Abstract
Background:
Post-traumatic headache (PTH) is persistent and highly disabling. Cognitive-behavioral therapy for headache (CBT-HA) reduces headache frequency and severity and improves people’s quality of life, yet it is underutilized and inaccessible to many. Leveraging technology to deliver evidence-based psychological treatments for headache may address barriers to treatment engagement.
Methods/design:
This single-arm, single-site pilot trial aims to test the feasibility, acceptability, clinical signal, and cost of a five-session CBT-HA intervention delivered via interactive voice response technology (IVR). Participants will include 35 Veterans with PTH receiving care within VA Connecticut Healthcare System. Participants will complete an intake interview and a 9-item, 30-day electronic headache diary during a baseline run-in period. The same diary will be done again by participants immediately after treatment completion. Following the baseline assessment period, eligible participants will receive CBT-HA via IVR for 10 weeks, including an automated daily assessment of patient-reported outcomes and retrieval of biweekly tailored feedback from a study therapist. In addition, participants will access an electronic patient workbook, and study therapists will visualize patient-reported data through a secure provider dashboard. Participants will complete validated and reliable assessment measures at baseline, immediately post-treatment completion (week 10), and 1-month post-treatment completion (week 14). The primary clinical signal outcome is the change in self-reported headache days from the 30-day baseline run-in period before treatment (weeks −4 to 0) to the 30-day post-treatment completion (weeks 10–14). Paired-samples t-tests will explore changes in outcomes from baseline. All cost analyses will be exploratory and will use micro-costing techniques.
Trial Registration:
Clinical Trials.gov: NCT05093556. Registered October 26, 2021.
Introduction
Over 450,000 United States service members sustained a traumatic brain injury (TBI) between 2000 and 2021. 1 Military personnel and Veterans are at increased risk of developing post-traumatic headache (PTH), a secondary headache disorder, following mild TBI, 2 –7 which is often persistent and highly disabling. 8 It is challenging to treat PTH, given its heterogeneous symptom presentation and associated medical and psychiatric comorbidities. 9
Available medical therapies for headache disorders are often underutilized. 10,11 Many individuals do not respond to medical headache treatments, experience unwanted side effects, have difficulties adhering to their prescribed medical regimen, 12 –14 or prefer non-pharmacological treatments. 15,16 More practitioners are embracing the biopsychosocial framework as part of comprehensive, multidisciplinary, headache care. 17 Consequently, patients are increasingly encouraged to prevent and ameliorate symptomatology through pharmacologic and non-pharmacologic treatment, which is especially relevant in people with PTH given the complexities of the disease. 7
Psychological therapies for headache are evidence-based interventions 18 that aim to reduce the frequency and severity of headache attacks and improve an individual’s quality of life. Research shows clinically meaningful benefits of psychological therapies, with moderate effect sizes, when given alone or combined with pharmacotherapy. 19 Despite decades of research supporting the effectiveness of psychological treatments for headache, they remain underutilized and inaccessible to many patients with headache. Common patient barriers to engagement include trepidation surrounding effectiveness, time commitment, cost, and difficulties accessing providers with specialized training. 20 Ultimately, follow-through on referrals for psychological treatment for headache management remains poor. 21
There is burgeoning interest in examining the delivery of evidence-based treatments for headache management via technology 22 and mobile health as an alternate means of delivery to traditional in-person care. 23 To date, one study provided support for the feasibility and acceptability of the delivery of progressive muscle relaxation via a smartphone-based application for patients with migraine within a tertiary headache center. 24 However, this study was limited to only one component of psychological treatment for headache, and providers did not tailor treatment plans to individual participants, as is common in traditional in-person care. Consequently, additional research examining technology-delivered evidence-based psychological interventions is warranted.
