Abstract
Background
Cognitive behavioral therapy for headache (CBT-HA) improves headache-related outcomes, but accessibility barriers limit its use. This pilot study evaluated the feasibility, acceptability and clinical signal of an interactive voice response (IVR)-delivered CBT-HA intervention for veterans with post-traumatic headache (PTH).
Methods
A single-arm pilot trial was conducted with 18 veterans diagnosed with PTH. Participants completed a 10-week IVR-CBT-HA program. Outcomes were assessed at baseline, immediately post-treatment and one-month follow-up. Primary outcomes included changes in headache days, interference, disability, feasibility and acceptability.
Results
Fifteen participants completed the study. Headache frequency, headache-related disability, depressive symptoms, anxious symptoms, sleep quality and headache catastrophizing were not statistically significant. Self-efficacy significantly improved from baseline to post-treatment (F2,12 = 8.71, p = 0.001), and remained stable at follow-up. Participants reported high satisfaction with the intervention (27.73/32, SD = 5.66) but low system usability (mean = 20.83/100, SD = 15.72). Study therapists rated the intervention as highly acceptable (acceptability of intervention: mean = 4.83/5, SD = 0.37) and feasible (feasibility of intervention measure: mean = 4.92/5, SD = 0.28). Interactive CBT for headache and relaxation training (i.e. iCHART) resulted in an approximately 33% cost savings compared to traditional CBT-HA.
Conclusions
Asynchronous, IVR-delivered CBT-HA was feasible, acceptable and cost-effective for veterans with PTH. Although headache reductions were not statistically significant, self-efficacy improvements suggest long-term benefits. Future research should explore technology refinements and larger randomized trials.
Trial Registration
ClinicalTrials.gov: NCT05093556 (registered 26 October 2021)
This is a visual representation of the abstract.
Introduction
Post-traumatic headache (PTH) is a prevalent and debilitating headache disorder attributable to neck or head injury, 1 which is particularly common in military personnel and veterans.2–9 The persistence and severity of PTH symptoms negatively impacts psychological well-being and functioning. 10 Despite its widespread occurrence, effective management of PTH remains a clinical challenge. 11
Cognitive behavioral therapy (CBT) has been shown to be an effective treatment for migraine and chronic tension-type headache.12–16 The treatment focuses on modifying thought patterns and behaviors to alleviate headache pain and associated symptoms, at the same time as enhancing self-efficacy and quality of life. 17 A recent randomized-clinical trial with 193 veterans with PTH showed that CBT for headache (CBT-HA) resulted in significant improvement in headache-related disability. 18 While CBT-HA shows promise for improving PTH outcomes, traditional in-person delivery of CBT-HA may not always be feasible or accessible, especially for the veteran population. Geographic barriers, transportation issues and the stigma surrounding psychological support often impede access to this evidence-based treatment.19–21
In response to these challenges, technology-based interventions have emerged as innovative solutions for expanding access to treatments. Interactive voice response (IVR) technology, which enables the delivery of therapeutic content via a touch-tone phone, offers a unique opportunity to address key barriers to treatment access. IVR-based CBT programs provide structured, yet flexible, CBT sessions, making this evidence-based treatment more accessible to veterans with PTH who might otherwise find it difficult to engage in traditional forms of therapy. Heapy et al.22 showed that IVR delivery of CBT for veterans with chronic low back pain was non-inferior to delivery of CBT in person. 22
The current single-arm, single-site, pilot clinical trial, based upon our previously published protocol, 23 aims to investigate the preliminary effectiveness of a CBT-HA program delivered via IVR to veterans with PTH. By leveraging technology to deliver behavioral interventions for headache, this study explores a novel approach to PTH treatment, potentially paving the way for more accessible and effective management strategies for this underserved patient population.
