Abstract
Background:
Anti-CGRP monoclonal antibodies have emerged as efficacious preventive therapies for some, but not all patients with migraine. It is not yet fully understood what predicts treatment response.
Objective:
To identify factors associated with good or poor response to erenumab, the first available CGRP monoclonal antibody.
Methods:
A chart review of patients with migraine from a large headache center who received at least three 4-weekly doses of erenumab between 2018 and 2020 was conducted. Clinical variables were compared between erenumab responders (defined as ≥30% reduction in monthly headache or migraine days at 3 months) and non-responders via logistic regression analyses.
Results:
Among 90 enrolled patients, 62.2% were erenumab responders and 37.8% non-responders. A significantly larger proportion of non-responders were unemployed (58.8% vs. 28.6%), had complex diagnosis (chronic migraine overlapping another primary or secondary headache) (47.1% vs. 14.3%), higher monthly headache days (30 vs. 25.5) and migraine days (20 vs. 12), a higher frequency of daily headache (76.5% vs. 48.2%), and failed more preventive therapies (5.5 vs. 3). Based on logistic regressions, erenumab responsiveness did not significantly associate with duration of migraine, presence of aura, medication overuse, number of concurrent preventives, response to onabotulinumtoxinA or triptans, or certain comorbidities and substance use.
Conclusions:
This work may help improve selection of patients who may benefit from erenumab, but further prospective research studies are needed.
Introduction
Notwithstanding its clinical heterogeneity and biochemical complexity, which is not yet fully understood, migraine is often treatable, using an arsenal of older and emerging pharmacological therapies. 1 Clinical efficacy and tolerability of the preventive drugs with different mechanisms of action is highly variable from person to person. A fairly high discontinuation rate for the conventional first line oral preventive agents (over 80% within the first year of treatment in one study 2 ), heightens the interest in the newly developed, effective, more targeted and better tolerated parenteral regimens. 3
Erenumab, the first in class and only human monoclonal antibody that inhibits the calcitonin gene-related peptide (CGRP) receptor, has been approved and clinically used in Canada for migraine prevention, for over 3 years. Previous randomized, double-blind, placebo-controlled studies have demonstrated safety, tolerability and efficacy of erenumab in both episodic and chronic migraine disorders, that are sustained over at least 12 months in the open label extension studies. 4,5 Overall, between 40% and 50% of patients ages 18–65 years treated with erenumab had 50% or greater improvement in their headache frequency in pivotal trials, 6 –8 whereas post marketing studies replicated comparable or even better drug performance in the “real-world”. 9 In studies or “real-world,” a proportion of patients are found to be non-responders (e.g., just over 50% of patients who did not achieve a 50% improvement threshold), and some patients show a “super-response” (e.g., between 3% and 9% patients with either EM or CM achieving 100% efficacy threshold). 9,10
It is largely unknown what headache or patient characteristics predict therapeutic response to erenumab. Mechanistically, multifunctional CGRP has been recognized as a key peptide in the complex network of pathways involved in migraine pathophysiology. 11 Older migraine treatments are known to modulate CGRP pathway in various ways, including onabotulinumtoxinA, 12 triptans, 13 a host of other nonselective treatment agents such as tricyclic antidepressants, and experimentally, cannabidiol 14 and serotoninergic psychedelics. 15 On the other hand, it is thought that CGRP may additionally signal via non-canonical receptors, while other ligands may have affinity for CGRP receptors 16 that likely adds to the intricacies of understanding headache pathophysiology and effects of anti-CGRP therapies.
While erenumab has emerged as a highly promising and long awaited migraine specific preventive medicine for both patients and their treating physicians, its high cost and a non-formulary status with many insurers in Canada at this point in time, is another call for better characterization of patients that may respond to this treatment. Thus, establishing an association between patient attributes and therapeutic efficacy of erenumab will help not only guide clinicians in optimizing and personalizing treatment regimens but also support patient advocacy, and further grow our understanding of migraine pathophysiology.
