Abstract
Background
Traumatic brain injury (TBI) commonly elicits acute (APTH) and/or persistent (PPTH) post-traumatic headache. Calcitonin gene related peptide (CGRP) has been implicated as a contributor to PTH pathophysiology. We explored the possibility of sexual dimorphism in the effects of CGRP receptor (CGRP-R) blockade in a preclinical model of PTH induced by a mild TBI (mTBI) in male or female mice.
Methods
Mice were lightly anesthetized and placed on a tissue paper stage prior to receiving a sham procedure or mTBI resulting from a closed-head weight drop injury. Behavioral responses to periorbital and hindpaw tactile (von Frey filaments) or thermal (hot plate) stimuli over the first 14 days post-mTBI were evaluated as measures of APTH. The PPTH phase was studied following the resolution of mTBI-induced APTH at days 14 and 28. PPTH was precipitated by exposure to bright lights (i.e., bright light stress, BLS). Olcegepant was delivered subcutaneously either repeatedly beginning 2 h after mTBI to produce a sustained block of CGRP-R signaling across the APTH phase, or as a single administration on days 14 or 28 post-mTBI to evaluate possible effects during the PPTH phase.
Results
mTBI, but not sham-procedure, produced periorbital and hindpaw tactile allodynia, as well as thermal hypersensitivity in mice of both sexes. APTH-related hypersensitivity was transient and resolved by day 14 post-injury. No sex differences were observed in the magnitude or duration of APTH-related pain behaviors. Sustained CGRP-R blockade was, however, significantly more effective in female than male mice in inhibiting pain behaviors in the APTH phase and in preventing the emergence of BLS-induced PPTH. CGRP-R blockade following the resolution of mTBI-induced APTH pain behaviors, on either day 14 or 28, minimally altered BLS-induced PPTH in either sex.
Conclusions
Sustained CGRP-R blockade starting soon after mTBI significantly inhibited APTH and prevented the expression of PPTH with greater analgesic effects in females compared to males. Delayed CGRP-R blockade beginning after resolution of APTH phase was minimally effective in preventing expression of PPTH in either sex. These data are consistent with previous observations that CGRP induces pain behaviors preferentially in females. Early and continuous CGRP blockade following mTBI may represent a viable treatment option for PTH treatment and the prevention of PTH persistence, especially in females.
This is a visual representation of the abstract.
Keywords
Introduction
Mild traumatic brain injury (mTBI), also known as concussion, is commonly associated with post-traumatic headache (PTH). PTH is a secondary headache that results from impacts to the head due to events such as a falls, motor vehicle collisions, assaults and sports (1–3). While PTH typically resolves over the first weeks to months following mTBI, 15–53% of individuals continue to experience PTH a year after mTBI (4–6). Patients who report headache amongst their initial symptoms of mTBI have a higher likelihood of having persistent symptoms than individuals who do not (7,8). PTH is classified as acute (APTH) or persistent (PPTH) depending on whether the headaches resolve or fail to resolve within three months, respectively.
The underlying pathophysiology driving PTH and its persistence remains unknown. PTH most commonly presents with a headache and associated symptoms (e.g., photophobia) that resemble migraine (9), but whether overlapping mechanisms can promote both PTH and migraine remains uncertain. Extensive evidence from clinical and preclinical studies has demonstrated a pivotal role of calcitonin gene-related peptide (CGRP) in the pathophysiology of migraine (see reviews by Tepper (10) and Edvinsson (11)), and CGRP has also been implicated in PTH (12,13). Preclinical studies have demonstrated that CGRP preferentially produces headache-like pain in female rodents (14,15), and increasing evidence supports sexual dimorphism in mechanisms promoting pain (14–26). Whether CGRP might contribute to PTH in a sexually dimorphic manner remains unknown. For that reason, we explored possible differences in the role of CGRP signaling in APTH-related pain and in promoting PPTH following mTBI in male or female mice.
We used a clinically relevant mTBI model characterized in our laboratories that replicates many of the biomechanical features associated with mTBI injuries. Lightly anesthetized mice were placed on a tissue paper stage and received a weight drop onto a closed-skull and unrestrained animal that imparts linear and rotational acceleration to the head and body (27–30). This method produces no observable radiologic, gross or histological brain damage, no skull fractures, minimal loss of consciousness, and no neurological deficits (27,28), similar to what is observed in humans following mTBI. As we have previously reported, the mTBI event produces transient periorbital and hindpaw cutaneous allodynia (CA) that indicate both peripheral and central sensitization and that was interpreted as modeling APTH. These initial pain behaviors resolve by day 14 post-mTBI whereafter mice display sensitivity to induction of pain behaviors by a second subthreshold trigger, including stress induced by bright light (i.e., bright light stress, BLS). Once developed, this sensitivity is long-lasting and persists for at least 67 days after a single mTBI (31). Our previous studies have shown that only mice with a previous mTBI event show BLS-induced periorbital and hindpaw CA that persists for a period of approximately 5 h (31–33). The emergence of long-lasting sensitivity to BLS-induced CA was interpreted as reflecting PPTH where a normally innocuous stimulus can induce pain behaviors during this sensitized state.
