Abstract
Background
Verapamil is recommended as a first-line preventive for episodic and chronic cluster headache, however, its use is limited by a wide range of adverse events. From clinical practice at a tertiary headache centre, we have observed that the initiation of verapamil may be associated with headache worsening. The aim of this service evaluation was to examine whether verapamil initiation was associated with headache worsening, and whether these exacerbations may be attributed to comorbid migraine in some patients.
Methods
Patients with a diagnosis of cluster headache from June 2014 to December 2023 were identified from the tertiary headache centre at King's College Hospital, London. Data including age, sex, headache phenotype and headache frequency was collected retrospectively in a cross-sectional design through the use of clinic letters followed by a telephone interview. A Wilcoxon signed-rank test was used to compare number of cluster headache attacks per day pre- and post-verapamil administration. A negative binomial generalized linear model was used to interrogate the relationship between the number of attacks post-verapamil and age, sex, verapamil dosing, comorbid migraine and baseline number of attacks.
Results
Of 168 patients included, the mean age was 31 years, 73% were male, 46% of patients had chronic cluster headache and 51% had comorbid migraine. During the latest ictal period, the median (interquartile range) frequency of cluster headache attacks per day was 3 (2–5) for the entire sample. The presence of comorbid migraine increased the likelihood of headache exacerbation by verapamil by an odds ratio of 1.616 (95% confidence interval: 1.059–2.465, χ21 = 4.955, P = 0.026). No differences were observed in the frequency of shadow attacks amongst those with comorbid migraine (n = 54/85, 64%) versus patients without migraine (n = 48/83, 58%) (Mann-Whitney U = 3728, z = 0.754, P = 0.451). No effect was seen on monthly migraine days pre- and post-verapamil administration (P = 0.141). Adverse events were reported in 62 of 109 (57%) of patients taking verapamil with the most common being PR interval prolongation (15.6%), lower limb oedema (8.3%), worsened headache (6.4%) and fatigue (6.4%).
Conclusions
It is likely that the association of cluster headache and migraine is more common than generally thought and co-existence may go under-recognised. Our results show comorbid migraine increased the likelihood of headache exacerbation during the initiation of verapamil. In patients with headache worsening, a dual diagnosis of migraine alongside cluster headache should be considered.
Introduction
The International Classification of Headache Disorders presents cluster headache (CH) and migraine as separate primary headache disorders, 1 affecting 0.1% and 15% of the general population, respectively.2,3 CH attacks are characterised by bouts of daily severe, strictly side-locked pain associated with prominent ipsilateral cranial autonomic symptoms, or with restlessness or agitation, or both. 1 Attacks occur with specific chronobiological features, such as circadian and circannual rhythmicity with a nocturnal preference, and can either be episodic or chronic, separated by a pain-free remission period of at least 3 months. 4 Migraine, on the other hand, is a recurrent headache disorder characterised by moderate to severe pulsatile headache associated with nausea, photophobia, phonophobia and worsened by routine physical activity. Attacks are commonly unilateral, although bilateral headache is reported in up to 40% of individuals with migraine, 5 and includes distinct phases, such as the premonitory, headache and postdrome phases. 6
CH and migraine share some mechanisms leading to attack initiation. 7 Pharmacological triggers, such as nitroglycerin, calcitonin-gene-related peptide (CGRP), pituitary adenylate cyclase activating polypeptide-38 (PACAP38) and vasoactive intestinal polypeptide (VIP), can trigger both migraine and CH.8–10 Likewise, in some studies, CGRP has been shown to be elevated in the cranial venous outflow of patients with CH, 11 an observation which is similarly seen in patients with migraine. 12 Recently, genome-wide association studies have found one shared locus for migraine and CH, located on chromosome 6 covering both FHL5 and UFL1, implying a potential genetic overlap between the conditions. 13 The associated symptoms of CH and migraine additionally imbricate, with some patients with migraine reporting periodicity in attack frequency and severity 14 as well as cranial autonomic symptoms, reported in 30% to 75% of patients, both adults and children,15,16 whilst 25% to 46% of patients diagnosed with CH experience clinical features typically associated with migraine, such as nausea, photophobia, phonophobia and even migraine itself. 17
For patients with CH, verapamil is considered the first-line preventive medication for both episodic and chronic subtypes, 17 however, its use is limited by a wide range of adverse events associated with high doses of the medication. 17 From observations in a tertiary headache centre, we have noted that the starting period of verapamil can be associated with a worsening of background pain or shadow attacks. For the purpose of this study, we have defined shadow attacks as a milder pain that mimic a regular CH attack with fewer and/or less intense additional symptoms that do not require abortive treatment. 18 We considered that this worsening may be attributed to comorbid migraine in some patients. The aim of this service evaluation is to characterise exacerbations of background pain and/or shadow attacks, and whether verapamil initiation is associated with this event. In patients with comorbid migraine, we have also documented the effect, if any, of verapamil on migraine.
