Abstract
Background:
Concomitant fremanezumab, a calcitonin gene-related peptide (CGRP) pathway monoclonal antibody (mAb), and onabotulinumtoxinA (onabotA) improve treatment response compared with onabotA alone in patients with chronic migraine (CM).
Methods:
This was a single-center, retrospective, observational study that assessed treatment response (change over time in monthly headache days [MHD] and pain intensity [PI]) in adult patients with CM receiving fremanezumab as add-on therapy to onabotA for CM prevention.
Results:
In the study population (N = 116, age 50.0 ± 13.1, female 85.3%, pre-index onabotA use 46.5 ± 34.2 months) receiving concurrent onabotA and fremanezumab for 17.5 ± 11.6 months, MHD decreased by 3.60 days (95% confidence interval [CI]: −5.26, −1.94, p < 0.001) and PI was reduced by 0.43 (95% CI: −0.77, −0.09, p = 0.012) at the final visit. Statistically significant reductions were seen in both MHD (−4.61, 95% CI: −6.84, −2.39; p < 0.001) and PI (−0.52, 95% CI: −0.84. −0.09; p = 0.017) among patients naïve to mAbs against CGRP or its receptor. No unexpected adverse events were observed.
Conclusion:
Concomitant fremanezumab and onabotA for CM prevention were effective at reducing the number of MHD and lessening PI, particularly in patients with difficult-to-treat CM who are naïve to mAbs against CGRP or its receptor.
Keywords
Introduction
Migraine is a very common headache disease in clinical practice and is one of the top three ranked disabling conditions (assessed as years lived with disability) among adults globally. 1 Between 2% and 8% of people with migraine have chronic migraine (CM), the management of which requires a more integrated approach including lifestyle modifications, trigger management, and both non-pharmacological and pharmacological interventions for acute and preventive treatments. 2 The goals of preventive migraine treatment include reducing frequency, severity, duration, and disability of migraine attacks, with the aim of improving the overall quality of life. 2,3
OnabotulinumtoxinA (onabotA; Botox®, Abbvie, North Chicago, Illinois) is indicated for the prevention of headache in adults with CM. 4 American Headache Society and European Headache Federation guidelines include additional preventive medication for patients with inadequate response to onabotA alone. 3,5 Calcitonin gene-related peptide (CGRP) is a neuropeptide and a mediator of key pathophysiologic processes involved in migraine, including neurogenic inflammation, cerebral vasodilation, and pain sensitization. 6 The trigeminovascular system is highly pertinent to migraine pathogenesis, where CGRP is released from C-fibers and CGRP receptors play a role in activating immune cells, blood vessels, and A-delta fibers. 6
Four monoclonal antibodies (mAbs) targeting the CGRP pathway are approved by the Food and Drug Administration (FDA) for the preventive treatment of migraine in adults and are increasingly used in the management of CM. 7 Fremanezumab (AJOVY®, Teva, Petah Tikva, Israel) is a mAb that targets the CGRP ligand and was FDA approved in September 2018. 8 Other CGRP pathway mAbs include the receptor-binding erenumab (Aimovig®, Amgen, Thousand Oaks, California), the ligand-binding mAbs galcanezumab (Emgality®, Eli Lilly, Indianapolis, Indiana), and eptinezumab (Vyepti®, Lundbeck, Deerfield, Illinois). 9 –11 Recent updates to migraine prevention/management guidelines in the US and Europe offer guidance on initiating and modifying treatment with CGRP pathway mAbs in patients with inadequate responses to other preventive treatments. 3,5
For patients with CM with a history of treatment failures, combinations of preventive treatments may offer the possibility of achieving adequate treatment response. Several outpatient retrospective studies reported good results with various oral preventives in combinations, 12,13 although a placebo-controlled study of adding propranolol to topiramate failed to demonstrate benefit. 14 Combination therapy with onabotA and a CGRP pathway mAb may provide an additive or synergistic effect on inhibiting the trigeminal nociceptive pathway, although clinical data to support evidence-based recommendations are limited. 5 A systematic review and meta-analysis of real-world evidence showed that combined onabotA and any CGRP pathway mAb could lead to an average reduction of 2.67 monthly headache days (MHD) versus onabotA alone. 15 Most individual studies have investigated either erenumab or galcanezumab in combination with onabotA, 16 –20 although two studies included the use of different CGRP pathway mAbs, including fremanezumab in 9% (14/153) 17 and 42.6% (63/148) of cases. 18 However, to date, there has been no dedicated study of the effectiveness of fremanezumab when concomitantly used with onabotA. Here we report the results of a retrospective study of the effectiveness of concomitant fremanezumab and onabotA in patients with CM at a tertiary headache center in the US.
Methods
The objective of this single-center, retrospective, observational study was to assess the treatment response in adult patients with CM on onabotA treatment for the prevention of migraine who receive fremanezumab as an add-on therapy in routine clinical practice using the electronic medical records (EMR) at the Jefferson Headache Center (JHC; Philadelphia, Pennsylvania). The study was divided into a pre-index period (from the date of first onabotA initiation to the index date [defined as the visit date of initiating fremanezumab treatment]) and a post-index period, i.e. from (but not including) the index date through the last visit (last captured observation while on combination treatment with onabotA and fremanezumab) (Figure 1). Data were analyzed at the index date, 3-, 6-, and 9-months post-index, and at the last visit.