Our study aims to compare individuals with migraine who respond to erenumab versus those that do not, to see if there are differences in sociodemographic, headache, and health characteristics. Specifically, we sought to identify factors associated with good response and, overall, describe previously unpublished post marketing clinical experience with erenumab in a tertiary headache clinic setting in Canada.
Methods
Data source
We conducted a retrospective chart review of patients meeting study criteria, treated at the Calgary Headache Assessment and Management Program (CHAMP), and assessed by the three staff headache neurologists at the clinic. Institutional ethics board approval was obtained (The Conjoint Health Research Ethics Board, University of Calgary, ID REB20-2203). Informed electronic consents for the chart reviews were obtained from the patients. Patients with the diagnosis of migraine (based on ICHD-III criteria) who received at least three 4-weekly doses of erenumab, either 70 mg or 140 mg, between December 2018 (when the drug first became available in Canada) and December 2020 for migraine prevention were included. No exclusion criteria were applied.
Outcome measures
Data were collected on the following variables: (1) sociodemographic (age, sex, employment), (2) headache phenotype (headache diagnosis, presence of aura, monthly headache and migraine days, presence of headache free days, years from onset, current and prior preventive use, acute medication use, response to onabotulinumtoxinA and triptans), and (3) patient health, including comorbidities (traumatic head/neck injuries, non-headache chronic pain, and mental health disorders) and substance use. Although initially proposed, we were not able to gather reliable retrospective data on patient-reported disability using validated questionnaires such as MIDAS and HIT-6, due to incomplete data in patients’ paper charts, and given limited in-person clinic visits under conditions of the COVID-19 pandemic.
Erenumab treatment response was determined based on reduction in monthly headache days (MHD) or monthly migraine days (MMD) at 3 months compared to the pre-treatment baseline. In our study, responders were defined as patients with migraine who achieved at least 30% (or greater) reduction in one or both measures. The 30% threshold, while less robust than 50% efficacy endpoint used in many headache trials, was selected based on a consensus of it translating into a clinically meaningful improvement in quality of life with chronic pain. 17 We have subsequently added a supplementary 50% response rate analysis for interested readers. It was our proposal to also utilize change in patient disability scores and monthly acute medication use as two other indicators of treatment response. However these data were not fully documented in charts in the context of reduced in-person appointments during the pandemic and hence paper documentation, such as headache diaries. Two study groups, erenumab responders and erenumab non-responders were created based on the definition above. Patient and headache characteristics were then compared between the groups.
Additional outcome measures included continuation rate of preventive therapy with erenumab for 12 months (or longer), reasons for discontinuing erenumab at any time and patient-reported adverse effects.
Statistical analysis
Participants’ characteristics were summarized for the overall group and by responders and non-responders, using means (with standard deviations) or quantiles, and frequencies and proportions, for continuous and categorical variables, respectively. Mann-Whitney U and Chi-squared test (Fisher's exact test for variables with cell size less than 5) were used for continuous and categorical variables, respectively, when comparing erenumab responders with non-responders. Logistic regression was used to calculate crude odds ratios (OR) for age, sex, employments status, headache diagnosis, baseline MHD and MMD, presence of daily headache, years from headache onset, aura, acute medication overuse, number of preventive medication failures, number of concurrent preventives, concurrent onabotulinumtoxinA therapy, response to onabotulinumtoxinA, response to triptans, presence of chronic pain, head or neck injury, or psychiatric comorbidity. In addition, a multivariable logistic regression was used to examine associations between multiple predictors and the outcome (i.e. response to erenumab) with OR and corresponding 95% confidence intervals (CI) reported.
Statistical analysis was performed using SAS 9.4 software and p < 0.05 was considered statistically significant. Statistical power calculation was not undertaken prior to the study, as this was an exploratory analysis of all available data.
Results
Patient enrollment and sociodemographic characteristics
A total of 221 patients were prescribed erenumab between December 2018 and December 2020 by the three participating CHAMP physicians and assessed for study eligibility. Of them, 92 consented to participate, while the remaining 129 were either unable to be contacted (n = 47), did not respond (n = 78), or did not consent to participate in the study (n = 4). Of the 92 study subjects, n = 2 were subsequently excluded for not meeting inclusion criteria (one patient did not complete all three erenumab treatments and another patient had the diagnosis revised to a secondary headache). Figure 1 summarizes patient enrollment.