Methods
Ethics
The data was compiled and analysed with the objective of carrying out a service evaluation, which does not require Research Ethics Committee review in the UK (http://www.hra-decisiontools.org.uk/research/). Informed consent was not required for the use of patient data for this study, as the data was acquired from patient clinic letters as part of an evaluation process; therefore, only local hospital approval was needed.
Participants
Patients with a diagnosis of episodic CH (eCH) or chronic CH (cCH) seen by the tertiary headache service offered by King's College London were invited to participate in this service evaluation up to the date of December 2023 (n = 168). All patients met the diagnostic criteria set by the International Classification of Headache Disorders Third Edition and clinical histories were validated by a headache specialist. 1 Patients with concurrent diagnoses of migraine were included. As a pre-requisite for inclusion, patients had to be able to distinguish their CH attacks from other headache entities, such as shadows, background pain and migraine attacks (defined below). Patients with other chronic primary or secondary headache disorders, significant psychiatric disease, probable diagnoses of CH and those later discharged from the service were excluded.
Study conduct
In a cross-sectional design, data was first retrospectively collected through clinic letters and later, followed by a telephone interview to clarify further details and missing information between the dates of June 2014 to December 2023.
For all patients included in the service evaluation, the last available clinic appointment letter was analysed to maximise information and ensure the information was accurate and up-to-date. Clinic appointments within the tertiary headache centre last approximately 1 hour, allowing a detailed history exploring headache phenotype, associated symptoms, shadows, medical history, medication history, family history and social history to be taken. Of particular relevance to this evaluation, patients had to differentiate between: (1) CH, (2) shadows (if any), (3) background pain (if any) and (4) migraine attacks (if applicable). CH attacks were defined as attacks of severe, strictly unilateral pain which is orbital, supraorbital, temporal or in any combination of these sites, lasting 15 to 180 minutes and occurring from once every other day to 8 times a day associated with ipsilateral conjunctival injection, lacrimation, nasal congestion, rhinorrhoea, forehead and facial sweating, miosis, ptosis and/or eyelid oedema, and/or with restlessness or agitation. 1 Whilst shadows were defined as a milder pain that mimics a regular attack in terms of location but is less intense, self-limiting, includes fewer and/or less severe additional symptoms and does not require abortive treatment in line with previously reported definitions. 18 Distinct to that, background pain was defined as any head pain that did not meet the requirements of the definitions of CH attacks and shadows. In individuals with migraine, migraine attacks were characterised by recurrent, unilateral ‘throbbing’ head pain associated with movement sensitivity, nausea and/or photophobia and phonophobia. 1 Patients self-reported the number of migraine days.
Demographic and clinical data, including sex, age of onset, disease duration, CH phenotype, the presence of shadows, concomitant diagnosis of migraine and verapamil administration was collected. Where verapamil was trialled, further information including total daily dose (mg), mean daily CH attacks on and off verapamil, monthly migraine days (MMD) on and off verapamil (if applicable), reason stopped, observational benefit of verapamil and time to benefit (measured in weeks) was collected.
