Abstract
Objective:
This study was designed to assess the efficacy and safety of galcanezumab in comparison with placebo for the prevention of migraine in Japanese patients with episodic migraine.
Methods:
In this double-blind, placebo-controlled study, which was conducted over 6 months, randomized adult patients received subcutaneous injections of galcanezumab (120 mg n = 115, 240 mg n = 114) or placebo (n = 230) once monthly. The primary endpoint was the overall mean change from baseline in the number of monthly migraine headache days. The key secondary outcome measures were response rates (≥50%, ≥75%, and 100%); the Migraine-Specific Quality-of-Life Questionnaire Role Function-Restrictive score; monthly migraine headache days requiring acute treatment; and Patient Global Impression of Severity (PGI-S).
Results:
The mean change from baseline in monthly migraine headache days over months 1–6 was significantly (p < 0.001) greater for the 120-mg galcanezumab dose (−3.60 days) and the 240-mg galcanezumab dose (−3.36 days) compared with placebo (−0.59 days). Both the 120-mg and 240-mg doses of galcanezumab were superior compared with placebo for each of the key secondary endpoints except for PGI-S (only the 240-mg dose was superior). The most commonly reported treatment-emergent adverse events were local injection-site reactions; erythema, swelling, pruritus, and pain were more commonly reported by patients who were treated with galcanezumab than those treated with placebo.
Conclusion:
The number of monthly migraine headache days was reduced with both doses of galcanezumab, and both doses were safe and well tolerated in Japanese patients with episodic migraine.
Keywords
Introduction
Clinical treatment guidelines from the Japanese Headache Society and Japanese Society of Neurology recommend preventive drugs for patients with migraine when considering attack frequency, disease severity, degree of migraine-related disability remaining with the use of acute medication, and risk of permanent neurological damage. 1 Similar to other countries, migraine burden and its impact on the workplace in Japan is high. 2,3 To date, antiepileptics, β-blockers, antidepressants, angiotensin receptor blockers/angiotensin-converting enzyme inhibitors, calcium channel blockers, and other medications are used for migraine prevention in clinical practice in Japan. 1,4 Despite this, the use of preventive treatments remains low in Japan, with many patients either switching treatments or discontinuing them altogether, primarily because of lack of efficacy. 4,5
The neuropeptide calcitonin gene-related peptide (CGRP) contributes to neurogenic inflammation, vasodilation, and nociceptive modulation in migraine. 6 The humanized monoclonal antibody galcanezumab, which binds to CGRP and prevents its activity, has been approved in many countries as a treatment for migraine prevention in adults. Two phase 3 randomized controlled trials (RCTs) in patients with episodic migraine 7,8 and a phase 3 RCT in patients with chronic migraine 9 conducted predominantly in Caucasian populations have demonstrated that treatment with once per month subcutaneous injections of 120-mg or 240-mg galcanezumab is safe, well-tolerated, and associated with clinically significant reductions in the number of monthly migraine headache days compared with placebo. This is the first report of the efficacy and safety of galcanezumab in Japanese patients with episodic migraine.
Materials and methods
Study design
This was a 6-month, phase 2, randomized, double-blind, placebo-controlled study of galcanezumab in Japanese outpatients with episodic migraine from 40 sites (December 2016 to January 2019). Local ethics review boards reviewed and approved the protocol and patients gave written informed consent before participating in the study. The study (ClinicalTrials.gov NCT02959177; https://clinicaltrials.gov) was conducted in accordance with the Declaration of Helsinki, Good Clinical Practice, and the Council for International Organizations of Medical Sciences International Ethical Guidelines.
There were four study periods (SP; Figure 1): SP I, a screening period that included a full clinical assessment and washout of migraine preventive treatments for ≥30 days (≥4 months for botulinum toxin A or B); SP II, a prospective baseline period to confirm patient eligibility and establish baseline number of migraine headache days using an electronic patient-reported outcome (ePRO) diary; SP III, a 6-month, double-blind treatment phase, where patients were required to complete the ePRO diary every day; and SP IV, a 4-month washout (follow-up) period (not a subject of the current manuscript). Patients took acute treatments for migraine headache as needed, except for opioids or barbiturates (limited to three times per month) and single-dose injectable steroids (allowed only once, in an emergency setting).

