Abstract
Background
Reversible cerebral vasoconstriction syndrome is normally triggered by vasoactive compounds or illicit drugs. A new type of migraine preventive medication blocks calcitonin gene-related peptide utilizing monoclonal antibodies. Calcitonin gene-related peptide is a potent vasodilator for the cerebrovascular system. Could blocking calcitonin gene-related peptide be a trigger for cerebral artery vasospasm in patients susceptible to developing reversible cerebral vasoconstriction syndrome (migraine patients) or in individuals using vasoactive compounds? We present a case of reversible cerebral vasoconstriction syndrome occurring after calcitonin gene-related peptide monoclonal antibody treatment.
Case report
A 43-year -old woman with a history of episodic migraine developed an acute headache with orgasm two days after taking her second injection of erenumab. Ten days after erenumab injection she developed a thunderclap headache while completing a high intensity workout. These new headaches were only left sided. Computed tomography angiography demonstrated mild to moderate areas of narrowing involving the left middle and anterior cerebral arteries, concerning for reversible cerebral vasoconstriction syndrome. She denied exposure to any known reversible cerebral vasoconstriction syndrome precipitant medication or illicit drugs. She did endorse recent exposure to high altitude prior to erenumab therapy. She was started on verapamil 40 mg three times per day and her headache ceased within 24 h of initiating treatment. A repeat CT angiogram completed 4 weeks after the initial study noted resolution of the areas of vessel stenosis.
Conclusion
A case of reversible cerebral vasoconstriction syndrome developing after treatment with a calcitonin gene-related peptide monoclonal antibody is presented. The timing of the new type of headache occurring 2 days post erenumab injection suggests a possible cause and effect relationship. Reversible cerebral vasoconstriction syndrome as a possible treatment-related complication to the usage of calcitonin gene-related peptide monoclonal antibodies needs to be studied further.
Reversible cerebral vasoconstriction syndrome (RCVS) is a disorder manifested by severe acute headaches and cerebral artery vasospasm (1). There appears to be a clinical phenotypic spectrum to RCVS presentation including recurrent thunderclap headaches over several months with resolution, recurrent thunderclap headaches evolving into a daily persistent headache, isolated thunderclap headaches, and finally a new daily persistent headache (NDPH) after a single thunderclap headache (2,3). There are various triggering precipitants to RCVS that are well documented in the literature, including vasoconstrictive medications and/or illicit substances, pregnancy/post-partum, exposure to blood products and intracranial hypotension to name a few (1). Female migraine patients may also have a heightened risk of developing RCVS (4). A new class of migraine preventive medications, monoclonal antibodies that block either the calcitonin gene-related peptide (CGRP) itself or its receptors, are becoming more regularly prescribed by neurologists and non-neurologists alike. CGRP is a potent vasodilator for both the cerebrovascular and cardiovascular systems (5). Thus, blocking CGRP for prolonged periods of time could potentially lead to ischemia in patients having mild oligemia with a TIA and conceivably more prolonged vasospasm if a patient was having a bout of RCVS (6,7). Could blocking CGRP alone be a trigger for cerebral artery vasospasm/constriction in patients susceptible to developing RCVS (migraine patients) or in individuals using vasoactive compounds? A case is presented of RCVS developing in a female patient shortly after an erenumab injection for migraine prevention.
Case report
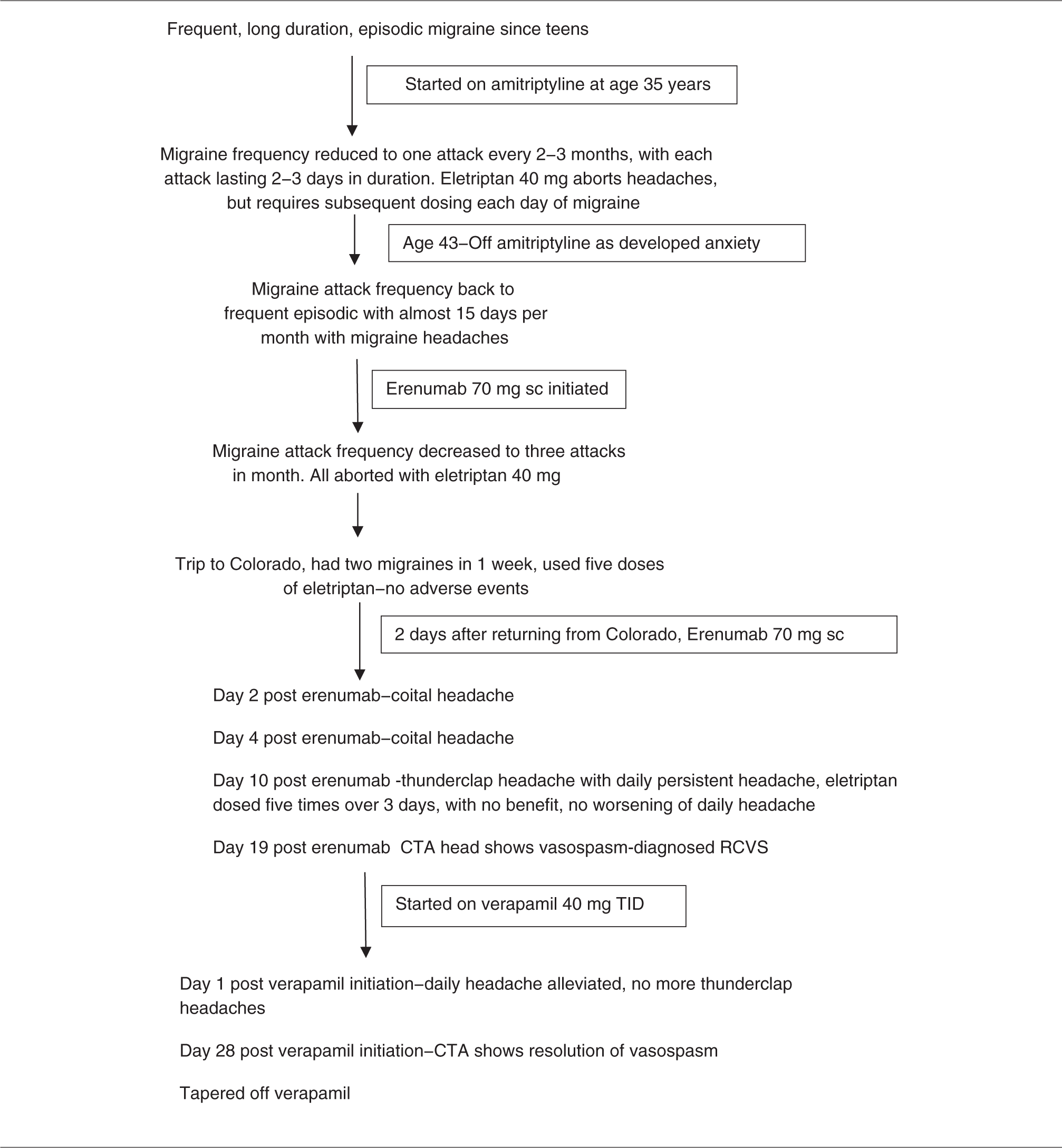
