Abstract
Background
Migraine patients unresponsive to calcitonin gene-related peptide (CGRP)(-receptor, -R) monoclonal antibodies (mAbs) may benefit from switching between CGRP(-R) mAbs. However, some patients do not tolerate or respond to any subcutaneous mAbs. This study evaluates the efficacy of the intravenous CGRP mAb eptinezumab in these therapy-refractory patients.
Methods
In this retrospective cohort study, patients with migraine who previously failed erenumab and at least one CGRP mAb (fremanezumab and/or galcanezumab) received eptinezumab 100 mg, followed by a second dose of 100 mg or 300 mg after 12 weeks. Monthly headache days, monthly migraine days, acute medication days, and migraine pain intensity were recorded from standardized headache diaries during the four weeks before the first infusion (baseline), and during weeks 9–12 and 21–24 of treatment. Patient-reported outcomes were analyzed at baseline, weeks 12, and 24.
Results
From January 2023 to February 2024, 41 patients received eptinezumab 100 mg. Of these, 38 (93%) received a second infusion after 12 weeks, with 29 (71%) increasing the dose to 300 mg. The percentage of patients with a ≥30% reduction rate in monthly migraine days was 23.1% at week 12 and 29.7% at week 24. Monthly migraine days decreased from 16.3 ± 8.0 at baseline to 15.4 ± 8.1 days during weeks 9–12 and 14.4 ± 8.0 days during weeks 21–24 (p = 0.07). During weeks 21–24, 38.5% reported a clinically meaningful reduction in HIT-6 scores and 52.4% in MIDAS scores. No adverse events were reported.
Conclusions
Eptinezumab may be an effective and well-tolerated option for some treatment-refractory migraine patients unresponsive to subcutaneous CGRP-(R) mAbs.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine is a frequent and disabling primary headache disorder that affects around 15% of the global population (1). Traditional first-line preventative treatments for migraine, such as antihypertensive, antidepressant, and antiseizure medications, were initially developed for other medical conditions and thus lack specificity for migraine. They often exhibit tolerability issues and insufficient efficacy, leading to poor and unpredictable therapy adherence (2,3).
The identification of Calcitonin Gene-Related Peptide (CGRP) as pivotal neuropeptide in migraine pathophysiology has led to the development of CGRP-targeted therapies for the acute and preventive treatment of migraine. In particular, monoclonal antibodies (mAbs) targeting CGRP and its receptor (CGRP-R) were demonstrated to be safe and effective in the preventive treatment of episodic migraine (EM) and chronic migraine (CM) (4,5), even in patients who did not respond to numerous unspecific first-line treatment options (6–10).
In real-world studies, CGRP(-R) mAbs led to a reduction of monthly migraine days (MMDs) of at least 50% in up to 77% of patients (11). Despite these generally favorable results, up to one-third of patients do not benefit from subcutaneous CGRP(-R) mAbs, showing no change or even an increase in migraine frequency (12). Depending on reimbursement- and insurance policies across different countries, patients in this group had typically failed multiple preventive treatments prior to the administration of the first CGRP targeted therapy (mAb), presenting a challenging-to-treat migraine population with limited treatment evidence (13).
Given the limited choices, switching between different CGRP(-R) mAbs classes is considered a suitable option for these patients. Real-world studies show highly variable 30% response rates to these switches. For example, Overeem et al. (14) reported a 32% response rate when switching from erenumab to galcanezumab or fremanezumab, with similar results of 35% in the vice versa switch after three months in a cohort with mainly CM. Ruiz et al. (15) reported a 53% response when switching from erenumab to galcanezumab and Straube et al. (16) 59% when switching from erenumab to fremanezumab in CM (14–17). Analyzing the ≥50% responder rates in both EM and CM after switching across any CGRP(-R) mAbs, Talbot et al. (18) observed 48% responders, while Hong et al. (19) reported a rate of 51% in a German cohort and Suliman et al. (20) 33% in the United Arab Emirates. However, approximately 10% of patients with migraine have an inadequate response to erenumab and galcanezumab and/or fremanezumab as well as all available first-line preventive treatments (21).
The marketing authorization for eptinezumab in the European Union has introduced a new treatment option, also for patients with refractory migraine. Due to the intravenous (IV) formulation, short time to onset of efficacy and distinct biochemical profile compared to the other CGRP mAbs (22,23), eptinezumab offers potential benefits for non-responders to erenumab, galcanezumab, and/or fremanezumab. Eptinezumab is approved for the intravenous treatment for both episodic and CM every three months in a dose of 100 mg or 300 mg (24). Initial results from real-world studies revealed that 40% to 50% of patients achieved a 30% response rate in the third month after eptinezumab treatment (25,26). However, a significant negative association was observed between the response rate and the number of prior preventive treatments (25,27). Consequently, analyzing a larger subgroup of treatment-refractory patients is warranted in order to better understand and address the challenges in this population.