We report findings for the primary aims of this study, which were to (1) evaluate whether participation in an IVR-based CBT-HA program is associated with changes in clinically relevant outcomes; (2) establish the feasibility and acceptability of interactive CBT for headache and relaxation training (iCHART); and (3) examine the preliminary cost-effectiveness of iCHART. We hypothesized that participants would demonstrate decreases in the number of headache days and improvements in headache-related disability, psychiatric symptoms and headache-related cognitions following treatment completion. Additionally, we hypothesized that iCHART would demonstrate high feasibility and acceptability, both from the patient and therapist perspectives. As an exploratory aim, we examined the cost of iCHART, comparing its provision to the standard of care and assessing changes in healthcare expenditures pre- and post-intervention.
Methods
Study design
The iCHART study was a single-arm, single-site, pilot trial aimed at evaluating a five-session CBT-HA intervention delivered via IVR technology.
Participants
We recruited 18 veterans from a single Veteran Affairs healthcare system (VA Connecticut Healthcare System) from November 2021 to November 2022 (Figure 1). Eligible participants were English speaking veterans, who were at least 18 years old, with a primary complaint of headache, with at least four headache days during the 30-day baseline diary run-in period, and an absence of hospice/palliative care, sensory deficits, significant cognitive impairment, active psychotic symptoms, suicidality or severe depressive symptoms. All participants met International Classification of Headache Disorders (ICHD-3) criteria for persistent PTH, which was documented in their electronic medical record and confirmed as current. We selected a more stringent diary completion threshold (≥28/30 days) to ensure participants were likely to adhere to the demands of the 10-week daily IVR protocol and to support data reliability. Detailed inclusion and exclusion criteria are previously published. 23

Study flow. PTH = post-traumatic headache; ICD = International Classifications of Diseases; VA = Veteran Affairs.
Procedures
Participants provided oral informed consent to participate and were enrolled in the study by a research assistant (OD). Participants completed an electronic headache diary for 30 days, via the VA Annie App, 24 which was time-stamped, to establish a baseline measure of their monthly headache days and headache interference, followed by a 10-week treatment period, with study assessments collected at baseline, immediately post-treatment (week 10) and one month after treatment completion (week 14). Finally, patients initiated a second 30-day electronic headache diary immediately post treatment completion. All assessment measures were administered electronically through Qualtrics, an online, HIPAA compliant, data capture system (https://www.qualtrics.com). There was a data safety monitoring board, comprised of a health psychologist, neurologist, veteran and statistician. Participants were eligible to receive a total of $50 for study participation. The protocol was prospectively registered at clinicaltrials.gov (NCT05093556). The VA Connecticut Healthcare System institutional review board granted approval for this study (1592868-24). No changes were made to the original protocol. There were no adverse events reported. A detailed description of the iCHART trial has been previously published. 23
Intervention
All participants received an electronic patient workbook, which was an adapted version of the VA Cognitive-Behavioral Therapy for Headache (CBT-HA) manual. 17 Treatment covered five cognitive and behavioral-based headache skills sessions over a 10-week treatment period, including relaxation training, managing contributing headache factors, acute management skills, cognitive restructuring and skill consolidation. The IVR technology was used for both daily patient-reported outcomes and treatment (biweekly delivery of personalized feedback from their study therapist). Daily calls lasted approximately five minutes and assessed five domains, headache pain, headache interference, reported stress, sleep quality and self-management skill practice.