Analysis
We used a convenience sample determined by the available patients. Data are summarised using the mean or median with appropriate dispersion measures according to the measurement type and its distribution. Wilcoxon signed-rank or Mann-Whitney tests, as appropriate, were used to compare groups with a level of significance set at P < 0.05, with a Bonferroni correction to P < 0.0125. Headache frequency was transformed into natural numbers to account for the presence of non-positive integers. 19 Patients with missing values for the pre- or post-period following verapamil administration were removed from the analysis. Propensity to have increased CH attacks post-verapamil was further evaluated using a negative binomial generalised linear model with a log link function, given over-dispersion of the data. The model included data on age, sex, use of verapamil, verapamil dose, baseline number of attacks/day and comorbid migraine. We compared this to a Poisson distribution using the Akaike information criterion, and thus report the negative binomial model. Analyses were conducted with SPSS (version 29.0) and P < 0.05 was considered significant.
Assessments
The primary outcome of the service evaluation was to characterise exacerbations of background pain and shadow attacks and to observe whether verapamil initiation is associated with this event. The effect of verapamil on migraine was explored as a secondary outcome.
Results
From June 2014 to December 2023, a total of 233 CH patient clinic notes were examined. Of those, 66 patients were excluded as a result of mixed headache phenotype, probable diagnoses and discharge from the service (Figure 1). The demographic and clinical characteristics of the included patient cohort can be seen in Table 1. Of 168 patients who met the inclusion criteria, the male to female ratio was ∼3:1 and 77 (45.8%) patients had cCH. The mean age of onset (SD) of CH was 31 ± 13 years with a mean CH duration (SD) of 17 ± 11 years. During the latest ictal period, the median (interquartile range, IQR) frequency of CH attacks per day was 3 (2–5), measured across the entire sample.

Flow chart of patient recruitment and inclusion.
Demographics and clinical characteristics of included patients.
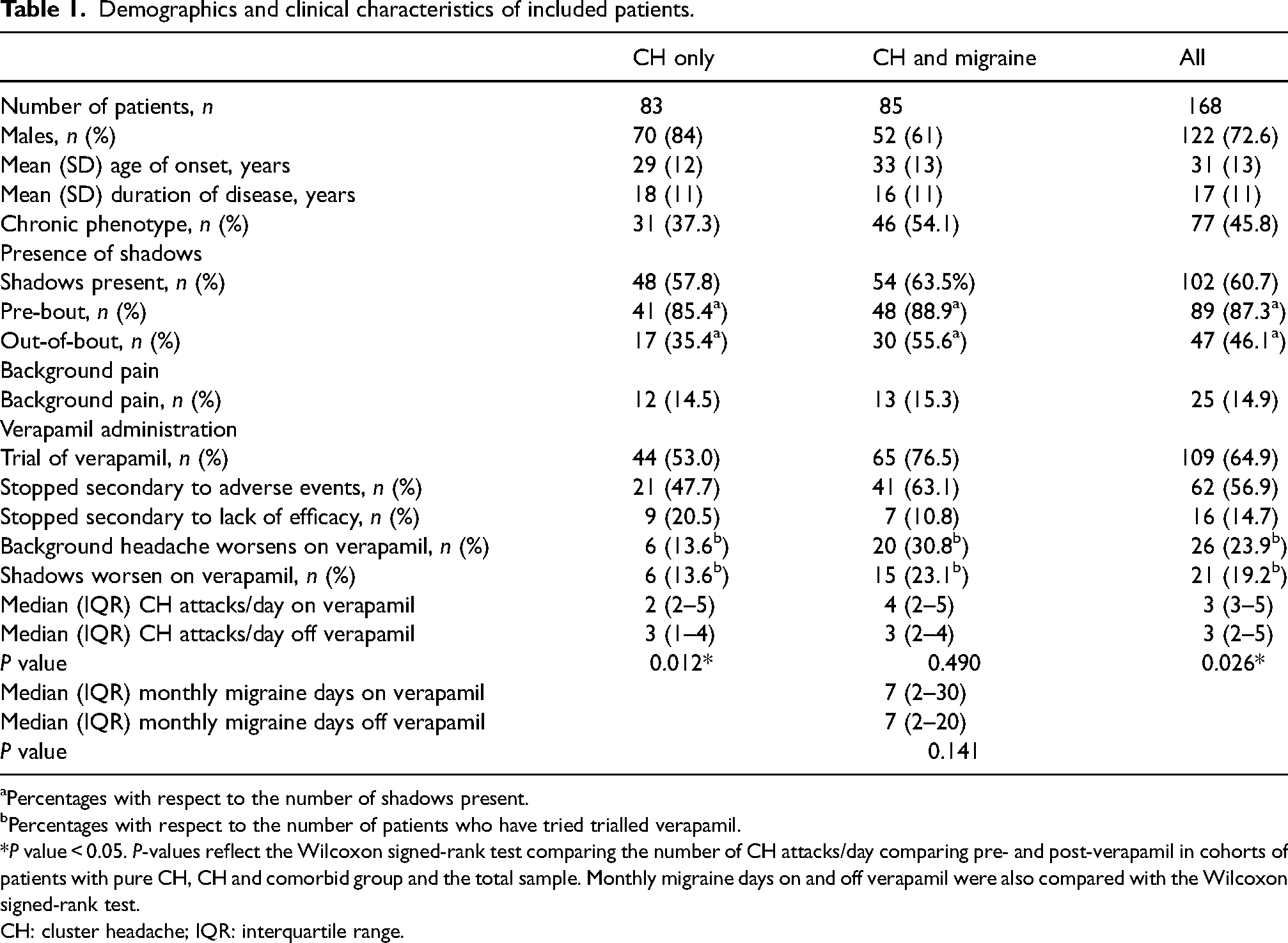
Percentages with respect to the number of shadows present.
