Abstract
Background
The Pan-European Real Life (PEARL) Phase 4 study evaluated real-world effectiveness and safety of fremanezumab for episodic migraine (EM) and chronic migraine (CM) prevention. This post-hoc analysis evaluated the effectiveness of fremanezumab in participants with three or more non-migraine-specific preventive treatment failures, including onabotulinumtoxinA.
Methods
Baseline daily headache diary data were compared with diary data following fremanezumab initiation. Primary endpoint: proportion of participants with ≥50% reduction in monthly migraine days (MMD) during the six months after fremanezumab initiation. Secondary endpoints included mean change from baseline in MMD at Months 1–12 and health-related quality of life. Safety was assessed through adverse events.
Results
Of 451 participants, 398 with three or more previous preventive treatment failures were included in the effectiveness analyses (EM, 40.2%; CM, 59.8%). Of the 290 participants with data available, the 50% responder rate was 53.8% (EM, 67.0%; CM, 46.5%) during the six months after fremanezumab initiation. The safety profile was consistent with previous findings.
Conclusions
This post-hoc analysis supports the effectiveness and safety of fremanezumab for migraine prevention in patients with three or more prior preventive treatment failures. These findings are consistent with those from a randomized controlled trial (RCT) in a similar population, illustrating the transferability of RCT data to real-world clinical practice.
Trial registration
encepp.eu: EUPAS35111.
This is a visual representation of the abstract.
Introduction
Non-migraine-specific oral preventive treatments can reduce the frequency of migraine attacks in patients with episodic migraine (EM) or chronic migraine (CM) (1), but discontinuation remains common because of poor adherence and persistence (2–4).
The BECOME study, involving 20,837 participants in Europe and Israel, reported that one or more non-migraine-specific oral preventive treatment had failed in 62% of participants. A subsequent analysis of 2419 participants with four or more monthly migraine days (MMD) revealed that 57% reported more than two failures, while 15% reported four or more failures (5).
The clinical definitions of resistant and refractory migraine further illustrate the challenges in management. Resistant migraine is defined as failure of three or more migraine preventive therapy classes with eight or more MMD for three or more consecutive months without improvement. Refractory migraine is defined by failure of all the available preventive therapies with eight or more MMD for six or more consecutive months (6). Patients with a history of multiple preventive treatment failures bear a significantly greater disease burden compared with treatment-naïve individuals (7). These patients face increased hospitalizations, impaired mental well-being and diminished day-to-day functioning at both home and in the workplace (7).
Randomized controlled trials (RCTs) of monoclonal antibodies (mAbs) targeting the calcitonin gene-related peptide (CGRP) pathway, such as fremanezumab, have demonstrated better tolerability profiles and lower discontinuation rates than non-migraine-specific oral preventive treatments (8). Multiple Phase 3 RCTs have demonstrated the efficacy and safety of fremanezumab when administered monthly or quarterly in adults with EM and CM (9–15). These studies also showed significant reductions in migraine frequency and migraine-related disability scores compared with placebo (9–15). Notably, the Phase 3b FOCUS study enrolled participants who had experienced previous failure of two to four migraine preventive therapy classes, and observed favorable outcomes over a 12-week period (15), suggesting that fremanezumab is a valuable option in this patient population.
It is important to note that, in RCTs of CGRP pathway mAbs conducted up to 2022, 46% of participants enrolled had limited previous exposure to preventive treatments, either having no previous treatment or not experiencing treatment failure (16). As a result, insights on patients with multiple previous treatment failures cannot always be inferred from these studies. Real-world data on the efficacy of CGRP pathway mAbs among patients with migraine and multiple previous treatment failures are therefore crucial in informing clinical decisions for this difficult-to-treat patient population.
So far, data from real-world studies have revealed that patients with more significant disability or higher headache frequency at baseline, and/or those who did not benefit from previous preventive treatments, have a reduced probability of responding to CGRP pathway mAbs (17,18). Previous treatment failure is also a common reimbursement criterion for CGRP pathway mAbs in Europe. For example, in Spain and Italy, patients must have experienced three or more preventive treatment failures (e.g. beta-blockers, tricyclic antidepressants, antiepileptics and onabotulinumtoxinA [the latter only for patients with CM]) due to a lack of efficacy and/or tolerability for three or more months to be eligible for CGRP pathway mAb reimbursement.