Study therapists were doctoral-level clinical psychologists (DGR, SWC and SCT), with one being pre-licensure and requiring supervision. They systematically reviewed daily patient-reported outcomes on a secure therapist dashboard specifically developed for this study. Participants received personalized biweekly feedback from study therapist based on their IVR-reported data, such as from comments on observed trends (e.g. “I notice your headache symptoms seem to increase in intensity after a few days of poor sleep”) to problem solving goals (e.g. “I know you mentioned not being able to complete your goal this week of eating three meals a day, SMART goals are a great way to organize your goals”). To encourage utilization of headache self-management skills, three weekly one-way standardized motivational enhancement messages were sent via the VA Annie App. 24
Measures
Participants completed an electronic headache diary for 30 days pre-treatment and 30 day-post treatment completion. A standardized assessment battery was administered electronically at pre-treatment (following the 30-day run in period), at week 10 (post-treatment) and at week 14 (follow-up). Patient domains assessed included headache-related disability (The Migraine Disability Assessment Measure (MIDAS) 25 and Headache Disability Inventory (HDI)), 26 feasibility (IVR call adherence), acceptability (The Client Satisfaction Questionnaire-8 (CSQ-8)), 27 the System Usability Scale (SUS), 28 depression (The Patient Health Questionnaire-8 (PHQ-8)), 29 anxiety (The Generalized Anxiety Disorder Screener (GAD-7)), 30 post-traumatic stress (Post-Traumatic Stress Disorder Checklist-5 (PCL-5)), 31 sleep quality (The Insomnia Severity Index (ISI)), 32 headache catastrophizing (The Headache Pain Catastrophizing Scale (HPCS)) 33 and self-efficacy (The Headache Self-Management Self-Efficacy Scale (HMSE)). 34 Additionally, study therapists completed measures of acceptability (Acceptability of Intervention (AIM)) 35 and feasibility (The Feasibility of Intervention Measure (FIM) 35 and Structured Assessment of Feasibility Questionnaire (SAFE)). 36 A detailed description of the assessment measures is included in the published protocol article. 23
Demographics
Baseline demographics, including age, gender, race, ethnicity, marital status, level of education and employment status, were gathered from the patient's electronic health record (EHR) and confirmed during a baseline interview (Table 1).
Baseline participant characteristics.

*Note: The three participants who were enrolled but did not complete the study reported blast injury-related post-traumatic headache.
Analyses
Given that this was a small pilot trial, no statistical power calculation was conducted in advance. We intended to recruit 35 participants for this trial; however, recruitment was halted due to changes in VA technology requirements, which would have necessitated a complete redevelopment of our dashboard and IVR system, rendering further enrollment infeasible within the study's timeline and resources. Analyses involving headache diary data included participants who provided at least partial post-treatment data. Headache days were assessed in a 30-day baseline run-in period at pretreatment (baseline, weeks −4 to 0) and the 30-day post treatment completion (post-treatment, weeks 10–14) from the daily headache diary and aggregated into the number of headache days over 30 days. Missing diary data were imputed proportionally for four participants with a mean of nine missing days of data (e.g. endorsed headache on 10 out of 20 reported days was imputed to headache on 15 of 30 days). Notably, imputation strategies of day-level missing data confer potential bias. Proportional imputation is susceptible to increased error if data are not missing at random and the findings should be interpreted with caution. Although not pre-specified in our protocol, we examined exploratory indicators of clinical significance commonly used in headache research including ≥50% reduction in headache days and interference and ≥10-point reduction in MIDAS scores, which are thresholds recognized as markers of clinical improvement in both clinical trials and practice.
Feasibility and acceptability outcomes are reported using descriptive statistics (mean, SD, range). Patient-reported outcomes were inspected for accuracy (e.g. MIDAS scores not exceeding upper range) and reviewed for violation of assumptions. Paired-samples t-tests evaluated changes in headache days and headache interference between baseline and post treatment; all secondary and tertiary clinical outcomes were also evaluated using repeated-measure analysis of variance and paired-samples t-tests (MIDAS, HDI, PHQ-8, GAD-7, PCL, ISI, HPCS and HMSE). For analyses that violated assumptions, non-parametric tests were conducted; however, these resulted in no-meaningful differences in the findings and therefore parametric results are reported throughout for consistency.
Cost analyses were limited to the patients who completed the study, including one participant who completed the intervention but was lost to follow up (n = 16). Costs were viewed from the perspective of the healthcare system and assessed the costs of IVR delivery of CBT-HA with in-person CBT-HA and baseline versus post-intervention pharmacy and emergency department (ED) visit costs. To examine the cost of the intervention, study therapists were asked to keep a log of their training and study therapy hours throughout, which was cross-referenced with the number of iCHART modules their participants completed. Salary information is publicly available and was used to calculate the overall cost of their time and their rate per hour. The intervention costs were compared to the hypothetical cost of delivering CBT-HA in-person (standard of care of six one-hour sessions) to each enrolled patient. The initial expenditure for purchase of the IVR system were assumed to be one-time already incurred and irrecoverable sunk costs, and not included in the cost analyses. Healthcare utilization, consisting of headache-related urgent care (UC) and ED visits, and headache prescriptions were extracted from the EHR for participants six months before and after the intervention.