Study design. AE, adverse event; CM, chronic migraine; MHD, monthly headache days; NRS, numerical rating scale; onabotA, onabotulinumtoxinA; PI, pain intensity.
The Institutional Review Board (IRB) determined the protocol to be exempt from IRB review pursuant to Title 45 Code of Federal Regulations Part 46.101(b) governing exempted protocol declarations. The request for EMR data was approved by the compliance officer prior to starting the review. All providers whose records were reviewed for this study were JHC headache specialists, including board-certified physicians in headache medicine, fellows in training, and nurse practitioners.
Patients were considered eligible for inclusion if, on the query of the EMR, they were identified as being ≥18 years of age with a migraine diagnosis according to the International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10) codes (G43.x), onabotA treatment at JHC (J0585 code or CPT64615 code) ≥3 cycles, with subsequent ≥3 months of concomitant use of onabotA and fremanezumab. Records of patients, along with basic demographic information, were obtained from the EMR following approval by the institution’s compliance officer. The medical record number (MRN) was used to access the medical records, while the list of MRNs and the link to the study number were kept in a separate password-protected file on a secure server to ensure confidentiality. Trained staff manually abstracted and analyzed records via chart review. Study data were collected and managed using REDCap® electronic data capture tools hosted at the JHC. 21
The data entry team and contributing physicians collaborated in data verification and correction. This included removal of incorrect or invalid entries for patients who never received onabotA or fremanezumab, or who were administered fremanezumab for less than 3 months. Data entries related to patients who stopped receiving onabotA before receiving fremanezumab, who started onabotA after receiving fremanezumab, and those without follow-up or who were non-JHC patients, were also removed.
Effectiveness outcomes assessed in the overall population were MHD and average pain intensity (PI; 0 = no pain to 10 = worst pain numerical rating scale) for each time point, including the index date. Clinically plausible, prespecified subgroup analyses were done to provide clinical insights among patients with relevant characteristics: by prior CGRP pathway mAb exposure; by daily (30 MHD) or non-daily (<30 MHD) headache (HA) at baseline; and by the HA frequency (≥15 MHD and <15 MHD) at baseline. Reasons for adding fremanezumab and for discontinuing fremanezumab and/or onabotA were also captured. Although details of all adverse events (AEs) were not routinely collected in the EMR system, all AEs that were noted are reported here.
Statistical analysis
The data were analyzed by SPSS® v28 (SPSS Inc., Chicago, Illinois). Analysis of effectiveness outcomes was completed by two predefined time series: (1) baseline, 3, 6, and 9 months, (2) baseline and last visit, which could occur before or after the 9-month time point. Descriptive data were presented as mean ± standard deviation (SD) or median (interquartile range [IQR]) if distribution was non-normal. Longitudinal repeated measure analysis used a generalized estimating equation (GEE) model with an unstructured correlation matrix, and the clustering unit was an individual patient. The main consequence from the effectiveness outcomes was assessed separately by each time series and adjusted for age, sex, and body mass index. Missing data were considered at random without any imputation. Outcomes from each time point were compared pairwise against baseline (least significant difference) and presented in estimated marginal mean (EMM) and standard error (SE). P-values were not multiplicity corrected, and a p-value of <0.05 was considered statistically significant.
