Abstract
Background
Calcitonin gene-related peptide plays an important role in migraine pathophysiology. Erenumab, a human monoclonal antibody that inhibits the calcitonin gene-related peptide receptor, is being evaluated for migraine prevention.
Methods
In this randomized, double-blind, placebo-controlled, phase 3 study, 577 adults with episodic migraine were randomized to placebo or 70 mg erenumab; 570 patients were included in efficacy analyses. Primary endpoint was change in monthly migraine days. Secondary endpoints were ≥50% reduction in monthly migraine days, change in acute migraine-specific medication treatment days, and ≥5-point reduction in Physical Impairment and Impact on Everyday Activities domain scores measured by the Migraine Physical Function Impact Diary. All endpoints assessed change from baseline at month 3.
Results
Patients receiving erenumab experienced −2.9 days change in monthly migraine days, compared with −1.8 days for placebo, least-squares mean (95% CI) treatment difference of −1.0 (−1.6, −0.5) (p < 0.001). A ≥ 50% reduction in monthly migraine days was achieved by 39.7% (erenumab) and 29.5% (placebo) of patients (OR:1.59 (95% CI: 1.12, 2.27) (p = 0.010). Migraine-specific medication treatment days were reduced by −1.2 (erenumab) and −0.6 (placebo) days, a treatment difference of −0.6 (−1.0, −0.2) (p = 0.002). The ≥5-point reduction rates in Migraine Physical Function Impact Diary – Physical Impairment were 33.0% and 27.1% (OR:1.33 (0.92, 1.90) (p = 0.13) and in Migraine Physical Function Impact Diary – Everyday Activities were 40.4% and 35.8% (OR:1.22 (0.87, 1.71) (p = 0.26). Safety and adverse event profiles of erenumab were similar to placebo. Most frequent adverse events were upper respiratory tract infection, injection site pain, and nasopharyngitis.
Conclusions
As a preventive treatment of episodic migraine, erenumab at a dosage of 70 mg monthly significantly reduced migraine frequency and acute migraine-specific medication use. (Funded by Amgen).
Trial registration
ClinicalTrials.gov, NCT02483585.
Introduction
Despite the availability of several oral preventive therapies for migraine, patients often discontinue treatment due to lack of efficacy or poor tolerability (1–5). Moreover, most of the drugs used in preventive treatment of migraine were initially designed for other purposes, reflecting the need for newer migraine-specific therapeutic agents. Calcitonin gene-related peptide (CGRP) is a neuropeptide implicated in migraine pathophysiology (6–10). Ample clinical evidence shows that CGRP and the CGRP receptor are suitable targets for the acute and preventive treatment of migraine (6,11–15). Erenumab (AMG 334) is a fully human monoclonal antibody that targets and blocks the canonical CGRP receptor (16,17).
ARISE (
Methods
Trial population
Adults (18 to 65 years old) with a history of EM (≥4 to <15 migraine days per month and < 15 headache days per month) (18) with or without aura for at least 12 months prior to the study were eligible. Patients older than 50 years of age at migraine onset, with a history of hemiplegic migraine or cluster headache, were excluded. Patients with prior treatment failures (based on efficacy or tolerability) were allowed in the study except when there was no therapeutic response to more than two classes of migraine prevention treatments (defined as an adequate trial of at least 6 weeks at the recommended dose without any benefit in terms of frequency, duration, or severity of the attacks, according to the investigator’s opinion). In the initial protocol, medications with possible migraine preventive effects were prohibited, though patients who had received preventive drugs could participate following washout for at least 2 months prior to baseline or 4 months for botulinum toxin (Supplementary Appendix). Subsequently, a protocol amendment allowed for concomitant use of only one migraine preventive medication if the dose was stable within two months before the baseline phase and was to remain stable throughout the study. Patients with medical conditions that might prevent study completion or interfere with interpretation of results were excluded (Supplementary Appendix). Women of child bearing potential had to use an effective contraceptive method.