While some European studies provide real-world insight into experiences with fremanezumab, the majority are either retrospective or limited to centers within a single country (19–24). Therefore, there is a need for large-scale, multinational, prospective, real-world studies that include patients with previous preventive treatment failures to guide clinical practice and healthcare policies. The Pan-European Real Life (PEARL; EUPAS35111) study is an ongoing Phase 4 study evaluating the real-world effectiveness, safety and tolerability of fremanezumab in a diverse European population from specialized headache centers and general hospitals (25). With a large cohort of individuals with migraine from multiple countries in Europe and a prospective observational period of 24 months, the PEARL study is the largest and most comprehensive real-world study for fremanezumab to date, with a long follow-up duration. The PEARL study will enable clinicians to make informed migraine management decisions by generating and disseminating up-to-date data from a diverse, real-world population.
The present post-hoc subgroup analysis has been conducted in participants who had experienced three or more migraine preventive treatment failures prior to enrollment in the PEARL study, to provide real-world evidence of the use of fremanezumab in this difficult-to-treat population.
Methods
The complete protocol for the PEARL study, including all endpoints and statistical analyses, has been published previously (26). Here, we report the methodology used for the subgroup analysis of participants with three or more previous preventive treatment failures.
Study oversight
The PEARL study protocol was approved by the Independent Ethics Committee/Institutional Review Board in the 11 participating European countries (Czech Republic, Denmark, Finland, Greece, Italy, Norway, Portugal, Spain, Sweden, Switzerland and the UK), as required by local law, and in line with all relevant local data protection policies. Informed consent was obtained from all participants as part of the study inclusion criteria. All participants agreed for their clinical data to be recorded anonymously and had the right to withdraw their consent at any point during the study (26).
Study design
The PEARL study was a 24-month, Phase 4, multicenter, Pan-European, prospective, observational study, conducted across 87 sites in 11 European countries (26). As the PEARL study was a non-interventional, prospective study, no medical procedures were performed beyond the real-world practice experienced by participants in the clinical setting. The present post-hoc subgroup analysis evaluated the effectiveness, safety and tolerability of fremanezumab treatment in adults with EM or CM for whom three or more preventive treatments for migraine had failed.
Participants
Eligible participants were adults (≥18 years) diagnosed with EM (<15 headache days per month) or CM (≥15 headache days per month for three or more months, eight or more of which meet the International Classification of Headache Disorders criteria for migraine (27)), who had been prescribed fremanezumab at subcutaneous doses of 225 mg monthly or 675 mg quarterly (28). Participants had ≥21 days of headache diary data in the 28 days before fremanezumab initiation and were willing to continue recording daily headache information throughout the study. Eligible participants for this post-hoc analysis were required to have eight or more headache days per month during baseline and have experienced three or more previous preventive treatment failures for migraine. For participants with CM, unsuccessful onabotulinumtoxinA treatment usually needed to be included as a previous treatment failure. Full inclusion and exclusion criteria are detailed in the published PEARL study protocol (26).
The PEARL study enrolled a total of 1140 participants. This post-hoc analysis was performed once all participants had completed six months of treatment. The last participants completed the study in early 2024, and the final analysis was completed in 2024 (26). At the time of this analysis, some participants had data available up to Month 12, as reflected in the results.
Study procedures
All participants maintained a daily headache diary as part of their routine disease management during the 28-day baseline period before enrollment and throughout the 24-month observational period of the study (26). These diary entries captured information about headache frequency, severity, duration and characteristics, as well as concomitant preventive and acute migraine treatments. The headache diary also captured participant-reported outcome measures and validated headache-related disability tools. Data were recorded and analyzed throughout the study and compared with baseline diary data. Full details on patient follow-up are detailed in the published PEARL protocol.
Clinical visits every three months (±15 days) totaling nine visits were recommended to reflect real-world routine clinical practice and disease management (see supplementary material, Figure S1); however, this was at the treating physician's discretion. Participants who discontinued fremanezumab treatment were followed until the end of the observational period according to the visit schedule in their local clinical practice and were encouraged by their treating physician to complete daily headache diaries as per guidelines and routine disease management (26). Participants who received a new preventive treatment following fremanezumab discontinuation were excluded from the study. Participants who switched from another CGRP pathway mAb were recommended to wait until their next scheduled dose before starting fremanezumab treatment.