The patient is a 43-year-old Caucasian woman with a history of frequent episodic migraine without aura since her teens. Typical headache location was bitemporal, bi-occipitonuchal or bi-retroorbital. Headaches were never unilateral. Her headaches could last 2–3 days with associated nausea, photo- and phonophobia as well as osmophobia. She never had brainstem or focal motor or sensory symptoms with her headaches. Migraines were well controlled on amitriptyline (30 mg) but because of anxiety issues she tapered off the medication. She had no prior response to propranolol or topiramate. Eletriptan (40 mg dose) was a very effective abortive, never causing a worsening of headaches. Typically, she would require only one 40 mg dose to abort a headache for that day, but as her headaches could last 2–3 days, she could require a single dose every day of the migraine attack. For a brief period, her headaches remained controlled off amitriptyline, but they then began to increase in frequency to almost half the month. As she had no contraindication to CGRP monoclonal antibodies (cardiovascular or cerebrovascular issues), erenumab 70 mg was initiated. She had no side effects from the first round of dosing and demonstrated great improvement with a reduction in frequency to only three migraine attacks during the month post injection. The migraines that did occur were easily aborted with eletriptan. She thus remained at the 70 mg dosing for the second injection. A new type of headache, however, began 48 h after her second administration of erenumab. The unique headache was first noted immediately post coitus at the time of orgasm. It was of acute onset, peaking in seconds, but only of moderate severity and she attributed it to neck irritation as the pain was localized to the occipito-nuchal junction on the left side. It was somewhat concerning to her as she had never had a left-sided only headache with her prior migraines. The headache lasted 1–2 h without treatment and had no associated migrainous symptoms and there was no neurologic dysfunction. Two days after the first coital-related headache, she had a second headache again with coitus/orgasm. This presented in the same manner with acute onset, moderate severity pain, left occipito-nuchal location and lasting 1–2 h in duration. Ten days after her erenumab injection she developed a thunderclap headache of maximum intensity without latency, while she was completing a high intensity workout. She described the pain as starting at the base of her head on the left side and shooting up the left side of her face, and subsequently becoming retro-orbital. When the headache first started, she reported “it was so bad I almost couldn't see”. From that point on she had a persistent moderate level intensity headache, only left-sided without a recurrent thunderclap headache. She tried five doses of eletriptan over several days, without a worsening of the headaches, but with no improvement. She was an avid runner, and reported that she was able to run with her new headache but when she pushed herself to a higher aerobic level the pain did worsen, leading her to stop. She contacted her treating headache neurologist 17 days after the erenumab treatment. Based on the patient’s clinical headache description she was diagnosed with thunderclap headache and headache associated with sexual activity by ICHD-3 criteria, but secondary evaluation was still required (8). Imaging studies were ordered. Within 48 h of her contact with her neurologist the patient had a CT head, CT angiogram (head and neck) and CT venogram (head) to rule out secondary etiologies for thunderclap headache. A negative pregnancy test was finalized prior to imaging. The CT angiogram was positive for mild to moderate areas of narrowing involving the M1 segment of the left middle cerebral artery and A2 segment of the left anterior cerebral artery, concerning for RCVS (Figure 1(a), 2(a)). There was no dissection or cerebral vein thrombosis noted. There was no subarachnoid blood or ischemia noted on the CT head. Her headaches now met ICHD-3 criteria for headache attributed to RCVS (8). No further testing was deemed necessary at the time as a secondary etiology for her thunderclap headache was discovered. She was admitted to the hospital to monitor blood pressure and neurologic status. She had a normal BMI. Her exam was non-focal except that she demonstrated both cervical and systemic