Study design. Patients randomized to the 120-mg dose received a loading dose of 240 mg at the first injection only (visit 3). Findings for SP IV are not included in the current manuscript. GMB: galcanezumab; M: month; SP: study period; X: visits when dosing occurred.
Study population
The study investigators evaluated and confirmed the eligibility of patients, obtained consent, and enrolled patients. Eligible patients were male or female, aged 18–65 years (inclusive), and had a diagnosis of migraine with or without aura, as defined by the International Headache Society (IHS) International Classification of Headache Disorders (ICHD)-3 β (1.1 or 1.2). 10 Patients had onset of migraine before age 50, had migraine for at least 1 year before entering the study and during the baseline period, and had 4–14 migraine headache days per month and at least two migraine attacks per month. Patients had to demonstrate ≥80% compliance (completion of daily entries) with the ePRO diary, and all patients agreed to use reliable methods of contraception during the study and for 5 months after the last dose.
Patients were not eligible if they were currently taking preventive treatments for migraine (patients had to discontinue any preventive treatment ≥30 days before entering SP II), had a history, on average, of ≥15 headache days per month during the 3 months before entering SP I, or were suspected as having chronic migraine, as defined by ICHD-3 β. In addition, patients were not eligible if they had failed to respond to at least three adequately dosed migraine preventive treatments from different classes (i.e. the maximum tolerated dose for ≥2 months). The definition of treatment failure did not include a failure to respond because of tolerability issues. Migraine preventive treatments were defined as drugs with a grade A or grade B recommendation from the Japanese Society of Neurology and the Japanese Headache Society guidelines for migraine 1 and botulinum toxin A or B. Botulinum toxins A and B are not approved for migraine prevention in Japan but were included as migraine prevention treatments to be consistent with the two global RCTs in patients with episodic migraine. 7,8 Furthermore, patients were excluded for any of the following reasons: had used galcanezumab or other CGRP monoclonal antibodies previously; in the past 12 months had taken a therapeutic antibody; had used barbiturates or opioids more than twice per month; had known hypersensitivity to medication, monoclonal antibodies, or therapeutic proteins; had a history of cluster headache, persistent daily headache, or migraine subtypes (including ophthalmoplegic migraine, hemiplegic migraine, and migraine with brainstem aura defined by IHS ICHD-3 β); or had a history of any psychiatric or medical illness that would prevent participation in the study.
Randomization, blinding, and treatment protocol
Patients were randomized (2:1:1) to one of three treatment groups to receive placebo, 120-mg galcanezumab or 240-mg galcanezumab, respectively, which were administered for 6 months by subcutaneous injection, once per month (Figure 1). Randomization was achieved using a computer-generated random sequence with an interactive web-response system, stratified by baseline monthly migraine frequency (<8 migraine headache days vs. ≥8 migraine headache days). At the first injection, patients randomized to 120-mg galcanezumab received a 240-mg loading dose (visit 3). Trained study personnel administered the subcutaneous injections and were blinded to treatment allocation during the treatment phase. To preserve blinding, all study treatments were supplied as identical 1-mL, single-dose, prefilled disposable syringes; at each dosing visit, all patients received two injections.
Study endpoints and assessments
The primary efficacy endpoint was the overall mean change from baseline in the number of monthly migraine headache days during the double-blind treatment phase (average of months 1–6). The definition of a migraine headache day was a calendar day on which a migraine headache or probable migraine headache 10 occurred.
Key secondary endpoints during the double-blind treatment phase were overall rates (≥50%, ≥75%, and 100%) of reduction in monthly migraine headache days; mean change from baseline over months 4–6 in the Migraine-Specific Quality-of-Life Questionnaire Role Function-Restrictive (MSQ-RFR) v2.1 domain score 11 ; overall mean change from baseline in the number of monthly migraine headache days requiring medication for acute treatment of migraine; and mean change from baseline in Patient Global Impression of Severity (PGI-S). 12 Multiple other secondary endpoints were assessed during the trial; of these, the mean change from baseline in migraine disability assessment (MIDAS) total score at month 6 is reported here. Additional endpoints included the proportion of patients with a ≥2-point improvement in PGI-S over months 4–6 and the number of weekly migraine headache days during the first month of treatment.
Assessment of safety included the frequency of adverse events (AEs), serious AEs (SAEs), discontinuations due to AEs, and treatment-emergent AEs (TEAEs), coded using the Medical Dictionary for Regulatory Activities (version 21.1). Additional safety measures comprised vital signs (including blood pressure), body weight, standard laboratory tests, electrocardiograms (ECGs), the Columbia-Suicide Severity Rating Scale, and antidrug antibody assessment (immunogenicity).
