Abstract
Background
Previous studies have reported notable differences in demographic and clinical features of cluster headache between Western and Asian populations, including lower prevalence of the chronic type and in women. Recently, prodromal symptoms of migraine and pre-attack symptoms of cluster headache have drawn attention regarding their potential pathophysiological implications and pre-emptive treatment. However, pre-attack symptoms of cluster headache have not been studied in the Asian population.
Methods
A total of 136 patients with cluster headache (21 first-onset, 110 episodic, and five chronic cases) were recruited in this multi-center study between October 2018 and December 2019. We evaluated the characteristics of pre-attack symptoms in a current bout using a structured questionnaire. Univariable and multivariable logistic regression analyses were performed to evaluate the factors associated with presence of pre-attack symptoms.
Results
Pre-attack symptoms were reported in 71.3% of our patients with cluster headache. When present, pre-attack symptoms occurred at a median of 20 minutes (range 1–120) before the attack. The prevalence of local and painful, local and painless sensory, autonomic, and general symptoms was 58.8%, 14.7%, 11.0%, and 30.1%, respectively. Multivariable logistic regression analysis revealed that higher number of bouts was associated with higher prevalence of pre-attack symptoms (OR = 1.464, p = 0.044).
Conclusions
Pre-attack symptoms were frequently observed in Korean patients with cluster headache, which was consistent with previous Western studies.
Introduction
Cluster headache (CH) is the most painful of the primary headaches, typically characterized by recurrent unilateral headache accompanied by cranial autonomic symptoms (1,2). Some patients with CH can predict upcoming attacks minutes or hours before, based on so-called pre-attack symptoms that are similar to the prodromal symptoms in migraine (3–8).
Pre-attack symptoms have been studied only in Western populations (3,5–8). Previous studies have demonstrated a 55% to 98% prevalence of pre-attack symptoms in CH. In a Danish prospective study using a diary, general symptoms such as concentration difficulties and mood changes occurred in almost half of all attacks, suggesting hypothalamic involvement in the pathophysiology of pre-attack symptoms (5). In addition to understanding the pathophysiology of CH, pre-attack symptoms may be the key to effective management. In migraine, several studies have suggested that treatment during the premonitory phase has the potential to be helpful (9,10). Although considerable differences in demographic and clinical features within the studies conducted on Western populations complicate direct comparison, the studies conducted in Asia have suggested several differences in demographic and clinical features between Western and Asian populations. Asians demonstrated a lower attack frequency per day, rare involvement of the lower half of the face, and a lower prevalence in women compared to the Western population. Moreover, the chronic type of CH, aura, and restlessness were less prevalent among Asians (11–14). From this perspective, the pre-attack symptoms in the Asian population might be different from those observed in the Western population.
However, to date, the prevalence and characteristics of pre-attack symptoms have not been reported in the Asian population. In this study, we aimed to investigate the prevalence and characteristics of pre-attack symptoms within active periods in Korean patients with CH.
Methods
Study setting and patients
This cross-sectional study was performed using data from the Korean Cluster Headache Registry (KCHR) version 2, which was a multicenter, prospective, and observational study that consecutively enrolled patients with CH. The KCHR version 1 comprehensively collected clinical characteristics during CH attacks and comorbidities. The detailed data of the KCHR version 1 have been described in our previous studies (15–17). The KCHR version 2 collected clinical characteristics before CH attacks or bouts. Patients who visited the participating hospitals were examined and interviewed by a headache specialist to confirm the diagnosis of CH based on the criteria of the third edition of the International Classification of Headache Disorders (ICHD-3) (1). Patients who consented to enrolment in the registry received a structured questionnaire. In this study, patients with CH within active periods were recruited from 12 hospitals in Korea from October 2018 to December 2019. We recruited patients with CH (ICHD-3 code 3.1), episodic CH (code 3.1.1) and chronic CH (code 3.1.2). Patients who had had a first episode of CH were classified as CH (ICHD-3 code 3.1). Patients with probable CH (ICHD-3 code 3.5) were excluded. The Institutional Review Board of each participating hospital approved this study. Both routine preventive and abortive treatments for CH were allowed. Written informed consent was obtained from all patients.
