Abstract
Background
Rasagiline mesylate, a monoamine oxidase-B inhibitor, is used in Parkinson's disease (PD) treatment. We report a case of reversible cerebral vasoconstriction syndrome (RCVS) developed after rasagiline mesylate initiation.
Case presentation
A 59-year-old woman was diagnosed with PD at the age of 54 years. She experienced frequent episodes of nausea after increasing the dose of levodopa-carbidopa hydrate, and her nausea worsened with the addition of rasagiline mesylate. Three months later, the patient experienced recurrent thunderclap headaches. Brain imaging revealed subarachnoid hemorrhage with segmental vasoconstriction in the main trunks of multiple cerebral arteries. The subsequent clinical course was consistent with RCVS. Her headache and nausea were completely resolved by the discontinuation of rasagiline mesylate and addition of verapamil hydrochloride.
Discussion
Worsened nausea was the only sign implicating the elevated monoamine levels, potentially linked to the development of RCVS. RCVS should be considered when a patient taking rasagiline mesylate experiences thunderclap headache.
Keywords
Introduction
Monoamine oxidase (MAO) has two isoforms: MAO-A, which representatively metabolizes serotonin, dopamine, norepinephrine, and tyramine, and MAO-B, which metabolizes phenylethylamine and dopamine. Monoamines, especially MAO-A substrates, can activate the sympathetic nervous system and increase blood pressure. 1 Rasagiline mesylate, a monoamine oxidase (MAO)-B inhibitor, is used in Parkinson's disease (PD) treatment. To the best of our knowledge, no reports have suggested an association between reversible cerebral vasoconstriction syndrome (RCVS) development and rasagiline mesylate. Herein, we report a case of RCVS developed during treatment for PD with rasagiline mesylate.
Case presentation
The case involved a 59-year-old woman with a history of episodic migraine. She developed PD with a complaint of rigidity of the left lower extremity at the age of 54 years and was started on oral levodopa-carbidopa hydrate. Five months before the first episode of thunderclap headache, the patient's physician had increased her dose of levodopa-carbidopa hydrate from 250 to 400 mg/day as she complained of freezing of gait. She then started experiencing frequent episodes of nausea, and oral domperidone (10 mg/day) was started. Two months later, oral rasagiline mesylate (1 mg/day) was added to resolve her complaint of freezing of gait, which made her nausea persistent. Although the dose of oral domperidone was increased from 10 to 20 mg/day, her nausea was not alleviated. Three months after the addition of rasagiline mesylate (day 0), the patient noticed a migraine-like discomfort in her right temple as usual after lunch while sitting and watching television, and she soon experienced a thunderclap headache (numerical rating scale [NRS] score of 10 in her occipital region). The pain gradually subsided within 30 min, with a throbbing pain persisting (NRS score 3–5), primarily in the right occipitoparietal region. Upon visiting a hospital that