Flowchart of patient enrollment.
Of our study population of 90 patients, 56 (62.2%) were classified as erenumab responders and 34 (37.8%) were erenumab non-responders based on the 30% or greater reduction in monthly headache or monthly migraine days as defined in the Methods section (Figure 2). Table 1 shows summary and crude comparisons of patient characteristics among those who responded to erenumab and those who did not. The mean age of the total cohort was 43.0 years (range 18–73 years; standard deviation 13.6) and 83.3% (75/90) were women. Age and gender did not significantly differ between responder and non-responder groups. There was a significantly higher unemployment rate among non-responders (58.8%) compared to responders (28.6%) (p < 0.01).
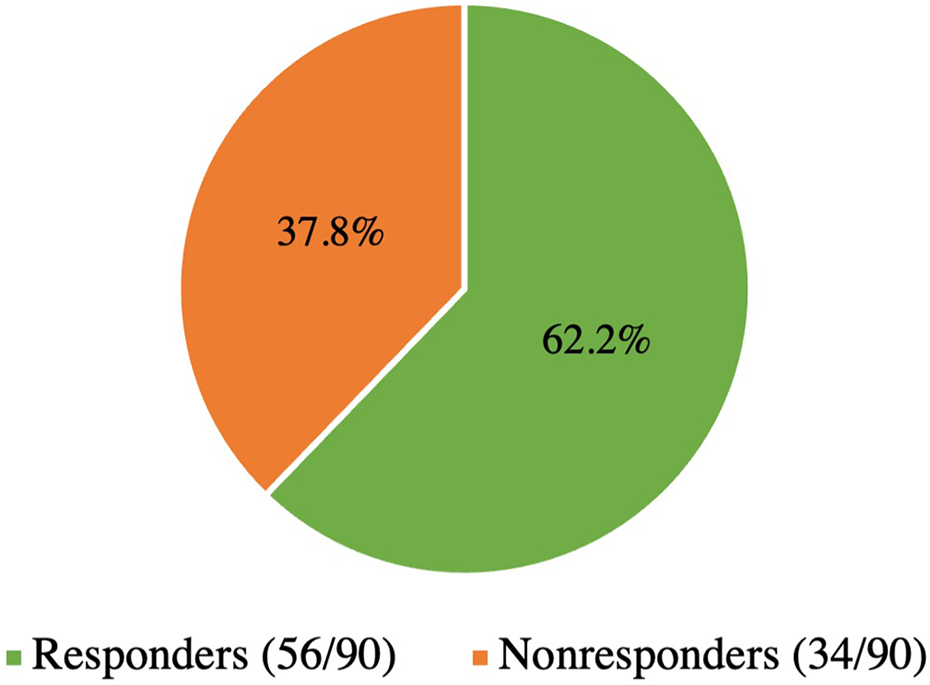
Distribution of erenumab responders and non-responders in the study population; total participants n = 90.