Clinical evaluation
All patients completed a structured questionnaire designed for the evaluation of CH. We collected demographic data and clinical information regarding current and previous bouts, including headache characteristics, disease duration, headache frequency (headache attacks per day), headache severity (numeric rating scale from 0 to 10), headache duration, diurnal rhythmicity, total number of bouts that was independent of disease duration, premorbid migraine, smoking history (current smoker, ex-smoker, or never smoked), and alcohol intake.
Assessment of pre-attack symptoms
A pre-attack symptom is defined as a symptom occurring minutes or hours before an attack during a bout of CH (6). In this study, pre-attack symptoms were defined as any symptoms preceding and forewarning of a patient’s usual severe CH attack by minutes or hours. To minimize recall bias, we assessed pre-attack symptoms during the current bout.
The clinical characteristics of any pre-attack symptoms during the current bout were assessed using the structured questionnaire. Only patients in active cluster periods were recruited in order to avoid recall bias. We selected 20 relevant symptoms and signs based on previous studies (3,7) and those validated by the KCHR study group. Patients could describe the symptoms that were not included in the list in their own words. The patients were further asked to indicate the presumed average interval of time (in minutes) from the pre-attack symptoms to onset of pain during the current bout. Patients who had taken any rescue medication during the pre-attack symptoms were questioned about the frequency of medication use and its effect compared to taking the medication after onset of the headache.
Pre-attack symptoms were grouped into four subtypes to investigate their characteristics: Local and painful (three items), local and painless sensory (two items), autonomic (six items), and general symptoms (11 items). Multiple pre-attack symptoms are defined, where the sum of any type that a patient has experienced before each cluster headache attack during the current bout comprises more than two types of symptoms.
Statistical analyses
Data are presented as number (percentage) or median (interquartile range, IQR). The Chi-square test or Fisher’s exact test and Student’s t-test, Mann–Whitney U test, or Kruskal-Wallis test were used for comparison of categorical and continuous variables, respectively in the groups depending on the normality of distribution. Bonferroni’s correction was applied for multiple comparisons. Univariable and multivariable logistic regression analyses were used to assess factors associated with the presence of pre-attack symptoms. Variables with univariable p-value of <0.20 were considered in the multivariable logistic regression analysis. We included age, sex, smoking, alcohol intake, diurnal rhythmicity, total bouts, premorbid migraine, diagnosis of CH, disease duration, and attack severity, and duration and frequency of the current bout as covariates. The Spearman correlation analysis was used to test the correlation among subtypes of pre-attack symptoms. Statistical analyses were performed using IBM SPSS 22.0 (IBM Corporation, Armonk, NY, USA). A two-tailed p-value of <0.05 was considered significant.
Results
Patients
Among the 157 patients recruited during the study period, 16 patients with probable CH and five patients who did not complete the structured questionnaire were excluded. A total of 136 patients were finally included in this study. Of these, 21 (15.4%) were diagnosed with CH, 110 (80.9%) with episodic CH, and five (3.7%) with chronic CH. Demographic data and baseline characteristics of the included patients are summarized in Table 1. The median age of patients was 36 (IQR, 30.0–42.8), with a male predominance (77.2%). Median disease duration was 7.1 (IQR, 2.9–11.4) years and the median of the total number of bouts was 7 (IQR, 4.0–10.0). Diurnal rhythmicity during a bout was reported in 89 (65.4%) patients. Higher total number of bouts were observed in the patients with pre-attack symptoms (p = 0.040) on comparison of the demographics and characteristics of patients with and without pre-attack symptoms. No significant differences were observed in the demographics, diagnoses (CH vs. episodic CH vs. chronic CH), disease duration, diurnal rhythmicity, and characteristics of the attack during a bout between the two groups.
Demographics and characteristics of patients with cluster headache according to pre–attack symptoms.