In the present study, we determined whether administration of a small molecule CGRP receptor (CGRP-R) antagonist would inhibit APTH pain behaviors, prevent stress-induced PPTH, and whether efficacy would be observed following the resolution of the APTH phase. Additionally, we expanded the assessment of outcome measures beyond tactile hypersensitivity indicative of CA to determine if increased gain would occur in other sensory modalities, and to assess if allodynia to thermal stimuli would be CGRP-dependent. Finally, we determined whether there were sex differences in the consequences of CGRP-R blockade on both APTH and PPTH pain behaviors.
Materials and methods
Animals
Male or female ICR adult mice (Jackson Laboratories) weighing 17–22 grams (six weeks-old) were housed five to a cage on a 14/10-h light/dark cycle (5 am-7 pm lights on) with food and water ad libitum. Experiments were conducted during the light cycle phase. The estrous cycle was not monitored in these studies. All experiments were performed in accordance with the ARRIVE reporting guidelines and with the approval of the Mayo Clinic Institutional Animal Care and Use Committee. A total of 231 mice were used in this study. Group size requirements to obtain significance at the α=0.05 and statistical power 0.9 were determined from previous experiments using power analysis. Mice were randomly divided into control and experimental groups and the experimenter was blinded to the treatments.
Induction of mild traumatic brain injury
The mouse closed-head weight drop injury model of experimental mTBI was adapted from Kane et al. (27), as reported previously by our group (14,30,32–35). Mice were lightly anesthetized with 5% (induction) and 2% (maintenance) isofluorane and then laid with their ventral surface on an elevated tissue paper stage capable of supporting body weight with the head unrestrained. The paper stage was situated over a plexiglass apparatus with a soft sponge at the bottom. A metal guide tube was directed to the top of the mouse skull between the ears to ensure standardized placement of the weighted drop. The weight (100 g), released from a height of 94 cm, results in a concussive impact to the head, pushing the mouse through the tissue paper and flipping it down to land on the soft sponge. All mTBI mice in this study experienced both rotational and linear forces to the head and body. After impact, the weight falls through the apparatus, avoiding a second impact with the animal. Following the procedure the mice were returned to their home cages and allowed to recover. Sham animals were anesthetized and placed on the tissue paper stage but did not undergo the weight drop or rotational flip. All mice awoke within 5 min of the procedure and were observed to confirm that no visual signs of neurological complications arose. Animals remained grouped in their same cohorts following the procedure.
Bright light stress (BLS) challenge
Unrestrained mice were exposed to BLS induced by LED strips (1000 lux output) that were placed on both sides of their home Plexiglass cages for 15 min as previously reported (14,32–34). The BLS protocol was designed to produce mild stress that arises from endogenous mechanisms and does not elicit significant CA in naïve or sham mice.
Drug administration
Olcegepant, a selective CGRP-R antagonist, was purchased from Tocris (Minneapolis, MN, USA) and dissolved using 20% DMSO in saline immediately before use. To optimize the administration regimen, the antagonist was administered subcutaneously (s.c.) in the flank: (a) at 1 mg/kg, 2 h after the mTBI or sham induction and daily up to day 14 after mTBI, referred as O1; (b) at 3 mg/kg, 2 h after the mTBI or sham induction and on days 3, 6, 9, and 12 after the induction, referred as O3. The antagonist was also administered at 3 mg/kg as a single injection 1 h before BLS on day 14 or 28 after mTBI or sham induction.
Behavioral assessment of tactile cutaneous allodynia
Prior to baseline behavioral assessment, mice were placed individually in elevated Plexiglass chambers with mesh flooring, located in a quiet area, and allowed to acclimate for 3 days for 2 h. Starting on day 0 (pre-mTBI baseline) and periodically thereafter, cephalic (periorbital) and extracephalic (hindpaw) cutaneous allodynia (CA) were measured in the same mice following a 2-h acclimation period. For assessment of periorbital CA, a 0.4 g (3.61) von Frey filament was applied with just enough force to cause the filament to display a slight arch to the periorbital region. This application was repeated every 20–30 s for a total of 10 times. A positive response was considered swiping of the face, shaking of the head, and/or turning away from the stimuli. Running away or rearing up were not considered positive responses. For assessment of hindpaw CA, a 0.6 g (3.84) von Frey filament was repeatedly applied (10 times total, once every 20–30 s) with just enough pressure to cause the filament to display a slight arch to the left hindpaw. Sharp withdrawal of the paw, shaking and/or licking the paw were considered a positive response, while lifting of the paw with the filament or running away were not. Frequency response was calculated as [(number of positive responses/10) * 100%] (14,23,32–35). The percent of analgesia used to evaluate possible sex differences was calculated as [AUC for each mTBI/olcegepant mice – average AUC for sham) ÷ (average AUC mTBI/vehicle – average AUC for sham) × 100]. Following mTBI induction, CA was measured for a time course of up to 14 or 28 days and in response to olcegepant or vehicle administration. Experiments with male or female mice were conducted on different days following thorough cleaning of experimental equipment. All CA behavioral data were collected by the same experimenter (J.O.).