Results
Patients, migraine, and treatment experience
In total, 236 patient records were screened, and 151 charts from patients with CM who were receiving onabotA and concurrent fremanezumab treatment from September 2018 to July 2021 were reviewed. Finally, 116 met the inclusion criteria for quantitative analysis (Figure 2).
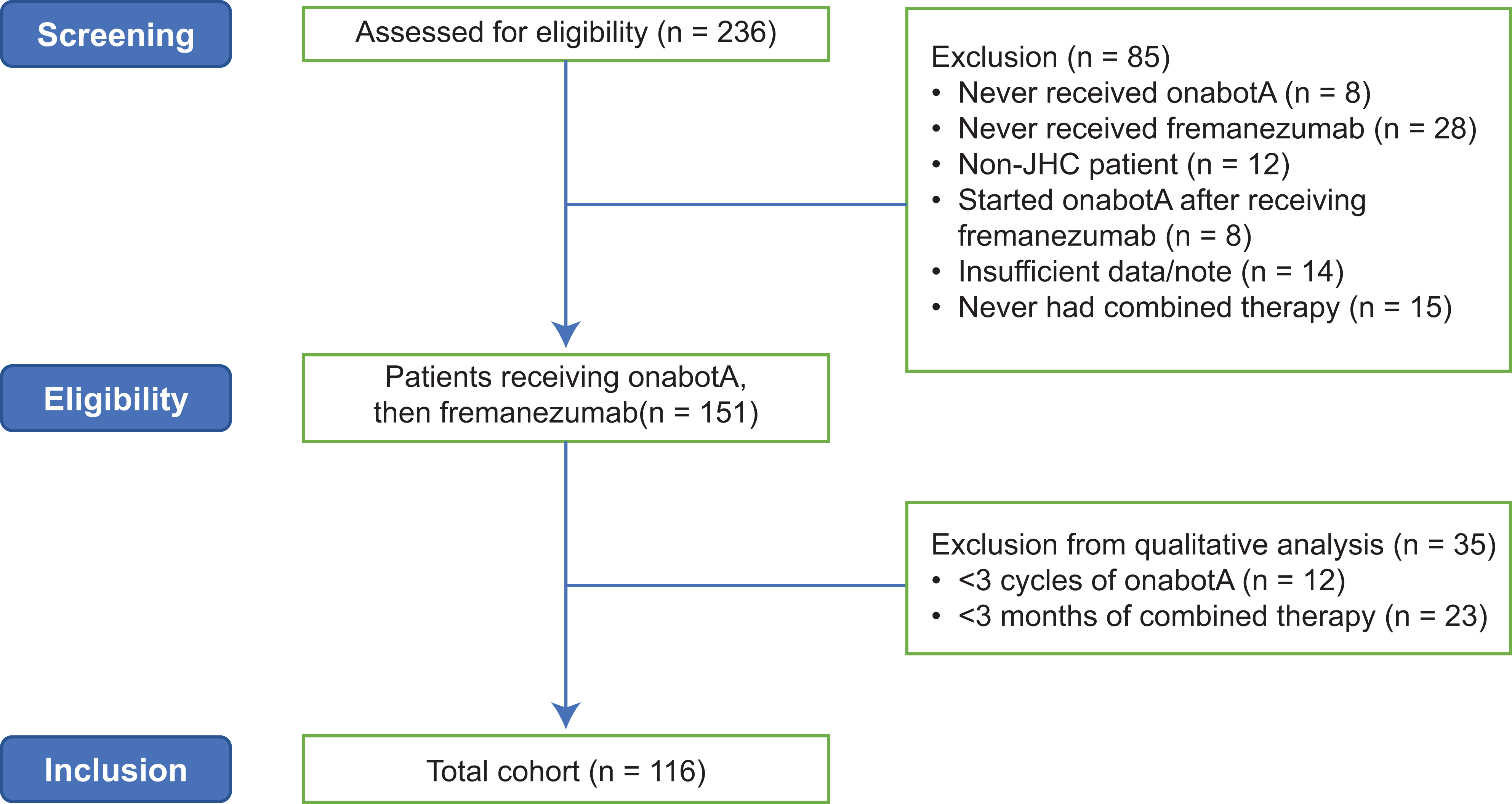
Patient disposition. JHC, Jefferson Headache Center; onabotA, onabotulinumtoxinA.
Patient demographics, migraine characteristics, onabotA usage, and treatment baseline are summarized in Table 1. The median duration of combined onabotA and fremanezumab was 13 (IQR, 23) months (mean ± SD [range] of 17.5 ± 11.6 [3–37]). After the initial three cycles of onabotA, 33 (28.4%) patients reported >50% improvement in either headache or migraine days or severity, 14 (12.1%) reported 30–50% improvement in either headache or migraine days or severity, 34 (29.3%) reported some other improvement and 35 (30.2%) had inadequate documentation of a response. During the study period, over 80% of patients received fremanezumab as monthly dosing: 97/116 (83.6%) at 3 months, 87/102 (85.3%) at 6 months, and 73/87 (83.9%) at 9 months (Table 2).
Patient and migraine characteristics at baseline, and prior treatment.

a Mean (range), median (IQR) unless stated otherwise. b n = 104.
BMI, body mass index; CGRP, calcitonin gene-related peptide; IQR, interquartile range; mAb, monoclonal antibody; MHD, monthly headache days; onabotA, onabotulinumtoxinA.
Treatment pattern and migraine characteristic in the pre-onabotA period, at the index date and across the study period.a,b

a The index date was the date of first fremanezumab initiation added to onabotulinumtoxinA treatment. bLast visit data were analyzed separately. cPercentage calculated based on data from 109 patients (MHD reduction >50%). dPercentage calculated based on data from 109 patients (MHD = 30).
HA, headache activity; MHD, monthly headache days; onabotA, onabotulinum toxin A.
Fremanezumab treatment initiation and discontinuation
Fremanezumab was initiated mostly due to inadequate response to onabotA (101/116, 87.1%). Other reasons recorded for initiating fremanezumab were: experiencing an erenumab side effect (4/116, 3.4%), problems with health insurance (1/116, 0.9%), or other/unknown (10/116, 8.6%).
During the study, five patients stopped onabotA (Table 2). Reasons recorded in the EMRs were: insurance denial (n = 1), experiencing a side effect (n = 1), “other” (n = 2; one patient reported wanting to cease preventive medication and the other decided to pause treatment until ready to restart), or unknown (n = 1). A total of 57 patients stopped fremanezumab treatment (Table 2). Reasons cited for stopping fremanezumab in the post-index period were: inadequate response (30/57, 52.6%), insurance denial (14/57, 24.6%), and side effects (9/57, 15.8%). EMRs also cited unknown (2/57, 3.5%) or “other” (5/57, 8.8%) reasons (patient preference or pregnancy) for ceasing treatment.
Overall response, MHD and PI changes in all patients
Across the study period, most patients (over 75%) reported that control of their migraine was the same or better than at the index date, and around one in four patients had a 50% response (Table 2). In the whole study population (N = 116), statistically significant reductions in MHD were observed 3 months after initiating fremanezumab (−3.80 days; 95% CI: −5.39, −2.19; p < 0.001) (Table 3), and improvements in MHD persisted across the study period (p < 0.001) (Figure 3A and 3B). At the last visit, the number of MHD decreased by 3.60 days (95% CI: −5.26, −1.94) from the index date (p < 0.001; Table 3).
