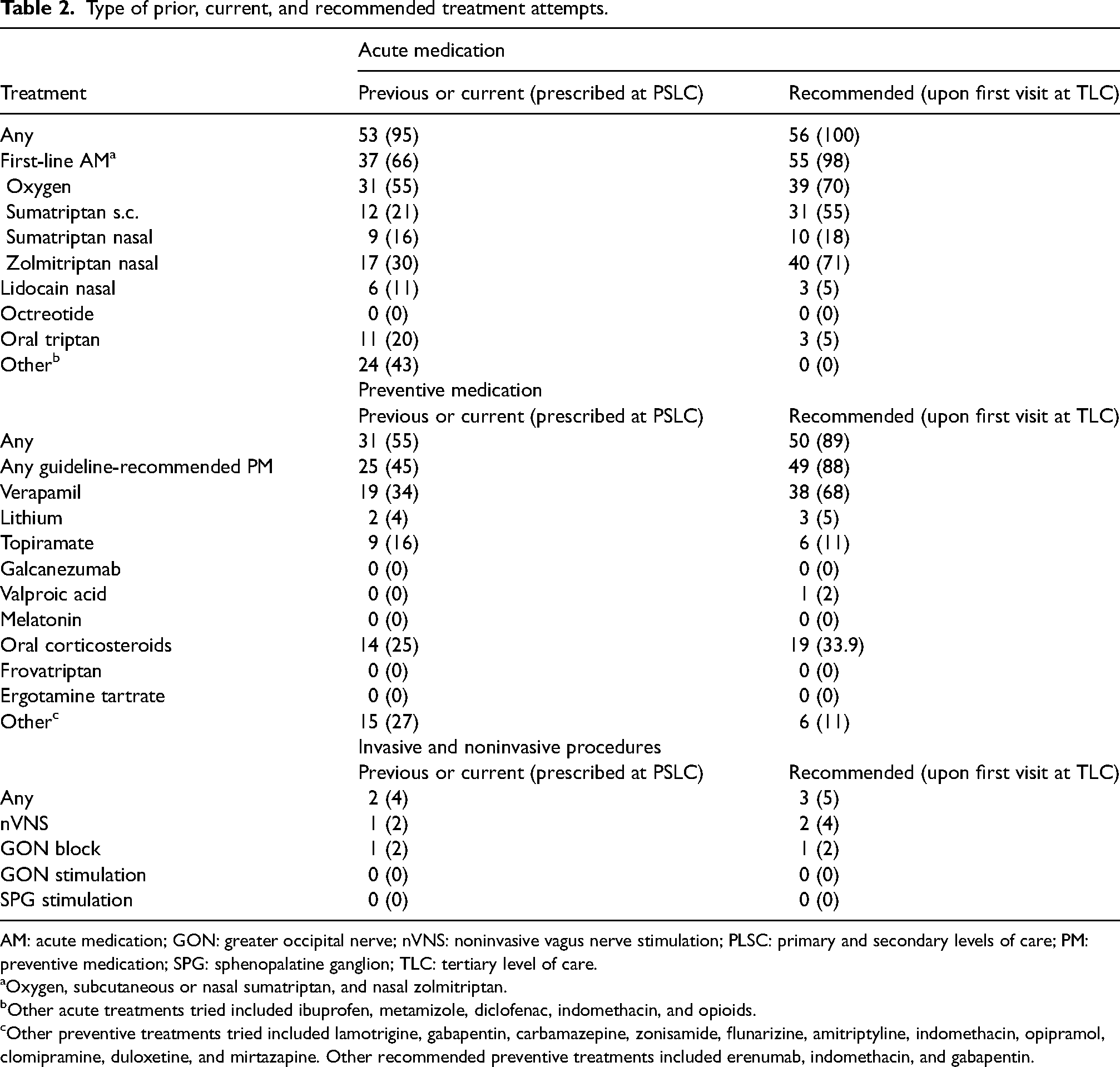
AM: acute medication; GON: greater occipital nerve; nVNS: noninvasive vagus nerve stimulation; PLSC: primary and secondary levels of care; PM: preventive medication; SPG: sphenopalatine ganglion; TLC: tertiary level of care.
Oxygen, subcutaneous or nasal sumatriptan, and nasal zolmitriptan.
Other acute treatments tried included ibuprofen, metamizole, diclofenac, indomethacin, and opioids.
Other preventive treatments tried included lamotrigine, gabapentin, carbamazepine, zonisamide, flunarizine, amitriptyline, indomethacin, opipramol, clomipramine, duloxetine, and mirtazapine. Other recommended preventive treatments included erenumab, indomethacin, and gabapentin.
Patient-reported outcomes.