hypermobility. She was started on an oral calcium channel blocker (verapamil 40 mg three times per day). On further questioning, she did endorse recent exposure to high altitude, noting that she was in Colorado for 1 week and she returned to Florida (her home state) 2 days prior to erenumab therapy. She did experience headaches while in Colorado, more reminiscent of her typical migraines, and these resolved with the use of eletriptan. She had taken five total doses of the triptan while she was on vacation without adverse events. She was on no hormonal supplementation. She had no exposure to ergotamine, pseudoephedrine, diet pills, energy drinks, cocaine, ecstasy, cannabis, LSD, tacrolimus, cyclophosphamide, or nicotine patches. The patient also denied a recent blood transfusion. Prior pregnancies were not marked by eclampsia. Her headache ceased within 24 h of starting verapamil. She remained headache free while on verapamil. A repeat CT angiogram was completed 4 weeks after the initial neuroimaging study and noted resolution of the areas of vessel stenosis in both the left middle cerebral and anterior cerebral arteries, helping to prove the diagnosis of RCVS. (Figure 1(b), 2(b)) She was thus instructed to taper off verapamil by 40 mg every 7 days. She did so without headache recurrence. She was directed to stop using erenumab as well as triptans to avoid possible RCVS recurrence. The timeline of events is in Table 1.

(a) (Images completed 19 days after erenumab injection): Axial CT angiogram image demonstrates narrowing and irregularity of the M1 and M2 segments of the left middle cerebral artery (arrow). (b) (Completed 4 weeks after initial imaging, patient on verapamil): Axial CT angiogram image demonstrates resolution of the previously seen narrowing of the M1 and M2 segments of the left middle cerebral artery (arrow).

(a) (Images completed 19 days after erenumab injection): Sagittal CT angiogram image demonstrates narrowing of the A2 segment of the left anterior cerebral artery (arrow). (b) (Completed 4 weeks after initial imaging, patient on verapamil): Sagittal CT angiogram image demonstrates resolution of the previously seen narrowing of the A2 segment of the left anterior cerebral artery (arrow).
Patient headache timeline.
Discussion
A patient is presented who developed a new type of headache 2 days after her second erenumab injection. Initial headache presentation was solely coital-based headaches, potentially thunderclap as of acute onset but only of moderate severity. She then developed a “true” ICHD-3 defined thunderclap headache of maximum severe intensity without latency during an intense work out, with a daily persistent headache ensuing thereafter (8). Nine days after the exercise-induced thunderclap headache, neuroimaging noted vasospasm side-locked to the patient’s headache and consistent with a diagnosis of RCVS. The vasospasm and new headache alleviated with a calcium channel blocker medication. Thus, this would appear to be a case of RCVS developing after a CGRP monoclonal antibody administration. The timing of the new type of headache developing 2 days post erenumab injection possibly suggests a direct cause and effect relationship, but it could still be coincidental (see Limitations section). The patient did travel to a higher altitude, which has been linked to RCVS onset in a prior case report, and she used triptans the week prior to RCVS initiation, which are also linked to triggering RCVS. Thus, it is possible that these events alone or in combination triggered RCVS without any added effect from blocking CGRP-induced vasodilation (9). It is also feasible that these events were necessary (lowering the threshold) for CGRP monoclonal antibody-triggered RCVS. Finally, it is conceivable that the RCVS was solely triggered by erenumab. There is a single case report of RCVS developing after dihydroergotamine infusion in a patient on erenumab, but the dihydroergotamine was the presumed precipitating factor not the CGRP monoclonal antibody. In addition, the reported patient was on multiple daily medications including other RCVS triggers (serotonin-noradrenaline reuptake inhibitor) (10).