There is growing evidence supporting the effectiveness of CBT delivered via telehealth and internet-based modalities for depression 25,26 and post-traumatic stress disorder. 27 Interactive Voice Response (IVR) is an automated telephone-based technology where people receive tailored messaging, track symptoms, target behavior change, and engage in disease self-management. 28,29 Delivery of cognitive-behavioral therapy via IVR technology was a non-inferior alternative to traditional in-person CBT delivery for patients with chronic low back pain. 30
Navigating life with a headache disorder can be daunting for patients, especially Veterans who routinely present with complex medical and psychiatric comorbidities and have limited access to providers with expertise in headache management. Delivery of traditional evidence-based psychological treatments for headache management through IVR holds promise to increase much-needed access to these treatments. Patients may be able to receive treatment at a time that is most convenient for them and not simply when therapists are available.
This current study is a single-arm, single-site, pilot clinical trial (iCHART—Interactive CBT for Headache And Relaxation Training) that aims to test a five-session Cognitive-Behavioral Therapy for Headache (CBT-HA) intervention delivered via interactive voice response technology (IVR). Participants will complete a 30-day run-in monitoring period to establish baseline headache days and ensure they meet study eligibility criteria. Follow-up assessments will be conducted immediately (week 10) and 1 month (week 14) post-treatment completion. We will compare changes from pre- to post-treatment for primary, secondary, and tertiary outcomes.
Methods
Study design
A single-arm, single-site, pilot clinical trial with a 30-day run-in period, 10-week treatment period, and follow-up assessments at week 10 and week 14 (see Figure 1). The primary clinical outcome is the change in self-reported headache days from the 30-day baseline run-in period prior to treatment to the 30-days post-treatment completion.

Study procedures from enrollment to follow-up period.
Study cohorts and participants
Eligibility
Participants will be 35 Veteran patients receiving care from VA Connecticut Healthcare System (VACHS), with an International Classification of Headache Disorders Third Edition (ICHD-3) diagnosis of PTH. Eligibility criteria include individuals who are (1) English speaking; (2) a Veteran at least 18 years of age; (3) not receiving hospice or palliative care; and; (4) have access to a touchtone telephone and computer/tablet; (5) lack of sensory deficits that would impair participation in the IVR telephone calls; (6) a diagnosis of an ICHD-3 PTH diagnosis per electronic headache record (EHR) ICD-10 codes and verified by the PTH screener; (7) a frequency of ≥4 headache days per month (as confirmed following completion of the baseline headache diary assessment period); (8) a primary pain complaint of headache per the participant; (9) an absence of significant cognitive impairment based on EHR ICD10 diagnosis of mild cognitive impairment, Dementia or Alzheimer’s Dementia, and confirmed by the Short Portable Mental Status Questionnaire (SPMSQ) 34 cognitive screener during the initial baseline assessment; (10) an absence of inpatient psychiatric hospitalization for any psychiatric or substance use diagnosis in the past 30 days; (11) an absence of active psychotic symptoms, suicidality, or severe depressive symptoms (PHQ score >20) per patient report and/or EHR notes; and (12) an absence of suicidal and/or homicidal ideation in the past 6 months.
We intend to recruit both men and women Veterans from racially, ethnically, and socioeconomically diverse groups who are at least 18 years of age. Participants will be eligible to receive a total of $50 for study participation; $20 following completion of the baseline intake and $30 for the final assessments at 1-month post-treatment completion.
Study description and procedures
Recruitment
Participants will be referred to the study from clinics within VACHS which treat a high volume of headache disorders (neurology, polytrauma, primary care, and women’s clinic), through self-referral, or via the Veterans Health Administration’s (VHA) Headache Data cohort (Figure 2). 35 The VHA constructed the Headache Data cohort using VHA electronic clinical and administrative data to identify all Veterans with a headache diagnosis receiving care in a VA facility.

Study recruitment, enrollment, and treatment procedures.
Enrollment
The study team will conduct a chart review of eligible participants’ EHR and then contact participants via phone to confirm study eligibility. Study team members will obtain verbal informed consent from all eligible participants.
Cognitive and headache diagnosis screener
Following consent, study staff will complete an additional screener to ensure participants are English speaking, interested in participating, have access to the required technology, lack sensory deficits, have a primary pain complaint of headache, do not have severe depression, and deny suicidal and/or homicidal ideation. The Short Portable Mental Status Questionnaire (SPMSQ) 34 will screen for severe cognitive impairment that may interfere with study engagement. The PTH screener will be used to verify that the participant meets the ICD-10 criteria for PTH at the time of screening.