For determination of cost attributable to ED visits for headache, we used the Medical Expenditure Panel Survey (MEPS), 37 identifying visits associated with International Classifications of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes G43 (migraine headache) and G44 (other headache). We applied MEPS complex survey design variables for weighted sampling, clustering and stratification to mean total cost per ED visit with Taylor series linearized standard errors. All costs were indexed to the 2022 calendar year using the Consumer Price Index. 38 Medication cost was determined using Veterans Health Administration medication pricing documents. Costs were reported as mean with standard error. Analyses were conducted using SPSS, version 27 (IBM Corp., Armonk, NY, USA) and STATA, version 18.0 (StataCorp, College Park, TX, USA). Alpha was set at 0.05 (two-tailed) for all inferential tests.
Results
Participant characteristics
Fifteen veterans completed the study. The mean age of the participants was 49.5 years (SD = 13.4). The majority was white (n = 14, 94%), non-Hispanic (n = 13, 87%) and male (n = 10, 67%). The three veterans who did not complete the study were all male, white, non-Hispanic and employed full-time, with a mean age of 45.0 years (SD = 1.0). They varied in terms of their marital status and highest education level completed. All participants met criteria for persistent PTH. Full participant characteristics are shown in Table 1.
Headache days and headache interference
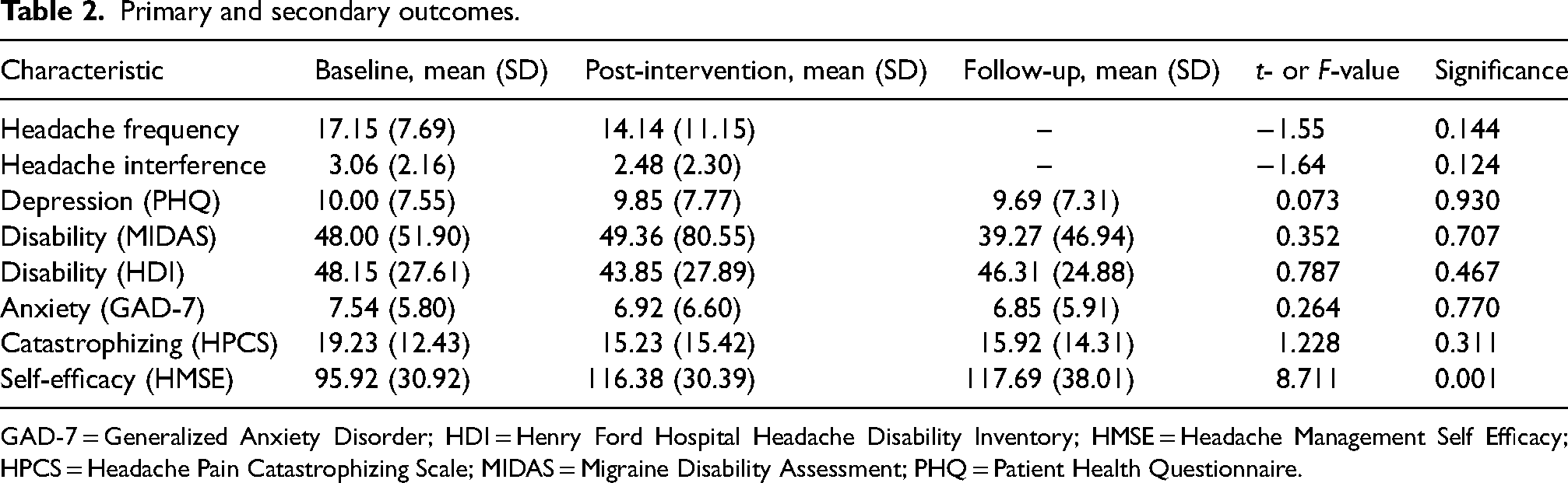
Headache frequency decreased from baseline to post-treatment by an average of three days per month (mean = 17.15, SD = 7.69 vs. mean = 14.14, SD = 11.15), although this change was not statistically significant (t14 = −1.55, p = 0.144), with a small effect size (Cohen's d = 0.40) (Table 2). Some 27% of participants (4/15) experienced a clinically meaningful reduction, with headache days decreasing by at least 50%.