Trial oversight
Institutional review boards at each center approved the study protocol. All patients provided written informed consent. Sites maintained compliance with the Health Insurance Portability and Accountability Act or relevant regional regulations. The study was conducted in accordance with the International Conference on Harmonisation Tripartite Guideline on Good Clinical Practice. An independent data monitoring committee reviewed and made recommendations regarding the safety of study participants throughout the double-blind treatment phase and until treatment assignment information was available to the study team for the primary analysis. Site investigators collected the data and Amgen conducted the data analyses according to a pre-specified statistical analysis plan. All authors interpreted the data and collaborated in manuscript preparation with support from a professional medical writer, funded by Amgen. All authors made the decision to submit the manuscript and attested to the veracity and completeness of data and analyses for their respective studies and the fidelity of this report to the study protocol. Amgen funded the study. A copy of the protocol is available at https://www.clinicaltrialsregister.eu EudraCT number 2014-004463-20.
Trial design
ARISE was a multicenter, randomized, double-blind, placebo-controlled, parallel-group, phase 3 trial conducted at 69 sites across North America and Europe (including Russia) from July 2015 (first patient randomized) to July 2016 (data cutoff date for primary analysis or primary completion date). The study was conducted in headache centers, neurology clinics, and clinical research sites. Eligible patients were recruited into the study by participating sites, where subjects were regularly followed for headache, and via advertisement.
The study comprised a screening phase (up to 3 weeks), a 4-week baseline phase, a 12-week double-blind treatment phase, and a 28-week open-label treatment phase. A safety follow-up visit was completed 12 weeks after last dose of investigational product. Primary results from the double-blind treatment phase of the study are reported here. Results from the open-label treatment and safety follow-up phases have not been completed and will be reported separately.
Patients were randomized 1:1 to erenumab 70 mg or placebo (monthly subcutaneous injection). Randomization was based on a computer-generated schedule created by the sponsor before study initiation and was centrally executed using an interactive response system. Eligibility was evaluated and confirmed by the principal investigator. Randomization was stratified by region (North America vs. other) and preventive migraine medication status ((a) current migraine preventive treatment; (b) prior migraine preventive treatment only; (c) no prior or current migraine preventive treatments). Patients, site personnel, and study sponsor personnel were blinded to treatment group assignment during the double-blind treatment phase. Erenumab and placebo were packaged identically in size and color to maintain the blind. At week 12, all patients continuing into the open-label treatment phase were to receive open-label 70 mg erenumab.
Assessments and safety evaluations
We conducted efficacy assessments throughout the baseline and double-blind phase of the study, with key assessments at week 12 (during the last month of the double-blind treatment phase). Patients used an electronic headache diary every day throughout the baseline phase and double-blind treatment phase to report information about their migraine and non-migraine headaches and acute headache medication use, including date and time of onset and resolution of headache, headache pain severity and pain features, associated symptoms, use of acute headache medication, and patient-reported outcomes (PRO) via the Migraine Physical Function Impact Diary (MPFID) (19). Additional PRO measures (including Headache Impact Test [HIT-6T] (20,21), Modified Migraine Disability Assessment [mMIDAS; measured as 4-week recall period vs. 3-month recall of standard MIDAS] (22), and Migraine-Specific Quality-of-Life Questionnaire [MSQ] (23,24)) were collected at pre-specified assessment times. We evaluated safety by monitoring adverse events (AEs) (Medical Dictionary for Regulatory Activities v19.0) (25), serious AEs (26), laboratory assessments, vital signs, electrocardiograms, and anti-erenumab antibodies.
Objectives and endpoints
The primary objective was to confirm the effect of erenumab compared to placebo for the primary endpoint of change from baseline in monthly migraine days (MMD) in the last month (month 3) of the double-blind treatment phase. A migraine day was defined as any calendar day recorded in the electronic headache diary on which the patient had an onset, continuation, or recurrence of a qualified migraine, defined as a migraine (with or without aura) lasting ≥ 30 minutes and presenting with at least two pain features (unilateral, throbbing, moderate to severe, or aggravated by physical activity) and/or at least one associated non-pain feature (nausea and/or vomiting, or both photophobia and phonophobia). Use of acute migraine-specific medication (triptans or ergotamine derivatives) on a calendar day was counted as a migraine day, irrespective of the duration of the migraine. Prespecified secondary endpoints included achievement of ≥50% reduction from baseline in MMD, change from baseline in monthly acute migraine-specific medication treatment days (MSMD), achievement of at least a 5-point reduction in monthly average Physical Impairment domain score as measured by the Migraine Physical Function Impact Diary (MPFID-PI), and achievement of at least a 5-point reduction in monthly average Impact on Everyday Activities (MPFID-EA) domain score. Exploratory PRO endpoints included change from baseline in HIT-6, modified (monthly) MIDAS, MSQ, and MPFID. All endpoints were assessed over the last month (month 3) of the double-blind treatment phase.