Mean change from index date in MHD and PI over time after initiating fremanezumab treatment in all patients and in subgroups by prior exposure to mAbs targeting CGRP or its receptor.a,b,c
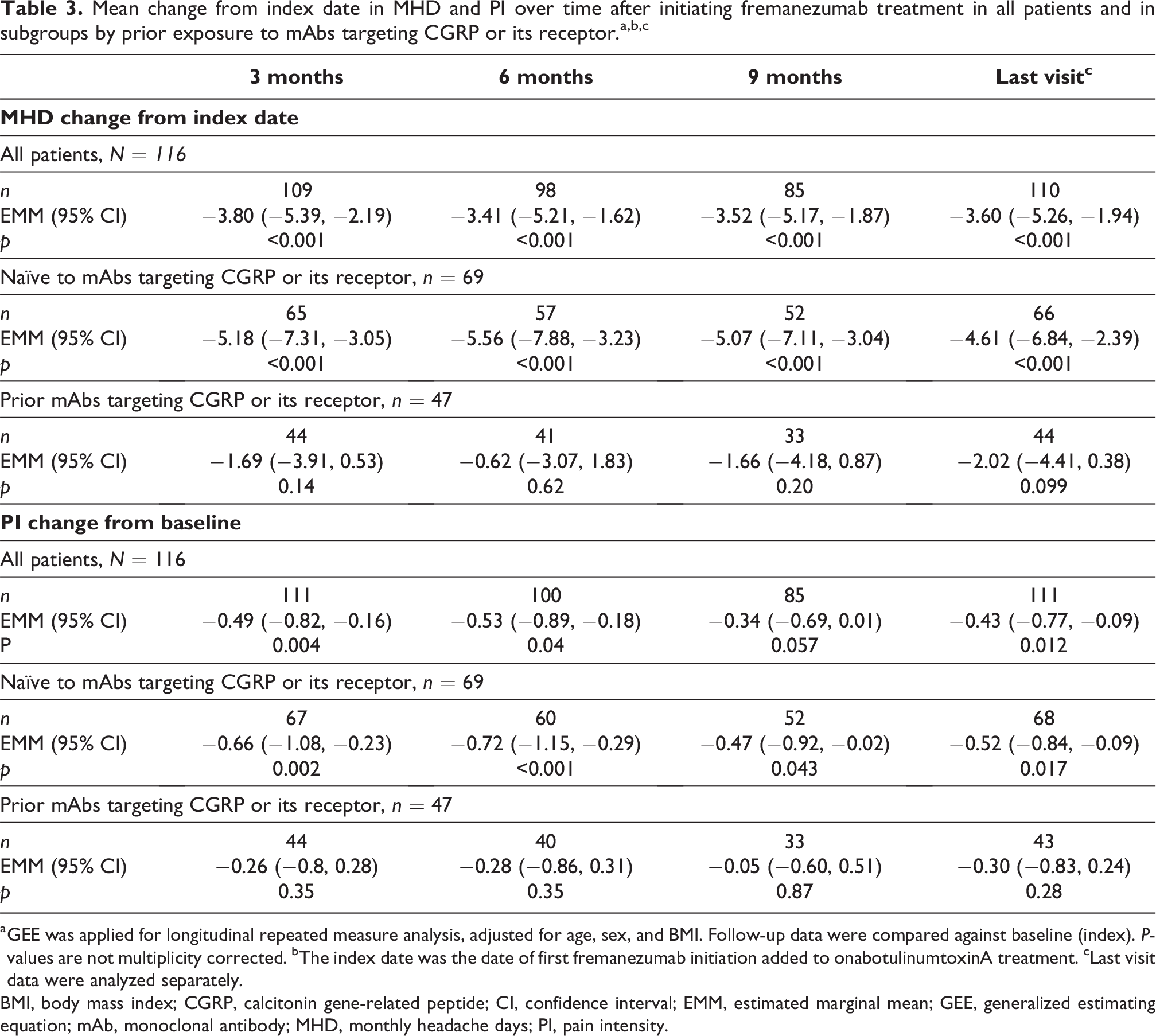
a GEE was applied for longitudinal repeated measure analysis, adjusted for age, sex, and BMI. Follow-up data were compared against baseline (index). P-values are not multiplicity corrected. bThe index date was the date of first fremanezumab initiation added to onabotulinumtoxinA treatment. cLast visit data were analyzed separately.
BMI, body mass index; CGRP, calcitonin gene-related peptide; CI, confidence interval; EMM, estimated marginal mean; GEE, generalized estimating equation; mAb, monoclonal antibody; MHD, monthly headache days; PI, pain intensity.

Mean MHD (3A, 3C, 3E) and PI (3B, 3D, 3F) over time after initiating fremanezumab treatment overall (3A and 3B) and in subgroups: prior exposure to mAbs targeting CGRP or its receptor (3A and 3B for MHD and PI, respectively); daily or non-daily HA at the index date (3C and 3D for MHD and PI, respectively); number of MHD (≥15 or <15) at the index date (3E and 3F, respectively). a,b,c *p < 0.001 versus baseline. † p < 0.005 versus baseline. ‡ p < 0.05 versus baseline. aGEE was applied for longitudinal repeated measure analysis, adjusted for age, sex, and BMI. Follow-up data were compared against baseline (index). P-values are not multiplicity corrected. bThe index date was the date of first fremanezumab initiation added to onabotulinumtoxinA treatment. cBased on the population with available MHD (top panel) or PI (bottom panel) data. MI, body mass index; CGRP, calcitonin gene-related peptide; CI, confidence interval; EMM, estimated marginal mean; GEE, generalized estimating equation; HA, headache activity; mAb, monoclonal antibody; MHD, monthly headache days; PI, pain intensity; SE, standard error.