Statistical analyses
The planned sample size was 451 enrolled patients (placebo: 225 patients, 120-mg galcanezumab monthly and 240-mg galcanezumab monthly: 113 patients each). The sample size was based on an assumed discontinuation rate of 15% and an effect size of 0.36 and was calculated to provide 88% power to detect a difference between at least one dose of galcanezumab and placebo, with a two-sided significance level of 0.05 based on simulations using the Dunnett test. These assumptions were based on findings from the global phase 3 studies for galcanezumab, 7,8 adjusted to account for the lower rates of patient discontinuations and less variability expected from a single-country study.
The intent-to-treat (ITT) population was used for all analyses and was defined as all randomized patients who received at least one dose of placebo or galcanezumab. The primary analysis and visit-wise continuous efficacy were analyzed using a restricted maximum likelihood-based mixed model for repeated measures (MMRM). Fixed categorical effects included treatment, month, and treatment-by-month interaction, and fixed continuous covariates included baseline number of migraine headache days and baseline number of migraine headache days-by-month interaction. The primary endpoint for each galcanezumab dose group compared with placebo was estimated as the main treatment effect from the MMRM analysis during the 6-month treatment phase, providing an estimate of the average treatment effect across 6 months of the double-blind treatment phase. Multiplicity adjustment (step-down Dunnett test) was used for the primary analysis only (galcanezumab dose groups vs. placebo). Comparison of the number of weekly migraine headache days in the first month between all patients who received galcanezumab or placebo was assessed using an ordinal repeated-measures analysis, while monthly migraine headache day response rates and PGI-S response rate were assessed using a generalized linear mixed model for repeated binary outcomes, which included fixed categorical effects (treatment, month, and treatment-by-month) and the continuous fixed covariate of baseline. Baseline number of migraine headache days was added as a categorical variable (<8 and ≥8 days) for analysis of PGI-S response rate and was added as a continuous covariate for analysis of monthly migraine headache day response rates. Categorical safety measures were reported using descriptive statistics. SAS 9.4 (SAS Institute, Cary, North Carolina, USA) was used for all statistical analyses.
Results
Disposition
Of the patients screened for inclusion, 459 were randomized, received at least one galcanezumab dose or placebo, and were included in the ITT population (Figure 2). The rate of study discontinuation was low across all treatment arms with 440 patients (95.9%), completing the double-blind treatment period. The most common reason for study discontinuation from the galcanezumab groups was AEs.

Patient disposition. GMB: galcanezumab; SP: study period.
Demographic and baseline clinical characteristics
These characteristics were well balanced across each treatment group (Table 1). Most patients were female (387 patients, 84.3%) and the mean age (standard deviation, SD) of patients was 44.1 (10.0) years; the mean duration (SD) of migraine (from diagnosis) was 21.4 (11.7) years, the mean number of monthly migraine headache days (SD) was 8.7 (2.9), and the mean number of migraine attacks per month (SD) was 5.5 (1.7). Approximately 60% of all patients had ≥8 migraine headache days per month. Most patients (60.6%) reported using migraine preventive treatment previously. Of these, 33.8% did not have a history of preventive treatment failure (i.e. 20.5% of all patients) and 66.2% had failed one or more preventive treatments (i.e. 40.1% of all patients). Across the treatment groups, baseline MSQ-RFR scores, PGI-S, and MIDAS total scores were similar (Table 1).