Assessment of outcomes
For all outcome measures, baseline data were defined as the data obtained from headache diaries in the 28-day period prior to fremanezumab initiation. The primary endpoint was the proportion of participants who reached ≥50% reduction from baseline in average MMD during the six-month period after fremanezumab initiation. Secondary endpoints included the mean change from baseline in MMD, migraine disability scores, as measured by the Migraine Disability Assessment (MIDAS), and six-item Headache Impact Test (HIT-6) at Months 1, 3, 6, 9 and 12, as well as average monthly days of acute migraine medication use. Exploratory endpoints included the mean change from baseline to Months 1, 3, 6, 9 and 12 in peak headache severity of remaining attacks, as measured by the 11-point Numeric Rating Scale (NRS). Full details of all PEARL study endpoints are provided in the published protocol (26). The safety of fremanezumab treatment was evaluated based on the documentation of adverse events (AEs) reported in clinical practice (26).
Statistical analysis
Data analyses were performed based on the data available from individual participant diaries. Participants with missing data for a specific effectiveness endpoint were excluded from the relevant analyses. The full analysis set (FAS) included all enrolled participants with ≥10 days of recorded data between fremanezumab initiation and the last documented follow-up. Effectiveness data were analyzed in the FAS using patient-reported outcome measures and validated headache-related disability tools (MIDAS and HIT-6), as recorded in headache diaries (26). The safety analysis set (SAS) included all participants who were enrolled in the study. Data were analyzed using descriptive statistics and no inferential statistics were performed. Full details of the statistical analysis are provided in the published PEARL study protocol (26).
Results
Study population
At the data cut-off for the third pre-planned interim analysis of the PEARL study (22 September 2022), 451 enrolled participants had three or more previous preventive treatment failures, meeting the inclusion criteria for this subgroup analysis. All 451 participants were included in the SAS and 398/451 were included in the FAS. In total, 53/451 participants were excluded from the FAS for one of the following reasons: the baseline visit was unsigned; less than four migraine days were documented at baseline; or less than 10 days of recorded data between fremanezumab initiation and the last documented follow-up. Only 14/398 (3.5%) participants withdrew from the study; 3/398 (0.8%) were removed due to non-compliance with the study procedure, 2/398 (0.5%) were lost to follow-up and 19/398 (4.8%) switched to a new preventive treatment for migraine.
More participants in the FAS had CM (59.8% [238/398]) than EM (40.2% [160/398]). Across all participants, the mean age was 47.7 years and 85.4% (340/398) were female (Table 1).
Participant demographics and baseline characteristics.

Of 315 participants with BMI recorded at baseline.
Preventive migraine therapy taken within five years prior to providing informed consent (multiple responses permitted).
BMI = body mass index; CM = chronic migraine; EM = episodic migraine; FAS = full analysis set; SD = standard deviation.
The three most common preventive treatment classes used prior to enrollment were anticonvulsants (81.7% [325/398]), tricyclics (74.6% [297/398]) and beta-blockers (71.9% [286/398]) (Table 1). These were used for a mean duration of 12.0, 10.3 and 9.5 months, respectively (Table 1). In total, 44/398 (11.1%) participants had previously used erenumab for a mean duration of 12.1 months and 4/398 (1.0%) had previously used galcanezumab for a mean duration of 4.0 months; no participants had previously used eptinezumab. The most common reason for treatment discontinuation across preventive regimens was lack of efficacy, ranging from 47.7% (21/44) of participants for erenumab treatment to 84.3% (220/261) of participants for onabotulinumtoxinA treatment.
Primary, secondary and exploratory endpoints
Responder rate
Of 290 participants with available data, 53.8% (156/290) reached the primary endpoint of ≥50% reduction in MMD during the six months after fremanezumab initiation (67.0% [69/103] for EM and 46.5% [87/187] for CM) (Figure 1a). Sustained ≥50% responder rates were also observed from Month 1 through to Months 3, 6, 9 and 12 (see supplementary material, Figure S2). Among participants with previous onabotulinumtoxinA treatment failure, 49.4% (83/168) reached a ≥50% reduction in MMD during the six months after fremanezumab initiation (63.6% [7/11] for EM and 48.4% [76/157] for CM) (see supplementary material, Figure S3). Among participants with prior CGRP pathway mAb exposure, 31.3% (10/32) reached a ≥50% reduction in MMD during the six months after fremanezumab initiation. Of those who switched due to lack of efficacy, 28.6% (4/14) achieved this endpoint. The mean ≥30% responder rate in participants with CM was 72.2% (135/187) during the six months after fremanezumab initiation (Figure 1b).

Proportion of participants with (a) average ≥50% reduction in MMD (primary endpoint) and (b) average ≥30% reduction in MMD during the six months after fremanezumab initiation by migraine type. Not all data were available at data cut-off (22 September 2022); missing data have been excluded. CM = chronic migraine; EM = episodic migraine; MMD = monthly migraine days.