Statistical analysis
Sample size was based on the primary endpoint of change from baseline in monthly migraine days in the last month of the double-blind treatment phase. Assuming a treatment effect compared to placebo of −1.12 for the erenumab 70 mg group and a common standard deviation of 3.78, the planned sample size of 270 subjects per group would provide 90% power using a two-sample t-test with a 2-sided significance level of 0.05. A 10% dropout rate was assumed. The assumed treatment effect of erenumab 70 mg was based on the mean difference compared to placebo and common standard deviation observed in the phase 2 trial (12).
Primary analyses were conducted after all patients completed the 12-week visit or prematurely terminated the study before the 12-week visit. The statistical significance levels of the primary and secondary endpoints were determined after multiplicity adjustment using a pre-specified hierarchical gate-keeping procedure and Hochberg-based testing procedures to maintain the 2-sided study-wise type I error rate at α = 0.05 (Supplementary Appendix). p values that were considered statistically significant are described as such in text and identified in tables. p values for exploratory endpoints were nominal and were not adjusted for multiple comparisons. Continuous variables were analyzed using a linear mixed effects model including treatment group, baseline value, stratification factors (region and preventive migraine medication status), scheduled visit, and the interaction of treatment group with scheduled visit, without any imputation for missing data. Dichotomous variables were analyzed using a stratified Cochran-Mantel-Haenszel test and using non-responder imputation, in which missing data was assumed to be non-response. The full analysis set included all patients who were randomized in the study. The efficacy analysis set included all patients who received at least one dose of randomized treatment and had at least one change from baseline measurement for MMD during the double-blind treatment phase, analyzed according to randomized treatment. The safety-analysis set, which included all randomized patients who received one or more doses of investigational product, was used to analyze adverse event incidence rates according to randomized treatment group unless a patient received an incorrect dose throughout the entire double-blind treatment phase.
Results
Patients
Demographics and baseline characteristics of the patients*.

Plus-minus values are means ± standard deviation.
Race was self-reported.
The body-mass index is the weight in kilograms divided by the square of the height in meters.
Failure due to lack of efficacy or poor tolerability.
MPFID scores range from 0–100, with higher scores indicating greater impact.
MPFID: Migraine Physical Function Impact Diary.
Primary efficacy endpoint
Primary and secondary endpoints at week 12*.

Data are least-squares means ± SE unless otherwise indicated. N values are the number of patients who were randomized and had at least one change from baseline measurement in monthly migraine day during the double-blind phase.
p values for pairwise comparison are unadjusted. Primary and secondary endpoints were tested with a hierarchical testing procedure with a prespecified order.
Primary and first tier secondary endpoints were significant after multiplicity adjustment.
The adjusted odds ratios and p values are obtained from a Cochran-Mantel-Haenszel (CMH) test after the missing data are imputed as non-response, stratified by stratification factors region and prophylactic migraine medication status.
CI: confidence interval; MPFID: Migraine Physical Function Impact Diary.
