Abstract
Introduction
In contrast to the premonitory phase of migraine, little is known about the pre-attack (prodromal) phase of a cluster headache. We aimed to describe the nature, prevalence, and duration of pre-attack symptoms in cluster headache.
Methods
Eighty patients with episodic cluster headache or chronic cluster headache, according to ICHD-3 beta criteria, were invited to participate. In this observational study, patients underwent a semi-structured interview where they were asked about the presence of 31 symptoms/signs in relation to a typical cluster headache attack. Symptoms included previously reported cluster headache pre-attack symptoms, premonitory migraine symptoms and accompanying symptoms of migraine and cluster headache.
Results
Pre-attack symptoms were reported by 83.3% of patients, with an average of 4.25 (SD 3.9) per patient. Local and painful symptoms, occurring with a median of 10 minutes before attack, were reported by 70%. Local and painless symptoms and signs, occurring with a median of 10 minutes before attack, were reported by 43.8% and general symptoms, occurring with a median of 20 minutes before attack, were reported by 62.5% of patients. Apart from a dull/aching sensation in the attack area being significantly (
Conclusion
Pre-attack symptoms are frequent in cluster headache. Since the origin of cluster headache attacks is still unresolved, studies of pre-attack symptoms could contribute to the understanding of cluster headache pathophysiology. Furthermore, identification and recognition of pre-attack symptoms could potentially allow earlier abortive treatment.
Introduction
Cluster headache (CH) is a severe, primary headache disorder affecting 1–4 in 1000 in the general population. A CH diagnosis is based upon these distinct clinical features: Severe, short lasting (15–180 minutes), unilateral, periorbital headache attacks accompanied by autonomic features and/or a sense of restlessness as defined in the international classification of headache disorders 3rd edition (beta version) (ICHD-3 beta) (1). Further characterizing the disease is the distinct chronobiological pattern, with attacks occurring in clusters often at the same time of the year and individual attacks occurring at the same time of day (2). The severity of the headache attacks is also a defining feature of CH (1), but there is no precise definition of the exact start of the attack due to lack of evidence in the area. Premonitory symptoms in migraine occur hours to days before the individual attack, and those most commonly described are changes in activity level, depression, yawning and neck pain (3). Some premonitory and prodromal symptoms are described in small CH studies but, in contrast to premonitory symptoms in migraine, these features and the nomenclature used to describe them are neither well defined nor described in the ICHD-3 beta (1).
In CH, previous studies have attempted to distinguish between prodromes, described to occur minutes before the anteceding attack, and premonitory symptoms occurring days or weeks before the upcoming cluster bout, but there is no clear consensus on these definitions. Prodromes reported in CH have been quite diverse, ranging from local painless or painful sensations in the area of the subsequent attack to generalized symptoms such as anxiety, nausea or yawning suggesting a more diffuse, central affection (4). Given that premonitory symptoms in migraine refer to symptoms in the hours or days before the attack and that premonitory symptoms in CH refer to symptoms in the days or weeks before the oncoming cluster, there is a need for better distinction between these entities. In the following, and to avoid confusion, we refer to symptoms occurring minutes or hours before an attack as pre-attack symptoms and symptoms occurring days or weeks before oncoming cluster periods as pre-cluster symptoms.
Since the pathophysiology of CH and the origin of CH attacks are still unresolved, studies on both the pre-attack and pre-cluster periods could contribute to the understanding of the underlying pathophysiology of CH. Furthermore, identification and recognition of pre-attack and pre-cluster symptoms could potentially allow earlier treatment with abortive and preventive treatments respectively.
The aim of this study was to describe the nature, prevalence and duration of pre-attack symptoms and signs in CH.