Headache diary
After completing the initial intake interview, eligible participants will complete a 9-item daily headache diary, via the VA Annie App, 36 for 30-days during the baseline monitoring period. They will also repeat the same 30-day headache diary immediately after treatment completion. The VA Annie App is a short-messaging system (SMS) where participants will receive daily automated text messages to gather patient-reported data about their headache symptoms and headache interference. Participants who do not respond to the initial SMS message sent via the Annie App will receive the same message twice more (20 minutes apart). To be considered eligible for the study, participants will be required to complete at least 28/30 days of the baseline headache diary. The Guidelines for Trials of Behavioral Treatments for Recurrent Headache 37 strongly recommends a minimum of 4 weeks for baseline and outcome assessment using headache diaries.
Semi-structured baseline interview
For participants who remain eligible for study participation after the 30-day baseline diary, study staff will complete semi-structured baseline interviews to determine final study eligibility. The interviews will cover participants’ demographic information, experience with headache, headache symptoms, healthcare utilization, past and present coping strategies, medications used for headache, confidence in managing headache symptoms, and satisfaction with current headache care.
Assessments
At baseline (after the 30-day headache diary run-in period), participants will complete validated assessment measures (Table 1) electronically through Qualtrics, a web-based, HIPAA compliant, survey tool that allows for data collection. Participants will complete assessments examining disability, quality of life, anxiety, depression, post-traumatic stress symptoms, insomnia, headache pain catastrophizing, and headache management self-efficacy at week 10 (post-treatment) and at week 14 (1-month post-treatment). Participants will complete measures of feasibility and acceptability at week 10 (post-treatment). Participants will be given a 10-day grace period to complete assessment measures, after that we will consider that data missing. Participants will complete the previously described 30-day headache diary for weeks 10–14.
Study outcome measures.

Intervention components
Patient workbooks
Participants will receive an electronic patient workbook, an adapted version of the VHA Cognitive-Behavioral Therapy for Headache (CBT-HA) manual 38 written at the sixth-grade reading level or below (see Table 2 and Figure 3). The intervention is entirely asynchronous, therefore using a website appears to be the most efficient and user-friendly way to provide the study materials. Treatment will cover the following five headache self-management skills: (1) relaxation training; (2) contributing factors (stress, sleep, hydration/nutrition, substances); (3) early warning headache sign identification and acute management strategies; (4) combating unhelpful thoughts (identification of unhelpful thinking patterns, coping statements, and cognitive restructuring); (5) planning ahead (skill consolidation and creating a headache self-management plan including both preventive and acute strategies). As participants progress through the study, they will be granted access to new headache self-management skills on the website. Each chapter of the participant workbook covers a new headache self-management skill. The introductory chapter provides an overview of the study, information about how the IVR system works, a copy of the daily and biweekly IVR questions the participant should expect to receive, a rationale for psychological interventions for headache management, an introduction to goal setting, and contact information for crisis support services.
Skill and topic content of intervention.