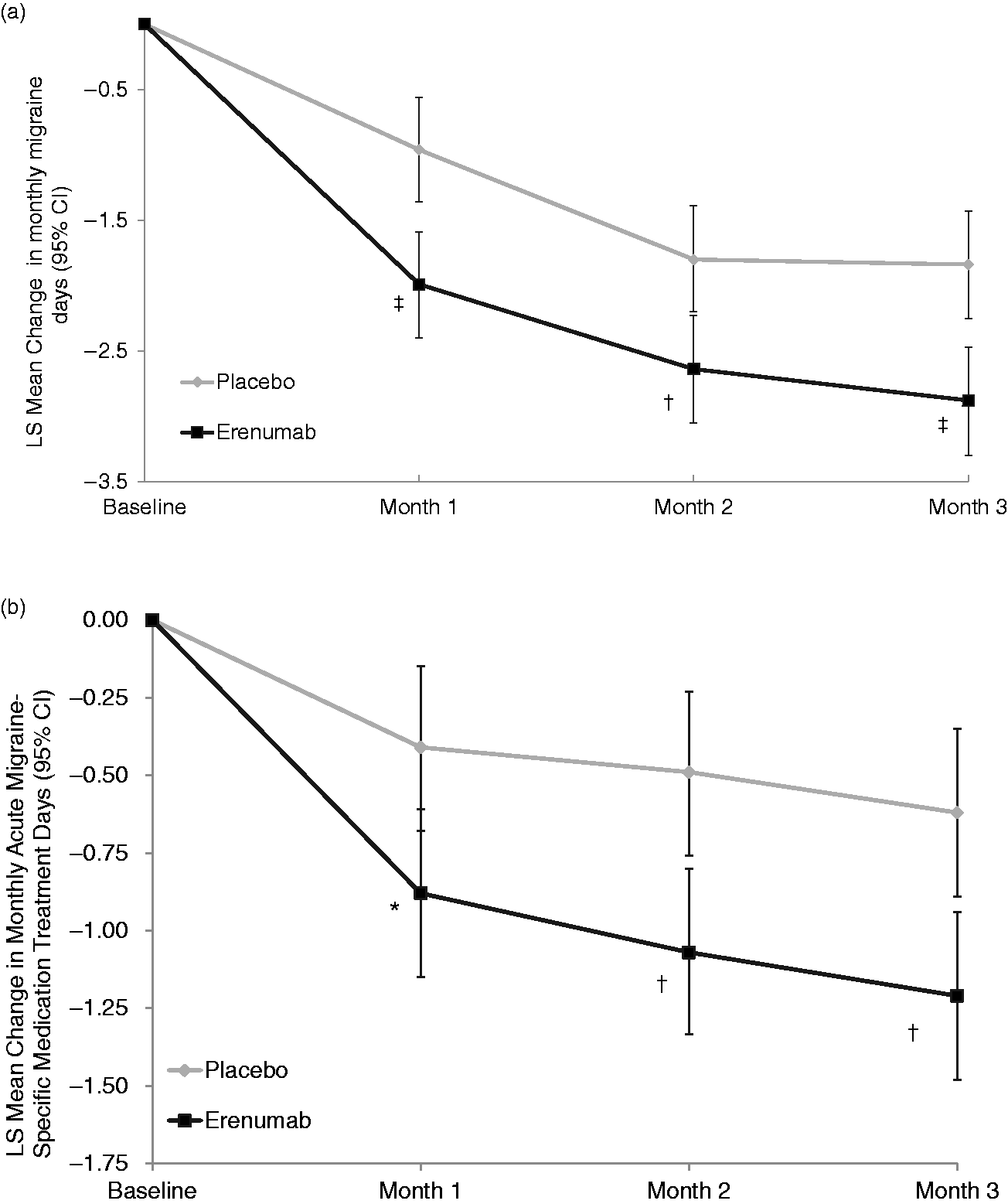
Efficacy over time. Least-squares mean changes from baseline in monthly migraine days (MMD) (Panel a) and monthly acute migraine-specific medication treatment days (MSMD) (Panel b) among patients with episodic migraine who were assigned to receive erenumab 70 mg or placebo every month. The error bars represent 95% confidence intervals (95% CI). Figure based on the efficacy analysis set. *p < 0.05, †p < 0.005, ‡p < 0.001 for erenumab vs. placebo (statistical significance established for month 3).
Secondary efficacy endpoints
Erenumab 70 mg was statistically superior to placebo for the key secondary endpoints of at least a 50% reduction in MMD and change in monthly MSMD (Table 2; p ≤ 0.01 for both) (Figures 1 and 2). A ≥ 50% reduction in MMD was achieved by 39.7% and 29.5% in the erenumab and placebo groups (odds ratio: 1.59; p = 0.010). Monthly MSMD was reduced by LSM −1.2 and −0.6 medication days, respectively (p = 0.002). In an ad hoc assessment among only those patients with baseline acute migraine-specific medication use, monthly MSMD was reduced by LSM −2.1 and −1.2, respectively (p = 0.002).