Materials and methods
Patients diagnosed with episodic cluster headache (eCH) or chronic cluster headache (cCH) according to the ICHD-3 (beta) criteria (1) seen at the Danish Headache Center, a national tertiary referral center, were invited to participate in the study. To be included, patients had to be able to distinguish their CH attacks from other types of headache and they were excluded if they were currently suffering from another chronic primary or secondary headache disorder. To avoid recall bias, only episodic patients in active cluster and chronic cluster headache patients were included in the study. A semi-structured interview was conducted to confirm the CH diagnosis and obtain relevant demographical data on the cohort. A questionnaire consisting of 34 items concerning signs and symptoms was constructed by the authors and its content was evaluated and validated by eight headache specialists at the Danish Headache Center. The questionnaire and structured interview were pre-tested on 10 CH patients prior to initiation of the study. Experiences and suggestions from the field testing and focus group were implemented in the final version of the diary. The 34 signs and symptoms included previously-reported CH pre-attack symptoms, premonitory migraine symptoms and accompanying symptoms of migraine and CH (3–8). Of the 34 signs and symptoms included in the questionnaire, 31 were found to be relevant to the pre-attack phase of CH and are the focus in this study. The excluded items were on the severity of pain during attack, on whether pain worsens with physical activity, and eventual constipation.
We chose not to allow metaphoric descriptions such as “a knife stabbing my eye” for the pre-attack, headache or post-attack phases. Patients were interviewed and asked specifically if they experienced any of the symptoms or signs prior to, during, or after a typical CH attack. The start of the attack itself was defined according to the ICHD-3 beta criteria (1) as the period of time where they experienced severe or extremely severe periorbital or temporal pain with accompanying symptoms and/or restlessness. Patients were explicitly instructed to report their symptoms according to this definition. The patients were furthermore asked to give an estimate of the duration of each symptom before and after an attack. If a patient indicated that they usually experienced a given symptom, but could not give a reliable time estimate for the symptom prior to or after the attack, this was allowed.
The study did not need formal approval by the local ethics committee according to Danish law, but the committee was informed of the study and had no objections to it (Ethics committee file name: 15021655).
Statistics
Statistical analyses were performed using SAS 9.4. Patients were divided with regard to sex and CH diagnosis for group comparisons. For every pre-attack symptom or sign, a subgroup analysis was made regarding previous or current migraine diagnosis. For statistical analyses, pre-attack symptoms were divided into three categories: Local and painful (four items), local and painless (seven items) and general symptoms (20 items). The prevalence of every pre-attack symptom was calculated and presented as a percentage. Given the non-normal distribution of the duration of pre-attack symptoms, data is presented with means (SD), medians, and interquartile ranges (IQR). For the prevalence of pre-attack symptoms, group comparisons were performed using Fisher’s exact test. The correlation between pre-attack symptoms was tested using Spearman’s rank correlation coefficient, but results were omitted when there were fewer than five observations. For group comparisons, a two-sided
Results
Demographics and clinical characteristics of included patients, presented with means and standard deviations (SD) and total number and percentage N (%).

eCH: episodic cluster headache; cCH: chronic cluster headache; VRS: verbal response scale; SPG stimulation: Sphenopalatineganglion stimulation; GON block: Greater occipital nerve blockade; min: minutes.
Number of pre-attack symptoms per patient from the specified categories, presented with means and standard deviations (SD).

eCH: episodic cluster headache; cCH: chronic cluster headache. No significant differences were found in group comparisons.
Prevalence of pre-attack signs and symptoms
Prevalence of individual pre-attack symptoms in included patients. Total number and percentage N (%) within each subgroup are presented.