Patient characteristics shown for the overall sample and erenumab responders and non-responders.a
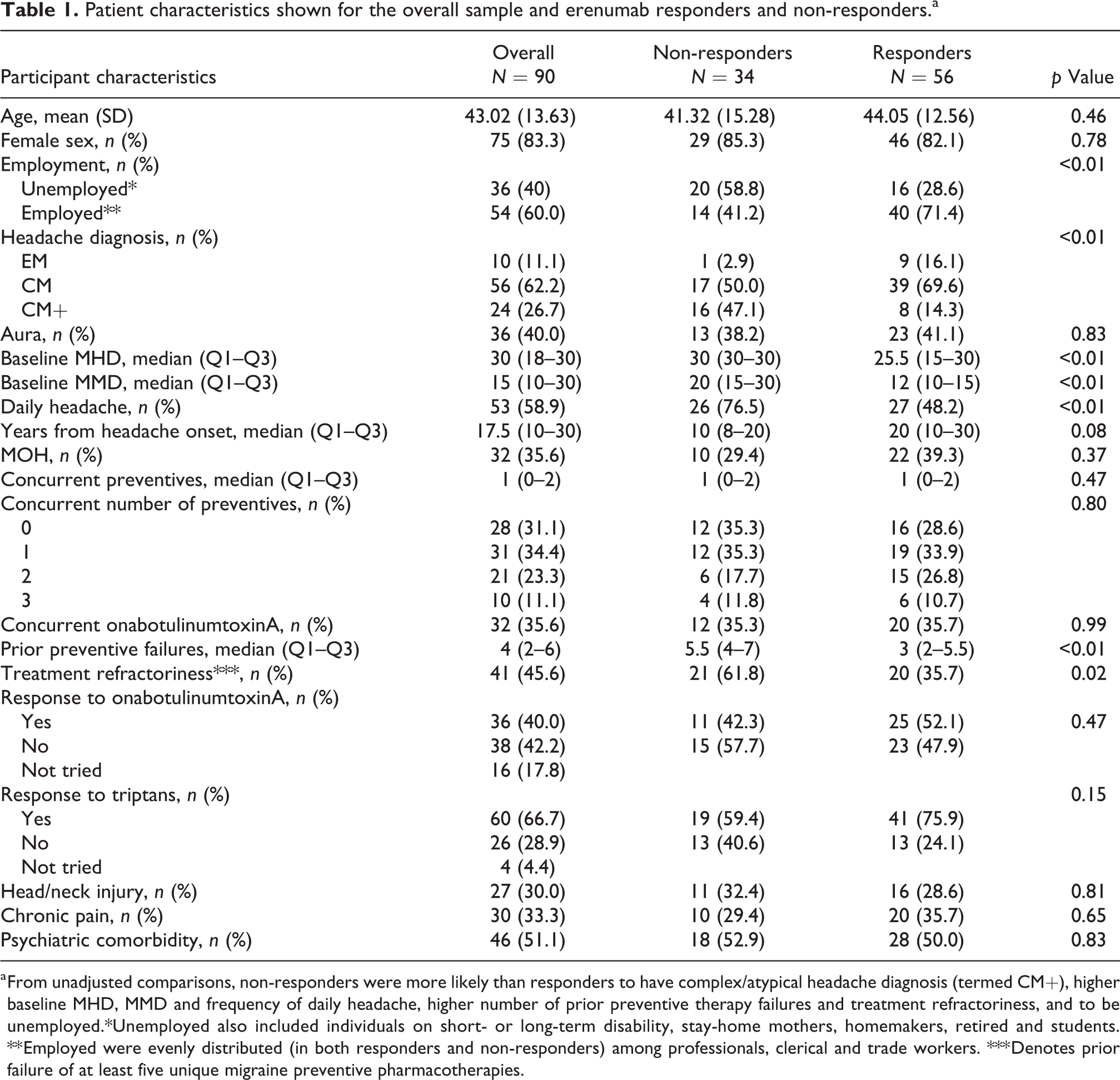
a From unadjusted comparisons, non-responders were more likely than responders to have complex/atypical headache diagnosis (termed CM+), higher baseline MHD, MMD and frequency of daily headache, higher number of prior preventive therapy failures and treatment refractoriness, and to be unemployed.*Unemployed also included individuals on short- or long-term disability, stay-home mothers, homemakers, retired and students. ** Employed were evenly distributed (in both responders and non-responders) among professionals, clerical and trade workers. *** Denotes prior failure of at least five unique migraine preventive pharmacotherapies.
Headache characteristics
Of the 90 study participants, 10 (11.1%) had the diagnosis of episodic migraine (EM), 56 (62.2%) chronic migraine (CM) and 24 (26.7%) were classified as “chronic migraine plus” (CM+), which included those diagnosed with CM and an additional primary or secondary headache syndrome such as trigeminal autonomic cephalalgia (TAC), posttraumatic headache (PTH), postural orthostatic tachycardia syndrome (POTS), idiopathic intracranial hypertension (IIH), new daily persistent headache (NDPH), cervicogenic headache (CGH), or spontaneous intracranial hypotension (SIH)/ rebound intracranial hypertension (RIH) (Figure 3). Aura symptoms were reported in 40% (36/90) of the total cohort, and similarly in either responders or non-responders.