Percentages with respect to the number of patients who have tried trialled verapamil.
*P value < 0.05. P-values reflect the Wilcoxon signed-rank test comparing the number of CH attacks/day comparing pre- and post-verapamil in cohorts of patients with pure CH, CH and comorbid group and the total sample. Monthly migraine days on and off verapamil were also compared with the Wilcoxon signed-rank test.
CH: cluster headache; IQR: interquartile range.
Shadows
Of 168 patients, 102 (60.7%) reported experiencing shadows distinct from their CH attacks, with the majority occurring before the CH attack (n = 89, 87.3%). Notably, 47 (46.1%) patients also reported experiencing spontaneous shadows occurring out-of-bout. Patients with eCH (n = 47) reported shadows more often than those with cCH (n = 40), however, this difference was not significant (χ2 (1, n = 168) = 1.451, P = 0.228). Furthermore, no differences were observed in the frequency of shadow attacks amongst those with comorbid migraine (n = 54/85, 64%) versus patients without migraine (n = 48/83, 58%) (Mann-Whitney U = 3728, z = 0.754, P = 0.451). This was true of both pre-bout (P = 0.458) and out-of-bout shadow attacks (P = 0.144), where pre-bout was referred to as shadows arising days or weeks before the first attack in an upcoming CH bout.
Background pain
Of 168 patients, 25 (14.9%) reported background pain which did not meet the criteria for CH attacks nor shadows. Similar proportions of patients in the CH group and CH with comorbid migraine group were affected by background pain, see Table 1.
Comorbid migraine
Of 168 patients, 85 (50.6%) also had comorbid migraine. Within this group, the number of males were twice that of the number of females, however, it was observed that females were more likely to have CH alongside comorbid migraine (χ2 (1, n = 168) = 10.705, P = 0.001). The mean age of onset of CH was 33 ± 13 (mean SD, years) in those with comorbid migraine, compared to 29 ± 12 in those without migraine, which was not different (χ21 = 55.444, P = 0.311, n = 163). Similarly, no differences were observed in disease duration between the two groups: CH and migraine: 16 ± 11 (mean SD, years) compared .to the non-comorbid group (18 ± 11; n = 168) χ21, = 52.178, P = 0.135). cCH was more common in patients with comorbid migraine (46 chronic vs. 39 episodic), which was significant (χ21 = 4.159, P = 0.041, n = 168). Over half (55%, n = 47) of the patients with CH and comorbid migraine reported no significant family history of headache in their first-degree relatives. In patients with a family history of headache, migraine (37%, n = 31) was more frequently reported compared to CH (2%, n = 2) in first-degree relatives. Five patients (6%) had a significant family history of both CH and migraine. The median (IQR) frequency of CH attacks per day in those with concomitant diagnoses of migraine was 3 (2–5).