Behavioral assessment of thermal allodynia
Thermal allodynia (TA) was assessed using the hot plate test (Columbus Instruments, Columbus, OH). Before baseline data collection, mice were acclimated to the equipment without heat being turned on. The plate was enclosed with round Plexiglas walls to prevent the animal from escaping. For testing, the plate temperature was set to 52°C, mice were individually removed from their home cages, placed in the center of the hot plate, and the latency to lick any of the hindpaws was assessed. Animals were removed from the plate immediately upon licking a hindpaw or if no response occurred within 30 s. All TA behavioral data were collected by the same experimenter (J.O.).
Optimization of sustained olcegepant treatment: Comparing daily vs. interval dosing
Two olcegepant dosing protocols, referred to here as O1 and O3, were tested in female mice to assess the effects of CGRP receptor blockade on behavioral outcomes. Mice were randomly separated into five groups: mTBI/control, mTBI/O1, mTBI/O3, sham/control and sham/O3. After baseline periorbital and hindpaw tactile measurements, mice underwent either the sham or mTBI procedure. The O1 protocol involved 14 s.c. doses of 1 mg/kg olcegepant, starting 2 h after mTBI and delivered daily thereafter. The O3 protocol included five doses, beginning 2 h after mTBI and followed by additional doses every three days (on days 3, 6, 9, and 12). Assessment of periorbital and hindpaw CA was performed periodically over a time course of 14 days. The mTBI/control group received vehicle control every three days following the O3 protocol. Two sham groups were given either olcegepant or vehicle according to the O3 protocol. As no differences were found between the sham groups, their data were combined for analysis and graphing.
Assessment of early and repeated CGRP-R antagonist treatment on the development of APTH and PPTH
Following baseline periorbital and hindpaw tactile and thermal measurements, mice underwent either the sham or mTBI protocol. Assessment of periorbital and hindpaw CA and TA were measured periodically over a time course of 14 days in different cohorts of female and male mice. The CGRP-R antagonist, olcegepant, was administered according to the protocol O3. On days 14 and 28, all mice were exposed to bright light stress (BLS) for 15 min, after which CA and TA were measured every hour for 5 h.
Assessment of delayed CGRP-R antagonist administration on the development of PPTH
After collecting periorbital and hindpaw CA baselines, mice underwent either sham or mTBI procedure. Periorbital and hindpaw CA were assessed periodically over 13 days to establish the return of sensory thresholds to pre-mTBI baselines. On day 14 and again on day 28, baseline measurements for periorbital and hindpaw CA were taken, animals received a single dose of either olcegepant or vehicle-control 1 h before the BLS. Mice were then exposed to BLS for 15 min, and CA was measured every hour for the subsequent 5 h. This experimental design resulted in three groups: mTBI/vehicle, mTBI/olcegepant, and sham (a combination of sham animals treated with vehicle-control or olcegepant).
Data analysis
Data are presented as the mean and standard deviation (SD). All statistical analyses were conducted in Graphpad Prism 9 (GraphPad Software, La Jolla; CA). Time courses were statistically analyzed using two-way repeated measures analysis of variance (ANOVA), performed with “time” as within subject factor to assess the main effects of time, treatment group, and the interaction between time and treatment on CA and TA. Tukey's post-hoc test for multiple comparisons was used to assess differences between the groups within each time point. Area under the curve (AUC) graphs were analyzed using one-way ANOVA with a Sidak's post-test and sex comparison of percent of analgesia was analyzed using Unpaired t-test. Statistical significance was established a priori at 95% (p < 0.05).
Results
Daily and interval dosing of olcegepant produced similar efficacy in preventing APTH in female mice
In mTBI mice receiving vehicle control, but not in the sham mice, CA developed in both the periorbital (Online Supplementary Figure 1a) and hindpaw (Online Supplementary Figure 1b) region beginning on day 1, peaking on day 3 and resolving by days 13–14 post-induction. Both daily (O1) and interval (O3) olcegepant treatment protocols effectively prevented the development of periorbital and hindpaw CA induced by mTBI (Online Supplementary Figure 1). Timecourses and AUC confirmed that there was no statistical difference between the O1 and O3 treatment protocols, evidenced by the similar efficacy on the blockade of allodynia in both periorbital and hindpaw regions. We therefore selected the O3 protocol for further study, which involves five intermittent doses of olcegepant starting 2 h after mTBI, with subsequent doses administered every three days.
Sexually dimorphic effect of early and repeated olcegepant treatment in cutaneous and thermal allodynia associated with APTH and PPTH
In vehicle treated female and male mice, mTBI induced a transient time-dependent development of periorbital (Figure 1(a) and 1(b)) and hindpaw CA (Figure 2(a) and 2(b)) or TA (Figure 3), demonstrated by the increased frequency of response after tactile stimulation and decrease of thermal latency, respectively. There were no sex differences in the development of either periorbital or hindpaw CA. Small sex differences were observed for TA likely due to differences in baselines. Repeated treatment with olcegepant reduced both periorbital and hindpaw CA, as well as TA in mice, with greater efficacy in females, evidenced by significantly higher percent of analgesia in comparison to males (Figures 1(c), 2(c) and 3(c)).