Statistically significant reductions in PI were also observed 3 months after initiating fremanezumab (−0.49; 95% CI: −0.82, −0.16; p = 0.004) (Table 3). PI continued to be reduced at 6 (p = 0.04) and 9 months (Figure 3A and 3B). Likewise, PI was reduced by 0.43 from baseline at the last visit (95% CI: −0.77, −0.09; p = 0.012; Table 3).
MHD and PI changes in prespecified subgroups
In a subgroup analysis, patients naïve to CGRP pathway mAbs experienced a significant reduction in MHD from the index date across the study period (p < 0.001; Figure 3C, Table 3). At the last visit, these patients experienced 4.61 (95% CI: −6.84, −2.39; p < 0.001) fewer MHD than at the index date (Table 3). Among those with prior exposure to CGRP pathway mAbs, the number of MHD did not change significantly from the index date (p = 0.099).
In addition, patients experienced significant reductions in MHD at all time points regardless of daily HA status at the index date (p < 0.02 versus the index date) (Figure 3C). At the last visit, MHD were reduced by 2.79 days (95% CI: −5.01, −0.58; p = 0.013) and 4.55 days (95% CI: −7.01, −2.09; p < 0.001) in patients with daily and non-daily HA at the index date, respectively (Table 4).
Mean change from index date in MHD and PI over time after initiating fremanezumab treatment for participants with daily HA at the index date versus those with non-daily HA.a,b,c

a GEE was applied for longitudinal repeated measure analysis, adjusted for age, sex, and BMI. Follow-up data were compared against baseline (index). P-values are not multiplicity corrected. bThe index date was the date of first fremanezumab initiation added to onabotulinumtoxinA treatment. cLast visit data were analyzed separately.
BMI, body mass index; CGRP, calcitonin gene-related peptide; CI, confidence interval; EMM, estimated marginal mean; GEE, generalized estimating equation; HA, headache activity; MHD, monthly headache days; PI, pain intensity.