Change in monthly migraine days
At baseline, the mean (standard deviation [SD]) MMD across all participants was 15.8(6.2) days. At Months 1, 3, 6, 9 and 12 after fremanezumab initiation, the mean change from baseline in MMD was −7.4, −7.8, −8.6, −8.7 and −8.2 days, respectively (Figure 2). Participants with EM had a mean change from baseline of −6.4 days at Months 1, 3 and 6, and −6.5 and −7.3 days at Months 9 and 12, respectively. Participants with CM had a mean change from baseline of −8.0, −8.8, −9.9, −9.8 and −8.6 days at Months 1, 3, 6, 9 and 12, respectively. Participants with a history of onabotulinumtoxinA treatment failure experienced a comparable MMD reduction (see supplementary material, Figure S4).

Mean change from baseline in MMD at Months 1, 3, 6, 9 and 12 by migraine type. Not all data were available at data cut-off (22 September 2022); missing data have been excluded. CM = chronic migraine; EM = episodic migraine; MMD = monthly migraine days; SD = standard deviation.
Change in migraine-related disability scores
For MIDAS score, the baseline mean(SD) for all participants was 87.3(57.2) and the mean change from baseline was −53.9, −61.2, −50.2 and −53.3 at Months 3, 6, 9 and 12, respectively. These reductions were −50.1, −56.1, −52.0 and −57.3 for participants with EM and −56.5, −64.3, −49.3 and −51.7 for participants with CM at Months 3, 6, 9 and 12, respectively (Figure 3a). For HIT-6 scores, the baseline mean(SD) for all participants was 67.1(4.8) and the mean change from baseline was −10.2, −11.5, −10.4 and −9.6 at Months 3, 6, 9 and 12, respectively. Mean reductions were −12.5, −13.4, −12.7 and −14.4 for participants with EM and −8.7, −10.3, −9.1 and −7.5 for participants with CM at Months 3, 6, 9 and 12, respectively (Figure 3b).

Change from baseline in migraine-related disability scores at Months 3, 6, 9 and 12 by migraine type. (a) MIDAS score. (b) HIT-6 score. Not all data were available at data cut-off (22 September 2022); missing data have been excluded. CM = chronic migraine; EM = episodic migraine; HIT-6 = six-item Headache Impact Test; MIDAS = Migraine Disability Assessment; SD = standard deviation.
Change in acute migraine medication use
The monthly average (SD) number of days with any acute headache medication use was 12.9(6.0) days across all participants at baseline. Mean reductions were −7.1, ‒7.2 and −7.4 days at Months 1, 3 and 6, respectively, and −6.5 days at both Months 9 and 12 (Figure 4a). A similar trend was observed for concomitant triptan use, for which the baseline mean(SD) number of days of use for all participants was 8.9(6.7) days with mean reductions of −5.2, −5.4, −5.4, −4.5 and −4.8 days at Months 1, 3, 6, 9 and 12, respectively (Figure 4b). In the subgroup of participants who had experienced previous failure of onabotulinumtoxinA as preventive medication, monthly average number of days with any acute migraine medication use at Months 1, 3, 6, 9 and 12 were −7.7, −7.8, −8.0, −6.5 and −6.2, respectively (see supplementary material, Figure S5).