Responder rates over time. Proportions of patients who achieved ≥ 50% reduction in monthly migraine days (MMD) from baseline during the double-blind treatment phase. Data were analyzed using imputation of missing data as non-response. Figure based on the efficacy analysis set. *p < 0.05 (statistical significance established for month 3).
Patient-reported outcome endpoints
With respect to patient-reported impact of migraine, differences in the proportion of patients who achieved ≥5-point reductions from baseline (improvement) in the two domains of the MPFID (pre-specified secondary endpoints) were not statistically significant between erenumab 70 mg and placebo (Table 2; p > 0.05). Respective ≥5-point reductions (improvement) in MPFID-PI in erenumab and placebo groups were achieved by 33.0% and 27.1% of patients (p = 0.13) and in MPFID-EA by 40.4% and 35.8% of patients (p = 0.26).
Prespecified, exploratory endpoints evaluated a series of PRO measures and their changes from baseline at month 3. Erenumab treatment was associated with improvements in health-related quality of life and clinically meaningful reductions in disability and headache impact when assessed using standard PRO instruments (Figure 3 and Table 3). At week 12, improvements in scores were greater in the erenumab group than in placebo in all three domains of the MSQ (role function-restrictive [MSQ-RFR], role function-preventive [MSQ-RFP], and emotional functioning [MSQ-EF]) (increase in scores indicates improvement) and HIT-6 (reduction in scores indicates improvement) (Figure 3 and Table 3) with treatment differences exceeding the established group-level minimally important difference (MID) for MSQ-RFR (≥3.2) and HIT-6 total score (≥1.5 points). Reductions from baseline to week 12 in modified (monthly) MIDAS total and presenteeism scores (for which no MID presently exists) were greater in the erenumab group as compared with placebo indicating greater improvement (Table 3). Reductions (improvement) from baseline to week 12 in the continuous MPFID-EA and MPFID-PI domain scores were numerically greater with erenumab 70 mg than placebo (Figure 3 and Table 3).
Patient-reported outcomes over time. Least-squares mean changes from baseline in MSQ-RFR, MSQ-RFP, and MSQ-EF scores (Panel a), HIT-6 total scores (Panel b), and MPFID-PI and MPFID-EA scores (Panel c) among patients with episodic migraine who were assigned to receive erenumab 70 mg or placebo every month. The error bars represent 95% confidence intervals (95% CI). Figure based on the efficacy analysis set. Exploratory PRO endpoints at week 12*. Data are least-squares means ± standard error unless otherwise indicated. Efficacy analysis set. p values for pairwise comparison are unadjusted. MPFID: Migraine Physical Function Impact Diary; HIT-6: Headache Impact Test; mMIDAS: Modified Migraine Disability Assessment; MSQ-RFR: Migraine-Specific Quality-of-Life Questionnaire role function-restrictive; MSQ-RFP: Migraine-Specific Quality-of-Life Questionnaire role function-preventive; MSQ-EF: Migraine-Specific Quality-of-Life Questionnaire emotional functioning; CI: confidence interval; HIT-6: Headache Impact Test-6; mMIDAS: modified Migraine Disability Assessment; MPFID: Migraine Physical Function Impact Diary; MSQ-EF: Migraine-Specific Quality-of-Life Questionnaire – emotional function; MSQ-RFP: Migraine-Specific Quality-of-Life Questionnaire – role function-preventive; MSQ-RFR: Migraine-Specific Quality-of-Life Questionnaire – role function-restrictive; PRO: patient-reported outcome.

Safety
Adverse events through week 12.

A serious adverse event was defined as an event that was fatal or life threatening, required inpatient hospitalization or prolongation of existing hospitalization, caused persistent or substantial disability or incapacity, caused congenital anomaly or birth defect, or was considered by the investigator to be medically important. Serious adverse events, all single events, were migraine, cholecystitis acute, flank pain, hypersensitivity, hyponatremia, and uterine leiomyoma in the placebo group and migraine, intervertebral disc protrusion, and urinary tract infection in the erenumab group.
Adverse events leading to discontinuation of study drug were irritable bowel syndrome, fatigue, allergy to arthropod sting, affect lability, and mechanical urticaria.
The severity of adverse events was graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.03. No adverse events greater than grade 3 were reported.