In this study, we aim to assess the efficacy of eptinezumab in patients with migraine who have previously failed both erenumab and at least one subcutaneous CGRP mAb (fremanezumab and/or galcanezumab) after three and six months of treatment.
Methods
Study design and patient selection
This was a monocentric retrospective longitudinal cohort study conducted at the Headache Center, Charité – Universitätsmedizin Berlin, Germany. We screened all patients who received eptinezumab between January 2023 and February 2024 at our outpatient headache center. Inclusion criteria for this analysis comprised a diagnosis of EM or CM according to the criteria of the International Classification of Headache Disorders 3 (ICHD-3) and previous treatment failure with erenumab and at least one subcutaneous CGRP mAb, i.e., fremanezumab, galcanezumab or both (28). Treatment failure was defined as either lack of efficacy (<30% reduction in MMD after three months of treatment according to German guidelines (29)) or poor tolerability as documented in the electronic patient record. ‘Poor tolerability’ in this context refers to any adverse effects or side effects associated with the CGRP mAbs that were significant enough to lead to treatment discontinuation. This includes, but is not limited to, injection site reactions, systemic allergic reactions, or other intolerable side effects that were documented in the electronic patient record.
Patients received a first infusion of eptinezumab 100 mg IV as per label and in accordance with the European Medicines Agency risk management plan (24). The administration of eptinezumab at both 100 mg and 300 mg IV doses is permitted for the first administration in Germany; the official product information recommends an initial dose of 100 mg and the evaluation for dose escalation after 12 weeks (24). After 12 weeks of treatment, patients had the option to increase the dose to 300 mg in case of good tolerability and an insufficient initial response defined as a reduction of ≤30% in MMD, or if they continued to experience a high migraine burden and relevant impact on their quality of life (24).
Definition of variables and data extraction
Demographic characteristics and migraine history (EM or CM, age at diagnosis, duration of the condition, and history of prior prophylactic treatments) were extracted from electronic patient records. We recorded the number of MMDs, monthly headache days (MHDs), monthly days with acute medication use (AMDs) and monthly average migraine pain intensity from standardized paper headache diaries, which we collect at every outpatient visit as part of the clinical routine. A headache day was defined as any day with documented headache. A migraine day was defined by clinical features according the ICHD-3 criteria for migraine or a positive response to triptans.
The ≥30% responder rates for MMDs and MHDs were calculated comparing the four weeks prior to treatment with eptinezumab (baseline) with weeks 9–12 and 21–24 after the first eptinezumab infusion. Patients with a ≥ 30% reduction in MMD during weeks 9–12 or weeks 21–24 were considered responders, while patients not meeting this criterion were classified as non-responders.
In addition, patients completed different questionnaires as part of clinical routine during their visits at baseline, week 12 and week 24. We used the Headache Impact Test (HIT-6) as a well-established measure to assess the effects of headaches on patients’ daily life (30) and the Migraine Disability Assessment (MIDAS) as self-administered questionnaire to quantify migraine-related disability over the last 12 weeks (31). The Depression, Anxiety and Stress Scale (DASS-21) was used to measure the emotional burden of depression, anxiety and stress (32). We used a visual analogue scale (VAS) to evaluate patients’ impressions of change. The scale ranges from 1, indicating ‘very much improved,’ to 10, indicating ‘very much worsened,’ with 5 representing ‘no change’.
Endpoints
The primary endpoint of this investigation was the percentage of patients with a ≥ 30% reduction of MMDs during weeks 9–12 and 21–24 of treatment with eptinezumab compared to baseline. Secondary endpoints were ≥30% response rates for MHDs at week 9–12 and 21–24 as well as the absolute reduction of MMDs, MHDs, AMDs and pain intensity at week 9–12 and 21–24. We further assessed the proportion of patients who discontinued treatment, the proportion of patients who chose to increase the eptinezumab dose for the second infusion, and documented side effects.
Patient-reported outcomes were analyzed as exploratory endpoints, focusing on absolute reductions at week 9–12 and 21–24 as well as percentage of patients achieving a clinically meaningful reduction. This was defined as a reduction of >2.3 points for the HIT-6 and >4.5 points for the MIDAS questionnaire (33,34). A minimum clinically meaningful change in DASS-21 scores was defined in an outpatient population as ≥ 6.0 (35).