Verapamil-worsening pain
Within the total sample, 109 (64.9%) patients had trialled verapamil as a preventive medication for CH with a mean (SE) total daily dose of 464 ± 31 mg. Prior to the trial, all patients were regarded as verapamil-naïve. Associated with the starting period of verapamil, 46.8% of patients (n = 51) reported no changes in their CH and/or background pain, whilst 23.9% of patients (n = 26) reported worsening. Given the retrospective nature of the study, 32 patients were unable to remember the effects of verapamil as a result of multiple medications tried and time passed since taking the medication. Of 26 patients who described worsening of their CH and/or background pain, 20 (76.9%) had comorbid migraine.
Of 109 patients who had trialled verapamil, 61 reported shadows. During the starting period of verapamil, it was observed that 45.9% (28/61) of patients reported no changes in the frequency of shadows, whilst 34.4% (21/61) reported worsening of shadows. In the patient group that reported worsening of shadows, 13 (61.9%) similarly reported worsening of CH and/or background pain, representing a deterioration in headache following verapamil initiation. Importantly, of the patients who reported worsening of shadows alongside worsening of CH and/or background pain, 84.6% (11/13) had co-existent migraine. Only five patients reported that verapamil helped shadows. The effects of verapamil were not remembered by 14 patients.
Adverse events
Adverse events were reported in 62 (56.8%) of 109 patients taking verapamil, which was further split by diagnoses: CH only: n = 21/44, 47.7% and diagnosis of CH and co-existent migraine: n = 41/65, 63.1%. Nine patients were unable to provide information relating to the adverse effects of verapamil. The most frequently reported adverse event was PR prolongation resulting in first-degree atrioventricular heart block (n = 17, 15.6%), consistent with previous studies.20,21 Other adverse events included lower limb oedema (n = 9, 8.3%), palpitations (n = 8, 7.3%), worsened headaches (n = 7, 6.4%) and fatigue (n = 7, 6.4%). A further 16 patients (14.7%) discontinued verapamil as a result of reduced efficacy (CH only: 9/44, 20.5% and CH with migraine: 7/65, 10.8%).
Verapamil – effects on CH attacks
Patients with CH and CH plus migraine
After accounting for missing data, 76 patients of both CH and CH with co-existent migraine were included for analysis. It was observed that 46.1% (35/76) of patients reported no change in CH attack frequency upon the initiation and maintenance of verapamil, whilst 34.2% (26/76) of patients reported a reduction in the number of attacks and 19.7% (15/76) of patients reported a greater number of attacks per day. The median (IQR) frequency of CH attacks per day in patients taking verapamil was reduced to 3 (3–5) compared to their original baseline (number of attacks per day prior to verapamil initiation) of 3 (2–5; W = 260; z = −2.22, P = 0.026), a change for only a proportion of the cohort.
A negative binomial generalised linear model was used to predict the number of attacks post-verapamil initiation as a function of age, sex, verapamil dosing, comorbid migraine and baseline number of attacks. The presence of comorbid migraine increased the likelihood of headache exacerbation following the initiation and maintenance of verapamil by an odds ratio of 1.616 (95% confidence interval: 1.059–2.465, χ21 = 4.955, P = 0.026). All other predictors were non-significant.
Patients with CH alone
Stratifying the sample by patients with a diagnosis of CH only, 33 patients were able to provide estimates for the number of CH attacks per day pre- and post-verapamil administration. Verapamil use was associated with a reduction in the number of CH attacks per day in 13 (39.4%) patients, whereas 16 patients (48.5%) saw no improvement in headache frequency and 4 patients (12.1%) experienced an increased number of CH attacks per day. The median (IQR) frequency of CH attacks per day in patients taking verapamil was 2 (2–5), which was less than those not taking verapamil 3 (1–4) (W = 24; z = −2.50, P = 0.012).
Patients with CH and comorbid migraine
Within the sample, 43 patients had a diagnosis of CH with comorbid migraine and were able to provide estimates for the number of CH attacks per day pre- and post-verapamil administration. Verapamil use was associated with a reduction in the number of CH attacks per day in 13 (30.2%) patients, whereas 19 (44.2%) patients saw no improvement in headache frequency and 11 (25.6%) patients experienced an increased number of CH attacks per day. The median (IQR) frequency of CH attacks per day in patients taking verapamil was 4 (2–5) compared to those not taking verapamil 3 (2–4), which was not different (W = 126; z = −0.69, P = 0.490).