Baseline demographics and disease characteristics.a
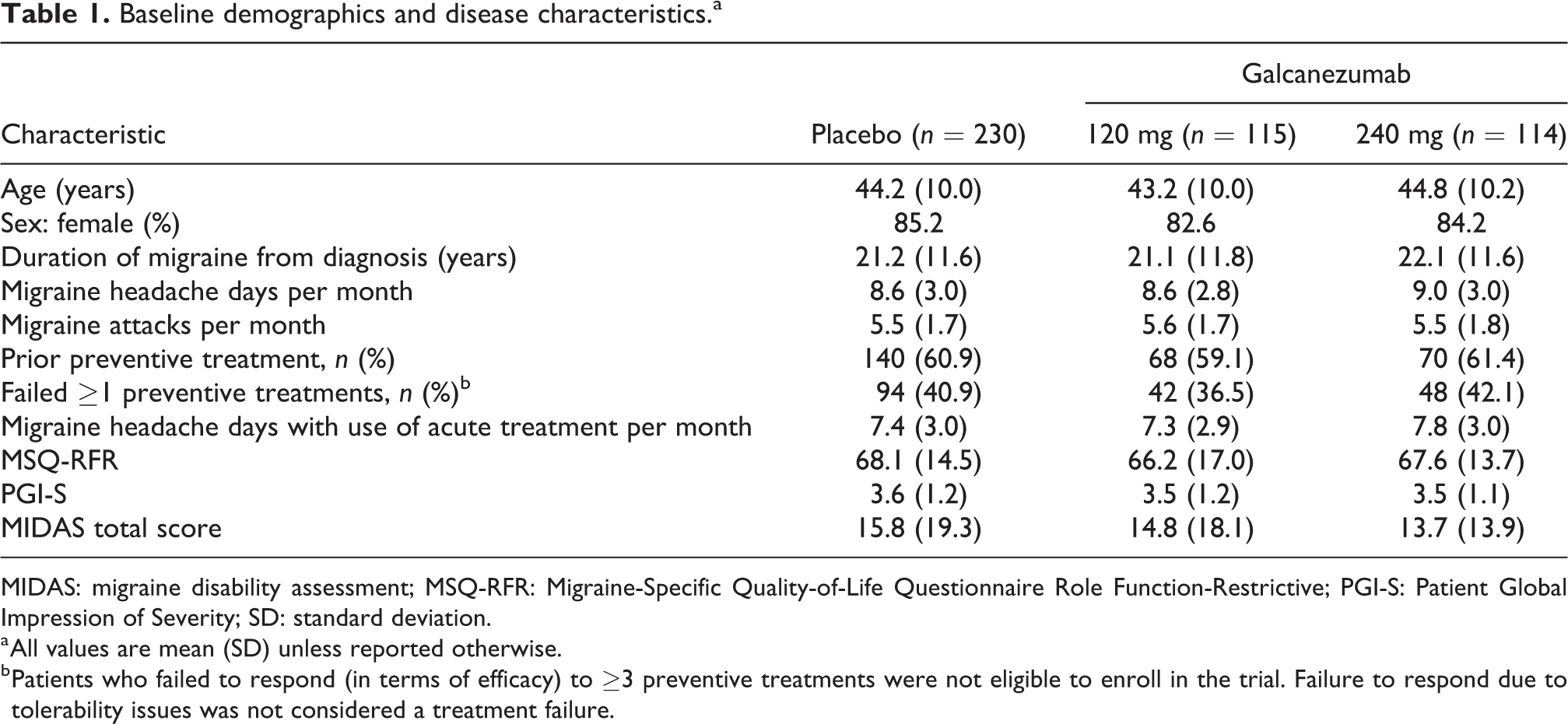
MIDAS: migraine disability assessment; MSQ-RFR: Migraine-Specific Quality-of-Life Questionnaire Role Function-Restrictive; PGI-S: Patient Global Impression of Severity; SD: standard deviation.
a All values are mean (SD) unless reported otherwise.
b Patients who failed to respond (in terms of efficacy) to ≥3 preventive treatments were not eligible to enroll in the trial. Failure to respond due to tolerability issues was not considered a treatment failure.
Efficacy outcomes
Compared with placebo, both the 120-mg and 240-mg doses of galcanezumab demonstrated statistically significant (p < 0.001) improvement in the overall mean change in monthly migraine headache days over months 1–6 (Figure 3; Table 2). The difference in the least-squares mean change from baseline in monthly migraine headache days compared with placebo was –3.01 days (95% confidence interval (CI) −3.80, −2.22; p < 0.001) for the 120-mg dose of galcanezumab and was −2.77 days (95% CI −3.56, −1.98; p < 0.001) for the 240-mg dose.

Overall LS mean change in monthly migraine headache days over months 1–6. ‡ p < 0.001 (vs. placebo) after multiplicity adjustment. GMB: galcanezumab; LS: least squares; SE: standard error.