Statistical analyses
Due to the retrospective nature of this study, no formal sample size estimation was conducted. Therewith, the sample size was based on the number of eligible patients. Normal distribution of data was assessed with the Kolmogorov–Smirnov test complemented by visual inspection of histograms. Since data of MMDs, MHDs and AMDs as well as patient reported outcome scores were not normally distributed, we used non-parametric tests for analysis. We used Friedman's 2-way ANOVA by ranks test (for k samples) for repeated measurements with all-time points as factors (baseline, weeks 9–12, weeks 21–24). In case of significant main effect, we used the Dunńs pairwise post-hoc test with Bonferroni correction for multiple testing. We used Kendall's W (coefficient of concordance) as a measure of effect size to analyze the level of agreement between subjects, following the interpretation guidelines for Cohen's d. A value of p ≤ 0.05 was considered statistically significant. We report our categorical variables in count (%) and continuous variables in mean ± standard deviation (±SD). All hypothesis tests were two-sided. Statistical analyses were performed with IBM SPSS Statistics, version 29.0 (IBM, Armonk, NY, USA).
Results
From January 2023 to February 2024, 41 patients underwent treatment with eptinezumab 100 mg following subcutaneous CGRP(-R) mAb treatment failures. After 12 weeks, 38 patients (93%) continued eptinezumab treatment, with 29 patients (71%) opting to increase the dose to 300 mg. Two patients (5%) were lost to follow-up, while one patient (2%) discontinued treatment due to lack of efficacy after the first infusion (Figure 1).

Second eptinezumab infusion at week 12.
Patient characteristics
Patient characteristics are summarized in Table 1. The mean age of the cohort was 49.6 ± 12.6 years and 78.0% were female. CM was diagnosed in 85.4% of patients, with 24.4% reporting a daily headache. Patients reported a long history of migraine, with an average disease duration of 27.1 years (±12.0 years).
Study population and headache characteristics.
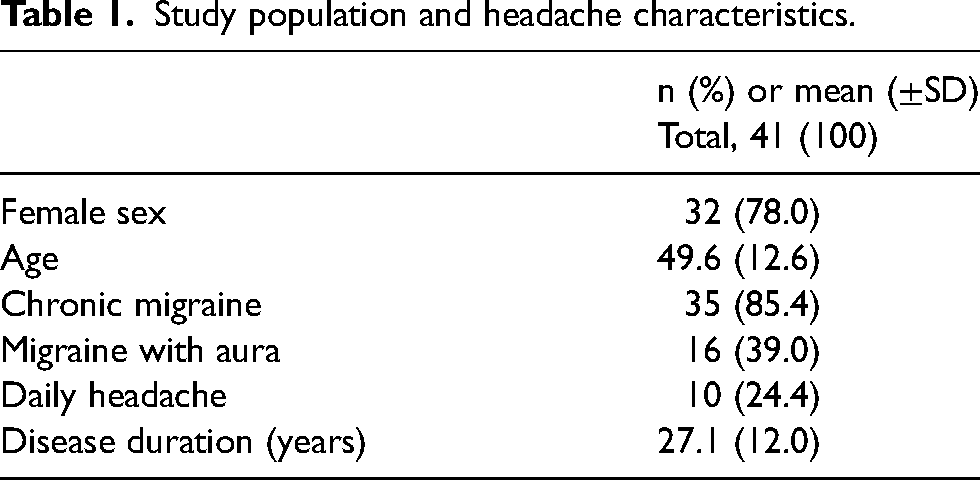
Prior prophylactic treatments.

individual treatment attempt, performed before referral to our outpatient clinic
Prior prophylactic treatment
Before initiating eptinezumab treatment, patients had undergone an average of 9.0 ± 2.2 unsuccessful prior preventive treatment attempts (Table 2). All 41 patients (100%) received erenumab. Thirty-one patients (75.6%) reported prior treatment attempts with all available subcutaneous CGRP(-R) mAbs, while six patients (14.6%) were treated with erenumab and galcanezumab and four patients (9.8%) with erenumab and fremanezumab. CGRP(-R) mAb treatment was primarily discontinued due to poor efficacy. Discontinuation due to poor tolerability was reported by two patients (4.9%; skin reaction; respiratory tract infections) for erenumab, one patient (2.7%; constipation) for galcanezumab, and one patient (2.9%; skin reaction) for fremanezumab.