Primary and secondary outcomes.
GAD-7 = Generalized Anxiety Disorder; HDI = Henry Ford Hospital Headache Disability Inventory; HMSE = Headache Management Self Efficacy; HPCS = Headache Pain Catastrophizing Scale; MIDAS = Migraine Disability Assessment; PHQ = Patient Health Questionnaire.
Headache interference also decreased from baseline to post-treatment (mean = 3.06, SD = 2.16 vs. mean = 2.48, SD = 2.30), although this change was not statistically significant (t14 = −1.64, p = 0.124), with a small effect size (Cohen's d = 0.42). A clinically meaningful reduction of 50% or more in headache interference was observed in 27% (4/15) of participants.
Secondary clinical outcomes
As shown in Table 2, participants showed significant improvements in headache management self-efficacy across all time points (F2,12 = 8.711, p = 0.001). Post-hoc analysis revealed that self-efficacy scores increased significantly from baseline to post-treatment (mean = 95.92, SD = 30.92 vs. mean = 116.38, SD = 30.39, p = 0.001, Cohen's d = 0.98 (large effect)) and remained elevated at follow-up (mean = 95.92, SD = 30.92 vs. mean = 117.69, SD = 38.01, p = 0.009, Cohen's d = 0.87 (large effect)). No significant changes were observed between post-treatment and follow-up (p = 0.816), indicating that these improvements were sustained over time.
Changes in headache-related disability (MIDAS) were not statistically significant across time points (F2,10 = .352, p = 0.707), with mean scores shifting from 48.00 (SD = 51.90) at baseline to 49.36 (SD = 80.55) post-treatment, and 39.27 (SD = 46.94) at follow-up. The effect sizes were small (Cohens d = 0.02 and 0.39, respectively). Six of 12 (50%) of participants with a valid MIDAS experienced clinically meaningful improvement, defined as a reduction of at least 10 points in MIDAS scores from baseline to post-treatment.
While exploratory, notably, there was minimal overlap among participants who achieved ≥50% reduction in headache days, ≥50% reduction in headache interference or a 10-point reduction in MIDAS scores, with nine out of 15 participants showing improvement in at least one of these domains.
Other clinical outcomes related to depression, anxiety and pain catastrophizing were non-significant across the three time points (p = 0.311 to 0.930) (Table 2).
Treatment feasibility and acceptability
Overall, participants were highly satisfied with the intervention, with a average score of 27.73 out of 32 (SD = 5.66) on the CSQ, indicating strong approval. However, the usability of the diary system and IVR received low ratings on the SUS (mean = 20.83/100, SD = 15.72, range = 0–50). Study therapists also rated the intervention as highly acceptable (AIM: mean = 4.83/5, SD = 0.37) and feasible (FIM: mean = 4.92/5, SD = 0.28), reinforcing the potential for implementation in clinical settings. Examination of IVR call completion revealed that participants engaged on 86.0% of IVR days (mean = 60.2, SD = 11.3). The findings from the SAFE questionnaire helped us identify barriers and facilitators to the intervention from the therapist perspective. Potential barriers include that the intervention requires specific training to deliver and is made up of several components. Several facilitators were endorsed, including that the intervention is applicable to veterans, is manualized, flexible, likely to be effective, cost-saving, has matched prioritized goals and can be piloted.