A similar pattern was observed for PI, with a significant reduction in PI from the index date among patients naïve to CGRP pathway mAbs (reduction in PI of 0.52, 95% CI: −0.84, −0.09; p = 0.017) but not those with prior exposure (p = 0.28). Patients with non-daily HA experienced reductions in PI over the post-index period (−0.53; 95% CI: −0.96, −0.10; p = 0.016 at the last visit; Figure 3D, Table 4).
Among patients with ≥15 MHD at the index date, there were significant reductions in both MHD and PI during the post-index period (Figure 3E and 3F, Table 5). At the last visit, MHD were reduced by 4.61 days (95% CI: −6.44, −2.78; p < 0.001) and PI by 0.36 (95% CI: −0.70, −0.02); p = 0.039) versus the index date (Table 5). There were no significant changes over the study in either MHD or PI among patients with <15 MHD at the index date (Figure 3E and 3F, Table 5), although this subgroup comprised a small number of patients (n = 27).
MHD and PI subgroup analysis (change from index date) for participants by CM status (≥15 MHD or <15 MHD) at the index date.a,b,c
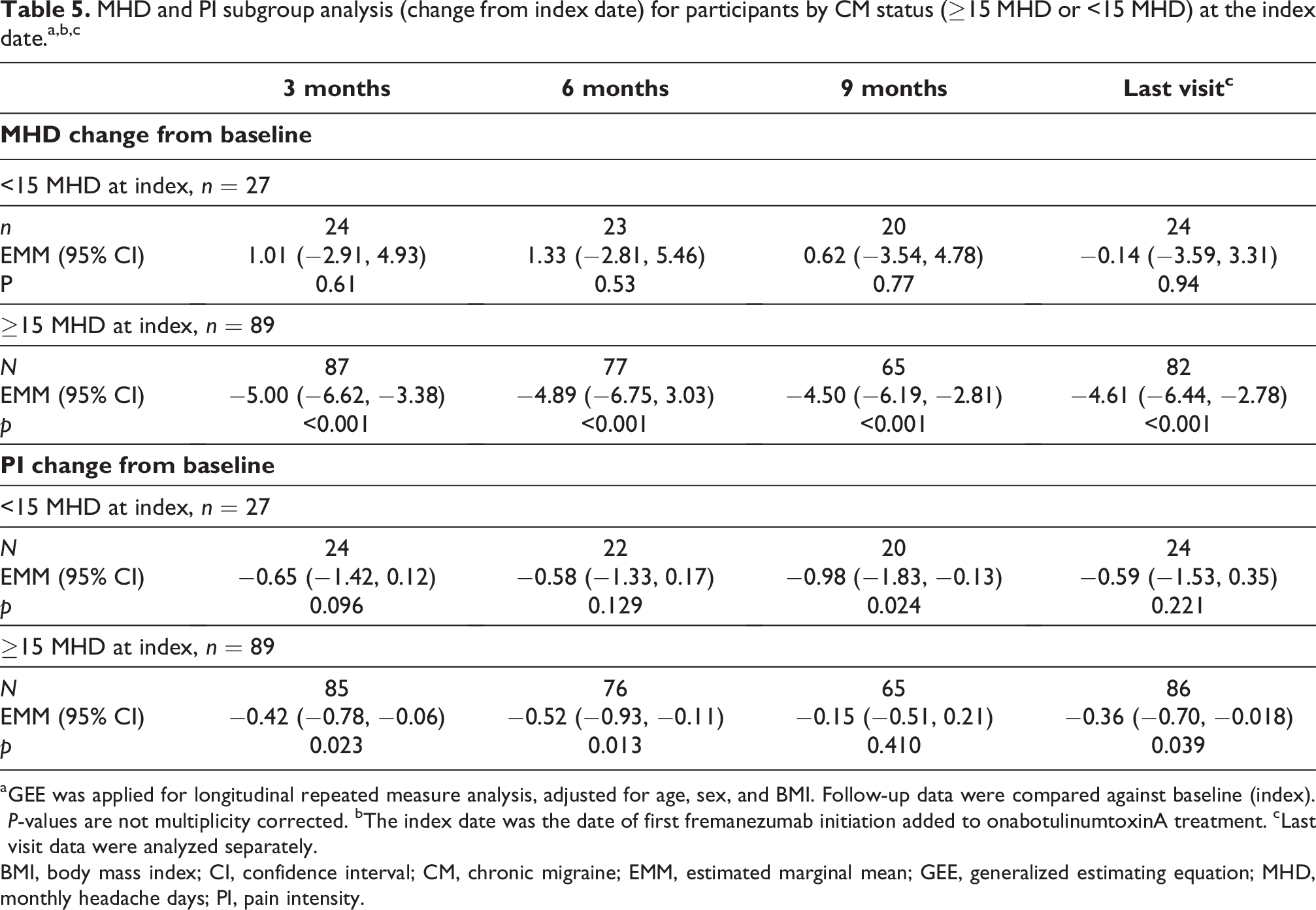
a GEE was applied for longitudinal repeated measure analysis, adjusted for age, sex, and BMI. Follow-up data were compared against baseline (index). P-values are not multiplicity corrected. bThe index date was the date of first fremanezumab initiation added to onabotulinumtoxinA treatment. cLast visit data were analyzed separately.
BMI, body mass index; CI, confidence interval; CM, chronic migraine; EMM, estimated marginal mean; GEE, generalized estimating equation; MHD, monthly headache days; PI, pain intensity.
Adverse events
In total, 19 AEs were recorded in the patients’ records following the initiation of fremanezumab. The most common events were injection site reactions, which are in line with the known safety profile of fremanezumab (Table 6). No serious AEs were recorded during the study period.
Adverse events reported by patients receiving concomitant onabotA and fremanezumab across the study period overall and within the first 3 months of treatment.
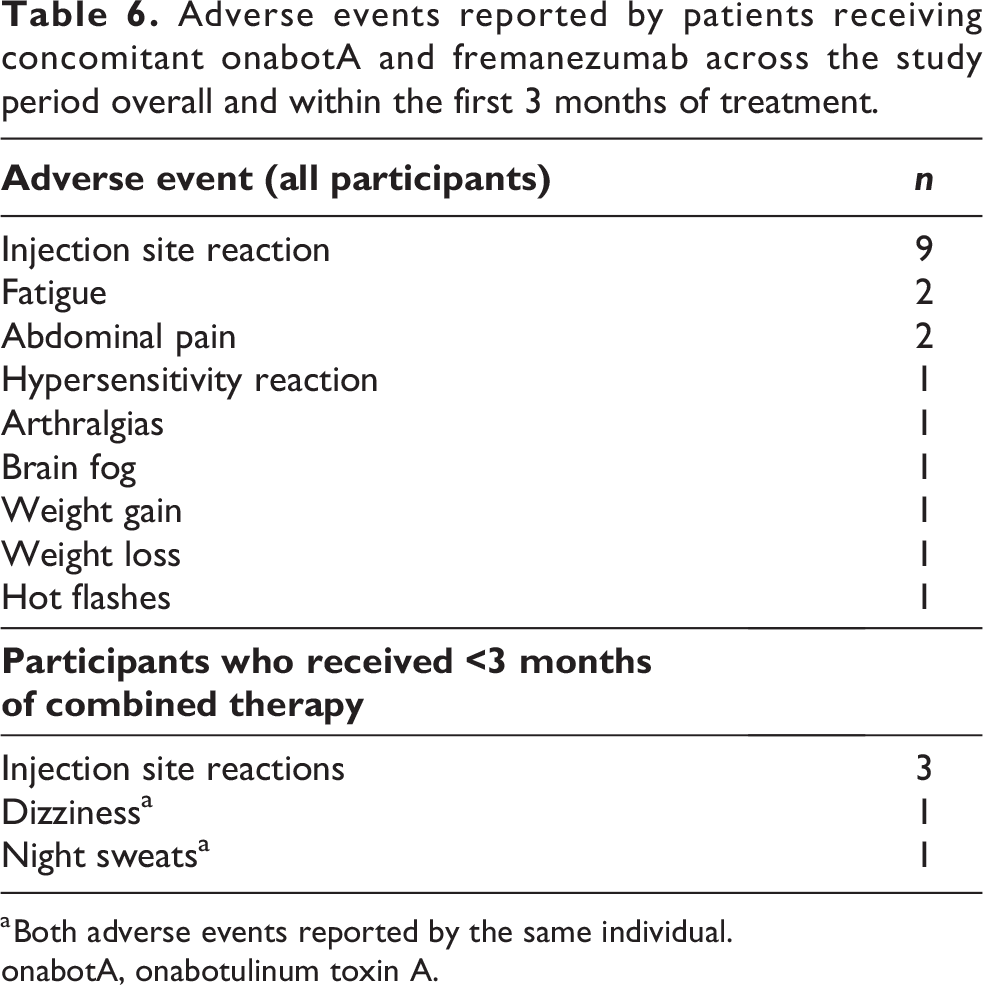
a Both adverse events reported by the same individual.
onabotA, onabotulinum toxin A.