CH: cluster headache; DASS-21: Depression, Anxiety and Stress Scale 21; HIT-6: Headache Impact Test-6; MCS: Mental Component Summary; PCS: Physical Component Summary; SF-12: Short Form 12; TTH: tension-type headache.
Values are given as n (%) for categorical data. P value from Pearson Chi² test for categorical variables. Effect sizes are reported as φ for Pearson's chi-square tests with 2 × 2 tables. Bold values denote statistical significance at the p < 0.05 level.
p for comparison between CH and migraine.
p for comparison between CH and TTH.
Among nonpharmacological treatments, reported by 32 patients (57%), the most frequently mentioned were physical exercise (n = 10, 18%), relaxation techniques (n = 9, 16%), and physiotherapy (n = 7, 13%; data not shown).
Patients with migraine had tried the same median number of acute treatments and preventive treatments, respectively. The detailed description of prior and recommended treatments in patients with migraine has been analyzed separately. 25
For patients with TTH, only 1 [1] prior acute treatment attempt was documented, which was significantly less than in CH (Z = −2.336, r = 0.22, p = 0.019). The number of prior preventive treatment attempts was nonsignificantly lower with 0 [1] medications tried in patients with TTH (Z = −1.071, r = 0.10, p = 0.284).
Substance use and comorbidities
Figure 2 displays the percentages of patients reporting current smoking, regular alcohol consumption, and illicit drug use. More than half of the patients with CH were current smokers (n = 29/56, 52%), and more than a quarter had a regular consumption of illicit drugs (n = 17/56, 30%). The latter corresponded to cannabis in 14/17 patients (82%), stimulants in 2/17 patients (12%) and dissociatives in 1/17 patients (6%). No patient reported the use of hallucinogens.

Prevalence of substance use and comorbidities in patients with cluster headache, migraine, and tension-type headache. * p < 0.05, ** p < 0.001. Alcohol, regular alcohol consumption; illicit drugs, and regular use of illicit drugs.
In patients with migraine, rates of current smoking and illicit drugs consumption were significantly lower (φ = 0.34, p < 0.001 and φ = 0.30, p = 0.003). Also, patients with TTH reported less current smoking (φ = 0.42, p < 0.001) and less illicit drug consumption (φ = 0.22, p = 0.037).
Cardiovascular, pulmonary, and psychiatric diseases were reported by 25% (n = 14), 18% (n = 10), and 27% (n = 15) of patients with CH, respectively. Sleep disorders were reported by more than one-third of patients (n = 20, 36%). No statistically significant differences were observed between groups, with a similarly high prevalence of comorbidities also noted among patients with migraine and TTH (Figure 2).
Patient-Reported Outcome Measures
Patients with CH reported moderate headache impact, with a mean HIT-6 score of 54.2 (±7.6). Substantial or severe impact was reported by 23 patients (41%, Table 3), and significantly more frequent in cCH compared to eCH (n = 18/28, 64% vs. n = 5/22, 23%, φ = 0.41, p = 0.003; data not shown). There were no differences between patients with eCH in-bout vs. out-bout.
The median DASS-21 scores for depression, anxiety, and stress were 14.0 [13.0], 9.0 [10.0] and 18.0 [14.0], respectively, with severe or extremely severe grades in 20% (n = 11), 20% (n = 11), and 23% (n = 13) of patients. Severe stress was more frequent in cCH than eCH (n = 11/28, 39% vs. n = 2/19, 11%, φ = 0.32, p = 0.031), but depression and anxiety rates did not differ between cCH and eCH and between patients with eCH in-bout versus out-bout.
The mean PCS and MCS scores were 39.9 (±10.9) and 40.4 (±11.9), respectively, corresponding to significant health-related physical and mental impairment in 27 (48%) and 25 (45%) patients, respectively. Rates of significant impact were higher in patients with cCH compared to eCH (n = 21/28, 75% vs. n = 6/22, 27%, φ = 0.48, p < 0.001 and n = 20/28, 71% vs. n = 5/22, 23%, φ = 0.48, p < 0.001). Patients with eCH in-bout had more mental impairment than those out-bout (n = 4/8, 50% vs. 1/14, 7%, φ = 0.49, p = 0.021). Our study was not sufficiently powered to examine predictors of unfavorable PROMs in patients with CH.
Patients with CH had higher rates of severe anxiety compared to patients with TTH (n = 11/48, 20% vs. n = 4/53, 7%, φ = 0.22, p = 0.030), while there were no differences in PROMs between patients with CH and with migraine.
Discussion
In this cross-sectional study, we characterized 56 patients with CH at the time of their first consultation in a tertiary headache center in comparison to newly referred patients with migraine or TTH matched for sex, age, and BMI.
One main finding of our study is that patients with accurately diagnosed CH at prior levels of care often do not receive adequate treatment. The high proportion of cCH in our sample suggests that referral to tertiary care has a high threshold, or that eCH is underdiagnosed at primary and secondary care levels, leading to fewer referrals.