Cost analyses
All cost-analyses are exploratory. Each study therapist was proficient in delivery of CBT-HA prior to this project. ASG provided a two-hour training session to each therapist and demonstrated how to use the IVR system, secure therapist dashboard, and offered examples for providing the biweekly feedback. Two therapists estimated spending approximately one hour per intervention module (therapist 1: modules delivered = 10, study hours = 10; therapist 2: modules delivered = 15, study hours = 14.5). The third therapist, who was under clinical supervision at the time, saw most participants, reported greater time efficiency (therapist 3: modules delivered = 55, study hours = 21.5), even when considering the time commitment of the supervisor (study hours = 7.15). Of note, the time log of the third therapist showed similar commitment as the other therapists early on (1 module = 1 h), with greater efficiency occurring with more study hours (1 module = 0.25 h.
In total, the therapists and supervisor spent a total of 52.15 intervention hours for iCHART. Adjusted for each therapist's and the supervisor's hourly rate, the mean total cost of therapist services for IVR CBT-HA was $144.25 (SE = $21.86). By contrast, the cost to deliver a standard six-week protocol to a patient would be $233.67 (SE = $16.36). Comparatively, iCHART results in approximately a 33% cost savings compared to traditional CBT-HA, with savings expected to improve as therapists continue to use the technology (Table 3).
Parameters and outcomes for cost analyses.

CBT-HA = cognitive behavioral therapy for headache; ED = emergency department; IVR = interactive voice response; MEPS = Medical Expenditure Panel Survey; VA = Veteran Affairs.
Costs are the mean and SE for subjects completing the study.
Additionally, healthcare utilization was examined through pre- and post-treatment urgent care visits, ED visits and medication costs. Throughout the observation period, no urgent care visits were noted in the EHR. Two emergency department visits occurred during the pre-treatment period, costing an estimated $948.23 (SE = $211.38) per visit, with none occurring post treatment. Participants were taking an average of 1.56 prescription headache medications during the six-month pre-treatment period, averaging $200.50 (SE = $102.28) compared to an average of 1.44 prescription headache medications during the six-month post-treatment period, with a mean cost of $192.53 (SE = $119.37).
Discussion
This pilot trial demonstrates the feasibility, acceptability and cost-effectiveness of delivering CBT-HA via IVR technology for veterans with PTH. Although the reductions in headache frequency and disability were not statistically significant and causal inferences are limited given the study design, participants experienced a clinically meaningful reduction in both. 39 Improvements in headache frequency and disability suggest that iCHART may enhance quality of life for veterans with limited access to traditional therapy.
A key finding of this study is the significant improvement in self-efficacy, which was sustained one month post-intervention. Self-efficacy is a well-documented predictor of long-term treatment adherence and symptom management in headache and chronic pain disorders.35,39 This suggests that IVR-CBT-HA may empower veterans to take an active role in managing their symptoms, potentially leading to improved long-term outcomes.
Both participants and therapists rated this intervention as highly feasible and acceptable, with high levels of satisfaction reported. This is especially important for people with headache, where barriers such as geographic distance and stigma often prevent individuals from seeking and receiving psychological interventions. The pilot study demonstrated that a CBT-HA program is implementable with minimal additional training for therapists already well versed in CBT-HA. Furthermore, variability in time efficiency among therapists suggests that increased experience with the intervention can enhance operational efficiency. This finding underscores the importance of ongoing training and support could further streamline the intervention process.
The use of novel technology allowed participants to engage in CBT-HA remotely, offering a scalable solution for delivering CBT-HA to populations that frequently face significant barriers to accessing traditional in-person treatment. Furthermore, providing flexible and accessible treatments may increase adherence and reduce treatment drop out in future studies.