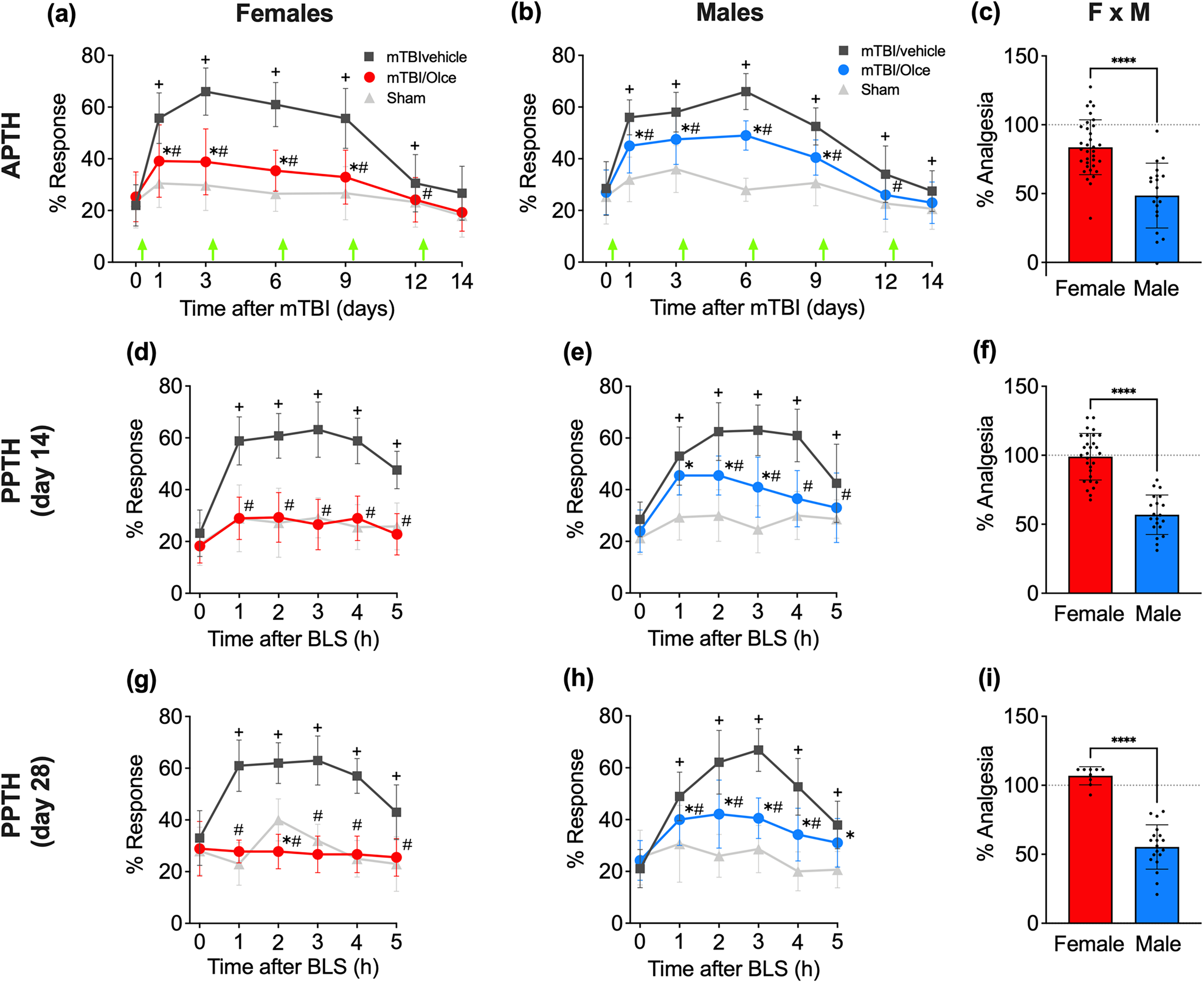
Repeated olcegepant treatment starting 2 h after a single mTBI is more effective for the management of APTH and PPTH in female than male mice. (a, b) After measuring baseline periorbital tactile responses, female (a) and male (b) mice were given a single mTBI or sham injury and were administered olcegepant or vehicle according to the O3 protocol (i.e., 2 h after the injury and readministered on days 3, 6, 9, and 12; green arrows). Periorbital allodynia as a measure of APTH was assessed over a time course of 14 days. (c) Percent of olcegepant-induced analgesia was calculated in female and male mice from the AUC of the 14-day time course (see Methods for details) to assess potential sex differences in olcegepant efficacy. (d, e) At day 14 after mTBI, all mice were exposed to BLS, and tactile allodynia was collected over a 5 h time course as a measure of PPTH. (f) Percent analgesia was estimated from the AUC of the 5-h time course to compare olcegepant efficacy in female and male mice. (g, h) At day 28 after mTBI, all mice were again exposed to BLS, and tactile allodynia was evaluated for 5 h. (f) Percent analgesia estimated from the AUC of the 5-h time course in female and male mice. (a, b, d, e, g, h) Two-way repeated measures ANOVA with Tukey's multiple comparison test. Significant differences (p < 0.05) between the mTBI/vehicle group and sham mice are indicated by a plus sign (+); differences between the mTBI/olce and sham groups by an asterisk (*) and differences between the mTBI/vehicle and mTBI/olce groups by a hashtag (#). (c, f, i) Unpaired t-test; **** p < 0.0001. Data are plotted as means ± SD. Statistical details are summarized in Online Supplementary Table 1.

