Abstract
Background
There is a growing awareness of constipation being a side effect of anti-calcitonin gene-related peptide (anti-CGRP) monoclonal antibodies (mAbs). This study aims to assess constipation as a side effect and explore its potential role as a predictor of treatment response in chronic migraine (CM) patients treated with anti-CGRP mAbs.
Methods
We conducted a retrospective analysis of patients with CM treated with anti-CGRP mAbs between January 2019 and December 2022 at a single center. Data on patient demographics, migraine characteristics, and constipation were reviewed.
Results
Among 317 patients, 192 received erenumab, 94 received fremanezumab, and 31 received galcanezumab. Constipation was significantly more common with erenumab (51.5%) compared to fremanezumab (4.2%) and galcanezumab (12.9%). Among erenumab patients, 24.4% switched due to constipation, while 19.2% switched due to lack/loss of effectiveness. Patients who switched treatment experienced milder symptoms (p < 0.0001) without compromising effectiveness, even when switching due to lack/loss of effectiveness (p = 0.0068). Importantly, severe constipation rates were higher in non-responders (p = 0.036).
Conclusion
Constipation is a notable side effect of anti-CGRP mAbs, particularly with erenumab in CM. Side effect monitoring is consequently crucial. Our findings suggest a potential link between poor treatment response and constipation, warranting further research into the underlying mechanisms.
Keywords
Introduction
Calcitonin gene-related peptide (CGRP) is a neuropeptide with a pivotal role in the pathophysiology of migraine. The observation of elevated CGRP levels during migraine attacks led to the discovery of therapies based on anti-CGRP monoclonal antibodies (mAbs) targeting either the receptor (erenumab) or the peptide itself (eptinezumab, fremanezumab, and galcanezumab), alongside small molecule CGRP antagonists known as gepants.1,2
Apart from its recognized involvement in migraine pathophysiology, CGRP assumes functions particularly within the gastrointestinal (GI) system, but also in the cardiovascular system, pain pathways, and so forth because of its widespread expression throughout the body. CGRP is produced in two isoforms in humans, α-CGRP and β-CGRP, each with distinct distributions and functions within the nervous system. α-CGRP is predominantly expressed in the peripheral nervous system, more specifically in the trigeminal ganglion while β-CGRP is mainly found in the enteric nervous system. 3 In the intestines, CGRP plays a role in facilitating the translocation of ions and water into the intestinal lumen while modulating intestinal motility. 4
Anti-CGRP mAbs have been used with remarkable success in chronic migraine (CM) patients for several years.
Double-blinded randomized clinical trials and phase IV studies indicate a favorable safety profile, including minimal GI side effects. For example, the constipation rates were reported as 4% in the phase 2 study and as 4.6% in the phase 3 study of erenumab used for preventive treatment of CM.5,6 However, constipation as a side effect of erenumab has gained widespread attention in clinical practice.7,8 Emerging real-world data revealed a significantly higher prevalence of constipation compared to early stage trials, ranging from 12% to 43%, which may lead to discontinuation.9–11 In addition, erenumab, being a CGRP-receptor-blocker, seems to cause constipation as a problematic side effect much more frequently than CGRP-ligand-blockers (eptinezumab, fremanezumab, and galcanezumab).12,13
Despite clinical observations suggesting that constipation may improve with a switch to other types of anti-CGRP mAbs, there is a notable lack of studies investigating this aspect. Furthermore, to date, no studies have explored the impact of constipation as a side effect on the clinical effectiveness of treatment in CM patients. Thus, we have conducted this retrospective study with the aim of evaluating constipation as a side effect and a potential predictor of treatment response in CM patients receiving different anti-CGRP mAbs in real-world settings.
Methods
This retrospective real-world study was conducted at SkåNeuro Neurology Clinic in Lund, Sweden. This clinic has a focus on the advanced treatment of headache disorders including CM. We have collected data from the electronic medical records between 1 January 2019 and 31 December 2022.
The study population consisted of patients older than 18 years of age diagnosed with CM with or without aura according to the International Classification of Headache Disorders, 3rd edition 14 and prescribed anti-CGRP mAbs as a preventive treatment. All patients reported treatment failure with at least two different oral preventive medications after at least three months period of drug administration before initiation of anti-CGRP mAbs, according to Swedish national guidelines.
The patient list for this study was obtained using the report function of our electronic database, which includes individuals receiving anti-CGRP mAb treatment for CM. Exclusion criteria included, having major medical and psychiatric disorders, and to use of concomitant medications that might lead to severe constipation such as opioids, unclear baseline headache days, and inadequate headache diaries with more than 10 missing days per month.