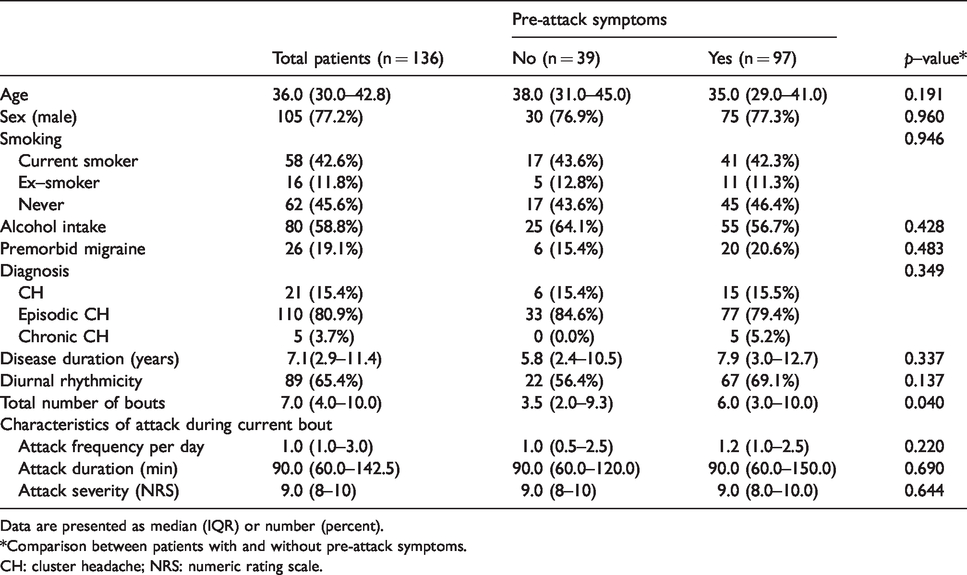
Data are presented as median (IQR) or number (percent).
*Comparison between patients with and without pre-attack symptoms.
CH: cluster headache; NRS: numeric rating scale.
Prevalence of pre-attack symptoms
Ninety-seven (71.3%) patients reported the presence of pre-attack symptoms. In addition to the items in the questionnaire, two items (eye blinking and chest discomfort) were included based on the patients’ own descriptions. The detailed prevalence of pre-attack symptoms in patients is shown in Table 2. The median time interval from the pre-attack symptom to headache was 20 (IQR, 10–30; range, 1–120) minutes. The symptoms occurred in a median of 90% (IQR, 80–100) of the attacks during a cluster bout. The most frequent pre-attack symptom was pain in the same area as the subsequent attack (41.2%), followed by neck pain (20.6%), sensory changes over the same area (13.2%), and yawning (11.0%). Based on the stratification into four subtypes, the prevalence of local and painful, local and painless sensory, autonomic, and general symptoms was 58.8%, 14.7%, 11.0%, and 30.1%, respectively. A weak correlation was found between the presence of local and painful symptoms and general symptoms (Rho = 0.289, p < 0.001).
Prevalence of pre-attack symptoms of patients.
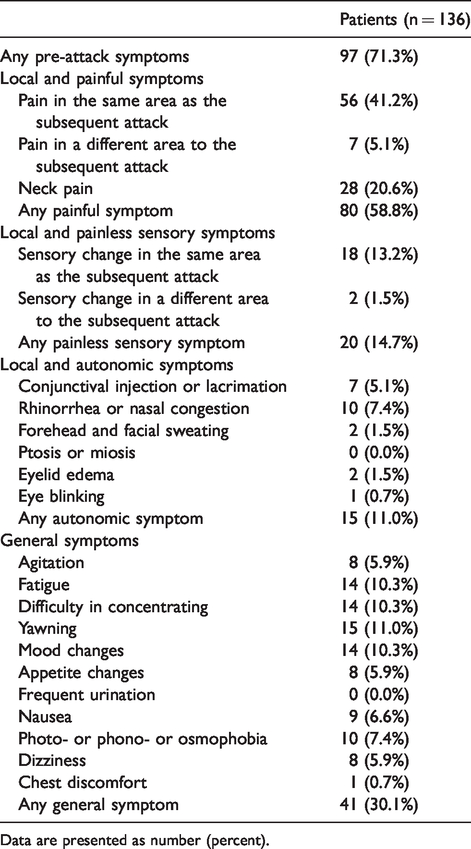
Data are presented as number (percent).
The mean number of pre-attack symptoms per patient was 2.4 (SD 2.1, range 1–13). Among them, 90.7% (88/97) reported one to four pre-attack symptoms. Fifty (36.8%) patients reported one pre-attack symptom during a bout, while 47 (34.6%) patients reported more than two symptoms (Table 3). Among the single pre-attack symptoms, the local and painful symptom (27.2%) was most frequent. Among the multiple pre-attack symptoms, local and painful plus general symptoms (17.6%) were most frequent.