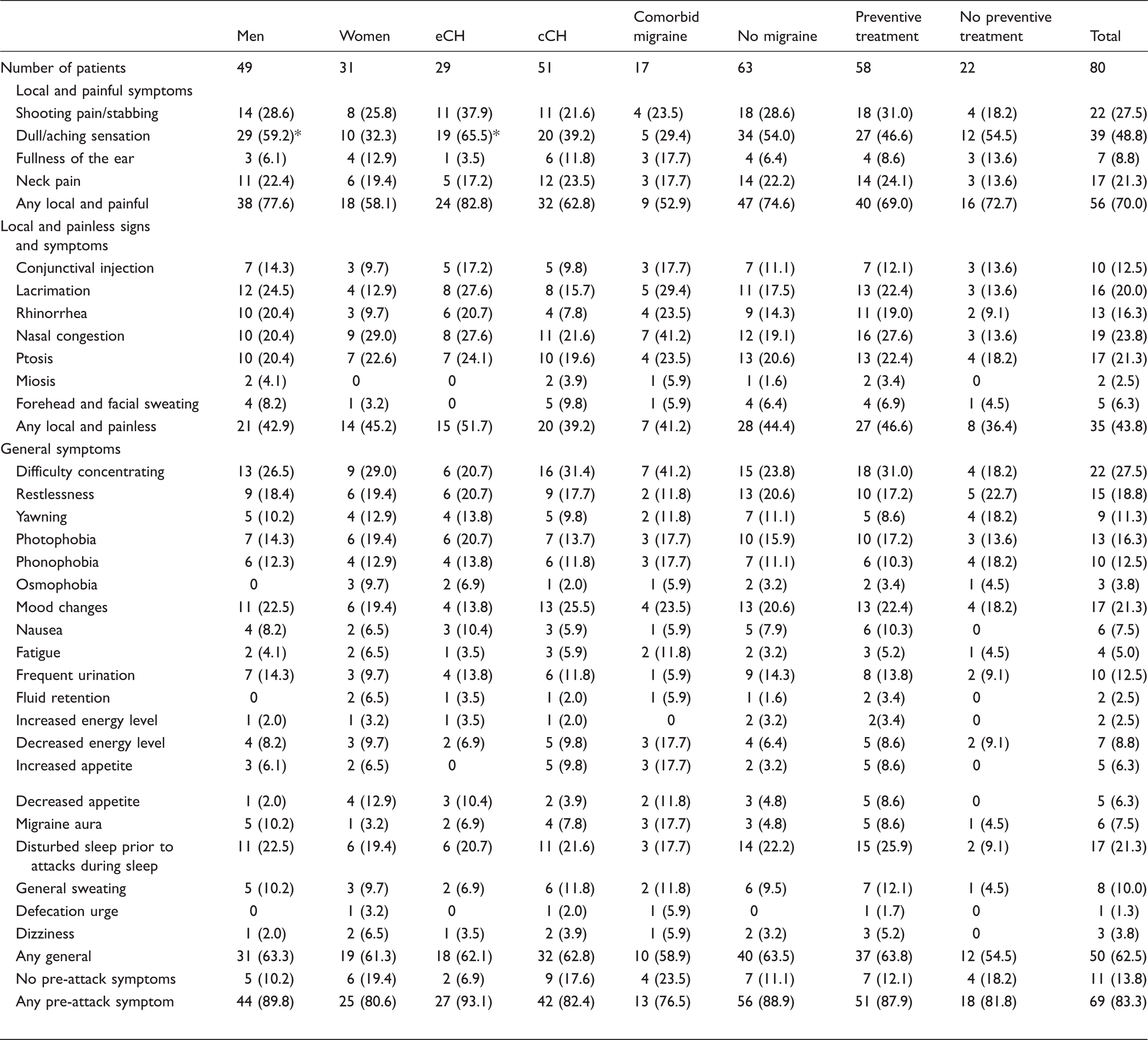
eCH: episodic cluster headache; cCH: chronic cluster headache.
Indicates significant difference between sub-group with
The presence of lacrimation was strongly correlated with the presence of other autonomic symptoms such as nasal congestion (r = 0.529) or rhinorrhea (r = 0.711). Strong correlations (p < 0.0001) were also found between photophobia and phonophobia (r = 0.551), as well as mood changes and restlessness (r = 0.612). Although the presence of one or more local and painful symptoms coincided with the presence of one or more local and painless symptoms or signs in 29 subjects, we did not see significant correlations between local and painful symptoms and cranial autonomic symptoms (CAS). Six (7.5%) patients reported the presence of CAS without the presence of any local and painful symptoms in the pre-attack phase.
Duration of pre-attack symptoms
Duration of pre-attack symptoms reported by patients. N (%) indicates the number and proportion of patients able to estimate the duration of a given pre-attack symptom.

SD: standard deviation; min: minutes; IQR: interquartile range.

Process of reported pre-attack symptoms and signs based on reported median durations (when reported by ≥5 patients). Median values of individual symptom durations before attack are shown in brackets. Min: minutes.