According to a standard protocol in our clinic, patient evaluations were scheduled at intervals of 3, 6, 12, 18, 24, and 36 months following the initiation of anti-CGRP mAb therapy within the clinical settings. Therapeutic outcomes and potential side effects of the prescribed medication were systematically assessed at each visit.
Methods for evaluating and reporting constipation and details on combination treatments
Collected data were categorized according to predefined chart questions. The chart included relevant demography, migraine features, and medical history of previous GI disorders including constipation. Most patients were undergoing combination therapies, including medications such as amitriptyline, candesartan, lamotrigine, and botulinum toxin. Except for amitriptyline, none of these substances have been associated with causing constipation. For patients treated with amitriptyline, we ensured that low doses were used and that all patients were on stable doses of their concurrent treatments before initiating or transitioning to anti-CGRP mAbs. Additionally, the proportion of participants on combination therapies was evenly distributed across the groups receiving different types of antibodies, further reducing the risk of confounding effects from other therapies on the evaluation of efficacy and side effects.
The degree of treatment-induced/worsened constipation was obtained from patients on visits as a routine practice, based on their self-assessment. A scoring system of 0 to 3 was used, where 0 indicated no constipation, 1 indicated a mild problem not requiring medication, 2 indicated a moderate problem requiring medical treatment/medication, and 3 indicated severe constipation unresponsive to medication and leading to a switch to other anti-CGRP mAbs. Response rates have been accepted with a minimum of a 30% decrease in monthly headache days (MHDs) compared to baseline, as the regulatory authorities in Sweden established this minimum response rate as the criterion for reimbursement of anti-CGRP-mAb treatments and it is also outlined in the International Headache Society guidelines for controlled trials of preventive treatment of CM in adults as applicable. 15
Data gathered from patient records during regular clinical activities was used. No missing data values were estimated, given the real-world design of the study. Descriptive statistics were performed to summarize the data initially. For categorical data, absolute numbers and percentages were used and the metric data results are presented as mean ± standard deviation (SD). The Kolmogorov–Smirnov test was used to assess data normality. Since MHDs were not normally distributed we used nonparametric tests. For statistical comparisons of MHDs, the Freidman test was used, and Dunn's multiple comparisons test was applied when significant differences were found. The Wilcoxon test was used for comparing changes in constipation rates. Fisher's exact test was used to compare treatment response rates between groups with severe constipation and those without. Analyses were performed using GraphPad Prism 10.1.2 (Boston, Massachusetts USA), with statistical significance set at p < 0.05, two-tailed.
The study was approved by the Swedish Ethical Review Authority with a diary number of 2022-05183-01 and all enrolled patients provided written informed consent.
Results
Patient characteristics
A total of 343 patients have been examined for eligibility. Of these, 20 were excluded due to other headache diagnoses, such as posttraumatic headache or cluster headache, in addition to CM (Figure 1). Additionally, six patients were excluded because of unsatisfactory headache data. Consequently, 317 patients were included, of whom 192 patients (60.6%) used erenumab, 94 patients (29.6%) fremanezumab, and 31 patients (9.8%) galcanezumab as their initial anti-CGRP mAb with a mean treatment duration of 21.9 ± 14.3 months. Patient characteristics are shown in detail in Table 1. The vast majority of patients were female (n = 266, 83.9%) and the mean age at the time of the treatment was 44.2 ± 12.2 years. The mean baseline MHD across all patients was 15.7 ± 6.3 days and was found to be comparable between the different treatment groups. Previous medical history of constipation before the initiation of treatment in the erenumab group was close to the whole group, 18.2% versus 18.9%, respectively. Similarly, the representation of prior GI system disorder history was comparable between groups. As a result, the pre-treatment characteristics of the patients effectively eliminated the presence of selection bias.