Prevalence of pre-attack symptoms according to types.
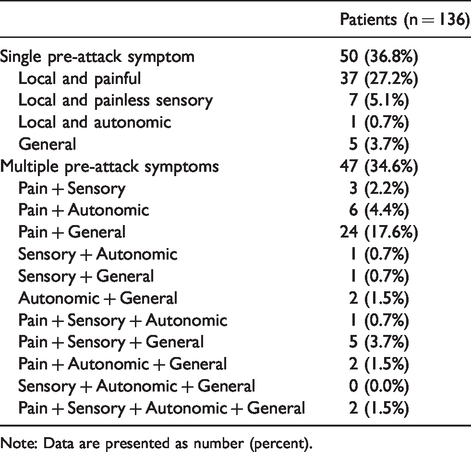
Note: Data are presented as number (percent).
Factors associated with pre-attack symptoms
The factor associated with the presence of pre-attack symptoms in the univariable and multivariable logistic regression analyses was the total number of bouts (Table 4). Higher total number of bouts was independently associated with higher prevalence of pre-attack symptoms (multivariable odds ratio = 1.464, 95% confidence interval = 1.010–2.124, p = 0.044). Greater male predominance and alcohol intake were observed in patients with a single pre-attack symptom on comparison of the data between patients with single and multiple pre-attack symptoms (Table 5). The median time interval from pre-attack symptoms to the attack, disease duration, and attack duration were significantly longer in patients with multiple pre-attack symptoms than in those with a single symptom (30.0 vs. 10.0, p = 0.013; 10.6 vs. 5.2, p = 0.018; and 120.0 vs. 60.0, p = 0.006; respectively). The total number of bouts, attack frequency, and severity did not differ between the groups.
Univariable and multivariable logistic regression analyses to assess associated factors with the pre-attack symptoms.

*Variables (p < 0.2 in univariable analysis) were included.
CI: confidence interval; OR: odds ratio.
Characteristics according to single and multiple pre-attack symptoms.
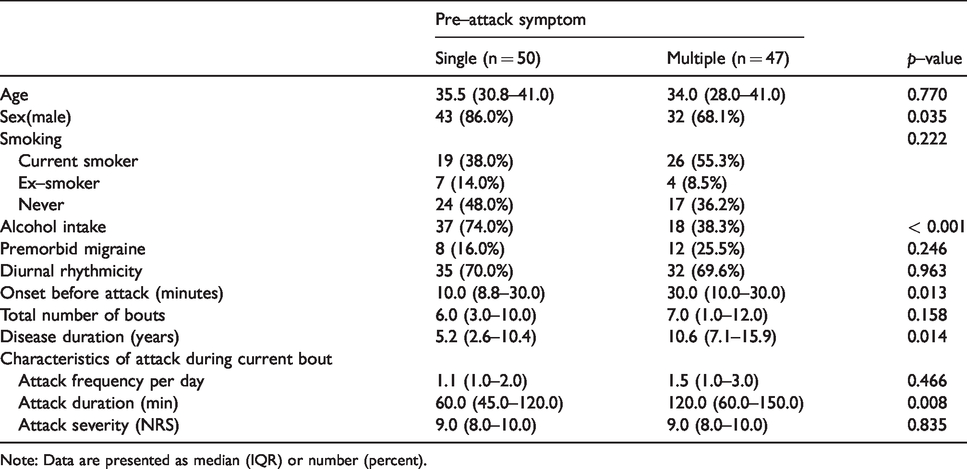
Note: Data are presented as median (IQR) or number (percent).
Rescue medication during pre-attack symptoms
A total of 42 (n = 42/97, 43.2%) patients tried rescue medications during the pre-attack symptoms. Among them, 39 patients reported taking rescue medications during more than half of the times that pre-attack symptoms occurred. Most patients (n = 36/42, 85.7%) experienced better response to the attack than on taking the medication during the attack.
Discussion
To the best of our knowledge, this cross-sectional observational study is the first in Asia to investigate the characteristics of pre-attack symptoms in CH. The main findings of this study were as follows: i) Pre-attack symptoms were observed in more than two thirds of patients with CH, which was independently associated with higher number of bouts. The prevalence of pre-attack symptoms in Korean CH patients was consistent with previous Western studies. ii) Local and painful symptoms of pre-attack symptoms were most frequent, followed by general symptoms. iii) Most patients reported better response to the attack when rescue medications were taken before, rather than during the attack.