Repeated olcegepant treatment starting 2 h after a single mTBI is more effective in relieving hindpaw allodynia associated with APTH and PPTH in female than male mice. The hindpaw data were collected from the same animals and in parallel with periorbital responses plotted in Figure 1. (a, b) Hindpaw allodynia in female (a) and male (b) mice over a time course of 14 days. (c) Percent of olcegepant induced analgesia in female and male mice calculated from the AUC of the 14-day time course in (a) and (b). (d, e) Tactile hindpaw allodynia induced by exposure to BLS on day 14 in female (d) and male (e) mice. (f) Percent analgesia estimated from the AUC of the 5-h time course in (d) and (e). (g, h) At day 28 after mTBI, all mice were again exposed to BLS, and tactile allodynia was evaluated for 5 h. (f) Percent analgesia estimated from the AUC of the 5-h time course in female and male mice. (a, b, d, e, g, h) Two-way repeated measures ANOVA with Tukey's multiple comparison test. Significant differences (p < 0.05) between the mTBI/vehicle group and sham mice are indicated by a plus sign (+); differences between the mTBI/olce and sham groups by an asterisk (*) and differences between the mTBI/vehicle and mTBI/olce groups by a hashtag (#). (c, f, i) Unpaired t-test; **** p < 0.0001. Data are plotted as means ± SD. Statistical details are summarized in Online Supplementary Table 1.

Olcegepant treatment is more effective in relieving mTBI-induced increased thermal hypersensitivity associated with APTH and PPTH in female than male mice. (a, b) Baseline paw withdrawal latency in the hot plate test were collected followed by a single mTBI or sham injury in female (a) and male (b) mice. Mice received olcegepant or vehicle treatment according to the O3 protocol (green arrows). Heat hypersensitivity was assessed over a time course of 14 days. (c) Percent of olcegepant-induced analgesia was calculated in female and male mice from the AUC of the 14-day time course (see Methods for details) to assess potential differences in olcegepant efficacy between sexes. (d, e) At day 14 after mTBI, all mice were exposed to BLS, and paw withdrawal latency was assessed over a 5-h time course. (f) Percent analgesia was estimated from the AUC of the 5-h time course to compare olcegepant efficacy in female and male mice. (g, h) On day 28 after mTBI, all mice were again exposed to BLS, and paw withdrawal latency was evaluated for 5 h. (f) Percent analgesia estimated from the AUC of the 5-h time course in female and male mice. (a, b, d, e, g, h) Two-way repeated measures ANOVA with Tukey's multiple comparison test. Significant differences (p < 0.05) between the mTBI/vehicle group and sham mice are indicated by a plus sign (+); differences between the mTBI/olce and sham groups by an asterisk (*) and differences between the mTBI/vehicle and mTBI/olce groups by a hashtag (#). (c, f, i) Unpaired t-test; **** p < 0.0001. Data are plotted as means ± SD. Statistical details are summarized in Online Supplementary Table 1.
On day 14, all mice were exposed to BLS for 15 min to assess reinstatement of periorbital and hindpaw CA (Figures 1(d), 1e, 2d and 2e) and TA (Figure 3(d) and 3(e)). Early and repeated injections of olcegepant prevented BLS stress-induced CA (Figures 1(d), 1e, 2d and 2e) and TA (Figure 3(d) and 3(e)) in both female and male mice as shown by the reversal of increased tactile response frequency and decreased thermal latency. Olcegepant analgesia was significantly greater in female mice compared to males (Figures 1(f), 2(f) and 3(f)).
Interestingly, the effects of early and repeated olcegepant treatment, as well as the observed sexual dimorphism in the efficacy, persisted even on day 28 post-mTBI induction, approximately 16 days after the cessation of treatment. On day 28, BLS reinstated periorbital and hindpaw CA (Figures 1 g, 1 h, 2 g and 2 h) and TA (Figure 3 g and 3 h) in mTBI female and male mice treated with vehicle. However, mTBI animals previous treated with olcegepant demonstrated significantly lower stress-induced CA and TA (Figures 1 g, 1 h, 2 g, 2 h, 3 g and 3 h). The olcegepant effect was notably more pronounced in females, as evidenced by a higher percent of analgesia observed in females compared to males (Figures 1(i), 2(i) and 3(i)).
Limited efficacy of delayed olcegepant treatment for preventing stress-induced Ca after PPTH development
Without any treatment following mTBI or sham induction, both female and male mTBI mice exhibited significant periorbital and hindpaw CA compared to sham animals on testing days 1–12, revealed by the increased frequency of response to tactile stimulation (Online Supplementary Figure 2a, 2b, 2c and 2d). The animals were then treated with either olcegepant or vehicle on day 14, and one hour after the treatment were exposed to BLS. In both vehicle- and olcegepant-treated mTBI female and male mice, BLS reinstated periorbital and hindpaw CA (Figure 4(a), 4(b), Online Supplementary Figure 3a and 3(b)). Olcegepant showed minimal to no efficacy in preventing the development of periorbital and hindpaw CA in either sex (Figure 4(a), 4(b), Online Supplementary Figure 3a and 3(b)). Only a few time points showed a slight difference between mTBI mice treated with vehicle versus olcegepant. A similar outcome was observed on day 28 after mTBI induction. BLS reinstated periorbital and hindpaw CA in female and male mTBI mice treated with vehicle (Figure 4(d), 4(e), Online Supplementary Figure 3d and 3(e)). Olcegepant showed negligible efficacy in preventing the development of periorbital and hindpaw CA in either female or male mice (Figure 4(d), 4(e), Online Supplementary Figure 3d and 3(e)). Additionally, no sex differences were observed with delayed treatment of olcegepant in mTBI mice, evidenced by comparable modest levels of analgesia in both female and male mice (Figure 4(c), 4(f), Online Supplementary Figures 3(f) and 3(c)).