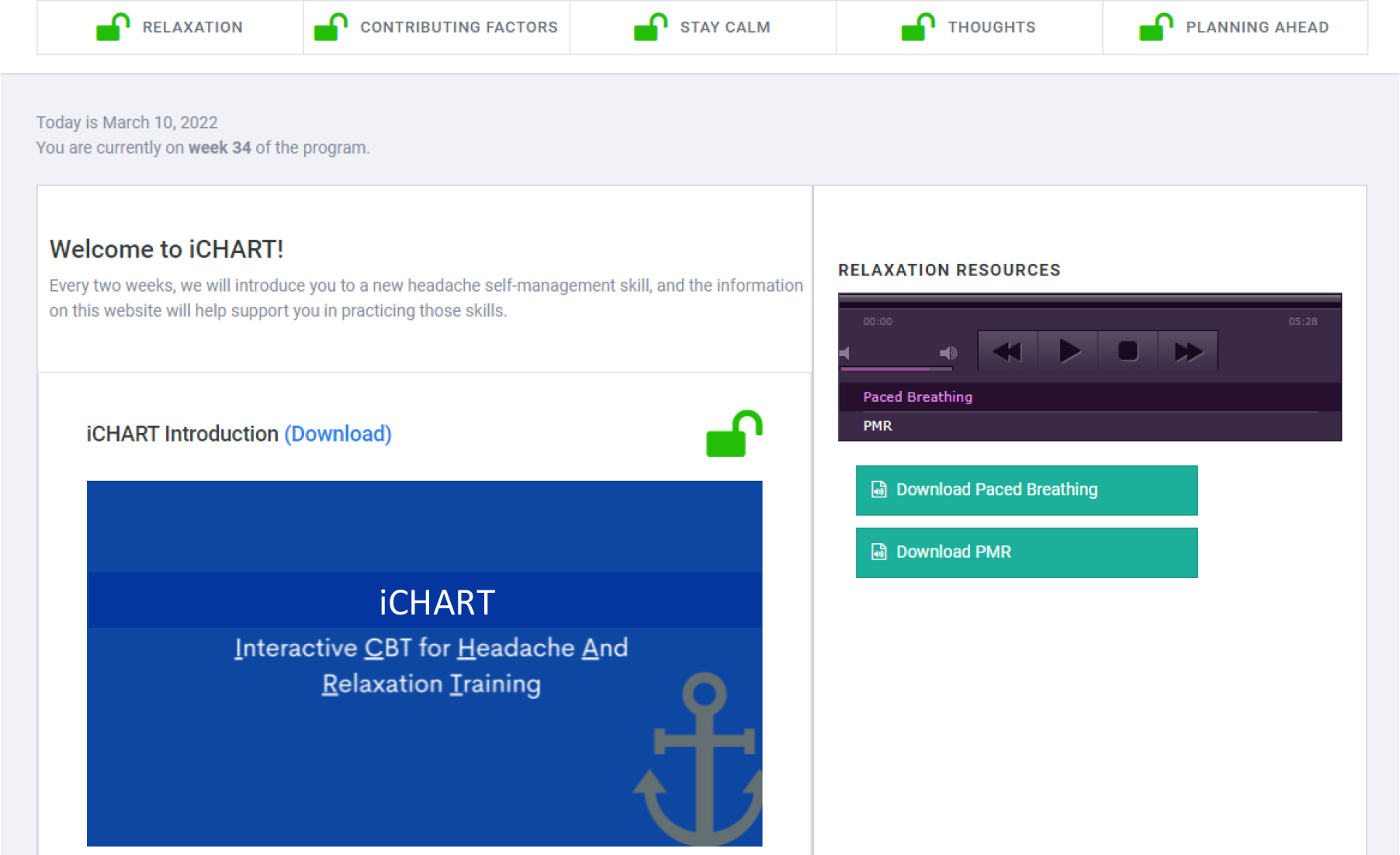
Example of patient-facing website housing patient workbook.
Study team members will be able to verify that participants have accessed the patient workbook materials via the provider-facing dashboard. At the end of each topic, participants will be asked a series of five true/false questions through the IVR system to assess their comprehension of the patient workbook contents. Participants will receive personalized feedback on the accuracy of their responses every 2 weeks during the intervention period.
IVR assessment and asynchronous therapist feedback
Daily IVR assessment calls
The IVR system is intended for assessment and treatment purposes. Patient-reported data will be collected daily through the IVR system (Table 3). Automated calls will last approximately 5 minutes and comprise a series of six questions related to; headache pain, headache interference, perceived stress, sleep quality, skipped meals, headache self-management skill practice, and menstruation (if applicable). Additionally, participants will rate their confidence and goal attainment every 2 weeks. Participants will select a preferred time to receive their daily calls to capture a 24-hour window. Participants who miss their initial daily call will receive two additional opportunities for a call that day. If participants know that they may be unavailable during their scheduled call time, they will be able to call into the IVR system. The intervention is extremely interactive in nature. Participants may elect to leave their study therapist a message at any point in time throughout the study if they need additional support or guidance and can also be directly connected to the VHA Veterans Crisis Line if they require immediate crisis support. Study therapists may also leave participants a message at any point in time during the study.