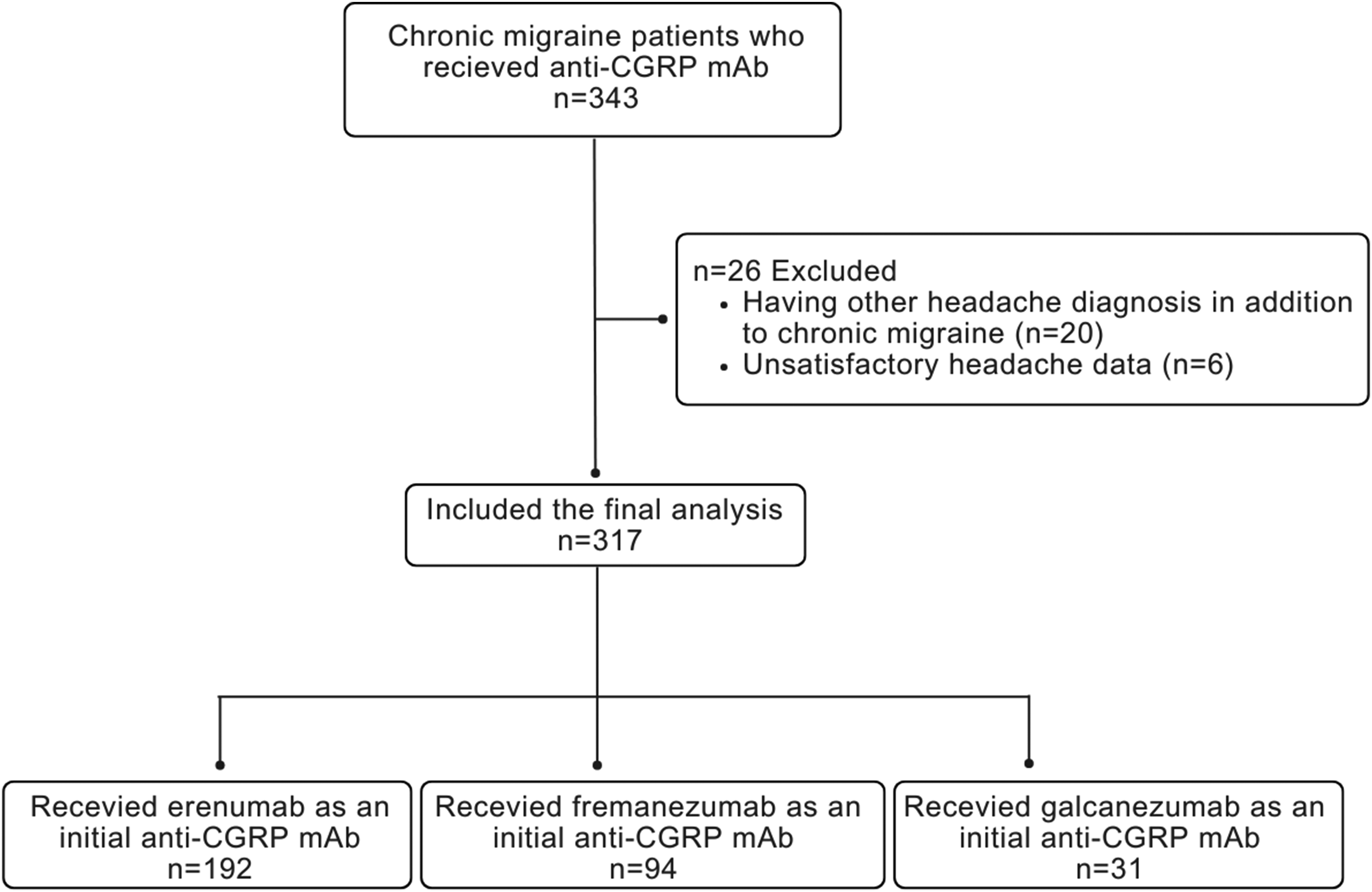
Flowchart of patients.
Characteristics of the study population (n = 317).

SD: standard deviation; GIS: gastrointestinal system; anti-CGRP: anti-calcitonin gene-related peptide; mAb: monoclonal antibody.
Constipation rates with anti-CGRP mAbs
Erenumab had the highest constipation rate of 51.5% (n = 99), with 24.4% (n = 47) of cases classified as severe constipation. On the other hand, fremanezumab had a significantly lower constipation rate of 4.2% (n = 4), with 1.1% (n = 1) classified as severe constipation. Among galcanezumab patients, 12.9% (n = 4) experienced constipation, with no severe cases (Figure 2).

Constipation rates. Panels (A) to (C) depict constipation rates in patients initially treated with erenumab, fremanezumab, and galcanezumab, respectively.
Constipation rates after switching anti-CGRP mAbs
A total of 104 patients switched to another anti-CGRP mAb during the observation period, either because of side effects (n = 63) or lack/loss of effectiveness (n = 41). Out of 63 patients, 47 (74.6%) switched from erenumab to a ligand-blocking anti-CGRP mAb (fremanezumab or galcanezumab) because of severe constipation and this switch led to highly significant improvements in constipation severity (p < 0.0001) (Figure 3(A) and (B)). Furthermore, constipation severity decreased even in milder cases, comprising 37 (19.2%) patients, who switched anti-CGRP mAb because of lack/loss of effectiveness (p = 0.0068) (Figure 3(C) and (D)).