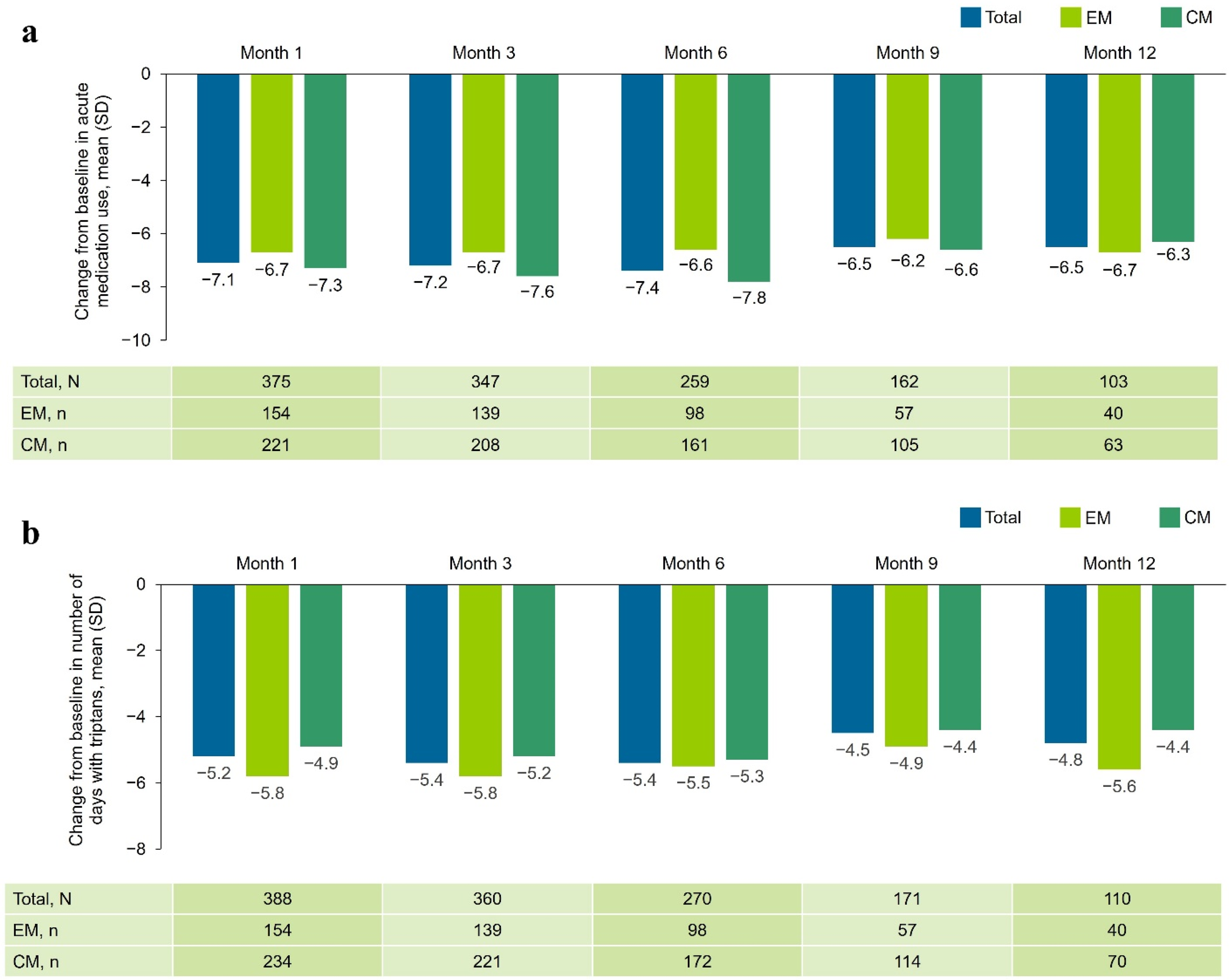
Change from baseline in average monthly days of (a) all acute migraine medication use and (b) concomitant triptan use at Months 1, 3, 6, 9 and 12 by migraine type. Not all data were available at data cut-off (22 September 2022); missing data have been excluded. CM = chronic migraine; EM = episodic migraine; SD = standard deviation.
Change in severity and duration of remaining migraine attacks
The mean(SD) NRS score for peak headache severity of attacks for all participants at baseline was 7.1(1.6). At Months 1, 3, 6, 9 and 12, the mean reduction from baseline in NRS score was −1.1, −1.4, −1.4, −0.7 and −0.9 for the remaining attacks, respectively (Figure 5a). The mean(SD) monthly duration of migraine attacks for all participants was 7.6(5.4) hours at baseline. At Months 1, 3, 6, 9 and 12, the mean reduction was −1.2, −1.2, −1.7, −1.8 and −2.2 hours for the remaining migraine attacks, respectively (Figure 5b).