Interactive voice response (IVR) call schedule for study intervention period

IVR: six daily questions (headache pain, interference, perceived stress, sleep quality, skipped meals, skill practice); Biweekly: two questions (confidence in headache management and goal attainment) via IVR; T/F: true false questions about each topic presented biweekly; Goals: patient recorded goals for each topic; Feedback: personalized therapist feedback via IVR; Patient goals: patient recorded SMART goals.
Asynchronous therapist feedback
Biweekly, study therapists will visualize and examine patient-reported data collected daily through the IVR system via a specialized secure provider-facing dashboard (see Figure 4). Participants will receive a 2- to 4-minute, pre-recorded, tailored, and personalized recording from their study therapist every 2 weeks during the intervention period, based on the patient-reported data captured through the IVR system. Participants will have the option to listen to the pre-recorded message from their study therapist, or they can save the message so that they can listen to it at a later time.

Example of patient-facing website housing patient workbook.
Motivational enhancement
Three weekly one-way standardized motivational enhancement/educational short-messaging service (SMS) messages will be automatically sent, via VA Annie App, to participants at intermittent times within each 14-day period to maintain engagement and encourage utilization of headache self-management skills.
Outcomes
Headache days
Participants complete a 30-day daily electronic headache diary during the pretreatment (weeks −4 to 0) and post-treatment (weeks 10–14) periods. Participants record days in which they experience headache and associated symptoms (e.g., photophobia, phonophobia, nausea/vomiting). The primary clinical outcome is the change in self-reported headache days from the 30-day baseline run-in period prior to treatment to the 30-days post-treatment completion.
Headache-related disability
The Migraine Disability Assessment (MIDAS), 39 is a 5-item, self-report measure of life disruption due to headache in the last 1 month. Cut points represent thresholds for little or no disability (0–5), mild disability (6–10), moderate disability (11–20), and severe disability (21+). The Headache-related disability inventory (HDI), 40 is a 25-item self-report questionnaire that assesses both the emotional and functional impact of headache on daily activities. Item response options include “yes,” “sometimes,” and “no.” Total scores range from 0 to 100. Higher scores indicate higher levels of reported disability. Both measures have demonstrated strong reliability and validity. 39,40
Treatment feasibility
The Feasibility of Intervention Measure (FIM) 31 is a 4-item measure of feasibility. Items are rated on a 5-point Likert scale (1 = completely disagree to 5 = completely agree). Scores are a calculated mean, with higher scores indicate greater feasibility The FIM has demonstrated high internal consistency (Cronbach α = 0.88–0.89) and test–retest reliability (r = 0.88). 31
The Structured Assessment of Feasibility Questionnaire (SAFE) 32 is a 16-item measure of feasibility of implementation of mental health services, organized into two sections. The first eight questions assess barriers to implementation; the final eight assess facilitators to implementation. Items are rated as either yes, partial, no, or unable to rate. The authors recommended against using a summary score since items within the sale may have unequal weight, therefore items will be examined individually. The SAFE has demonstrated excellent interrater (k = 0.84) and test–retest reliability (k = 0.89). 32
Feasibility variables will also be pulled directly from the provider-facing dashboard across the duration of the intervention study period, including: (1) total number of IVR calls completed (out of 70); (2) total number of IVR calls completed per topic module; (3) total number of study therapist feedback calls completed (out of 5).