Distribution of headache diagnoses among erenumab responders (in green) and non-responders (in orange) (panel (a)). There was a significantly higher number of patients with CM+ diagnosis in the non-responder group, p < 0.01. Additional or overlapping headache diagnoses constituting CM+ in non-responders (panel (b)) and responders (panel (c)) shown in absolute numbers. PTH: posttraumatic headache, TAC: trigeminal autonomic cephalalgia, NDPH: new daily persistent headache, POTS: postural orthostatic tachycardia syndrome, CGH: cervicogenic headache, IIH: idiopathic intracranial hypertension, SIH/RIH: spontaneous intracranial hypotension with rebound intracranial hypertension.
As shown in Table 1, there was a larger proportion of people with EM or CM among erenumab responders and fewer CM+, i.e. those with an additional headache diagnosis, which was a statistically significant observation (p < 0.01). When we further calculated OR of being an erenumab responder with an EM or CM diagnosis, it was 5.3 times (95% CI = 1.95–14.60) the odds of being an erenumab responder with the CM+ diagnosis, and this unadjusted association was significant (p < 0.01). Table 2 and Figure 4 shows that after adjusting for age, sex, years from headache onset, and prior preventive failures, the associations between the headache diagnosis and erenumab responsiveness, and employment status and erenumab responsiveness, were still significant statistically. For example, the odds of being a responder with the EM or CM diagnosis was 7.2 times the odds of being a responder with CM+ diagnosis, after adjusting for other covariates.
Adjusted associations of headache diagnosis and employment status with erenumab responsiveness; controlling for age, sex, years from headache onset, and prior preventive failures.


After adjusting for age, sex, years from headache onset, and prior preventive failures.
The number of baseline MHD was significantly higher in non-responders, with the median of 30 (interquartile range (IQR) 30–30 or range 12–30 days), versus 25.5 (IQR 15–30 or range 9–30 days) headache days per month in responders (p < 0.01). Similarly, baseline MMD count was higher in non-responders with the median of 20 (IQR 15–30 or range 4–30 days) compared to 12 migraine days per month (IQR 10–15 or range 2–30 days) in responders (p < 0.01). Daily headache was seen in 58.9% (53/90) of all study participants, with the higher prevalence among non-responders at 76.5% (26/34) compared to responders 48.2% (27/56) (p < 0.01). These data are shown in Figure 5.
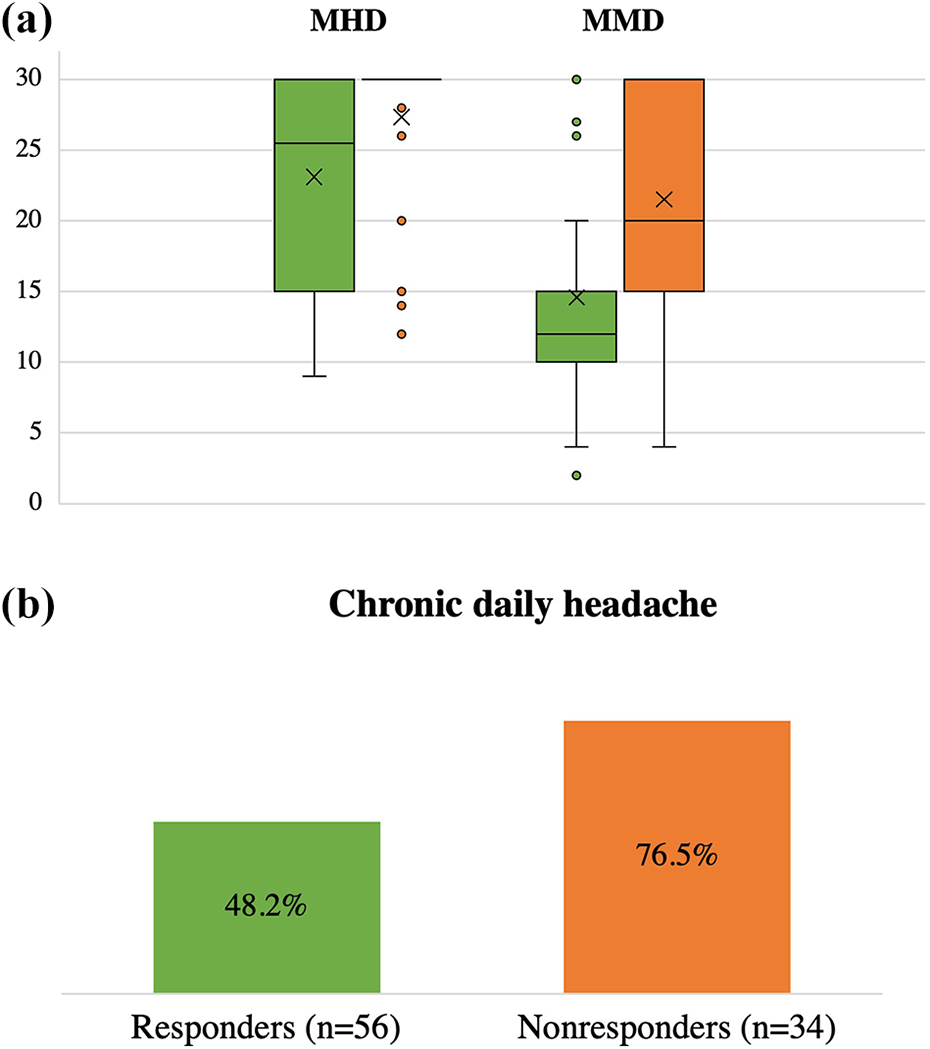