A recent statement from the European Headache Federation and Lifting The Burden outlines criteria for headache care at primary, secondary, and tertiary levels of care.17,26 Referral to a tertiary headache center is recommended for the ∼1% of most difficult-to-treat headache patients, which may include patients with TAC even at their first visit. The majority of our patients (53.6%) had cCH, significantly exceeding the overall estimated rate of 10–15% and the reported range of 0–37% reported in studies from other tertiary headache centers.1,27–31 This indicates that, in terms of chronicity, our sample was more severely affected than the average. Although our study design does not allow us to determine which patients are not referred, our findings suggest that many severely affected individuals may lack access to specialized care. One possible explanation could be that patients with eCH are not diagnosed in outpatient settings, particularly when they present during an interictal period. Another possibility is that eCH patients are not referred because they receive satisfactory outpatient treatment. However, this seems unlikely given that referred cCH patients were offered relatively few treatment options.
Patients with CH were referred four times earlier in their disease course compared to those with migraine. Similarly, a study assessing characteristics of patients referred to a Hungarian tertiary headache center found a delay between onset and referral of approximately six years in CH and chronic TTH, respectively, compared to 14 years in migraine. 32 Other studies describing patients from tertiary headache centers have not indicated the disease duration at referral33,34 or have indicated longer disease durations of nine years for CH data from China, 29 10 years from Japan, 35 13 years from Italy, 28 and 14 years from Denmark. 27 While it is encouraging that our patients with CH requiring tertiary care were referred earlier, a delay of 5.5 years remains substantial and can lead to significant impairment for the patients.
At prior levels of care, most patients with CH received the correct diagnosis but inadequate treatment. More than one-third had not tried first-line acute medications, and 44.6% had not tried oxygen, despite EAN guidelines strongly recommending oxygen, subcutaneous/nasal sumatriptan, and nasal zolmitriptan. 14 In a Danish study, only 16% of patients were oxygen-naive, 27 while an Italian study found 16.7% without oxygen use. 28 For preventive treatment, verapamil is the preventive method of first choice. 14 Oral corticosteroids and galcanezumab (only for eCH) have a weak recommendation, while lithium, topiramate, ergotamine tartrate, frovatriptan, valproic acid, and melatonin are recommended based on expert consensus. 14 In our study, 55.4% of patients had not tried any first-line preventive medication, and almost two-thirds had not tried verapamil. In the Danish Cluster Headache Survey, only 22% of patients were verapamil naive. 27 Patients admitted to an Italian tertiary headache center were using verapamil in all but 28.7%. 28 These findings emphasize the importance of raising awareness, firstly, to improve CH care at the primary and secondary levels, and secondly, to ensure seamless access to tertiary care for CH patients when the complexity of their needs exceeds the capacity of primary and secondary care services. Furthermore, accurate diagnosis requires a careful and often time-consuming interview to assess the detailed characteristics of the headache. The same applies to the evaluation of the need for additional diagnostic procedures, and to the initiation and monitoring of appropriate treatment. These processes are particularly challenging due to limited time resources, especially in primary and secondary care settings. 36
The recommendations from our center were in line with the guidelines, with notable findings including a higher recommendation rate for nasal zolmitriptan compared to oxygen and sumatriptan, as well as a higher rate for topiramate compared to lithium, likely driven by patient preferences.
Patients with CH had higher rates of smoking and illicit drug use than patients with migraine and TTH, but not significantly higher prevalence of manifest cardiovascular or respiratory disease. The lack of statistically significant differences might be due to the relatively small sample size. Nevertheless, in our CH patients with a median age of 44 ± 9 years, the 25% rate of cardiovascular disease was significantly higher than the age-specific average of 3.2% for men aged 40–44 in Germany. 37 There is some evidence for an elevated cardiovascular risk in patients with CH.38,39 This topic needs further research into both pathophysiological mechanisms and preventive measures.