After the establishment of PPTH, olcegepant has minimal to no efficacy for preventing BLS-induced allodynia in both sexes. Female and male mice were given a single mTBI or sham treatment and development of acute allodynia was confirmed (Online Supplementary Figure 4). (a, b) On day 14, mice were treated with olcegepant or vehicle 1 h before BLS exposure. Periorbital tactile responses were assessed in female (a) and male (b) mice over a 6 h time course after BLS. (c) Percent of olcegepant-induced analgesia was calculated from the AUC of the 6-h time course (see Methods for details) to assess olcegepant efficacy in both sexes. (d, e) At day 28 after mTBI, mice received the same treatment with olcegepant or vehicle as assigned on day 14 and were again exposed to BLS. Tactile allodynia was evaluated for 6 h. (f) Percent analgesia estimated from the AUC of the 6-h time course in female and male mice. (a, b, d, e) Two-way repeated measures ANOVA with Tukey's multiple comparison test. Significant differences (p < 0.05) between the mTBI/vehicle group and sham mice are indicated by a plus sign (+); differences between mTBI/olce and sham groups by an asterisk (*) and differences between the mTBI/vehicle and mTBI/olce groups by a hashtag (#). (c, f) Unpaired t-test; ns = not significant. Data are plotted as means ± SD. Details of statistical analyses are summarized in Online Supplementary Table 1.
Discussion
We explored the potential sexually dimorphic role of CGRP signaling in APTH and PPTH following mTBI in mice. First, we observed that mTBI produced generalized transient periorbital and hindpaw CA, suggesting APTH-related pain likely reflecting both peripheral and central sensitization. Notably, after the initial CA has resolved, stress, one of the most common triggers for headache, reinstated the periorbital and hindpaw CA only in mTBI mice, suggesting persistent central sensitization and susceptibility to PPTH attacks. Second, early and sustained CGRP-receptor antagonism effectively prevented both mTBI-induced APTH and stress-induced PPTH, with greater efficacy observed in female mice. These data highlight a sexually dimorphic role of CGRP in APTH and transition to PPTH induced by mTBI. Third, the assessment of TA in response to heat stimuli following mTBI demonstrated a similar behavioral and temporal effect as CA. In addition to CA, BLS also triggered TA in animals with mTBI, indicating that increased sensitivity extends beyond the tactile modality. The onset and chronification of TA following mTBI also appear to depend on CGRP, with this effect being more pronounced in females. Fourth, at the PPTH phase, which likely reflects established central sensitization, CGRP-receptor antagonism showed limited to no effect in alleviating PPTH-related pain behavior in either sex. Altogether, these results suggest that early and sustained CGRP-targeted interventions following mTBI could be beneficial in preventing and managing APTH and PPTH, particularly in females.
Currently, there are no medications specifically approved for the treatment of APTH or prevention of PPTH (36–40). Most patients with PTH experience frequent headache episodes consisting of symptoms that closely resemble migraine (36–41). As a result, current treatment strategies are primarily adapted from pharmacological therapies shown to be effective for migraine (13,36–41). The precise localization of peripheral CGRP contribution to migraine attacks remains unclear. It is well documented that CGRP signaling in vascular smooth muscle cells elevates intracellular cyclic adenosine monophosphate (cAMP) levels, leading to vasodilation of blood vessels in the intracranial meninges (see reviews by Ashina et al. (37,38)). Another study revealed that the nociceptive CGRP-signalling between C- and Aδ fibers may be mediated via Schwann cells (42). Additionally, CGRP has been implicated in mast cell degranulation and in the direct activation and sensitization of peripheral neurons within the trigeminal system, including those in the dura mater (43,44). Recent clinical and preclinical studies have also supported involvement of this peptide in PTH (13,33,35,41,45,46). A randomized, double-blind, placebo-controlled, two-way crossover study provided evidence that intravenous infusion of CGRP induces headaches with migraine-like phenotypes in patients with persistent PTH, even in those without a history of pre-existing migraine (45). Increased CGRP immunoreactivity within the trigeminal system was observed in a model of TBI in male mice (12). Our group and others have previously demonstrated that continuous early sequestration of CGRP with a monoclonal antibody prevented both APTH and PPTH (13,33), as well as the loss of descending control of nociception, a correlate of diffuse noxious inhibitory controls (DNIC), following mTBI in male mice (35). Systemic treatment with fremanezumab at 2 h and again on days 7 and 14 after mTBI fully prevented the development of APTH and PPTH revealed by BLS in male mice (33) or by systemic administration of nitroglycerin in male rats (13). Systemic fremanezumab treatment at 2 h post mTBI also fully prevented the loss of DNIC in male mice (35). In contrast, delayed anti-CGRP mAb treatment following establishment of central sensitization was ineffective in preventing PPTH (33). Those studies, however, did not evaluate potential effects in female animals, precluding potential conclusions about possible sex differences in the efficacy of anti-CGRP mAb treatment. Herein, we observed that early and sustained administration of a small molecule CGRP-R antagonist also significantly reduced APTH and the subsequent expression of PPTH in both female and male mice.