Baseline headache frequency (panel (a)) and prevalence of chronic daily headache (CDH) (panel (b)) in erenumab responders (green) and non-responders (orange). Number of monthly days (panel (a), y axis) shown as median and IQR. p < 0.01 for MHD and CHD in responders vs. non-responders, p < 0.001 for MMD in responders vs. non-responders
There was no significant difference in the years from the headache onset between the two groups, with the median 17.5 years (IQR 10–30) for the total cohort. Medication overuse was present in 35.6% (32/90) of all participants, and this was not significantly associated with the response to erenumab. Both responders and non-responders, similarly, were receiving the median of one other preventive agent at the time of erenumab treatment (IQR 0–2); where 35.7% responders and 35.3% non-responders were on a dual therapy with onabotulinumtoxinA.
However, non-responders were found to have failed more prior preventive agents with the median of 5.5 (IQR 4–7) versus responders who failed the median of 3 (IQR 2–5.5) preventives (p < 0.01) (Figure 6). The overall treatment refractoriness in our cohort was 45.6% (41/90), defined as a failure of 5 or more unique migraine preventive agents 18 prior to erenumab treatment. Of interest, there were 35.7% (20/56) of previously refractory patients among the erenumab responders. Not surprisingly, there were significantly more (21/34 = 61.8%, p = 0.02) previously refractory patients among erenumab non-responders (Table 1). OnabotulinumtoxinA non-responder rate was 47.9% (23/48) in erenumab responders while 57.7% (15/26) in erenumab non-responders, which was not statistically different. There was 24.1% (13/54) triptan non-responder rate among erenumab responders compared to 40.6% (13/32) among erenumab non-responders, which was also not statistically significant.

Prior preventive failures in erenumab responders (green) and non-responders (orange). Number of agents (y axis) shown as median and IQR. p < 0.01.
Comorbid conditions
The analyses of patient comorbidities in erenumab responders and non-responders revealed no statistical difference in the frequency of mild traumatic head or neck injuries (28.6% versus 32.4%, respectively), non-headache chronic pain conditions (35.7% versus 29.4%, respectively) or rates of anxiety and depression (50.0% versus 52.9%, respectively). There were similar rates of regular cannabis use in the two groups (26.8% in responders and 20.6% in non-responders), and 2 patients in each group were current cigarette smokers.
Other measures
Of all study participants who were commenced on erenumab, 56.0% (51/91) continued past 12 months; of them, 86.3% (44/51) were classified as responders. Reasons for discontinuing erenumab at any time are shown in Figure 7(a) where absolute numbers are given.