Effect on migraine days in patients with CH
Additionally, 34 patients were able to quantify the number of migraine days experienced in 1 month (MMD) pre- and post-verapamil administration. Verapamil use increased the number of MMDs in 12 (35.3%) patients, whereas 15 (44.1%) patients observed no change and 7 (20.6%) patients reported a reduced number of MMDs. The median (IQR) number of MMDs in patients using verapamil as a CH preventive was 7 (2–30) days, which was no different to those not using verapamil (7, 2–20; W = 131.5; z = 1.470, P = 0.141).
Discussion
We conducted a cross-sectional service evaluation to investigate whether the initiation and maintenance of verapamil is associated with any previously unrecognised side effects that patients should be aware of, particularly worsening of shadows and increased background pain. From observations in clinical practice, we considered this worsening may be secondary to co-existent migraine. Our evaluation demonstrates that the presence of comorbid migraine increases the likelihood of headache exacerbation following the initiation and maintenance of verapamil by an odds ratio of 1.616. No differences were observed in the number of monthly migraine days pre- and post-verapamil initiation.
Pre-cluster symptoms in the CH literature are not well-described. Pederson and colleagues found in a sample of 100 patients, 86% patients reported at least one pre-cluster symptom, with two of the most frequent symptoms in the pre-cluster period reported as a dull/aching sensation (62%) and shadow attacks (60%). 18 Out-of-bout symptoms were common and reported in 58% of the eCH patients. 18 Earlier studies reported the prevalence of pre-cluster symptoms to be 10%, 22 33% 23 and 40.5%, 24 respectively. Consistent with recent literature, we found that 61% of patients experience shadow attacks, with 56% of patients reporting spontaneous out-of-bout shadows. No statistical differences were observed in the frequency of shadow attacks amongst those with comorbid migraine (n = 54/85, 64%) versus patients without migraine (n = 48/83, 58%).
The literature on the occurrence of CH and migraine together is modest. Early studies by Solomon and Cappa suggested that migraine often developed before CH, and disappeared after the onset of CH. 25 In contrast, D’Amico and colleagues reported 9 of 10 patients had CH and migraine that persisted together. 26 Here, we report that approximately half of the patients with CH had underlying migrainous biology (n = 85/168, 51%), with significant associations shown for female gender and cCH. Despite the different clinical manifestations of CH and migraine, it is unsurprising to acknowledge their co-existence given how common migraine is in the community, 3 suggesting that the diagnosis of migraine may be overshadowed by the diagnosis of CH. Genome-wide association studies provide further support for their co-existence. Recent studies have identified one locus on chromosome 6, covering FHL5 and UFL1, that overlaps between the two primary headache disorders,13,27 notably, with a greater effect size for CH (OR≈1.30) than for migraine (OR≈1.09), implying an increased risk of developing CH. 13 Further similarities can also be observed in the proposed anatomical structures underlying CH and migraine, with both placing a particular pathophysiological importance on the activation of the trigeminovascular system.28, 29
Our evaluation found that the presence of comorbid migraine increased the likelihood of headache exacerbation following the initiation and maintenance of verapamil by an odds ratio of 1.616. Exacerbations of background head pain and shadow attacks were reported more often in patients with co-existent migraine and CH (30.8%; 23.1%) compared to those without migraine (13.6%; 13.6%). No differences were observed in the median number of monthly migraine days pre- and post-verapamil. Although there is no previous evidence that links verapamil initiation and maintenance with worsening of headache, subjective patient reports can be found in both clinical practice and patient support sites, such as OUCH UK. 30 As such, we advocate that in patients experiencing headache worsening during the starting period of verapamil, physicians look for a comorbid diagnosis of migraine that may have been missed.
Adverse events were common, and occurred in 62 (56.9%) of 109 patients taking verapamil. We observed that in patient with CH with co-existent migraine, the incidence of adverse events was higher (63.1%) compared to patients diagnosed with CH only (47.7%). This suggests that alongside an increased likelihood of headache exacerbation, patients with comorbid migraine may also exhibit reduced tolerability to the medication, resulting in medication failure. Further research is warranted to explore this association further in order to optimise CH care and prevent unnecessary harm.