Sex is an established biological variable in pain conditions, with both human and preclinical evidence demontrating sexually dimorphic mechanisms promoting pain (14–26). Although males experience mTBI more frequently than females, the incidence of PTH after mTBI is higher in females (47,48), which suggests the potential for mechanistic (i.e., qualitative) sex differences in PTH pathophysiology. In the present study, we did not observe any meaningful sexual differences in the magnitude and duration of mTBI-induced APTH and PPTH. We note, however, that a previous study reported that female rats exhibit a prolonged state of cephalic hyperalgesia and greater sensitivity to headache triggers compared to males in a head-fixed model of mTBI (49). This discrepancy may be attributed to the animal model of mTBI and species used in these studies.
Previous reports have indicated that dural application of CGRP elicits female-selective pain-like responses in rodents (14), suggesting that CGRP may be more efficient at eliciting migraine headaches in women. These findings are consistent with clinical data demonstrating the effectiveness of small-molecule CGRP receptor antagonists in the treatment of acute migraine in women while at present, no data support efficacy of CGRP-R antagonists for the treatment of acute migraine in men (50,51). We note that in contrast to acute (i.e., abortive) treatment, prevention therapies with small molecule CGRP-R antagonists or CGRP-targeting monoclonal antibodies are clinically effective in both men and women (51) possibly reflecting different roles of CGRP in patients with varying frequencies of migraine attacks. Interestingly, despite reports of female-selective pain behaviors induced by CGRP (14) and the greater PTH incidence in females following mTBI (47,48), previous studies evaluating the efficacy of anti-CGRP therapy have almost exclusively used male mice (13,33,35). Consequently, whether CGRP may play a more prominent role in mTBI-induced APTH and PPTH in female animals has not yet been thoroughly evaluated. Our study demonstrated that treatment with olcegepant, administered 2 h following mTBI and then every three days, effectively prevented the development of APTH and PPTH in both female and male mice. However, we observed that olcegepant treatment had greater analgesic actions in female mice.
In contrast to our findings, previous studies using a fixed head weight-drop model TBI in rats reported reduced effectiveness of an anti-CGRP mAb against periorbital CA in females compared to males (49). We note that the fixed head weight-drop model used in that study did not allow for contributions of linear and rotational forces typical of closed head impact injuries and produced only periorbital but not hindpaw CA (13,33,35,49). In contrast, the mTBI model used in the present study allowed both linear and rotational head movement during the weight drop, resulting in the development of both periorbital and hindpaw CA following mTBI, suggesting the development of central sensitization states, which might mimic human conditions more closely. These methodological differences may account for the discrepancies between our findings. Consistent with previous preclinical studies using different models of TBI (52,53) we also observed transient thermal allodynia that could be reinstated by stress, supporting the development of a centrally sensitized state by the mTBI. Earlier and sustained treatment with olcegepant also prevents the development and the reinstatement of TA in mice, with greater efficacy in females, suggesting that CGRP-dependent mechanisms may play a role in both APTH and the progression to PPTH states across different pain modalities, with notable differences between sexes.
On the other hand, once central sensitization was established, the current and previous studies (33) revealed that CGRP signaling did not appear to play a significant role in sustaining PPTH-related pain behavior in either sex. Patients with persistent post-concussion symptoms (most of whom had PPTH), especially females, have been reported to have five times higher circulating levels of CGRP than healthy controls (54). Notably, elevated CGRP levels were observed primarily at earlier stages of PPTH, approximately 4 months after concussion, with a subsequent decrease at later stages, around 11 months post-concussion (54). These findings align with animal studies that showed increased CGRP levels in female compared to male rats after mTBI (49,55). Lower levels of circulating CGRP in PPTH patients compared to healthy controls have also been observed at late time points, i.e., approximately 49 months after concussion (56). Together, these findings support the idea that CGRP may play a role in establishment of PTH, but not in maintaining mTBI-induced central sensitization associated with PPTH. It seems likely that once central sensitization is established (i.e., PPTH), CGRP-dependent mechanisms might become less prominent while CGRP-independent mechanisms become more prominent. In this regard, a single-center, non-randomized, single-arm, open-label study performed in adults aged 18–65 with established PPTH demonstrated that the anti-CGRP receptor monoclonal antibody, erenumab, offered only limited therapeutic benefits in preventing headache attacks (57). Moreover, in a Phase 2 randomized placebo-controlled trial, fremanezumab, an anti-CGRP monoclonal antibody, failed to achive its primary or any of the prespecified secondary endpoints in patients with PPTH (58). In conclusion, the results of this study increase understanding of the mechanisms involved in mTBI-related PTH and highlight a significant sex difference in the role of CGRP. Our findings support the conclusion that CGRP-dependent mechanisms initiate and promote both APTH and the progression to persistent states that can be revealed with multiple pain modalities, including mechanical and thermal stimulation. Our data also support the conclusion that once a state of central sensitization has been established, CGRP does not directly contribute to stress-induced headache-like pain, supporting the involvement of CGRP-independent mechanisms. These data provide insights into the possible use and timing of anti-CGRP therapy for treating APTH and preventing the onset of PPTH in patients with mTBI. Therefore, therapies targeting CGRP signaling may potentially address some of the unmet treatment needs of individuals who are at risk of developing PPTH after mTBI, while the CGRP-independent mechanisms associated with PTH remain to be elucidated. Importantly, the sexually dimorphic actions of CGRP in PTH revealed in this study indicate that early and sustained targeting of CGRP signaling might provide greater benefits in treating APTH and preventing the development of PPTH in females. These findings underscore the importance of consideration of patient sex in the treatment of PTH as well as for future research and clinical trials. Recognizing and leveraging these differences in pain mechanisms could lead to more effective and personalized therapies for PTH following mTBI.