Prevalence of pre-attack symptoms
Prevalence of pre-attack symptoms in the Korean population was 71.3%. Although methodological differences complicated precise comparison of our data with previous studies conducted on Western populations, our results were consistent with the prevalence rates reported in Western studies (Table 6). Blau and Engel were the first to investigate warning symptoms of CH, and reported that 61.3% patients had prodromata in the minutes before the pain (3). The authors raised an open-ended question but excluded painful symptoms as pre-attack symptoms. Torelli et al. and Donnet et al. exclusively investigated episodic and chronic CH, respectively (7,8), and reported a significant difference in the prevalence rate between episodic and chronic CH (98% vs. 55%). This difference could probably be attributed to the methodological differences between the two studies. An Italian study on episodic CH investigated pre-attack symptoms based on a 65-item list (7), whereas a French study on chronic CH asked patients to describe symptoms in their own words (8). There was no difference in the prevalence rate between episodic and chronic CH in a Danish study (6). In our study, 70% of the patients with episodic CH and all five patients with chronic CH had pre-attack symptoms. In two Danish cohort studies conducted in the same clinic, the prevalence rate was a bit higher (86% and 95%) than that reported in our study (5,6). The difference in the prevalence may be attributed to there being fewer symptoms on the list of the questionnaire and shorter disease duration in Korean patients with CH. The disease duration in patients in our study was 7.1 years, whereas previous studies on Western patients reported durations of approximately 14 to 16 years (5–7). Table 6 presents an up-to-date summary of the studies investigating pre-attack symptoms in CH.
Summary of studies investigating the pre-attack symptoms in cluster headache.

*Data are presented as mean or as otherwise specified.
†Excluding pain symptoms.
‡Including pain symptoms.
cCH: chronic cluster headache; N: number of subjects; NA: not available.
The median time interval from the pre-attack symptom to the attack was 20 minutes, which was remarkably shorter compared to migraine, wherein the premonitory symptoms were reported to begin 2 to 48 hours prior to the headache phase (18,19).
In our study, a higher number of bouts was observed in patients with pre-attack symptoms. Total number of bouts was independently associated with higher prevalence of pre-attack symptoms. This implies that the pattern of the symptoms of CH can be predicted based on the repetition of cluster bouts. However, it is uncertain whether it is associated with the pathophysiological changes of CH over time or not. It is possible that patients with a larger number of bouts had a better memory of pre-attack symptoms. This issue can be solved by study using a diary consecutively for each bout.
Characteristics of pre-attack symptoms
In our study, painful symptoms occurred in 58.8% of the patients. Previous studies reported that 36% to 90% of patients experienced painful symptoms (5–8). The variations in rates among studies may be attributed to methodological differences. No two studies have used the same methodology and really investigated the same question. This heterogeneity of the studies complicates direct comparison between the previous Western studies and the current study. In Danish studies, dull and aching sensation, and fullness of the ear were included as painful symptoms, while these were considered painless symptoms in an Italian study. In our study, we categorized the painless sensory symptom as an independent subtype. Among the painful symptoms, neck pain was observed in one-fifth of the patients, a similar rate to that reported in a previous study (6). Autonomic and general symptoms occurred in 11% and 30% of our patients, respectively, while the reported rates in Western cohorts were 43% to 70% and 50% to 79%, respectively (5–7). Although we added an open-ended question, our questionnaire had fewer items than in other studies. This discrepancy might have affected the lower rates observed in our study. The most common pre-attack symptoms in order of frequency were painful, general, and autonomic symptoms, which was the same order as that reported in Western studies (5–7).
Pathophysiological implication of pre-attack symptoms
Understanding premonitory symptoms of migraine offers great insight into the pathophysiology of the disease. Correlation of symptoms with functional changes by neuroimaging study during the premonitory phase has suggested involvement of the hypothalamus and brainstem before onset of the headache phase (20,21). Since premonitory symptoms are not specific to migraine, functional imaging during the pre-attack symptoms of CH might provide insights into the pathophysiology of CH. However, there are limited functional imaging studies on the pre-attack phase of CH because of its short duration.