Reasons for discontinuing erenumab at any time during the treatment (panel (a)) and adverse effects shown if more than 1 account reported (panel (b)). Absolute numbers are given.
Adverse effects were reported in 26.1% (24/92) of participants, of whom 33.3% (8/24) discontinued the drug because of them. Adverse effects are shown in Figure 7(b) in absolute numbers. No differences in both total adverse effects were seen in erenumab responders (12/24) versus non-responders (11/24) and adverse effects leading to the drug discontinuation, where 3/8 non-responders and 4/8 responders (of note, with modest to moderate degree of response, between 30% and 50%) discontinued (one participant being excluded from the primary analysis for not completing 3 months of erenumab treatment).
All primary analyses comparing study variables among erenumab responders and non-responders were repeated for a more robust, 50% response rate, which was 53.3% (48/90). These data can be found in a Supplementary File (Table S).
Discussion
To our knowledge, this study is among the first to explore the association between patient characteristics and efficacy of erenumab, a relatively new migraine preventive agent for which post-trial clinical experience is still being gathered.
Patient population
Our study cohort was formed from a large urban tertiary referral center that routinely sees large volume of patients with migraine under the care of headache neurologists who have treated many with erenumab since its inception in Canadian clinical practice over 3 years ago. The participants’ demographics, i.e. age and sex, were generally representative of the migraine population (in the U.S.). 19 We included all ages with a range of 18–73 years, which extends beyond the trial selected population of 18–65 years. Everyone with the diagnosis of migraine who met the inclusion criteria and consented to the study was enrolled with no exclusion criteria imposed. This yielded an inclusive, diverse and complex cohort where most had a chronic migraine diagnosis (88.9%) with high baseline headache frequency (median MHD 30 with MMD 15) where daily headache was present in 58.9%, acute medication overuse in 35.6%, prior preventive refractoriness in 45.6%, and 68.9% being co-treated with one to three other preventive agents, of whom half were receiving onabotulinumtoxinA, together with erenumab therapy.
Erenumab responder rate and treatment adherence
Erenumab responders (i.e. at least 30% response rate) constituted 62.2% of our 90 study participants, which is generally comparable to the treatment success rates found in the hallmark trials, accounting for the difference in methodology where 50% responder rate was commonly reported by others (of note, our 50% responder rate in supplementary analysis was 53.3%). Of all who started erenumab in our practice, 56.0% continued past 12 months. It is well recognized that 12-month adherence to oral preventive therapies is considerably lower. 2,3 One recent descriptive study on “real-world” erenumab experience conducted over a 6-month treatment period, reported 27.7% participant dropout rate before completing 6 months of treatment. 9 In the above study with a complex patient population, akin ours, 54.8% patients had at least 50% reduction in MMD as a measure of erenumab response. 9 The top three reasons for discontinuing erenumab therapy in our cohort were lack of desired response, difficulty securing drug coverage, and adverse effects, in this descending order. These are similar to findings reported by other investigators 9 with the exception of drug coverage issues, which have posed a major barrier to continued treatment in Canada past the initial complimentary trial period.
Predictors of erenumab response
With interest, we found that patients with mixed headache syndromes that included CM with atypical features or overlap with TAC, PTH, POTS, IIH, NDPH, CGH, SIH/RIH were less likely to respond to erenumab, and this association remained significant after adjusting for possible refractoriness (i.e. prior preventive failures) of these headaches to therapies in general. This requires further understanding as CGRP has also been implicated in the pathogenesis of certain non-migraine headaches, particularly PTH, 20 IIH, 21 and cluster headache. 22 Preliminary data on erenumab has possibly shown some promise in the above three syndromes, 20 –22 though conclusions cannot be fully drawn for most of these studies due to methodological limitations in the open label studies or case series. Randomized and double-blind clinical trials are required in these conditions to better understand the efficacy of CGRP targeting therapies.