Summary of primary outcomes, key secondary outcomes, PGI-S response rates, and MIDAS total score.a

GLIMMIX: generalized linear mixed model; LS: least squares; MIDAS: migraine disability assessment; MMRM: mixed-model repeated measures;
MSQ-RFR: Migraine-Specific Quality-of-Life Questionnaire Role Function-Restrictive; PGI-S: Patient Global Impression of Severity.
a All values are the LS mean change from baseline unless reported otherwise.
b Restricted maximum likelihood-based MMRM.
c Adjusted for multiplicity.
d GLIMMIX for binary outcomes.
e p < 0.01; p-values versus placebo.
fp < 0.001; p-values versus placebo.
The onset of effect of galcanezumab was rapid. A statistically significant improvement (p < 0.001) in the mean change in migraine headache days from month 1 was observed for both doses of galcanezumab compared with placebo and was maintained for up to 6 months (Figure 4(a)). Statistically significant differences between treatment groups were evident from week 1. The likelihood of fewer weekly migraine headache days with galcanezumab in the first month of treatment compared with placebo was statistically significant (all p < 0.001) at week 1 (odds ratio (OR) 4.0, 95% CI 2.7–6.0), week 2 (OR 2.5, 95% CI 1.7–3.7), week 3 (OR 2.2, 95% CI 1.5–3.3), and week 4 (OR 2.5, 95% CI 1.7–3.8).

(a) LS mean change in migraine headache days at each month over months 1–6; (b) response rates: mean percentage of patients with ≥50%, ≥75%, and 100% reduction in monthly migraine headache days over months 1–6; (c) LS mean change in the number of migraine headache days requiring acute medication; (d) LS mean change in the number of migraine headache days requiring triptans; (e) LS mean change in the MSQ-RFR domain average over months 4–6; and (f) LS mean change in the PGI-S score average over months 4–6.
The response rates for a ≥50%, ≥75%, and 100% reduction in monthly migraine headache days over months 1–6 were statistically significantly greater (p < 0.001) for patients who received 120-mg and 240-mg galcanezumab compared with placebo (Figure 4(b)). For galcanezumab 120 and 240 mg, 49.8% and 48.2% of patients, respectively, reported ≥50% reduction in monthly migraine headache days over months 1–6, approximately 25% for both groups reported ≥75% reduction in monthly migraine headache days, and 9.0% and 8.1%, respectively, reported 100% reduction in monthly migraine headache days. For placebo, 20.3%, 9.6%, and 2.8% of patients reported ≥50%, ≥75%, and 100% reduction in monthly migraine headache days, respectively. The reduction in the number of migraine headache days that patients required acute treatment, including triptans, was statistically significantly greater for both galcanezumab doses compared with placebo (Figure 4(c) and (d); Table 2). The difference in the overall mean change from baseline in monthly migraine headache days with acute treatment compared with placebo was −2.90 days (95% CI −3.61, −2.19; p < 0.001) for 120-mg galcanezumab and −2.70 days (95% CI −3.41, −1.99; p < 0.001) for 240-mg galcanezumab over months 1–6.
Concurrent with the improvements in monthly migraine headache days, patients receiving galcanezumab reported improvements in functioning (MSQ-RFR scores), self-reported disease severity (PGI-S), and migraine-related disability (MIDAS total score; Table 2). The change in the average MSQ-RFR scores over months 4–6 from baseline was statistically significantly (p < 0.001) greater for both the 120-mg and 240-mg doses of galcanezumab compared with placebo (Figure 4(e); Table 2). The change in average PGI-S rating over months 4–6 from baseline was statistically significantly greater for 240-mg galcanezumab compared with placebo (p < 0.01) but not 120-mg galcanezumab (Figure 4(f); Table 2). Similarly, the proportion of patients with ≥2-point improvement in PGI-S rating at months 4–6 was statistically significantly greater for 240-mg galcanezumab compared with placebo (p = 0.001) but not 120-mg galcanezumab (p = 0.20; Table 2). Improvement in the overall mean change in MIDAS total score at month 6 was statistically significant (p = 0.002) for 120-mg galcanezumab compared with placebo but not 240-mg galcanezumab (p = 0.061; Table 2).
Safety and tolerability measures
Overall, the frequency of TEAEs was numerically higher for the 120- and 240-mg galcanezumab groups compared with placebo (Table 3). At least one TEAE was reported by 85.2% of patients for 120-mg galcanezumab, 81.6% for 240-mg galcanezumab, and 64.8% for placebo. Four patients treated with galcanezumab reported a SAE (sudden hearing loss, meniscus injury, and tooth impacted: galcanezumab 120 mg, one each; and nasal septum deviation: galcanezumab 240 mg) during the double-blind treatment phase. Seven patients treated with either 120-mg galcanezumab (five patients) or 240-mg galcanezumab (two patients) were discontinued because of an AE (Table 3); no deaths were reported.