Responder rates
During weeks 9–12 of eptinezumab treatment, a ≥ 30% reduction in MMDs was observed in nine patients (23.1%) compared to baseline, with two patients (5.1%) experiencing a reduction of ≥50% (Figure 2). Eleven patients (29.7%) reported a ≥ 30% reduction in MMDs during weeks 21–24 compared to baseline, with two patients (5.4%) achieving a reduction of ≥50%. When excluding patients with daily headaches, the response rates for a ≥ 30% reduction in MMDs were 30.0% during weeks 9–12 and 35.7% during weeks 21–24. No patients achieved a reduction rate of ≥75% in MMDs. A ≥ 30% reduction in MHDs was observed in six patients during both weeks 9–12 (15.4%) and 21–24 (21.4%).

Relative reduction of MMDs during weeks 9–12 and 21–24 compared to baseline after treatment with eptinezumab.
Of the nine responders at three months, three remained on the 100 mg dose of eptinezumab, while six opted to increase the dose to 300 mg, seeking an enhanced response. Among those who increased the dose, three maintained their responder status at six months, whereas the other three experienced a reduction in efficacy, with their response dropping to ≤30% reduction in MMDs. Notably, the three patients who initially responded at three months and chose not to increase the dose all maintained their responder status at six months. Of the 23 patients who did not initially respond to eptinezumab 100 mg but continued treatment with 300 mg, five (21.7%) reported a ≥ 30% reduction in MMDs after six months (see Figure 3).

Flowchart of eptinezumab treatment with response and drop out rates after three and six months of treatment.
Evolution of migraine and headache frequency
The number of MMDs decreased from 16.3 (±8.0) in the month before initiation of eptinezumab treatment to 15.4 (±8.1) during weeks 9–12 and 14.4 (±8.0) during weeks 21–24. This numerical trend did not reach statistical significance (p = 0.07, W = 0.07, Table 3). Accordingly, there were no differences comparing the MMDs for weeks 1–8 or 13–20 (see online supplementary data, Tables S1 and S2).
Headache and migraine details.
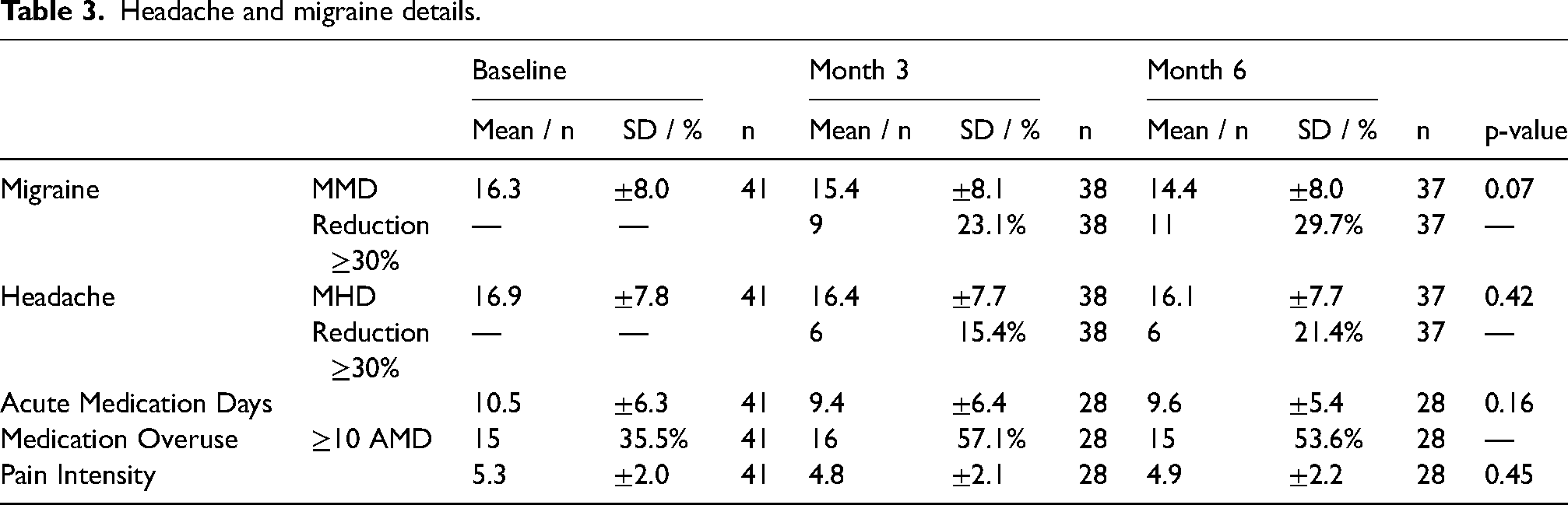
The number of MHDs was 16.9 (±7.9) at baseline, 16.4 (±7.7) during weeks 9–12 and 16.1 (±7.7) during weeks 21–24 (p = 0.42, W = 0.02). We observed a slight and not statistically significant decrease in migraine pain intensity, with 5.3 (±2.0) on a numerical analogue scale (NAS) at baseline, 4.8 (±2.1) during weeks 9–12 and 4.9 (±2.2) during weeks 21–24 (p = 0.45, W = 0.03). Similarly, acute medication days (AMDs) decreased from 10.5 (±6.3) at baseline to 9.4 (±6.4) during weeks 9–12 and 9.6 (±5.4) during weeks 21–24, without reaching statistical significance (p = 0.16, W = 0.07). The absolute number of patients with a medication overuse (MO) defined as intake of acute medication on more than 10 days per month did not change from Baseline (15 patients) to Month 6 (15 patients).