Discussion
Erenumab treatment resulted in reduction in migraine frequency, use of acute migraine abortive medications, disability, and headache impact. Erenumab demonstrated superior efficacy to placebo for the primary endpoint (change from baseline in MMD at month 3) at a dose of 70 mg monthly, which had been established as the minimally effective dose in the EM population (14). In addition to the reduction in MMD, erenumab treatment resulted in reduced MSMD (triptans and ergotamine derivatives) and significantly greater odds of achieving at least a 50% reduction in MMD compared with placebo at month 3. The treatment effects on reduction of MMD and MSMD were apparent from the first planned time point (4 weeks) of analysis and were maintained throughout the double-blind phase (Figure 1). The least-squares difference versus placebo in monthly MSMD at month 3 (−0.6 days) was based on the total study population, which included 39% of patients who did not use acute migraine-specific medication during the baseline phase (Table 1) and therefore may underestimate the effect of erenumab in reducing the consumption of acute migraine-specific medication. When only users of acute migraine-specific medications at baseline were considered, the actual change from baseline in MSMD was −0.92 days.
These efficacy results are consistent with the results of a phase 2 erenumab study in EM testing doses as high as 70 mg, which showed a reduction of 3.4 MMD in the 70 mg erenumab group compared with 2.3 days for placebo (a placebo-adjusted reduction from baseline of 1.1 days) (14). In the STRIVE phase 3 trial in EM, which tested 70 mg and 140 mg erenumab doses, at 3 months there was a reduction in MMD of 3.0, 3.5, and 1.7 in the erenumab 70 mg, 140 mg, and placebo groups respectively (a placebo-adjusted reduction from baseline of 1.3 and 1.8 days) (27). The STRIVE trial had a 6-month double-blind phase, with reductions in MMD in the last 3 months of the 6-month double-blind treatment phase (i.e. the average over months 4 to 6) of 3.2 and 3.7 days in the erenumab 70 and 140 mg groups compared with 1.8 days for placebo (27). The magnitude of these placebo-adjusted treatment effects is dependent in part on the placebo response rates, which are known to be high in clinical trials of patients with migraine (28,29).
To help determine the meaningfulness of the treatment difference in reduced migraine frequency, it is important to measure patient-reported assessments of migraine impact to provide insight into patient perception of erenumab’s treatment effect. Supporting the patient perspective of the response to erenumab, the observed reductions in MMD were accompanied by reductions in the patient-reported outcomes assessed by standard PRO instruments such as HIT-6, modified (monthly) MIDAS, and MSQ. The differences in MSQ-RFR and HIT-6 total score exceeded the established group-level minimally important difference (MID). These PRO results underline the benefit obtained with erenumab compared to placebo in terms of meaningful improvements in terms of headache impact.
The MPFID is a novel migraine-specific instrument developed and validated to assess the impact of migraine on physical function, both on days with and without migraine. In contrast to standard headache PRO instruments, it is assessed daily with an electronic diary, and for any given month an average score is derived for each patient. Work is ongoing to establish the MID for change in MPFID domain scores. As an initial assessment, the current study used a categorical analysis based on a 5-point threshold change in MPFID-EA or MPFID-PI domain scores. However, a high proportion of the patients in the study population had scores <5 at baseline, limiting the sensitivity of the analysis. The results of the analysis assessed as continuous scores are consistent with the results in STRIVE, in which the mean between-group changes in MPFID domain scores were statistically significant (secondary endpoints in that study) (27). Further work is needed to determine the optimal approach to assessing change on the MPFID.
Our decision to use a dosage of 70 mg erenumab monthly in this study was based on the results of our phase 2 study in EM (14). A higher dosage of erenumab was examined in the STRIVE phase 3 pivotal study, which assessed both 70 mg and 140 mg monthly erenumab treatment over 6 months, and additional efficacy was apparent with the higher 140 mg dosage across the primary and secondary endpoints, and at all time points (27).