Discussion
This is to date the most detailed study investigating the prevalence and time profile of pre-attack signs and symptoms in a cohort of CH patients. Blau and Engel were first to describe pre-attack symptoms (called prodromata) in CH patients, and found that of 150 patients interviewed, 61.3% had prodromata in the minutes before the attack (4). Their described prodromata were quite diverse, reflecting the fact that patients were asked to describe symptoms in their own words. Torelli et al. conducted a prospective study in which 42 patients were asked to fill out a questionnaire immediately after their next typical CH attack. Choosing from a 65-item list they found that 97.6% of patients experienced one or more pre-attack (prodromal) symptoms with a mean duration of 6.3 minutes before their attack (5). The lower prevalence of pre-attack symptoms in our cohort is likely due to the fact that the present data is based on recall in relation to a typical attack and not linked to a specific attack, as was the case in the Torelli study. In the interest of comparison, we also divided the pre-attack symptoms and/or signs in this study into three categories, namely local and painful, local and painless, and general.
Local and painful symptoms
We found that the most common pre-attack symptom was a dull/aching sensation in the area of the subsequent attack experienced by almost half of the patients (48.8%), completely in line with the findings in the Torelli study (50%). Adding neck pain, ear fullness or shooting pain/stabbing, 70% of our patients had one or more local, painful symptoms. Similar to migraine, we found that CH patients can experience neck pain in the pre-attack phase; the prevalence in our CH cohort (21.3%), however, was lower compared with those reported in migraine ( ≤ 50%) (3,8–10). The cause of neck pain is speculated to be a consequence of hypothalamic dysfunction, resulting in the loss of the inhibitory effect on nociceptive trigeminal input. Due to central connections between the trigeminal and cervical spinal nerves, this leads to contraction of neck muscles (11). There have been recent discussions about whether ear fullness should be included as an accompanying symptom in the diagnostic criteria of CH, since up to 20% of patients experience this during a CH attack, although the symptom was not found to add value to the diagnostic criteria (12). We found that 8.8% of our patients had a sense of ear fullness in the pre-attack phase, which could represent a build-up phenomenon for patients experiencing this symptom. Ear fullness has also been described in the premonitory phase of a migraine attack, but the prevalence was not specified in the one study reporting this (3). Blau and Engel also reported local and painful symptoms in the area of the subsequent attack, but these were interpreted as a build-up of pain rather than a pre-attack symptom. Indeed, this could be the case for all the pre-attack symptoms, which raises the ever-pending question: When does the CH attack begin? It is well recognized that a typical migraine attack can be divided into five phases from prodromes (I) to recovery (V), acknowledging the fact that a migraine attack is not merely a headache of 4–72 hours (13). A similar model of the typical CH attack has yet to be developed, but is highly warranted.
Local and painless signs and/or symptoms
More than 40% of our patients experience one or more local and painless signs or symptoms, such as lacrimation, nasal congestion etc. prior to their attack. Although reported with lower prevalence, autonomic symptoms were also reported in the Torelli and Blau studies (4,5). Cranial autonomic symptoms are not uncommon in migraine, but in contrast to CH they have not been reported in the premonitory phase of migraine (14–16). Local and painful symptoms frequently coincided with autonomic symptoms or signs, but the fact that six patients experienced only CAS and no pain in the pre-attack phase suggests that, for this group of patients, parasympathetic outflow is increased prior to pain mediating mechanisms (17). This could lead to the speculation that autonomic activation, in this group of patients, is independent of the trigemino-autonomic reflex (18).
General symptoms
The central role of the hypothalamus in CH pathophysiology is unquestionable, and reflected in the chronobiological features (2), neuroendocrine disturbances (19) and imaging findings (20,21). As in migraine, we found that various general symptoms implicating the hypothalamus, such as mood changes, concentration difficulties, restlessness and so on are frequent in the pre-attack phase. In a recent study on 2219 migraineurs by Laurell et al., about a third of the cohort experienced yawning, mood changes, restlessness and light sensitivity in the premonitory phase before their migraine attack (22). With regards to restlessness and mood changes, the prevalence was comparable with our findings, whereas yawning, fluid retention, increased energy level and light sensitivity are more frequent in migraineurs (22). Depending on the specific symptom, the presence of the above-mentioned symptoms points to involvement of the hypothalamus or its neuroanatomical connections. Mood changes and concentration difficulties suggest involvement of the limbic system, whereas yawning, altered energy levels and appetite changes are mediated via the central dopaminergic system (23,24). Neuroimaging studies have demonstrated hypothalamic activation in the premonitory phase of migraine (25), but similar studies have not yet been done in CH, and would probably, owing to the much shorter pre-attack phase, be difficult to conduct.