In our study, some symptoms that occur during an attack were also observed in the pre-attack symptoms. Previous studies hypothesized that the trigeminovascular system, parasympathetic nerve fibers, and the hypothalamus are involved in CH attacks (22). Combined with the previous observations of pre-attack symptoms, we hypothesize that these structures could be involved in the early stages prior to CH attacks. In our study, most of the painful or sensory pre-attack symptoms occurred on the ipsilateral side of the attack. These ipsilateral pre-attack symptoms are similar to so-called “shadow attacks”, which consist of a low-level ipsilateral pain that can occur outside of the CH bouts (23,24). Our result may support the hypothesis that the hypothalamus has a fundamental role in generation of a permissive state that allows the initiation of a CH attack (22). Moreover, a recent study using diary data of patients showed that general symptoms occurred in the early preictal phase and were followed by painful and autonomic symptoms, which suggested the central role of the hypothalamus in the pre-attack symptoms (5).
Treatment implications
Several studies have suggested that pre-emptive treatment during the premonitory phase of migraine might effectively prevent the upcoming headache phase (9,10,25). In our study, among the patients who tried rescue medications, 86% reported satisfaction with use of the medications during the pre-attack symptoms. Pre-attack symptoms in CH might enable patients to use abortive treatment more effectively. However, in the present study, we evaluated neither the therapeutic effect for each attack nor types of rescue medication. Further randomized, placebo-controlled studies are needed to assess the exact therapeutic effect during pre-attack symptoms.
Strengths and limitations
To the best of our knowledge, this is the first study to investigate the characteristics of pre-attack symptoms in a large number of patients diagnosed with CH in Asia. The diagnosis and characterization of CH were well investigated by experienced neurologists. Nevertheless, this study has limitations. First, since this cross-sectional study included data based on the patients’ memory, the results could be associated with recall bias. Because the patients completed a single questionnaire for all their bouts, we could not assess individual pre-attack symptoms. In addition, in this study, timing of filling out the questionnaire was not predetermined. Recall errors for multiple attacks can make calculating the time between pre-attack symptoms and the start of the attack a difficult task. Therefore, we could not calculate interval of time from the pre-attack symptoms to onset of attack exactly. Although we recruited patients during the active cluster period in order to minimize the recall bias, further prospective studies using a diary would be warranted to validate our results. Second, the predetermined 20-symptom checklist could be considered insufficient to incorporate all the pre-attack symptoms. Previous studies considered more symptoms in their checklists, which were mainly adopted from studies on migraine. However, there has been no consensus on the list of symptoms associated with CH. In addition, patients are more likely to choose symptoms irrelevant to CH in cases of longer checklists. We tried to select common symptoms linked to CH by validating previous pre-attack symptom lists prior to conducting this study, and allowed patients to describe symptoms in their own words. Third, we did not evaluate how many patients experienced pre-attack symptoms during sleep. One of the hallmark features of CH is its circadian periodicity, with attacks often occurring at night. Studies during sleep may provide potential pathophysiological and therapeutic implications. Fourth, we could not recruit an adequate number of patients with chronic CH compared to other cohorts. This could be attributed to the low prevalence of chronic CH in the Asian population. Future studies with large samples of patients with chronic CH are necessary to generalize our findings. Lastly, although pre-attack symptoms occurred in a median of 90% of the attacks during a cluster bout in our study population, we could not verify whether patients experienced the same symptoms in all attacks. Prospective studies using a diary will resolve this limitation.
Conclusion
Pre-attack symptoms were frequently observed in Korean patients with CH, which was consistent with results of previous studies on Western populations. A higher number of cluster bouts was associated with higher prevalence of pre-attack symptoms. Patients believed that taking rescue medications during the pre-attack symptoms helped to relieve upcoming CH attacks. Our findings may offer an understanding of the pathophysiology of CH and a new therapeutic strategy for CH attacks.
Clinical implications
Pre-attack symptoms were observed in 71.3% of CH patients, which was independently associated with a higher number of bouts. The prevalence of pre-attack symptoms in Korean CH patients was consistent with previous Western studies. Local and painful pre-attack symptoms were most frequent, followed by general symptoms.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors have no financial support for the research, authorship, and/or publication of this article.