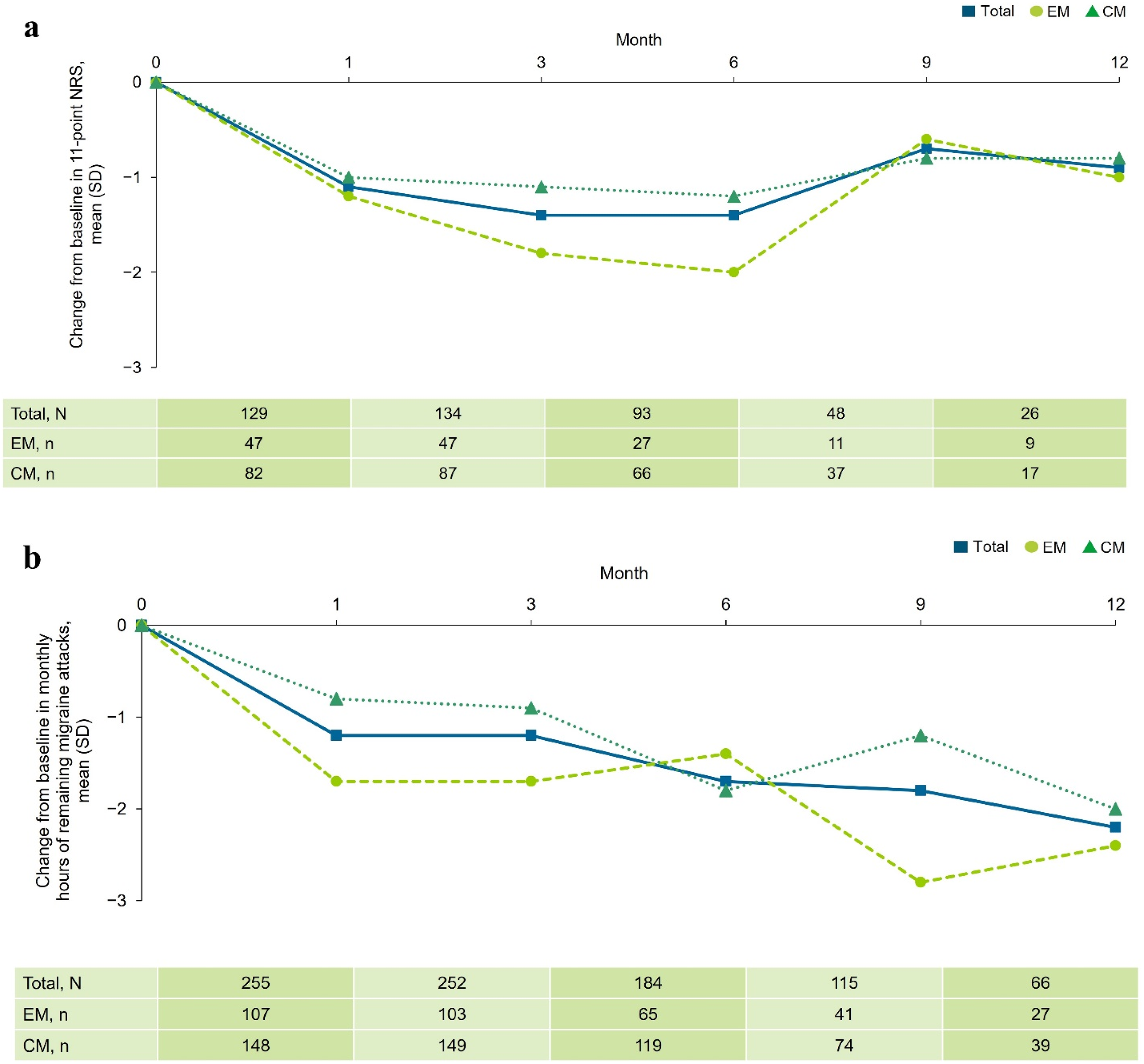
Change in (a) peak headache severity of attacks according to the 11-point NRS and (b) duration of remaining migraine attacks at Months 1, 3, 6, 9 and 12 by migraine type. Not all data were available at data cut-off (22 September 2022); missing data have been excluded. CM = chronic migraine; EM = episodic migraine; NRS = Numerical Rating Scale; SD = standard deviation.
Safety
Of 451 participants in the SAS, 21.5% (97/451) had a drug-related AE, as defined by the investigator (Table 2). The most common drug-related AEs were general disorders and administration site conditions (14.0%, 63/451). Drug-related gastrointestinal AEs were reported by 6.0% (27/451) of participants, with constipation only reported by 3.8% (17/451) of participants. One participant (0.2%) reported the following serious drug-related AEs: injection site disorder, oral/tongue pruritus and drug hypersensitivity. Overall, 2.9% (13/451) of participants discontinued fremanezumab treatment due to any AE. The most common AEs leading to discontinuation included drug ineffectiveness (1.6%; 7/451), injection site erythema (0.4%; 2/451) and injection site warmth (0.4%; 2/451). There were no new or unexpected safety signals observed.
Safety analysis.

Multiple responses were permitted for participants with more than one AE. Participants with more than one report for one specific AE were only counted once.
Includes system organ classes reported in ≥1% of the study population and specific disorders reported in ≥1% of the study population.
Drug-related means the investigator has classified the AE to be related to fremanezumab treatment. Only one participant was reported to have serious drug-related AEs.
AEs leading to drug discontinuation were all AEs with “Drug withdrawn” reported as the action taken.