Overview of AEs.

AE: adverse event.
Injection-site reactions were the most commonly reported TEAEs across all groups (Table 4). Compared with placebo, injection-site erythema, swelling, pruritus, and pain were reported more frequently in both the 120- and 240-mg galcanezumab groups (Table 4). Among galcanezumab-treated patients, the proportions of patients reporting injection-site erythema and pruritus were higher for the 240-mg dose group than the 120-mg dose group (Table 4). Most injection-site TEAEs occurred on the day of injection, were mild to moderate in severity and were self-limiting. Two galcanezumab-treated (240-mg dose) patients reported severe injection-site events: one reported severe injection-site erythema and injection-site pruritus and discontinued study treatment because of the injection-site erythema; the other reported severe injection-site pruritus, injection-site erythema, and injection-site induration but did not discontinued study treatment.
Most common TEAEs.a

AE: adverse event; TEAE: treatment-emergent adverse event.
a AEs were coded by the Medical Dictionary for Regulatory Activities (version 21.1). Common events were defined as those with an incidence in either galcanezumab dose group ≥1.5% before rounding and greater than placebo.
There were no clinically relevant changes between the treatment groups in other measures of safety—including vital signs, body weight, laboratory tests, immunogenicity, and ECGs—and no findings related to treatment-emergent suicidal behavior.
Discussion
This is the first study to investigate the efficacy and safety of 120- or 240-mg galcanezumab once monthly compared with placebo for migraine prevention in Japanese patients. Both doses of galcanezumab met the primary objective after adjusting for multiplicity. Treatment with galcanezumab was associated with a reduction of approximately 3 monthly migraine headache days over and above placebo, representing the largest therapeutic gain among all galcanezumab studies conducted to date. 7 –9 The onset of effect of galcanezumab was rapid, and compared with placebo, patients reported fewer migraine headache days, fewer migraine headache days requiring acute treatment, including triptans, and improvements in functioning (MSQ-RFR), disease severity (240-mg dose only; PGI-S), and migraine-related disability (120-mg dose only; MIDAS total score). In addition, the rate of TEAEs among Japanese patients treated with galcanezumab was low and similar to those observed in the phase 3 clinical trials conducted in predominantly Caucasian populations. 7 –9
Almost half of the patients who were treated with galcanezumab in the current study reported a 50% or greater reduction in monthly migraine headache days, which is widely considered a clinically significant level of improvement. 13 Similar to the global trials, significant reductions in the number of monthly migraine headache days were evident from the first week of treatment with galcanezumab, and there did not appear to be any difference in the pattern of reduction in migraine headache days between Japanese patients and patients in the global trials. Pooled analysis of data from two global trials of patients with episodic migraine has confirmed the rapid onset of effect of galcanezumab and has also shown that more than one-quarter of patients who do not respond in the first month may respond within 3 months of starting treatment. 14
Several studies have shown higher placebo effects with placebo injection compared with oral placebo, 15 and findings from a meta-analysis of migraine prevention trials have also shown greater placebo effects in trials with sham acupuncture or surgery arms compared with oral administration of placebo. 16 Despite the current study being a placebo-controlled study of an injectable drug, a very low placebo effect was observed. One potential explanation may be that the study investigators were a highly selected group of clinicians who were specialized in the treatment of migraine and who had considerable clinical trial experience in migraine. As such, this may have led to the recruitment of highly motivated patients who understood the protocol well and who also understood the importance of the blinded, placebo-controlled phase of the study. An even lower placebo response was observed in a recent study by a different sponsor, in which there was an overlap of approximately half the number of study sites with our study. 17
In Japan, migraine is among the main causes of chronic pain 18 and is associated with significant levels of disability. 3,19 In this study, the reductions in monthly migraine headache days with galcanezumab were associated with clinically meaningful improvements in functioning (MSQ-RFR) 20,21 for both galcanezumab doses and improvements in PGI-S for the 240-mg dose compared with placebo. However, compared with the global galcanezumab studies, 7,8 patients in this study had MSQ-RFR scores and MIDAS total scores that indicated less functional impairment and migraine-related disability. Although the numbers of baseline monthly migraine headache days in Japanese patients in the current study and in patients in the global studies were similar, the mean MSQ-RFR and MIDAS total scores differed between these populations. For Japanese patients, MIDAS total scores among the groups ranged from 14 to 16, and MSQ-RFR scores ranged from 66 to 68. In contrast, for the global trial populations, MIDAS total scores ranged from 31 to 36, and MSQ-RFR scores ranged from 49 to 53. The higher MSQ-RFR scores for Japanese patients compared with the global trial populations are consistent with the findings from a cross-sectional study of 99 Japanese adults, which showed that although the pattern of MSQ scores for patients in Japan was similar to those for patients in the United States, mean scores for MSQ-RFR, role function-preventive, and role-emotional in Japan were 5–20 points higher than those in the United States. 22 Cultural differences in perception and coping with pain may, at least partly, explain this finding.