It would make sense that CGRP monoclonal antibodies could trigger RCVS, as administration may preclude any reflex vasodilation that is needed during a state of cerebral artery vessel constriction. There are CGRP receptors on smooth muscle cells of cerebral arteries (11). In isolated human cerebral arteries, CGRP monoclonal antibodies inhibit CGRP-induced vessel relaxation (12). In subarachnoid hemorrhage, CGRP is released to counteract blood-induced cerebral artery vasospasm (5). Edvinsson, in his work on CGRP, noted that in an environment of intracranial arterial vasoconstriction, the trigemino-vascular reflex is activated to release CGRP to counter-balance the vasospasm with a potent vasodilator (5).
There are probably only a select few migraine patients who will develop cranial artery vasospasm with medication or illicit substances. In the same vein, there will probably be only a select few migraine patients who can develop RCVS after CGRP monoclonal antibody administration. Further study to define that specific subgroup of patients will be important, as RCVS can cause significant morbidity including stroke and cerebral hemorrhage. The present case patient with possible erenumab-triggered RCVS was systemically hypermobile. Hypermobile female migraine patients could be at higher risk for developing RCVS as they have autonomic dysregulation and vessel laxity at baseline. One of the authors has already reported on NDPH after a single thunderclap headache as a possible RCVS variant and most of those reported patients (three of four) had hypermobility issues (2).
Limitations to the hypothesis
There are several items about erenumab that may question a cause and effect relationship with RCVS.
Pharmacokinetics: The patient’s initial coital headache, which was presumed to be the onset of her RCVS, occurred on day 2 post administration of erenumab. However, the time needed to reach maximal plasma levels (Tmax) after subcutaneous injection of erenumab 70 mg is between 3 to 11 days (13). Thus, was the timing of her coital headache too early for it to be related to erenumab? One possibility is that the RCVS truly started with the exercise-induced thunderclap headache on day 10 post erenumab administration, which fits well with the Tmax kinetics of the monoclonal antibody and that the coital headaches were either set up by another secondary etiology (possibly elevated CSF pressure after her recent trip to Colorado, or cervicogenic-based head pain associated with her cervical hypermobility issues) and/or they were just primary headache associated with sexual activity. Luminal versus abluminal monoclonal antibody placement: The animal model literature on CGRP demonstrates that CGRP blockade with monoclonal antibodies administered abluminally prevents CGRP-induced vasodilation, while luminal application does not prevent it (14). The main issue is that CGRP receptors are located on the smooth muscle cells and that luminal CGRP monoclonal antibodies are prevented from reaching these abluminal receptors because of the blood-brain barrier (BBB) (11,14). In the experimental models looking at erenumab and human meningeal arteries, the erenumab was present luminally as well as abluminally, but clinically when erenumab is given to patients the antibody would primarily be present on the luminal side (15). Thus, luminal CGRP monoclonal antibodies may not be able to influence abluminal CGRP-induced vasodilation and thus it may not have been possible for luminal erenumab to induce RCVS. One possible scenario, however, is that the patient developed some BBB breakdown secondary to the migraines she experienced just prior to erenumab treatment (15). Then, the resultant RCVS (starting with the coital-based headaches) led to further BBB disruption (16). It has recently been documented that the BBB breakdown secondary to RCVS peaks earlier than the phase of vasoconstriction/vasospasm (16). This may then explain the delayed thunderclap headache occurring on day 10 post erenumab treatment, at the time of true Tmax concentration. The hypothesis is that the cumulative BBB disruption allowed luminal erenumab to influence abluminal CGRP receptors to block vasodilation and induce or enhance vasospasm.
Conclusion
Based on this case report, it is suggested to warn patients of RCVS symptomatology when providing CGRP monoclonal antibody treatment as migraine patients may consider a worsening headache, even a thunderclap headache, as just a severe migraine and utilize triptans, which could potentially make their neurologic situation worse with added cerebral arterial vasoconstriction. RCVS as a possible treatment-related complication to the usage of CGRP monoclonal antibodies needs to be studied further.
Clinical implications
CGRP monoclonal antibody treatment has been shown to be effective for migraine prevention; however, there could be inherent risks in blocking a major vasodilator to the human circulatory system. Patients should always be aware that a thunderclap headache (head pain that reaches maximum intensity without latency) is abnormal until proven otherwise. Any new headache after CGRP monoclonal antibody treatment should be reported to the treating physician so that evaluation for RCVS can be obtained if deemed necessary.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Consent
The patient gave consent for the case report.