AE = adverse event; SAS = safety analysis set.
Discussion
Over the first six months of fremanezumab treatment, 53.8% of participants with a history of three or more preventive treatment failures achieved a ≥50% average reduction in MMD. The ≥50% response rate was 67.0% for EM and 46.5% for CM. In addition, 72.2% of participants with CM achieved a ≥30% average reduction in MMD during the six-month period. This finding is particularly notable as a ≥30% reduction in MMD is considered a clinically meaningful improvement for patients with CM (29,30), especially in the context of multiple preventive treatment failures. No new or unexpected safety signals were observed during the study. In this difficult-to-treat patient population, reductions in MMD were also associated with improvements in migraine-related disability scores. Moreover, decreases in acute migraine medication use, as well as in the severity and duration of remaining migraine attacks, were also observed at Months 3, 6 and 9, and persisted across 12 months.
Despite recent advancements in migraine therapy, multiple preventive treatment failures remain common. These failures contribute to disease burden and reduced responsiveness to CGRP pathway mAbs (2,17,18,31,32). A recent analysis of the 2020 National Health and Wellness Survey in Europe found that patients with migraine and two or more preventive treatment failures had worse migraine-related disability scores and a lower quality of life compared with treatment-naïve patients (7). This highlights the need for preventive treatments with a reliable success rate across a diverse patient population.
The present analysis suggests that the real-world effectiveness of fremanezumab treatment is consistent with the positive outcomes observed in RCTs (9–15). In the Phase 3b FOCUS RCT, participants with two to four previous preventive treatment failures experienced mean MMD reductions of −3.7 and −4.1 with quarterly and monthly fremanezumab, respectively (15). Furthermore, the ≥50% responder rate was 34% over the 12-week period (15). This subgroup analysis of the PEARL study reported a mean MMD reduction of −8.6 days and a ≥50% responder rate of 53.8% during the six months of treatment. These results align with the second interim analysis of the PEARL study, which reported a ≥50% responder rate of 55.9% in all participants, irrespective of previous preventive treatment failures (25).
The improvements in disability scores observed in this subgroup analysis of the PEARL study further reinforce those documented in the FOCUS RCT. In the FOCUS study, mean reductions from baseline in MIDAS scores were −19.7 (quarterly fremanezumab) and −24.7 (monthly fremanezumab), and in HIT-6 scores were −5.2 (quarterly fremanezumab) and −6.1 (monthly fremanezumab) (15). This PEARL subgroup analysis demonstrated a mean reduction from baseline in MIDAS scores of −53.9, and HIT-6 scores of −10.2, which underscores the robust impact of fremanezumab on disability outcomes. A consistent decrease in mean acute migraine medication use was also observed, including in the use of triptans, throughout the 12-month period. The mean change from baseline in monthly average number of days of acute headache medication at Month 3 was −3.7 (quarterly fremanezumab) and −3.9 days (monthly fremanezumab) in the FOCUS study, and −7.2 days in this subgroup analysis of the PEARL study. Overuse of acute migraine medication, defined as 10–15 days of medication use per month, is a known risk factor for migraine chronification (33,34), with CM being associated with a substantially higher rate of migraine-related disability than EM (35). Effective prevention with fremanezumab may therefore ultimately lower the overall migraine burden.
Safety findings in this subgroup analysis were consistent with previous fremanezumab RCTs, confirming the favorable tolerability of fremanezumab in this complex population with multiple prior preventive treatment failures. The rate of drug-related AEs in this study was 21.5%, which is comparable to the 21% and 19% reported for quarterly and monthly fremanezumab, respectively, in the FOCUS study. Importantly, the incidence of constipation was only 3.8% in the present study. This is notably lower than the 20.0–41.3% reported for CGRP receptor-targeting treatments, such as erenumab, in real-world studies of patients with CM (36,37) and lower than the incidence observed with atogepant in a pharmacovigilance study, with a reporting odds ratio of 12.86 (the threshold for this measure was three or more) (38).
Study strengths and limitations
A major strength of the PEARL study is its real-world setting. Participants were treated with fremanezumab according to routine clinical practice, which captures the diversity of the migraine population, including the high prevalence of comorbidities (35,39). Although RCTs have provided critical efficacy and safety data (9–15), observational studies such as PEARL offer additional insights into the patient experience in every day clinical settings (40).