Contrary to previous studies, headache impact as assessed by PROMs was not significantly worse in patients with CH compared to migraine and TTH.4,40 This likely reflects the severe disease burden across all groups referred to our center. More precisely, patients with migraine had long disease duration before referral, and patients with TTH had high chronicity rates. The absolute mean SF-12 scores in our patients with CH were worse than in previous studies, which also suggests a severely affected cohort. 41 Moreover, while PROMs provide valuable insights into the patient's perceived disability, their accuracy in reflecting clinical severity can be compromised by factors such as the timing of completion, individual circumstances, and role expectations.42,43
This study has several limitations. First, the small sample size may explain the lack of statistical significance despite numerically notable differences between groups. As the comparisons between patients with different headache disorders and CH subgroups were exploratory analyses, no correction for multiple testing was applied; therefore, due to the small sample sizes, the results should be interpreted carefully, particularly with regard to statistical significance. Second, we have not used PROMs specifically designed for the assessment of CH, but general PROMs applicable to different kinds of headache disorders. Third, there is a potential for recall bias, though patients were encouraged to bring prior medical records. Although all consecutively referred patients were invited to complete the standardized questionnaire, only those who did so and had a doctor's letter available were included. We were not able to assess whether patients who declined to participate differed systematically in their diagnoses, which may introduce selection bias. Further, information on referral pathways was limited to the physician who issued the referral to our center, without capturing prior consultations at other levels of care. This introduces a potential source of bias, as patients referred from primary care may have already consulted specialists (e.g., neurologists) prior to referral. Therefore, referral categories may not fully reflect the entirety of a patient's diagnostic trajectory. Also, detailed information on the diagnostic trajectory prior to referral, including the timing and sequence of previous misdiagnoses and investigations, was not available. Therefore, conclusions about the temporal relationship between (mis)diagnosis, imaging, and referral must be interpreted with caution. While a one-time cranial imaging is recommended as part of the diagnostic work-up for CH, 15 other diagnostic procedures like repeated ophthalmologic exams might lead to diagnostic and therapeutic delays. In patients who had already received a diagnosis of CH prior to referral, it was not always possible to determine whether prior treatments had been prescribed before or after the diagnosis was established, which may affect the interpretation of treatment adequacy. With regard to illicit drug use, we did not assess whether the use was recreational or intended as a form of self-medication for headache, limiting interpretation of these findings. Information on cardiovascular comorbidities was based on general categories (e.g., “cardiac disease” and “stroke”) and may lack detail on specific conditions such as arrhythmia, heart failure, or stroke subtypes, which were not systematically assessed. The same applies to subcategories of sleep disorders. It is important to note that differences between our findings and those of previous studies may be influenced by contextual factors, including differences in healthcare systems, referral practices, diagnostic criteria, or data collection methods.
The strengths of our study include a more precise assessment compared to survey-based studies, as it incorporates not only patient recollections but also prior medical reports and detailed medical history documentation at the time of referral by experienced headache specialists. Comparisons with matched patients with migraine and TTH provide more meaningful results than comparison with healthy controls. While we acknowledge the limitations related to sample size, we believe our study offers timely insight into real-world care and supports the ongoing need for structured quality improvement in headache management. Regular reassessment of CH care in real-world settings is essential—both to evaluate the impact of growing awareness and advocacy efforts and to address ongoing challenges in implementing guideline-based treatment amid increasing demands on healthcare providers.
In conclusion, our findings suggest inadequate acute and prophylactic treatment for CH patients in primary and secondary cares, along with a high referral threshold. Greater efforts are needed to raise awareness and enhance CH care at primary and secondary levels, while ensuring access to tertiary care when required.
Clinical implications
More than a third of CH patients had not received guideline-recommended acute treatment, and over half had not received guideline-recommended preventive therapy before referral to a tertiary headache center.
The high proportion of cCH in this cohort suggests that episodic cases may be underdiagnosed or that access to specialized care is delayed.
Greater efforts are needed to improve awareness, enhance adherence to treatment guidelines, and ensure timely referral for patients requiring specialized care.
Footnotes
Acknowledgments
Dr Kristin Sophie Lange and Dr Bianca Raffaelli are participants in the BIH Charité Clinician Scientist Program. Dr Mira Pauline Fitzek is a participant in the BIH Charité Junior Clinician Scientist Program.
Author contributions
KSL contributed conceptualization; investigation; formal analysis; visualization; writing—original draft preparation. JD contributed investigation; writing—review & editing. LHO contributed investigation; writing—review & editing. MPF contributed investigation; writing—review & editing. CLH contributed investigation; writing—review & editing. JBH contributed investigation; writing—review & editing. YS contributed investigation; writing—review & editing. MU contributed investigation; writing—review & editing. UR contributed conceptualization; supervision; writing—review & editing. BR contributed conceptualization; investigation; project administration; writing—original draft preparation.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KSL reports a research grant from the International Headache Society and personal fees from Teva, Organon; MF reports personal fees from Teva, Novartis; UR reports personal fees from Amgen, Allergan, Abbvie, Lilly, Lundbeck, Novartis, Pfizer, Medscape, StreaMedUp, Springer, Teva, and research funding from Novartis; BR reports research grants from Lundbeck, Novartis, the German Research Foundation (DFG), and Else Kröner-Fresenius-Stiftung and personal fees from Abbvie/Allergan, Eli Lilly, Lundbeck, Novartis, Organon, Perfood, Teva.
Ethical approval
The study was approved by the ethics committee of the Charité – Universitätsmedizin Berlin (EA4/246/23). Due to the retrospective nature of this study, written informed consent from enrolled patients was not required under local regulations.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