Replicating a recent report that suggested a good response to erenumab (30% response rate in 49% of patients at 3 months) in refractory chronic migraine with multiple preventive therapies failures (a mean of 8.4 agents), 23 we redemonstrate the 30% response rate in 48.8% (20/41) of refractory cases that we defined as 5 or greater unique preventive failures (based on a recently proposed criteria for redefining refractoriness 18 ). On the whole though, our erenumab responders had lower preventive failures compared to non-responders (a median of 3 versus 5.5 agents) and a lower migraine chronicity overall (in numbers of headache/migraine days per month and daily headache, but not the disease duration). These latter results precisely agree with the newly published data on predictors of sustained efficacy of erenumab after a 12-month treatment period. 24 Of interest, prior response to onabotulinumtoxinA did not appear to correlate with the response to erenumab despite the plausible mechanism of action within the CGRP pathway, and synergistic clinical effect of the dual therapy. 25 Some explain this by the selective inhibitory actions of these two drugs on different nociceptive nerve fibers (C- and Aδ-fibers, respectively). 26,27 Similarly, when examining association between response to triptans and erenumab, although we observed more triptan responders among erenumab responders and vice versa, it was not statistically significant. On the other hand, one study found an OR of 3.64 (95% CI = 1.25–10.64) for erenumab responders who responded to triptans, compared to those who did not, although their study was limited by a small size with high proportion of both triptan and erenumab responders. 28
Study limitations
The results of our study might be limited by the retrospective design that carries an inherent likelihood of incomplete data, which was further impacted by the shift to virtual care in the context of the pandemic that affected regular charting of headache diaries and disability questionnaires. Another disadvantage is a relatively small sample size, possibly underpowering our research and limiting multivariate analyses. In addition, this exploratory work has identified an association rather than causality between patient variables and erenumab response, which nonetheless provides important preliminary data and a foundation for further prospective studies. Lastly, CHAMP physicians are headache fellowship-trained neurologists highly experienced in treating migraine, while the CHAMP population of migraineurs, being limited to a single center, is more complex, with a higher burden of disease, relative to the community population of migraine patients. This may affect the generalizability of the study results, however, imparts valuable insight into the potential factors affecting treatment response in more complex migraine patients. In addition, this is the first published Canadian experience and perspective on this topic to our knowledge.
Conclusion
In this retrospective observational study based on a complex tertiary clinic cohort of patients with migraine treated by headache neurologists, we demonstrate that non-response to erenumab is potentially associated with the following individual factors: higher rate of unemployment, complex/atypical chronic migraine diagnosis, higher monthly headache/migraine frequency, including percentage of chronic daily headache, and higher number of preventive therapy failures. Larger prospective research studies could build on this pilot work to determine stronger, causal inferences predictive of response to erenumab, in an ultimate move toward precision medicine and efforts at reducing numbers of treatment non-responders overall.
Clinical Implications
Efficacy of erenumab for migraine prevention ranges from super- to no-response, where further understanding may help individualize therapies.
In this study, erenumab responsiveness (≥30% reduction in monthly headache or migraine days) was associated with a higher rate of employment, lower migraine frequency, including prevalence of chronic daily headache, and fewer prior preventive failures. However, about 50% of refractory patients still responded to erenumab.
Supplemental material
Supplemental Material, sj-docx-1-rep-10.1177_25158163221128185 - Predictors of clinical response to erenumab in patients with migraine
Supplemental Material, sj-docx-1-rep-10.1177_25158163221128185 for Predictors of clinical response to erenumab in patients with migraine by Olga Lekontseva, Meng Wang and Farnaz Amoozegar in Cephalalgia Reports
Footnotes
Abbreviations
Acknowledgments
Thank you to CHAMP physicians (Drs Lara Cooke and Jeptha Davenport) and administrative/nursing/research support staff (Lisa LeBlanc, Caryn Diaper, Kristin Hayes and Dorota Biggs) who kindly agreed for their patients to be considered for inclusion in the study and supported the study process wherever was needed.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FA has received speaker’s honoraria and research support from Novartis, Eli Lilly, Teva, Abbvie, and Lundbeck.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. OL received a 1-year fellowship support from Canadian Headache Society and Frontiers in Headache Research Award from American Headache Society for this project proposal.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