Treatment acceptability
The Client Satisfaction Questionnaire-8 (CSQ-8) 41 is an 8-item self-report measure of treatment acceptability and satisfaction with mental health treatment. Items are coded on a 4-point scale with responses tailored to questions regarding quality and satisfaction of services (1 = lowest degree of satisfaction to 4 = highest degree of satisfaction). Total scores range from 8 to 32. The CSQ-8 has demonstrated high internal consistency (Cronbach α = 0.93) and moderate-to-high correlation with measures of client satisfaction (r s = −0.34), client outcomes (r = −0.40), and service utilization (engaged in therapy; r bp = 0.57; sessions per month: r rb = 0.56). We will consider treatment acceptable as indicated by a score of >24 when given immediately after treatment completion. The System Usability Scale (SUS) 33 is a 10-item measure of treatment acceptability that assesses participants’ perceived usability of technology systems. The SUS. For the current study, the IVR stem for treatment delivery will be the technology examined by the SUS. Response options range from 1 to 5 (1 = strongly agree to 5 = strongly disagree), with 50% of the items being positively worded and 50% negatively worded. Scores are converted to an overall score from 0 to 100. Higher scores indicate higher perceived usability. The Acceptability of Intervention (AIM) 31 is a reliable and validated 4-item measure of treatment acceptability. Items are scored on a 5-point scale (1 = completely disagree to 5 = completely agree). Scores are a calculated mean. The AIM has demonstrated high internal consistency (Cronbach α = 0.83–0.85) and test–retest reliability (r = 0.80). 31
Depression
The Patient Health Questionnaire 8-item (PHQ-8) 42 is an 8-item, self-report measure of depressive symptom severity based on Diagnostic and Statistical Manual of Mental Disorders, Fifth edition (DSM-5) criteria. The PHQ-8 is a modified version of the Patient Health Questionnaire 9-item 43 which drops the final item assessing suicidality. Items are rated from 0 to 3 (0 = not at all to 3 = nearly every day) with total score ranging from 0 to 24 for the PHQ-8. The PHQ-9 provides thresholds for severity of depressive symptoms: none-minimal (0–4), mild (5–9), moderate (10–14), moderately severe (15–19), and severe (20–27) depressive symptoms. The PHQ-8 has demonstrated exceptionally high correlation with the PHQ-9 (r = 0.997) and identical cutoffs are advised. 43 The PHQ-9 has demonstrated high internal consistency (Cronbach α = 0.86–0.89) and high sensitivity and specificity (88%; 88%) compared to structured mental health interviews. 43
Anxiety
The Generalized Anxiety Disorder Screener (GAD-7) 44 is a 7-item, self-report measure of anxiety. Items are rated from 0 to 3 (0 = not at all to 3 = nearly every day). Total scores range from 0 to 21. Cut points represent thresholds for none-minimal (0–4), mild (5–9), moderate (10–14), and severe (15–21) anxious symptoms. The GAD-7 has demonstrated high internal consistency (Cronbach α = 0.92) and test–retest reliability (r = 0.83), and high sensitivity (0.66–0.89) and specificity (0.80–0.81) across multiple anxiety disorders compared to structured mental health interviews. 44
Post-traumatic stress disorder
The Post-Traumatic Stress Disorder Checklist-5 (PCL-5) is a 20-item, self-report measure of symptoms of PTSD based on DSM-5 criteria. 45 Items are rated from 0 to 4 (0 = not at all to 4 = extremely). Total scores range from 0 to 80, with higher scores indicating higher post-traumatic stress symptoms. The PCL-5 has demonstrated high internal consistency (Cronbach α = 0.94) and test–retest reliability (r = 0.82), and strong convergent validity with other measures of PTSD (r = 0.84–0.85). 45
Sleep quality
The Insomnia Severity Index (ISI) 46 a 7-item self-report measure of insomnia. Items are rated from 0 to 4 with responses tailored to the question (e.g., symptom severity rated 0 = none to 4 = very severe), with total scores ranging from 0 to 28. Cut points represent thresholds for no clinically significant insomnia (0–7), subthreshold insomnia (8–14), clinical insomnia (moderately severe) (15–21), clinical insomnia (severe) (22–28). The ISI has demonstrated strong reliability (Cronbach α = 0.90–0.91) and good convergent validity with other measures of insomnia (r = 0.80) and sleep diaries (r = 0.54–0.59), and is particularly well-suited to detecting changes in perceived insomnia. 46,47
Headache catastrophizing
The Headache Pain Catastrophizing Scale (HPCS) 48 is a modified version of the Pain Catastrophizing Scale (substitutes “headache” for “pain” in the questions), The HPCS is a 13-item self-report measure of catastrophic thinking related to headache with three subscales: rumination, magnification, and helplessness. Item responses range from 0 to 4 (0 = not at all to 4 = all the time). Total scores range from 0 to 52, with higher scores indicating higher levels of catastrophizing. A total score of 30 or higher indicates clinically significant catastrophizing. 48 Meta-analysis of the PCS’s psychometric properties found the total score to have high internal consistency (Cronbach α = 0.89) and test–retest reliability (r = 0.88). 49 The PCS is moderately correlated with related measures of depression, anxiety, negative affect, and fear of pain and uniquely predicts pain intensity above and beyond those constructs. 48