Changes in constipation rates after switching. Panel (A) shows the patients who switched from erenumab to either fremanezumab or galcanezumab due to constipation while (B) shows a constipation rate following this switch. Panels (C) and (D) illustrate the constipation rates of patients who switched from erenumab to fremanezumab or galcanezumab due to lack/disappearance of effect while on erenumab, before and after switching, respectively.
Treatment response and constipation
The overall treatment response rate to the first anti-CGRP mAb was 70.6% (n = 224). The response rate for erenumab as the initial anti-CGRP mAb was 68.2% (n = 131), for fremanezumab it was 70.2% (n = 66), and for galcanezumab, it was 80.6% (n = 25). Among patients who switched from erenumab to fremanezumab, the response rate 3 months after the switch was 68.9% (n = 51), and for those who switched to galcanezumab, it was 70.5% (n = 12). Due to the limited number of patients (n = 6), response rates for those who switched from fremanezumab or galcanezumab to erenumab were not analyzed.
We further analyzed the relationship between treatment response and severe constipation (Figure 4). Among the patients who did not experience severe constipation response rate to erenumab was 76% (n = 101) (Figure 4(A)). For those who either did not respond or lost the effect of erenumab, 24% (n = 31) switched to another anti-CGRP mAb (Figure 4(B)). These patients, who switched due to lack/loss of effectiveness, experienced a significant reduction in MHDs after switching (p = 0.0277).

Treatment effectiveness in relation to severe constipation. Panel (A) shows changes in MHD among patients who did not experience severe constipation but responded to erenumab, while panel (B) depicts patients who neither experienced a response to erenumab nor severe constipation. Panel (C) illustrates MHD changes in patients who had both severe constipation and a satisfactory response to erenumab. Finally, panel (D) shows patients who did not respond to erenumab but experienced severe constipation.
Among patients who did experience severe constipation, the response rate to erenumab was 60% (n = 28), and these patients maintained treatment effectiveness after switching (p > 0.99) (Figure 4(C)). Those who did not respond to erenumab and switched due to both severe constipation and lack/loss of effectiveness (n = 19) also experienced significantly lower MHDs following the switch (p = 0.0147) (Figure 4(D)).
Additionally, we compared treatment response rates between groups with and without severe constipation (Figure 5). Out of the patients treated with erenumab as an initial anti-CGRP mAb, 132 patients did not experience constipation as a side effect (Figure 5(A)). Of these, 101 patients (76.5%) responded to the treatment, while 31 patients (23.5%) did not respond. In the group of patients who reported constipation as a side effect, which consisted of 47 patients, 28 patients (59.6%) responded to erenumab, while 19 patients (40.4%) did not show a response (Figure 5(B)). The comparison of treatment responses revealed significantly higher rates of non-responders among patients experiencing severe constipation (p = 0.036), highlighting a possible inverse correlation between the occurrence of constipation and effectiveness.

Treatment response in groups with and without severe constipation. “Effect (+)” indicates a positive treatment response, while “Effect (−)” indicates no response to treatment. Panel (A) represents patients who did not experience severe constipation, and panel (B) represents patients who did experience severe constipation.
Discussion
In our study, we investigated the frequency of constipation as a side effect of anti-CGRP mAbs in a large sample of 317 patients with CM, with a particular focus on erenumab. Our findings reveal a high prevalence of constipation with a rate of 51.5% in patients who received erenumab as their initial anti-CGRP mAb. This rate was 4.2% with fremanezumab and 12.9% with galcanezumab.
Of particular interest, erenumab-induced constipation was not only more frequent but also more severe, leading to treatment switch in a significant number of cases (24.4%). Conversely, the treatment-induced/worsened constipation rates with fremanezumab and galcanezumab were significantly lower and patients switching from erenumab to these anti-CGRP mAbs experienced substantial improvements in constipation severity without compromising treatment response.