day, her blood pressure was 125/75 mmHg, and the patient was prescribed oral combined analgesic medications, which did not improve her headache. The following day (day 1), she experienced a second episode of thunderclap headache after feeling “something like a surging wave” in the occipital region for a 20–30 s while having breakfast. The patient revisited the hospital after the pain gradually subsided (NRS score 3–5) within 30 min (subsequent thunderclap headaches also started and subsided in the same pattern). She was prescribed pramipexole hydrochloride (0.125 mg/day) for suspected worsening of the posterior neck rigidity due to PD; however, this medication also did not improve her headache. On day 5, she experienced a third episode of thunderclap headache throughout the head while watching television after lunch and visited the hospital. She was intravenously administered acetaminophen (500 mg). However, the headache was not relieved. Brain computed tomography on day 8 showed no abnormalities, and the dosage of pramipexole hydrochloride was increased to 0.25 mg/day. On day 11, the patient experienced a fourth episode of thunderclap headache throughout her head while watching television in the afternoon and visited our hospital on the same day. Her blood test showed that hepatic and renal functions were normal, and the serum C-reactive protein level was not elevated. Brain magnetic resonance imaging (MRI) revealed subarachnoid hemorrhage in the ambient cistern and left parieto-occipital lobe (Figure 1(a)-1,2). Segmental vasoconstriction in the distal main trunks of the anterior, middle, and posterior cerebral arteries was observed on brain magnetic resonance angiography (MRA) (Figure 1(a)-3). The patient was tentatively diagnosed with RCVS based on the recurrent episodes of thunderclap headaches with subarachnoid hemorrhage. She was admitted to our hospital and conservatively treated. Rasagiline mesylate and pramipexole hydrochloride were discontinued from day 12. On that morning, she noticed a continuous cloudy haze in the lower-right quadrant of her visual field. In the afternoon, she experienced a fifth episode of thunderclap headache throughout the head when lying in bed, and oral verapamil hydrochloride 120 mg/day was initiated. On day 13, the headache and nausea had completely resolved, and the haze had mildly improved. On day 15, oral domperidone was discontinued. Brain MRI revealed gradual resolution of the subarachnoid hemorrhage (Figure 1(b)-1,2). Brain MRA revealed extensive segmental vasoconstrictions in the main trunks of multiple cerebral arteries, suggesting centripetal propagation of the vasoconstrictions (Figure 1(b)-3). Arterial spin labeling (ASL) revealed hyperperfusion of the left occipital lobe (Figure 1(b)-4,5). Electroencephalography revealed neither epileptiform discharges nor continuous focal slowing. Nausea recurred on day 16, and it was alleviated with 10 mg intravenous metoclopramide. The patient gradually noticed throbbing pain in the right parietal area on the morning of day 17. The hemorrhage in the ambient cistern revealed