Self-efficacy
The Headache Management Self-Efficacy Scale (HMSE) 50 is a 25-item self-report questionnaire related to a person’s confidence in their ability to manage their headache symptoms using behavioral strategies (e.g., relaxation and stress management). Item responses range from 1 to 7 (1 = strongly disagree to 7 = strongly disagree). Total scores range from 25 to 175 with higher scores indicating higher levels of headache management self-efficacy. The HMSE has demonstrated high internal consistency (Cronbach α = 0.90) and good convergent validity based on moderate-to-high correlations with related constructs of locus of control (internal subscale: r = 0.40), coping efforts (prevention: r = 0.54; tolerance: r = 0.55), and disability (r = −0.24). 50
Perception of change
The Patient Global Perception of Change (PGPC) 51 is a single-item scale measures the participant’s perception of improvement in their symptoms since the start of the study. Patients respond to the single-item on a 7-point scale (1 = no change or worse to 7 = a great deal better). Single-item measures of global perceived change are frequently included as an indicator of clinically significant change, with scores >5 (e.g., “better” and “a great deal better”) reflecting improvement. 51
Plans for analysis
SAS 9.4 will be used for all quantitative analyses. We will visually inspect all data and evaluate all continuous study variables for normality of distribution, skewness, and kurtosis. A p-value of <0.05 (two-tailed) will be used to indicate statistical significance. Baseline demographic variables (age, race, ethnicity, marital status, sex), headache days, psychiatric comorbidities (PHQ-9, GAD-7, PCL, PSS), and psychosocial variables (ISI, HPCS, HMSE) will be described using mean and standard deviation, median and interquartile range, or number and percent.
Aim 1—Assessment of clinical signal
All analyses will first be conducted in an Intent-to-Treat sample, which includes all enrolled participants who were eligible to begin treatment in the study. For all clinical outcomes, missing values at follow-up will be imputed using multiple imputation including baseline values and demographic predictors in the imputation model. Headache days which will be assessed at pretreatment baseline 30-day baseline run-in period (baseline, weeks −4 to 0) and the 30-day post-treatment completion (post-treatment, weeks 10–14) from the daily headache diary, and aggregated into the number of headache days/30 days. Paired-samples t-tests will evaluate changes in headache days between baselines and post-treatment; all secondary and tertiary clinical outcomes will also be evaluated using paired-samples t-tests (Headache Disability Inventory, Patient Health Questionnaire, Generalized Anxiety Disorder Screener, Post-Traumatic Stress Disorder Checklist, Insomnia Severity Index, Headache Pain Catastrophizing Scale, Headache Management Self-Efficacy Scale). For Migraine Disability Assessment Scale (MIDAS), which is typically non-normally distributed, we will use a Wilcoxon signed-rank test to assess differences between baseline and follow-up MIDAS scores. We will use the same strategy for any other outcomes that are non-normally distributed.
Aim 2—Feasibility
Treatment adherence will be defined by the number of IVR calls the participant answers out of 70 total calls. Descriptive statistics (mean and standard deviation) will be calculated and compared to standard treatment acceptability (CSQ-8 > 24).
Aim 3—Cost data
All cost analyses will be exploratory. We will evaluate costs from the Veteran and healthcare system perspective, with a 6-month time horizon. Micro-costing techniques will assess (a) start-up costs and (b) post-start-up intervention costs for treatment delivery, and aggregate post-treatment acute healthcare utilization costs. Start-up costs will include development costs for the IVR system, patient-facing website, and provider dashboard. Intervention costs will monetize (wages/benefits per unit time) the time providers are logged into the dashboard and time recording tailored personalized feedback, and time Veterans, are accessing treatment. We will sum costs of acute headache-related healthcare utilization events (emergency room, unplanned medical visits, and outpatient pharmacy), for downstream expenditures, assigning costs for services and medications from the Managerial Cost Accounting System (MCA) for VA. We will compare the downstream costs for the 6 months before study enrollment and 6 months after completing the study. We will assess health-related quality of life changes from health utilities derived from the VR12 at enrollment and completion of the study.