One limitation of this study is that the data were collected via participant-reported headache diaries, which rely on individual accuracy and may be subject to human error. Furthermore, the observational design resulted in a higher percentage of missing data compared with RCTs. Consequently, and considering the number of participants who discontinued fremanezumab, data collected at Month 6 and later should be interpreted with caution due to the smaller sample sizes. Additionally, in this post-hoc analysis, the average number of prior preventive treatment failures was not recorded. These data are available for the overall PEARL population and will be reported in the final study analysis. Despite these limitations, the PEARL study remains the largest prospective study of fremanezumab, including in patients with previous preventive treatment failures. Future interim and final analyses will further expand the real-world evidence base in an even larger patient population.
Conclusions
Fremanezumab significantly reduced MMD in a difficult-to-treat subgroup of adults with CM and EM who had a history of three or more preventive treatment failures. Over the first six months of treatment, participants experienced reductions in migraine frequency, improvements in migraine-related disability scores, decreased use of acute medications and reductions in the severity and duration of residual migraine attacks. These benefits contributed to an overall improvement in health-related quality of life. This subgroup analysis of real-world data demonstrates that fremanezumab provides effective and long-acting migraine prevention with a favorable safety and tolerability profile across a broad range of patients with multiple previous preventive treatment failures, thereby supporting the applicability of RCT findings to clinical practice.
Clinical implications
High disease burden and management challenges persist among individuals with multiple previous migraine preventive treatment failures.
This subgroup analysis demonstrates the effectiveness of fremanezumab in preventing migraine following failure of previous preventive treatments, including onabotulinumtoxinA.
The findings support the transferability of clinical trial data and confirm that outcomes observed in RCTs are reflected in everyday practice, underscoring the clinical value of fremanezumab.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251395029 - Supplemental material for Addressing unmet needs in migraine: Real-world fremanezumab effectiveness in participants of the PEARL study with at least three prior preventive treatment failures
Supplemental material, sj-docx-1-cep-10.1177_03331024251395029 for Addressing unmet needs in migraine: Real-world fremanezumab effectiveness in participants of the PEARL study with at least three prior preventive treatment failures by Messoud Ashina, Cristina Tassorelli, Pinar Kokturk, Hasan Akcicek and Patricia Pozo-Rosich in Cephalalgia
Footnotes
Acknowledgments
Medical writing support and editorial support for the development of this manuscript, under the direction of the authors, was provided by Frida Sommer and Niamh Farnan of Ashfield MedComms, an Inizio company, and funded by Teva Pharmaceuticals.
Author contributions
MA and PK conceptualized and designed the study, analyzed the data and reviewed the manuscript. HA and CT analyzed the data and reviewed the manuscript. PP-R reviewed the manuscript.
Consent to participate
Written informed consent was obtained from all participants as part of the study inclusion criteria.
Data availability statement
The data sets used and/or analyzed for the study described in this manuscript are available upon reasonable request. Qualified researchers may request access to patient level data and related study documents including the study protocol and the statistical analysis plan. Patient level data will be de-identified and study documents will be redacted to protect the privacy of trial participants and to protect commercially confidential information. To make your request, please visit: www.clinicalstudydatarequest.com.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: MA has received personal fees and declares PI/collaborator in clinical trials for AbbVie, Amgen, AstraZeneca, Eli Lilly, GlaxoSmithKline, Lundbeck, Novartis, Pfizer and Teva Pharmaceuticals; as well as research grants from the Lundbeck Foundation, the Novo Nordisk Foundation, Lundbeck and Novartis. CT declares advisory boards, symposia (personal fees) and/or PI/collaboration in clinical trials (institutional fees) for AbbVie, Chordate, Dompé, Eli Lilly, Ipsen, Lundbeck, Pfizer and Teva Pharmaceuticals, as well as research grants from the European Commission, the Italian Ministry of Health, the Migraine Research Foundation and the Italian Multiple Sclerosis Foundation. PK and HA are employees and/or shareholders of Teva Pharmaceuticals. PP-R declares grant/research support from AbbVie, AGAUR, EraNet NEURON, Instituto Investigación Carlos III, Novartis, RIS3CAT FEDER and Teva Pharmaceuticals, as well as consultancy or education for AbbVie, Dr Reddy's, Eli Lilly, Lundbeck, Medscape, Novartis, Pfizer and Teva Pharmaceuticals.
Ethical statement
The PEARL study protocol was approved by the Independent Ethics Committee/Institutional Review Board in the 11 participating European countries (Czech Republic, Denmark, Finland, Greece, Italy, Norway, Portugal, Spain, Sweden, Switzerland and the UK), as required by local law, and in line with all relevant local data protection policies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by Teva Pharmaceuticals.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