Overall, the incidence of adverse events was similar in the erenumab and placebo groups, and these analyses did not suggest any particular safety risk with erenumab administration. In line with the good safety and tolerability profile, the double-blind treatment phase completion rate was almost 95%, considerably higher than those observed for oral preventive migraine treatments, such as topiramate (30,31). As expected, given the non-hepatic metabolism of monoclonal antibodies, erenumab treatment did not result in any observable effect on liver enzymes, in contrast to the hepatotoxicity observed with small molecule CGRP receptor antagonists such as telcagepant (MK-0974) (32), which is thought to be due to off-target effects of the small molecules and not due to CGRP receptor inhibition. The rate of binding antibodies against erenumab was low (4.3% of subjects exposed) and only one subject (0.4%) was transiently positive for neutralizing antibodies (testing negative at the end of the study). Data from other studies with erenumab similarly indicate that anti-erenumab antibodies occur infrequently, do not increase over time, and remit in the majority of cases (14,15,27). Given the low frequency of anti-erenumab antibodies in individual studies and their frequently transient nature, the incidence and potential impact of anti-erenumab antibodies will be the subject of a future publication, using pooled data across erenumab studies.
Because CGRP is among a number of mediators with vasodilatory properties (e.g. substance P, neurokinins, nitric oxide) released during ischemia (33), there is a theoretical risk that patients with cardiovascular conditions might be at increased risk with inhibition of the CGRP pathway. However, during the 12-week double-blind phase, the occurrence of cardiovascular adverse events was low and did not substantially differ between erenumab and placebo groups (0.4% vs. 1.0%). These results supplement those from a placebo-controlled study of erenumab in a high risk population of patients with stable angina with a median age of 65 years, in which inhibition of the canonical CGRP receptor with erenumab did not adversely affect total exercise time in a treadmill test, among other safety endpoints (34). A lack of cardiovascular effects with inhibition of the CGRP receptor is also supported by studies with small-molecule CGRP receptor antagonists in which there is no evidence of vasoconstriction after CGRP receptor blockade when used for acute migraine relief (35,36). More data from migraine patients with comorbid cardiovascular conditions in a real-world setting may help further assess the theoretical cardiovascular risk of blocking the CGRP pathway.
With 69 sites in eight countries in North America and Europe, the ARISE study included a large, diverse population representative of the population with EM in these regions. However, further studies in a real-world setting may be required to determine the efficacy of erenumab in more diverse patient populations, including patients with no therapeutic response to multiple preventive medication categories who were not allowed into this study. Beyond the 3-month tolerability and safety data presented here, the 28-week open-label extension phase of this study will provide additional mid-term safety data on patients treated up to 40 weeks during the study. However, longer follow-up periods in large numbers of patient will be necessary to detect rare AEs and fully understand the safety profile of erenumab. Lastly, durability of the treatment effect will require longer studies, as this is not adequately assessed in a 3-month study.
There remains a considerable unmet need for safe and well-tolerated treatment options for the prevention of migraine that are more effective and have better tolerability than currently available preventive treatment options, as this can enable treatment adherence.
These results, together with other phase 2 and 3 studies of erenumab in episodic and chronic migraine (14,37), support the use of erenumab as a migraine preventive treatment. Compared to currently available therapies, the good safety and tolerability profile of erenumab may help promote better adherence to treatment in a condition with high unmet needs.
Clinical implications
Erenumab is a fully human monoclonal antibody that targets and blocks the canonical CGRP receptor. Erenumab treatment resulted in reductions in monthly migraine days, increases in the proportion of patients achieving at least a 50% reduction from baseline in monthly migraine days, and reductions in the number of days per month that acute migraine-specific drugs were used. This phase 3 study provides evidence that once-monthly subcutaneous injection of erenumab is a potential new preventive therapy in patients with episodic migraine.