Article highlights
A mouse model of mild traumatic brain injury (mTBI) produced generalized periorbital and hindpaw cutaneous allodynia as well as thermal hypersensitivity, which suggest central sensitization in promoting multiple PTH-related pain modalities. Following the resolution of mTBI-induced pain, stress triggered cutaneous and thermal allodynia only in previously injured mice, suggesting sustained central sensitization and increased vulnerability to PPTH. Sustained blockade of CGRP signaling with olcegepant prevented mTBI-related APTH as well as stress-induced PPTH more effectively in female than in male mice. CGRP-receptor antagonism after the establishment of central sensitization was minimally effective in blocking PPTH-related pain in either sex. Early and sustained targeted intervention following mTBI with CGRP antagonism may prevent debilitating PTH and its persistence, particularly in females.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251321087 - Supplemental material for Sex differences in effectiveness of CGRP receptor antagonism for treatment of acute and persistent headache-like pain in a mouse model of mild traumatic brain injury
Supplemental material, sj-docx-1-cep-10.1177_03331024251321087 for Sex differences in effectiveness of CGRP receptor antagonism for treatment of acute and persistent headache-like pain in a mouse model of mild traumatic brain injury by Edita Navratilova, Caroline M. Kopruszinski, Janice Oyarzo, Kara R. Barber, Trent Anderson, David W. Dodick, Todd J. Schwedt and Frank Porreca in Cephalalgia
Footnotes
Acknowledgments
We kindly thank Dr Xu Yue for the technical support.
Author contributions
E.N. and F.P. conceived and designed the study. J.O. collected the data. E.N. analyzed the data. C.M.K., E.N. and F.P. drafted the manuscript. E.N., and C.M.K. prepared the figures. E.N., C.M.K., J.O, K.R.B., T.A., T.J.S., D.W.D. and F.P read, revised and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.N., C.M.K., J.O., K.R.B., and T.A. declare that they have no personal, financial, or relational conflicts of interest with this work. D.W.D. reports the following conflicts within the past 12 months: Consulting: AEON, Amgen, Clexio, Cerecin, Ctrl M, Allergan, Alder, Biohaven, Linpharma, Lundbeck, Promius, Eli Lilly, eNeura, Novartis, Impel, Satsuma, Theranica, Vedanta, WL Gore, Nocira, XoC, Zosano, Upjohn (Division of Pfizer), Pieris, Revance, Equinox. Honoraria: CME Outfitters, Curry Rockefeller Group, DeepBench, Global Access Meetings, KLJ Associates, Academy for Continued Healthcare Learning, Majallin LLC, Medlogix Communications, MJH Lifesciences, Miller Medical Communications, Southern Headache Society (MAHEC), WebMD Health/Medscape, Wolters Kluwer, Oxford University Press, Cambridge University Press. Research Support: Department of Defense, National Institutes of Health, Henry Jackson Foundation, Sperling Foundation, American Migraine Foundation, Patient Centered Outcomes Research Institute (PCORI). Stock Options/Shareholder/Patents/Board of Directors: Ctrl M (options), Aural analytics (options), ExSano (options), Palion (options), Healint (Options), Theranica (Options), Second Opinion/Mobile Health (Options), Epien (Options/Board), Nocira (options), Matterhorn (Shares/Board), Ontologics (Shares/Board), King-Devick Technologies (Options/Board), Precon Health (Options/Board), Axon Therapeutics (Options/Board). Patent 17189376.1-1466:vTitle: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis. Within the past 24 months, T.J.S. has received compensation for consulting with AbbVie, Amgen, Eli Lilly, Linpharma, Lundbeck, Scilex, and Theranica and royalties from Up To Date. He has held stock options in Aural Analytics and Nocira. Research support has been received from the American Heart Association, Henry Jackson Foundation, National Headache Foundation, National Institutes of Health, Patient Centered Outcomes Research Institute, Pfizer, Spark Neuro, and United States Department of Defense. F.P. has served as a consultant or received research funding from Voyager, SiteOne Therapeutics, Nektar, Amgen, Acadia, Blackthorn, Teva, Eli Lilly, Hoba, Allergan, Ipsen, and Proximagen and is a founder of Catalina Pharmaa and Axon Therapeutics.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant W81XWH-19-1-0534 from the United States Department of Defense to Todd Schwedt, Edita Navratilova, Trent Anderson and Frank Porreca.
Data availability
All data are available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