Discussion
Overall, the results of this real-world study support the effectiveness and safety of concomitant onabotA and fremanezumab in the management of CM. A statistically significant difference was seen in reduced numbers of MHD and average PI at multiple time points compared with the index date. Subgroup analyses revealed that patients who were naïve to CGRP pathway mAb treatment, experienced non-daily HA, or had ≥15 MHD at the index date, reported statistically significant reductions in MHD. Those who had prior treatment with CGRP pathway mAbs or had non-daily HA at the index date also showed significant reductions in PI.
Although 155 units (U) of onabotA is the approved dose for CM management, 4 monotherapy is not always sufficient for adequate treatment of CM. Similar to our study, higher doses of onabotA (up to 195 U as per the PREEMPT studies 22 –24 ) are often used in real-world practice, and doses of 200 U might prevent CM more effectively than 155 U. 25 Adding other drugs with an alternative mechanism of action, such as those targeting the CGRP pathway, could offer clinical benefits to reduce suffering in these patients with CM. Several observational studies have determined that concomitant onabotA and CGRP pathway mAbs can improve clinical outcomes, mostly measured as the number of MHD. 16 –20,26,27 However, not all studies have demonstrated clinical benefits from concomitant CGRP pathway mAbs and onabotA, and some patients may derive more benefits than others, e.g. those who are partial responders to onabotA versus non-responders (reduction of MHD by between 30% and 50% versus less than 30% from baseline, respectively). 19,26,28 Outcomes of treatment with either fremanezumab alone or onabotA alone were not analyzed in this study, and therefore, comparisons between combination therapy and monotherapy were not made.
Both clinical trials and real world studies of onabotA with individual CGRP pathway mAbs have mostly included erenumab to date and have been of short duration (up to 6 months and mostly up to 3 months). 7,27 In our study, which is the largest and longest real-world assessment of fremanezumab and onabotA to date, we observed that most patients required the addition of fremanezumab due to inadequate response to onabotA. Where assessed, the reduction in MHD across these studies was broadly similar to those observed elsewhere. 16 –18,20 For example, a pooled analysis of eight studies showed that combined onabotA and any CGRP pathway mAb could lead to an average reduction of 2.67 MHD versus onabotA alone. 15 A retrospective medical record review by Cohen et al. demonstrated that fremanezumab added to onabotA reduced MHD by 8.8 days over onabotA alone after 2 to 3 months, more than either erenumab (5.5 days) or galcanezumab (5.3 days). 17 Data from studies of concomitant erenumab and onabotA suggest that peak effects of this modality on MHD may be in the first 3 months, whereas our data show persistent improvements with fremanezumab at 9 months after its initiation. 27
The JHC is a tertiary referral center providing care to a patient population with many types of headache disorders, most of whom have previously received outpatient and inpatient treatment at other centers without satisfactory response. Effectiveness in this population may translate into benefits in non-specialty settings to help expedite early headache prevention and consequent improvements to patients’ quality of living. 29 Our demonstration of effectiveness in patients with CM shows that concomitant fremanezumab and onabotA can help meet the currently unmet needs of patients with a high burden of CM. 30
The discontinuation rate in this study was considered favorable for a real-world study, with higher rates reported in other observational investigations of CM prevention. 31,32 The most commonly stated reasons for fremanezumab discontinuation were inadequate response to fremanezumab or lack of insurance coverage. However, most patients (over 75%) reported that overall treatment response with concomitant fremanezumab was the same as or better than onabotA alone. On the other hand, safety was not a common reason for discontinuation and AE reporting was low across the study. Anecdotal evidence and experience suggest that multiple preferences may influence the decisions of individual patients regarding continuing treatment. These include: each person’s experience and comfort with onabotA, preference for self-administration of fremanezumab versus onabotA injections administered by the clinician, and the potential for cosmetic effects of onabotA. Our results reflect those of a US claims database analysis showing that adherence to onabotA alone or concomitantly with CGRP pathway mAbs was three times greater than adherence to the index mAb administered concomitantly. 33 Other factors that could influence treatment decisions beyond patient choice include the evolving reimbursement landscape and the availability of other anti-CGRP or CGRP receptor mAbs. These factors are not routinely captured in EMRs so no firm conclusions can be drawn regarding reasons for discontinuation in this cohort of onabotA-experienced patients.