Supplemental Material
Supplemental material for ARISE: A Phase 3 randomized trial of erenumab for episodic migraine
Supplemental material for ARISE: A Phase 3 randomized trial of erenumab for episodic migraine by David W Dodick, Messoud Ashina, Jan Lewis Brandes, David Kudrow, Michel Lanteri-Minet, Vera Osipova, Kerry Palmer, Hernan Picard, Daniel D Mikol and Robert A Lenz in Cephalalgia
Supplemental Material
Supplemental Figures - Supplemental material for ARISE: A Phase 3 randomized trial of erenumab for episodic migraine
Supplemental material, Supplemental Figures for ARISE: A Phase 3 randomized trial of erenumab for episodic migraine by David W Dodick, Messoud Ashina, Jan Lewis Brandes, David Kudrow, Michel Lanteri-Minet, Vera Osipova, Kerry Palmer, Hernan Picard, Daniel D Mikol and Robert A Lenz in Cephalalgia
Footnotes
Acknowledgements
The ARISE trial was funded by Amgen. Erenumab is co-developed in partnership with Amgen and Novartis. We thank the study investigators for the ARISE trial and patients for their participation and commitment to this work. Medical writing support was provided by Jon Nilsen, PhD, of Amgen.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David W Dodick has served and has been compensated for his role on advisory boards and/or has consulted within the past 5 years for Allergan, Amgen, Alder, Arteaus, Pfizer, Colucid, Merck, NuPathe, Eli Lilly and Company, Autonomic Technologies, Ethicon J&J, Zogenix, Supernus, Labrys, Boston Scientific, Medtronic, St Jude, Bristol-Myers Squibb, Lundbeck, Impax, MAP, Electrocore, Tonix, Novartis, Teva, Alcobra, Zosano, Insys, GBS/Nocira, Acorda, eNeura, Charleston Laboratories, Gore, Biohaven, Bioventric, Magellan, Theranica, Xenon, Dr Reddy’s/Promius Pharma. Dr Dodick owns equity in Epien, GBS/Nocira, Second Opinion, Healint, and Theranica. Dr Dodick has received funding for travel, speaking, editorial activities, or royalty payments from IntraMed, SAGE Publishing, Sun Pharma, Allergan, Oxford University Press, American Academy of Neurology, American Headache Society, West Virginia University Foundation, Canadian Headache Society, Healthlogix, Universal Meeting Management, WebMD, UptoDate, Medscape, Oregon Health Science Center, Starr Clinical, Decision Resources, Synergy, MedNet LLC, Peer View Institute for Medical Education, Medicom, Chameleon Communications, Academy for Continued Healthcare Learning, Haymarket Medical Education, Global Scientific Communications, HealthLogix, Miller Medical, MeetingLogiX, Wiley Blackwell, SAGE publishing, Cambridge University Press. Consulting use agreement: NeuroAssessment Systems, Myndshft. Board position: King-Devick Technologies, Inc. Epien Inc. Patent 17189376.1-1466: Title: Botulinum Toxin Dosage Regimen for Chronic Migraine Prophylaxis. Messoud Ashina: Personal fees from Alder BioPharmaceuticals, Allergan, Amgen, Alder, ATI, Eli Lilly, Novartis and Teva. Principal investigator for Alder ALD403-CLIN-011 (Phase 3b), Amgen 20120178 (Phase 2), 20120295 (Phase 2), 20130255 (OLE), 20120297 (Phase 3), GM-11 gamma-Core-R trials, Novartis CAMG334a2301 (Phase 3b), and PAC1 20150308 (Phase 2a). Jan Lewis Brandes: Research grants from Allergan, Amgen, Clinivest, Teva, CoLucid, Zozano, Arteus; Consulting fees from Amgen, Supernus, Promius; Speakers’ bureau fees from Amgen, Supernus, Promius, Depomed, Pernix, Teva, Avanir; Advisory board for Amgen, Avanir, Supernus, Teva, Lilly, Promius. David Kudrow: Personal fees from Amgen, Ely Lilly, Novartis, Alder. Research support from Alder, Ely Lilly, Amgen, Teva, Allergan, CoLucid, Zosano, Genentech-Roche, VMBiopharma, UCB.
Michel Lanteri-Minet: Received honoraria for advisory boards, speaker panels or investigation studies from Allergan, Amgen, Astellas, ATI, BMS, Boehringer, Boston Scientific, CoLucid, Convergence, Glaxo-SmithKline, Grunenthal, Lilly, Medtronic, Menarini, MSD, Novartis, Pfizer, ReckittBenckiser, Saint-Jude, Sanofi-Aventis, Teva, UCB, Zambon. Vera Osipova: Personal fees from Allergan, Pfizer and Takeda. Principal investigator for Amgen 20120297 (Phase 2). Kerry Palmer, Hernan Picard, Daniel D Mikol, and Robert A Lenz are employees and stockholders of Amgen Inc.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