Consistent with previous studies of galcanezumab, 7,8,23,24 the most frequently reported TEAEs in the Japanese population were injection site-related and included erythema, swelling, pruritus, and pain. However, most events occurred early during treatment, very few were severe, and very few patients discontinued either the 120-mg or 240-mg dose of galcanezumab. Given that currently available preventive treatments are not well tolerated by patients 25 and that persistence with oral preventive treatments is low, 25,26 including in Japan, 5 the tolerability of galcanezumab in this trial, together with the increase in the number of migraine-free days, suggest that, from the point of view of adherence, galcanezumab may be an attractive option.
Findings from this study are strengthened by the randomized double-blind study design, low rate of discontinuation from the study, and inclusion of multiple clinically relevant measures for assessment of treatment outcomes. 27 –29 Moreover, the consistency of the findings between the global clinical trials and Japanese patients suggests that the effect of galcanezumab may be generalized between populations. Future studies that assess the effect of galcanezumab in clinical practice, where treatment adherence and persistence may not be as well controlled and where patients may present with comorbidities and be on multiple medications, will further contribute to the use of preventive treatment for patients with episodic migraine.
In conclusion, this study demonstrated that both the 120- and 240-mg doses of galcanezumab were superior to placebo in terms of (i) reducing monthly migraine headache days and the timing of onset of efficacy and (ii) improvements in patient functioning and disability. The safety and tolerability of galcanezumab in Japanese patients were consistent with the findings from global studies conducted primarily in Caucasian populations. Overall, no new safety concerns were identified, most AEs were injection site-related, and very few patients discontinued treatment.
Key findings
Treatment with galcanezumab, at either the 120- or 240-mg dose administered subcutaneously once a month, was associated with a clinically significant reduction in the number of monthly migraine headache days.
Patients treated with galcanezumab reported a reduction in the use of acute migraine treatments, including triptans, and an improvement in functioning and disability.
Galcanezumab treatment was safe and well tolerated by Japanese patients with migraine.
Footnotes
Acknowledgements
The authors would like to thank all study participants and Dr Atsushi Kuga, MD, PhD, formerly of Eli Lilly Japan K.K, for his substantial contributions to the conception and design of the study and preparation of the manuscript.
Author contributions
All authors participated in the study design, interpretation of study results, and the drafting, critical revision, and approval of the final version of the manuscript. FS was an investigator in the study, and AO conducted the statistical analysis.
Data sharing statement
Lilly provides access to all individual participant data collected during the trial, after anonymization, with the exception of pharmacokinetic or genetic data. Data are available to request 6 months after the indication studied has been approved in the United States and European Union and after primary publication acceptance, whichever is later. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, blank, or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at ![]() .
.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AO is an employee of Eli Lilly Japan K.K. and VS is an employee of Eli Lilly and Company. AO and VS are minor shareholders in Eli Lilly and Company. FS has received funding as an advisor for Eli Lilly Japan K.K. Medical writing assistance was provided by Serina Stretton, PhD, CMPP, and Yuriko Kikuchi-Rech, PhD, of ProScribe—Envision Pharma Group, and was funded by Eli Lilly Japan K.K. ProScribe’s services complied with International Guidelines for Good Publication Practice (GPP3).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Eli Lilly and Company, manufacturer of galcanezumab, sponsored the study. Eli Lilly Japan K.K. was involved in the study design, data collection, data analysis, and preparation of the manuscript.