by MRI had disappeared, and the vasoconstriction in the main trunks of multiple cerebral arteries improved (Figure 1(c)-1,2,3). ASL revealed more prominent hyperperfusion in the left parietal lobe than in the left occipital lobe (Figure 1(c)-4,5). The oral dose of verapamil hydrochloride was increased from 120 to 240 mg/day. On day 19, both throbbing pain and haze were completely relieved. On day 21, subarachnoid hemorrhage had almost resolved on MRI (Figure 1(d)-1,2). Vasoconstrictions of the cerebral arteries on brain MRA and findings of hyperperfusion of the cerebral blood flow on ASL persisted (Figure 1(d)-3,4,5). By day 37, there were no abnormal findings on brain MRI/MRA and ASL. The clinical course (Figure 2) and imaging findings were consistent with the manifestations of RCVS. 2 Her recurrent thunderclap headache was retrospectively diagnosed as acute headache attributed to RCVS (6.7.3.1) based on the third edition of the International Classification of Headache Disorders. 2 Her motor function related to PD was controlled using oral levodopa/carbidopa hydrate (400 mg/day) alone without causing nausea and freezing of gait. The patient was discharged on day 39. Nine months after onset of RCVS, she experienced rigidity of the left upper extremity, and the addition of oral opicapone (25 mg/day) was effective. RCVS had not recurred to date (17-month follow-up from its onset). She had not experienced any triggers of RCVS such as psychological strain or sneezing during the clinical course.
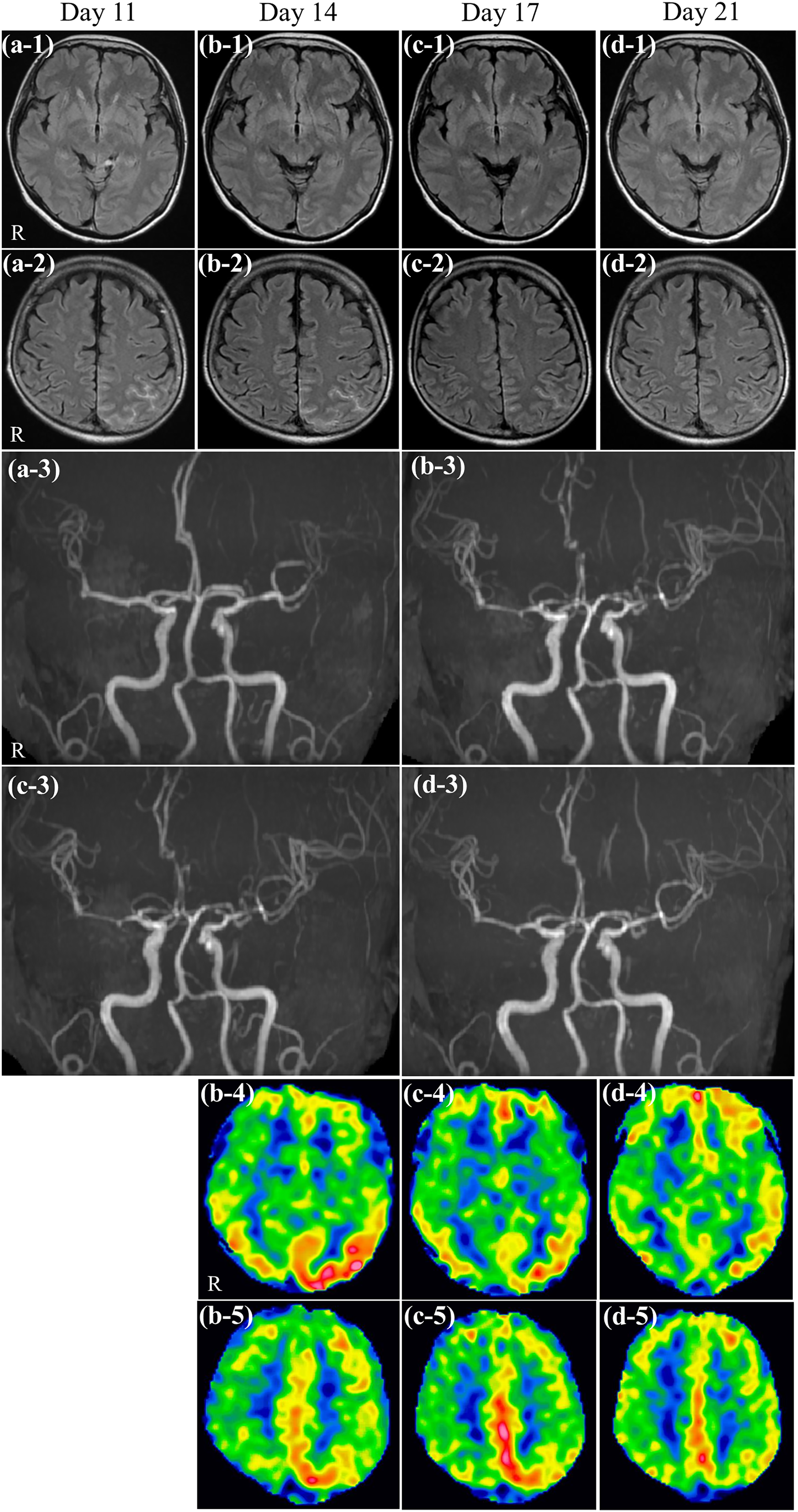
Serial evaluations on brain MRI. FLAIR: (a) hemorrhage in the ambient cistern and left parieto-occipital lobe appeared on day 11 from the onset of RCVS (a-1,2). The hemorrhage in the ambient cistern disappeared on day 17 and that in the left occipitoparietal lobe decreased gradually over time from day 14 to 21 (day 14, b-1,2; day 17, c-1,2; day 21, d-1,2). Conditions: SIGNA Artist 1.5 T; TR 10000 ms, TE 100 ms, thickness 5 mm, spacing 1.5 mm. MRA: Segmental vasoconstrictions appeared in the distal main trunks of the anterior, middle, and posterior cerebral arteries on day 11 from the onset of RCVS. Cerebral aneurysm was not observed (a-3). On day 14, extensive segmental vasoconstrictions developed in the main trunks of multiple cerebral arteries, and centripetal propagation of the vasoconstrictions appeared (b-3). They gradually alleviated over time, as observed on days 17 (c-3) and 21 (d-3). Conditions: SIGNA Artist 1.5 T; TR 28 ms, TE (out of phase). ASL: Hyperperfusion appeared in the left occipital lobe on day 14 (b-4,5). On day 17, more prominent hyperperfusion appeared in the left parietal lobe than in the left occipital lobe (c-4,5). On day 21, alleviation of hyperperfusion of the cerebral blood flow was observed (d-4,5). Conditions: SIGNA Artist 1.5 T; post-labeling delay 2025 ms. ASL, arterial spin labeling; FLAIR, fluid attenuated inversion recovery; MRA, magnetic resonance angiography; MRI, magnetic resonance imaging; R, right; RCVS, reversible cerebral vasoconstriction syndrome; TE, echo time; TR, repetition time.