Our findings add to the growing evidence base for the clinical benefits of concomitant onabotA and an anti-CGRP or CGRP receptor mAbs to improve clinical outcomes, although medical insurance coverage was an acknowledged limitation to accessing fremanezumab for some patients in this setting. EMRs were typically not considered suitable for a cost and healthcare resource investigation, but it is reasonable to infer that effective migraine symptom prevention, particularly among sufferers of CM, might result in decreased access to healthcare services and resource use over time. 34 Evidence suggests that concomitant onabotA and CGRP pathway mAbs may be considered cost-effective. 35 Potentially, the greatest economic benefits will relate to reducing the burden of non-medical costs influencing work productivity. 29
Strengths and weaknesses
This is the first observational study of onabotA treatment with concomitant fremanezumab exclusively that applied longitudinal data analysis (GEE model). Other studies have investigated small numbers of patients with fremanezumab in the context of other CGRP pathway mAbs. It also has a long duration of follow-up compared to many other studies. Adopting a longitudinal study design including time points longer than 3 months, allows for the study of effectiveness in migraine sufferers who may want to add CGRP pathway mAbs to their preventive regimen per treatment guidelines. 3,5 Inevitably, there were some limitations to this study. Patients seen at JHC typically experience CM that is more difficult to treat than in other settings, so may not reflect those usually seen in other, non-specialty settings (e.g., primary care). Although an analysis of outcomes by dosing frequency (monthly versus quarterly) is desirable, there were insufficient data in this case. The study also did not consider outcome data for patients who discontinued fremanezumab; as a result, the study population may be enriched for patients that responded to treatment. This approach aligns with that used in other studies investigating real-world treatment responses. 20,36 In addition, this study did not report the duration of wash-out periods for those patients who switched from other CGRP pathway mAbs, such as erenumab, to fremanezumab. Although this reflects real-world clinical practice, in which wash-out periods are not typically observed, there is a need to be mindful of the carry-over effects from previous CGRP pathway mAbs when considering these data.
Using real-world evidence, data collection efforts are often limited by the scope of information routinely collected in medical charts, particularly those that relate to safety and the severity and duration of the impact of symptoms on individuals. Also, the accuracy of patient self-reported outcomes that is not necessarily verified by providers may introduce a degree of bias that is difficult to adjust for in such a setting. Additionally, study conditions were not controlled, so selection and reporting bias cannot be excluded, although this might also be considered a strength regarding applications of the findings in routine practice. The impact of the COVID-19 pandemic on data collection should also be noted. There was a higher proportion of patients with treatment delays from March 2020 to the end of the study due to the COVID-19 pandemic than previously seen. During this time, in-person visit duration was limited to adhere to social distancing protocols and minimize spread of infection. Although the JHC clinic typically favors patients seeing the same provider at each visit to ensure continuity of care, this was not always possible during the pandemic. During that period, the clinic continued to offer procedure visits for administering onabotA, but inevitably the pandemic resulted in unusual delays in contact, mostly due to cancellations by the patients or the clinic. More visits were conducted remotely via telemedicine during this period than is typical. While this facilitated a degree of follow-up contact, the virtual setting was subject to specific limitations, notably, the inability to collect vital signs or perform a complete physical examination. Finally, the study used data from a single tertiary headache clinic in the US, so the generalizability of results is limited, and other countries may not currently approve, or health systems reimburse, concomitant use of onabotA and CGRP pathway mAbs.
In conclusion, this real-world study provides data supporting the effectiveness and safety of the concomitant use of fremanezumab with onabotA for the prevention of CM that is difficult to treat. According to study results, patients with CM who were naïve to CGRP pathway mAbs, experiencing non-daily HA, or with frequent (≥15) MHD, may benefit most from dual treatment with fremanezumab and onabotA. Further study in large, multi-center, prospective trials is required to confirm these findings and help inform treatment selection and personalization protocols.
Clinical implications
In US clinical practice, combinations of onabotA and one of the mAbs targeting CGRP or its receptor are commonly used in the treatment of patients with difficult-to-treat CM.
Currently, evidence for the effectiveness and tolerability of concomitant fremanezumab with onabotA from real-world observational studies is scarce.
The current retrospective chart review described here shows that concomitant fremanezumab and onabotA helps achieve a statistically significant reduction in the number of MHD and PI in the full study population without unexpected adverse events.
The benefits of adding fremanezumab to onabotA in CM are most pronounced among patients naïve to mAbs targeting CGRP or its receptor, who have frequent (≥15) monthly headache days, or do not experience headache attacks every day.
This observational study is the largest evaluation of concomitant fremanezumab and onabotA to date and demonstrates that the combination is effective and well tolerated.
Footnotes
Acknowledgments
This study was funded by Teva Branded Pharmaceutical Products R&D, Inc. Medical writing support was provided by David Floyd on behalf of Ashfield MedComms, an Inizio company, and editorial support was provided by Olivia Morris of Ashfield MedComms, an Inizio company, funded by Teva Pharmaceuticals USA, Inc. The authors acknowledge the support of Kathryn Dent, Matthew Beucherie, and William Connolly, who were medical students at the Thomas Jefferson University at the time of the study and Dr Stephanie Nahas-Geiger, MD of the Jefferson Headache Center, for her initial work on the project.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Within the past 24 months, H. Yuan has received funding from NIH (R44NS115460); institutional support for serving as an investigator from Teva, Abbvie, Trillen, and Thermaquil; consultant fees from Silva, Clexio, and Pfizer; and royalties from Cambridge University Press and MedLink. F. Cohen and M. Hopkins have no conflicts of interest to report. M.T. Driessen, L.J. Krasenbaum, and M. Ortega are employees of Teva Pharmaceuticals. M.J. Marmura received compensation for providing consultation to Alder/Lundbeck, Axsome, Supernus, Satsuma, and Theranica; received speakers bureau fees from Eli Lilly and Amgen/Novartis; received institutional support for serving as principal investigator from Teva Pharmaceuticals, gammaCore, and Allergan/AbbVie; and received payments for authorship or royalties from Demos Medical, Cambridge University Press, and MedLink.
Ethical approval
The protocol is exempt from IRB review and no consent required.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