Limitations
A major limitation to the evaluation is the retrospective study design, particularly as it limited the amount of information patients were able to supply or accurately recall. Moreover, the study design reduced the ability to differentiate easily among the four different pain types considered by the evaluation, notably, CH, shadows, background pain and migraine. This was further hindered by the absence of diary data, which may further reduce the accuracy of headache frequency and phenotyping. Considering the above, we acknowledge the work needs to be repeated using a prospective study design with a combination of diary and patient self-reported information to increase reliability and accuracy of the findings.
To combat missing information in the evaluation, one researcher contacted the patients via telephone to clarify and gain further details missing from clinical documentation. Open-ended questions without hints were used to decrease the risk of suggestibility from the interviewer to the patient. However, considering the evaluation duration (June 2014–December 2023), some patients were not able to remember the effects of verapamil whilst others confused verapamil with other medication tried for CH. As a result, some data, such as the time spent on verapamil, etc., were unable to be analysed in the current evaluation.
Furthermore, it is important to consider that all patients were recruited from a tertiary headache service, which may have resulted in selection bias and limited generalisability as a result of patients having more severe forms of headache disorder not seen as frequently within the wider public. Additionally, some patients with dual diagnoses of both CH and migraine did not acknowledge they also were affected by migraine, prioritising their CH diagnosis above it due to its increased severity, frequency and impact on their quality of life. Although, our evaluation reports that comorbid migraine increases the likelihood of headache exacerbation post-verapamil administration, it must be acknowledged that an increased number of shadows may also account for positive pharmacological effects of verapamil and thus, improvement of a CH bout.
Limited data exists regarding patients with multiple diagnoses of primary headache disorders, and we believe that our study offers insights linking comorbid migraine to headache worsening during the starting period of verapamil, making it worthy of wider sharing. Further strengths include that a single physician interviewed all patients via telephone and careful phenotyping and application of ICHD-3 1 were used, ensuring the validity of diagnoses.
Conclusion
It is likely the association of CH and migraine is more common than generally thought, and co-existence in clinical practice may go under-recognised, reducing the effectiveness of clinical management and symptom optimisation. We found that that comorbid migraine increases the likelihood of headache exacerbation, particularly shadow worsening and background pain, following the initiation and maintenance of verapamil, which is in keeping with observations from clinical practice and subjective patient reports. Patients who experience headache worsening in these instances should be considered for migraine. We recommend that physicians take a direct approach to exploring a comorbid diagnosis of migraine, as it is possible that patients may overlook their symptoms of migraine or be less willing to share information regarding migraine in order to prioritise their CH diagnosis. Going forward, we consider the importance of informing patients of the risk of headache worsening as part of our routine practice.
Highlights
The association of cluster headache and migraine is more common than generally thought and co-existence may go under-recognised. The presence of comorbid migraine increases the likelihood of headache exacerbation following the initiation and maintenance of verapamil for cluster headache in keeping with observations from clinical practice and subjective patient reports. Going forward, physicians should consider the importance of informing patients of the risk of headache worsening as part of routine practice when starting verapamil.
Footnotes
Declaration of conflicting interests
R.W. reports no conflicts. P.J.G. reports, over the last 36 months, grants from Kallyope and personal fees from Aeon Biopharma, AbbVie, Aurene, CoolTech LLC, Dr Reddy's, Eli-Lilly and Company, Linpharma, Lundbeck, Pfizer, PureTech Health LLC, Satsuma, Shiratronics, Teva Pharmaceuticals, Tremeau and Vial, and personal fees for advice through Gerson Lehrman Group, Guidepoint, SAI Med Partners, Vector Metric, and fees for educational materials from CME Outfitters and WebMD, and publishing royalties or fees from Massachusetts Medical Society, Oxford University Press, UptoDate and Wolters Kluwer, and a patent magnetic stimulation for headache (No. WO2016090333 A1) assigned to eNeura without fee.
Ethical approval
Informed consent was not required for use of patient data for this study, as the data was acquired from patient clinic letters as part of an evaluation process; therefore, only local hospital approval was needed.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Our work is funded by the South London and the Maudsley NIHR Biomedical Research Centre (NIHR203318).