Patient's clinical course. A thunderclap headache developed and recurred 5 times in 12 days with continuous headache and nausea. She had been taking rasagiline mesylate (1 mg/day), levodopa-carbidopa hydrate (400 mg/day), domperidone (20 mg/day), and Macrogol 4000 during the above clinical course. Brain magnetic resonance imaging was performed on days 11 (a), 14 (b), 17 (c), and 21 (d). The letters correspond to the same timepoints used in Figure 1.
Discussion
Overactivation of the sympathetic nervous system by sympathomimetic vasoactive substances or Valsalva maneuver-like triggers may cause both abrupt dilation of the distal arterioles and stretching of the perivascular nociceptive nerve with a thunderclap headache, resulting in RCVS development. 3 To counteract both excessive pulsatile flow related to blood pressure surge and distal arteriole dilation, the main trunks of multiple cerebral arteries subsequently constrict, manifesting as centripetal propagation of vasoconstrictions. 3 On ASL, the affected brain regions exhibit hypoperfusion at RCVS onset, followed by a switch to hyperperfusion after 1–2 weeks. 4 Most symptoms gradually improve for several weeks even if accompanied by stroke, and few have residual deficits. 5 Oral verapamil hydrochloride is one of the medications used to treat RCVS, 6 as well as nimodipine, which is not available in Japan. In the present case, the clinical course and image findings were consistent with those of typical RCVS.
There are two reports of RCVS occurring while taking MAO inhibitors (phenelzine and tranylcypromine). 7 In those cases, the inhibitors’ amphetamine-like structure and increased monoamine levels were considered to cause hyperactivation of the sympathetic nervous system and to have triggered RCVS development. 7 Rasagiline mesylate, a selective MAO-B inhibitor, does not have an amphetamine-like structure and is well studied in terms of its safety. 1 However, it has a dose-dependent MAO-A inhibitor effect and may contribute to serotonin syndrome 8 or elevated blood pressure 1 when the monoamine levels increase beyond an individual's tolerance in a certain condition.
In the present case, we hypothesized that rasagiline mesylate may potentially linked to the development of RCVS, after carefully considering medications or triggers that could cause RCVS. The relationship between RCVS and serotonin is well studied. 5 The improvement of nausea after discontinuing rasagiline mesylate, while continuing the increased dose of levodopa-carbidopa hydrate, may suggest not only elevated dopamine levels but also excessive serotonin levels. Particular single-nucleotide polymorphisms in patients with PD have been reported to be associated with individual differences in clinical responses to rasagiline mesylate. 9 This patient may have had some specific predisposition to MAO sensitivity, which could result in excess serotonin levels.
Our hypothesis has several limitations. First, we could not objectively demonstrate elevated serotonin levels. However, serotonin toxicity is assessed based on clinical symptoms, not based on serum or cerebrospinal fluid serotonin levels. 8 In the present case, nausea may be the only sign implicating elevated serotonin levels. Second, the mechanism by which RCVS developed 3 months after starting oral rasagiline mesylate remains unknown. The inhibition rate of platelet MAO-B peaks at 6 days after starting oral rasagiline mesylate (2 mg/day). 10 In another study, half of the patients who started taking oral rasagiline mesylate reported that they experienced some adverse effects between 2 and 6 months, including dyskinesia, orthostatic hypotension, and headache. 11 Increased serotonin levels caused by both rasagiline mesylate and other potential triggers may be required for RCVS development. Third, we could not totally exclude the possibility that RCVS occurred independently of rasagiline mesylate. Our hypothesis is based on the premise that the patient had not experienced any triggers of RCVS such as psychological strain or Valsalva strain.
RCVS should be considered when a patient taking rasagiline mesylate experiences thunderclap headache, especially when accompanied by nausea.
Article highlights
Rasagiline mesylate potentially linked to develop reversible cerebral vasoconstriction syndrome (RCVS) by increasing serotonin levels.
RCVS should be considered when a patient taking rasagiline mesylate experiences a thunderclap headache, especially when accompanied by persistent nausea.
With RCVS in mind, it is important to pay attention to the onset episode of the headache; by the time patients come to the hospital, their headache may have already alleviated.
Footnotes
Acknowledgments
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yasuhiko Matsumori reports lecture fees/honoraria from Amgen K.K., Daiichi Sankyo Co., Ltd, Eli Lilly Japan K.K., Otsuka Pharmaceutical Co., Ltd
Ethical statement
The patient provided written informed consent for the publication of this report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