Sensitivity analysis
We will conduct sensitivity analyses in the group of individuals who complete the intervention (“completer analyses”) to understand the impact of the interventions on our outcomes among individuals who receive a full treatment dose.
Study administration
The Institutional Review Board at VA Connecticut Healthcare System (VACHS) approved this study. All participants will provide informed consent. The Data and Safety Monitoring Board will contain up to six members which represent multiple disciplines, including a health psychologist, neurologist, biostatistician, and Veteran advocate, who will provide ongoing evaluation of the study’s progress and monitoring of adverse events.
Discussion
Interactive CBT for Headache And Relaxation Training (iCHART) is a single-arm, single-site, pilot trial that aims to test a five-session Cognitive Behavioral Therapy for Headache (CBT-HA) intervention delivered via interactive voice response technology (IVR) over 10-weeks. While CBT-HA is considered an evidence-based intervention that results in decreased headache activity and improved quality of life, this is the first trial to examine the delivery of CBT-HA for PTH via IVR technology.
Patients face several challenges in receiving psychological treatment for their headache disorders, 20 including being able to find time to attend in-person appointments, difficulties finding providers who specialize in psychological interventions for headache, cost of treatment, and stigma associated with receiving treatment, which often results in suboptimal follow-through on referrals for psychological treatment. 21 In the past decade, there has been an increased interest in examining mobile and telehealth delivery of psychological interventions for headache, 23,52 with many patients expressing particular interest in these modes of treatment delivery. 16,53 The current study aims to address barriers to treatment engagement, allowing patients to receive care at a most convenient time. In addition, outcomes are assessed at two time points, allowing for examination of this intervention’s immediate and long-term effects.
Findings from this study may address an important knowledge gap regarding the feasibility, acceptability, clinical signal, and costs of CBT-HA provision through IVR technology, which may ultimately still allow patients to receive access to evidence-based care if they are unable to or prefer not to attend appointments in-person. In examining the costs to the healthcare system for provision of headache-related services before and after IVR, we will have the foundation for cost-benefit analysis to determine if IVR is cost-neutral or even cost-saving in the short and medium term. 54 Additionally, we will derive pre-intervention and post-intervention health utilities, which can be used to evaluate changes in quality-adjusted life years (QALYs) for cost-effectiveness analysis. 55 The incremental cost-effectiveness ratio (ICER) measuring change in cost per change in QALY may further provide useful information for stakeholders interested in adopting this technology. In addition to gathering information about IVR treatment delivery, we will also be able to examine the potential benefits of using IVR technology for the capture of patient-reported outcomes, which may result in less recall bias than traditional data capture methods. 56 Future studies should evaluate subgroup response and dose to determine optimal characteristics of the population and intervention before a definitive trial.
This single-arm pilot trial is uniquely positioned to add much-needed support for delivering evidence-based psychological headache interventions via interactive voice response technology.
Clinical implications
Post-traumatic headache (PTH) is persistent and highly disabling.
Cognitive-behavioral therapy for headache reduces headache frequency and severity and improves people’s quality of life, yet it is underutilized and inaccessible to many.
Delivery of CBT-HA through interactive voice technology may hold promise to increase much-needed access to this treatment.
Footnotes
Acknowledgment
The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States Government.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ASG, OD, DGR, SWC, SCT, JPN, and BTF report no conflicts of interest. EKS has consulted for GlaxoSmithKline, Eli Lilly, and Click Therapeutics. JJS has received grants support from the Department of Veteran Affairs, the American Academy of Neurology, and the American Heart Association/American Stroke Association.
Ethical approval
The study team will conduct a chart review of eligible participants’ EHR and then contact participants via phone to confirm study eligibility. Study team members will obtain verbal informed consent from all eligible participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Veterans Affairs Headache Centers of Excellence. VA Connecticut Healthcare System Bell-Kern’s research award (ASG) supported participant compensation.