Group comparisons
Apart from a dull/aching sensation being experienced significantly more among men and episodic patients, we did not see any differences between genders or eCH/cCH groups in the prevalence of pre-attack symptoms or mean number of pre-attack symptoms experienced. In a recent study by our group, we have found that the clinical features of CH, overall, are the same between genders (26), which supports the lack of differences in pre-attack symptoms. Whether the patients were episodic or chronic CH sufferers or had a comorbid migraine diagnosis played no role in prevalence of pre-attack symptoms, in line with previous studies (27,28). We did not perform any detailed group comparisons within demographics, as this was not the aim of the present study.
Duration of pre-attack symptoms
When reporting a pre-attack symptom, the majority of patients were able to estimate the duration prior to their attack. Most difficult for the patients to estimate was, understandably, the duration of disturbed sleep prior to attacks occurring during sleep. The duration of the various pre-attack symptoms was reported with wide ranges, which could be explained by either individual variances or the fact that the present data is based on recall. To present a more reliable estimate of pre-attack symptoms, one would have to observe the patients prospectively up to a CH attack and register the pre-attack symptoms in a standardized manner. This would, owing to the fickle nature of CH, be difficult to obtain for spontaneous attacks experienced by a study cohort, but perhaps possible in an experimental CH provocation model. Nevertheless, when examining median durations of pre-attack symptoms experienced by five or more patients in our cohort, we get the impression that the pre-attack phase is initiated with general symptoms roughly 60 minutes prior to the attack. Succeeding these are local and painful sensations represented by neck pain or a dull/aching sensation in the area of the subsequent attack and finally lacrimation, rhinorrhea and nasal congestion, which are reported with a median of 5 minutes before the attack. Given the scarcity of pre-existing data on this topic, we chose to approach the time-aspect of the pre-attack phase in an exploratory manor presenting all reported time-estimates of the individual pre-attack symptoms.
Strengths and limitations
This is to date the most comprehensive study on the pre-attack phase of a larger cohort of well-characterized CH patients, but compared to similar studies in migraine our sample size is still relatively small. Some of the pre-attack symptoms suggested in our interview were only reported by a few patients, which could question the validity of these particular findings. We only included patients who were in an active disease state in an attempt to limit the effect of recall bias. Based on previous similar studies in CH and migraine, we developed a comprehensive list of signs and symptoms, which we assessed relevant to the present study; there is, of course, the possibility that some patients experienced additional symptoms not represented. To accommodate these limitations, a prospective study investigating not only the validity but also the consistency of pre-attack symptoms experienced by the individual CH patient should be conducted. A prospective diary study with patients registering symptoms prior to, during and after an attack seems feasible and could furthermore form basis for a model of the entire CH attack as in migraine.
Conclusion
This study demonstrates that pre-attack symptoms are very frequent in CH. We identified several symptoms in the early pre-attack phase of CH confirming the central role of the hypothalamus in CH pathophysiology. More than 40% of our patients experience autonomic symptoms prior to onset of the severe pain characteristic of CH which may have pathophysiological implications. If the pre-attack phase of CH can be confirmed in prospective studies, the symptoms could hold a key to early treatment and understanding of the unresolved issues of CH pathophysiology.
Article highlights
Pre-attack symptoms occur in over 80% of cluster headache (CH) patients. General symptoms suggestive of hypothalamic involvement are experienced by more than 60% of patients prior to an attack. More than 40% of our patients experience autonomic symptoms prior to onset of the severe pain characteristic of CH. Pre-attack symptoms can offer insight into unresolved issues of CH pathophysiology.
Footnotes
Declaration of conflicting interests
The authors declare following potential conflicts of interests with respect to research, authorship and/or publication of this article: Agneta Snoer has received honoraria from AstraZeneca for lecturing and is a sub-investigator on clinical trials sponsored by Eli Lilly. Nunu Lund is an investigator on clinical trials sponsored by Autonomic Technologies and Xenon Pharmaceuticals. Rigmor Højland Jensen has given lectures for Pfizer, Berlin-Chemie, Norspan, Merck and Autonomic Technologies (ATI); has been a member of the advisory boards of ATI, Medotech and ElectroCore; and a principal investigator on clinical trials sponsored by Eli Lilly. Mads Barloese has received honoraria from ATI.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work has been supported by an unrestricted grant from Tryg Foundation.