The high frequency of constipation with erenumab compared to fremanezumab and galcanezumab, may be attributed to differences in their mechanism of action, with erenumab targeting CGRP receptors and the others targeting the CGRP ligand. Notably, patients experiencing severe constipation demonstrated lower treatment response rates compared to those without constipation, thus suggesting that the constipation severity might be inversely correlated with the treatment response in CM patients receiving anti-CGRP mAbs. This implies a potential interplay between GI side effects and treatment effectiveness, additionally a possible result of different mechanisms of action. Further investigation into the underlying mechanisms and whether this side effect could serve as a predictor of treatment response is warranted.
The discrepancy between constipation rates reported in early phase studies and real-world evidence highlights the importance of post-marketing surveillance in capturing rare or previously unrecognized side effects. Our findings align with recent real-world studies indicating higher constipation rates with erenumab than initially reported in clinical trials.7–11 To date, several studies have been published examining the effectiveness of switching therapies; however, no studies have specifically addressed the impact of switching on constipation rates.16,17 This emphasizes the need for continued monitoring of treatment outcomes and side effects in clinical practice to ensure patient safety and optimize treatment decisions. Additionally, considering the severity of constipation when deciding to switch anti-CGRP mAbs may be useful for physicians as this approach has the potential to improve treatment effectiveness while minimizing side effects.
Taken together, constipation emerges as a significant concern in clinical practice, even though its occurrence appears relatively low in clinical trials. This implies that we need to study underlying mechanisms, but also that it is crucial to develop guidelines to manage severe constipation in clinical settings. Based on our findings, we propose a treatment algorithm (Figure 4) that offers a practical roadmap for clinicians to guide treatment decisions in CM patients undergoing anti-CGRP mAb treatment. This algorithm suggests that patients with no constipation and a good treatment response did not require switching to another anti-CGRP mAb (Figure 4(A)). In contrast, switching becomes necessary for patients with either severe constipation or inadequate treatment response. Notably, the switch to another anti-CGRP mAb leads to a marked improvement in MHDs in a significant proportion of patients (Figure 4(B) and (D)). Importantly, our analysis demonstrates that switching due to severe constipation does not compromise treatment effectiveness (Figure 4(C)).
Our study has some limitations worth mentioning, in addition to being a retrospective single-center study. The limited number of patients receiving galcanezumab makes it challenging to make comparisons with other anti-CGRP mAbs. The constipation data collected from patients relied solely on their individual subjective assessments and were not formally evaluated by a gastroenterologist or diagnosed based on established criteria such as ROME IV. 18 This reliance on subjective reporting may introduce variability in the interpretation of constipation severity. Moreover, the medical records include information about pre-existing GI disorders but lack data on other systemic conditions that could potentially contribute to constipation. To address this limitation, we utilized data related to the patient's medical history of constipation. However, it is important to acknowledge that this approach may not capture the full spectrum of factors influencing constipation within our cohort. Further prospective, multicenter studies using standardized constipation assessment tools are required to definitively evaluate the role of constipation on treatment response.
Conclusion
In conclusion, constipation represents a significant side effect of anti-CGRP mAbs, particularly with erenumab in CM patients. This highlights the importance of continuous monitoring for side effects and the need for different treatment strategies to optimize therapeutic outcomes. Individualized dynamic treatment by switching between anti-CGRP mAbs in cases of severe constipation or/and lack of effectiveness could improve treatment success in clinical practice. Another important read-out of this study is that the treatment response to anti-CGRP mAbs might be associated with the presence of constipation. Further research is needed to elucidate the mechanisms underlying constipation associated with anti-CGRP mAbs.
Clinical implications
Constipation is a more common side effect with erenumab than with fremanezumab and galcanezumab.
Switching from erenumab to another anti-CGRP mAb leads to improvement in constipation symptoms while maintaining treatment effectiveness in cases of severe constipation.
Severe constipation was more prevalent among non-responders, suggesting a potential correlation between treatment response and the occurrence of constipation.
Footnotes
Acknowledgments
We would like to thank our colleagues Anna Zenthio and Britt Frejvall for their great contribution to data collection.
Author contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: UF has received consulting and speaking fees from Allergan, Lilly, Lundbeck, Novartis, and Teva. He participated as a principal investigator in clinical trials sponsored by Amgen, Lundbeck, Novartis, and Teva. GS has received consulting and speaking fees from Allergan, Abbvie, Lundbeck, Novartis, Pfizer and Teva. He participated as a principal investigator and a national coordinator in clinical trials sponsored by Abbvie, Lundbeck, Pfizer, and Teva. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
The study was approved by the Swedish Ethical Review Authority with a diary number of 2022-05183-01 and all enrolled patients provided written informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PP has received funding from The Swedish Research Council (grant no. 2021-01769).