Although participants reported high satisfaction with treatment itself (asynchronous delivery of CBT-HA), usability ratings for the diary and IVRs system were notably low, which suggests limitations with the technology utilized to deliver the treatment A limitation of the usability measure is the inability to determine whether low satisfaction was attributed to the VA Annie App daily diary, the IVR system or both. However, given the prevalence of web-based and mobile applications, touch-tone technology may feel outdated and cumbersome. Future iterations of iCHART could explore alternative treatment delivery modalities, such as app-based interventions, which may improve engagement and applicability, at the same time as retaining the benefits of remote delivery. Addressing these technology usability concerns while retaining the core therapeutic components of the intervention will be beneficial for future larger-scale trials. Additionally, future studies should incorporate qualitative methods to capture patients’ perspectives on asynchrony delivery of CBT-HA compared to traditional in-person psychological care.
The cost analyses revealed a 33% reduction in therapist expenses compared to traditional in-person CBT. The use of IVR technology reduced therapist time and operational costs, enabling the delivery of evidence-based care at scale. Although reductions in prescription medication usage were modest, they reflect a movement towards more cost-efficient care. Of note, access to CBT-HA is concurrent with access to higher-level headache care in our tertiary headache centers. Most patients post-intervention were on less costly prescription medications than during the pre-intervention period, although two patients received treatments consistent with refractory headache and comprised approximately 85% of post-intervention medication costs amongst our sample. Future studies should investigate the sustainability of these savings over longer periods and assess broader economic implications of scaling this intervention.
Limitations
Several limitations of the present study warrant discussion. The small sample size limited statistical power, making it difficult to detect significant changes in clinical outcomes despite observed trends toward improvement. Additionally, the single-arm design precludes direct comparisons to a control group, limiting causal conclusions about the relative effectiveness. Additionally, while we used widely recognized markers of clinically meaningful improvements, these were not pre-defined in our protocol; therefore, all treatment-related improvements should be interpreted as exploratory. However, prior trials have shown that IVR delivery of CBT for chronic back pain in a veteran population was non-inferior to in-person delivery, 22 suggesting that the observed trends in this study warrant further exploration. The cost analyses were exploratory and were taken from the perspective of the healthcare system, and did not include savings to the patient such as caretaker costs or hypothetical driving time costs, nor lost wages and benefits for patients. The limited number of patients allowed for focus on differences in costs due to acute headache healthcare utilization, headache medication and costs of providing the intervention. We were reliant on reporting in the VA EHR for pharmacy data and UC and ED visits and non-VA costs were not included. A longer time horizon might also show greater cost-savings. Of note, while the results of the cost analysis were encouraging, the sample size was not sufficient to determine whether the intervention would consistently be cost-saving relative to the comparator. As a pilot study, the cost analysis was exploratory, not hypothesis-driven, but does provide an avenue for future examinations of cost-drivers and downstream cost evaluations for this intervention. Lastly, the study was conducted at a single site, which may limit generalizability to other healthcare settings or populations. Despite our limitations, this small trial was feasible, acceptable in terms of patient satisfaction, and the preliminary data can support future, larger randomized controlled trials.
Conclusions
This pilot trial demonstrates the feasibility and acceptability of an asynchronous, technology-assisted, delivery of CBT-HA for veterans living with PTH. The significant improvements in headache management self-efficacy and the high levels of patient and therapist satisfaction with the treatment suggest that this intervention holds promise as an accessible and effective treatment option for this underserved population. Future research should focus on larger, multi-site comparative effectiveness trials that examine technology-assisted CBT-HA with synchronous delivery of CBT-HA by therapists among patients with PTH and other chronic headache disorders. Additionally, it will be beneficial to explore the long-term effects of interventions and refine the technology to optimize usability.
Footnotes
Clinical implications
Acknowledgments
The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States Government.
Ethical statement
Participants provided oral informed consent to participate and were enrolled in the study by a research assistant (OD).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States Government.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/orpublication of this article: ASG, OD, DGR, SWC, SCT, JPN, and BTF report no conflicts of interest. EKS has consulted for GlaxoSmithKline, Eli Lilly, and Click Therapeutics. JJS has received grants support from the Department of Veteran Affairs, the American Academy of Neurology, and the American Heart Association/American Stroke Association.