Patients with daily headache
We analyzed the subgroup of patients who experienced daily headaches before starting eptinezumab treatment. Of the 10 patients, one (10%) responded with a sustained reduction of more than 30% in MMDs after three months of treatment. MMDs decreased from 28.0 (±0) at baseline to 25.3 (±6.1), though this reduction was not statistically significant (p = 0.16). Similar trends were observed for MHDs, which decreased from 28.0 (±0) to 27.2 (±2.0) after six months (p = 0.32), AMDs, which declined from 11.7 (±7.7) to 10.0 (±9.6) (p = 0.91), and pain intensity, which reduced from 6.3 (±2.5) to 5.7 (±2.2) (p = 0.14) (see Table 4).
Patients with daily headache: Headache and migraine details.

Tolerability
No side effects were reported in the 24-week treatment period.
Patient reported outcomes
MIDAS score
Patients presented with an average MIDAS score of 96.1 (±73.9) at baseline, followed by scores of 93.2 (±74.1) during weeks 9–12 and a decrease to 73.6 (±76.9) during weeks 21–24. However, these changes did not reach statistical significance (p = 0.255, W = 0.07). During weeks 9–12, 22 patients (61.1%) reported a clinically meaningful reduction in MIDAS Scores (>4.5 points) (33). During weeks 21–24, 11 patients (52.4%) showed meaningfully improved MIDAS Scores (Table 5)
Patient reported outcomes.

1-very much improved, 5-nochange, 10 very much worsened.
HIT-6
At baseline, the mean HIT-6 score was 65.1 (±5.4), which decreased to 63.7 (±6.8) during weeks 9–12 and 62.7 (±7.0) during weeks 21–24, albeit without achieving statistical significance (p = 0.093, W = 0.09). During weeks 9–12, 11 patients (28.9%) reported a clinically meaningful reduction (>2.3 points) in their HIT-6 scores. Ten patients (38.4%) reported a clinically meaningful reduction during weeks 21–24 (34) (Table 5).
DASS-21
The average DASS-21 scores over time are presented in Table 5. A numerical trend was observed in the DASS-21 scores for depression (p = 0.1, W = 0.1) and anxiety (p = 0.06. W = 0.12). A significant difference was noted for the stress scale (p = 0.04) across all observation points; however, this significance was lost after post hoc analysis and adjustment using the Bonferroni correction (p = 0.098, W = 0.14). During weeks 9–12, 11 patients (29.7%) exhibited a clinically meaningful reduction (≥6.0) in the DASS-21 scores for depression, six patients (16.2%) for stress, and four patients (10.8%) for anxiety. Seven patients (30.4%) displayed a clinically meaningful reduction in the depression scale, and six patients (26.1%) exhibited reductions in both the stress and anxiety scales during weeks 21–24 (35) (Table 5).
VAS of patients’ impression of change
The patients’ impression of change, as measured by a 1–10 VAS (1 = ‘very much improved’; 10 = ‘very much worsened’) was 4.8 (±1.8) after three months and 4.3 (±1.8) after six months of treatment. After three months of treatment, 47.3% of patients reported a VAS score of less than 5, indicating an improvement in their condition. This percentage increased to 61.5% after six months of treatment with eptinezumab. (Table 5).
Discussion
The findings from this real-world analysis suggest that some treatment-refractory migraine patients who did not respond to multiple subcutaneous CGRP(-R) mAbs may benefit from switching to the intravenous CGRP-mAb eptinezumab. Approximately one-third of patients demonstrated a clinically relevant reduction in MMDs by more than 30% following the transition to eptinezumab. Although there was a slight numerical decrease in mean MMDs and MHDs, these changes did not achieve statistical significance in our cohort. This suggests a reduced efficacy of eptinezumab in these highly treatment-refractory cohort. Notably, patients did not report any side effects, indicating a favorable safety profile for eptinezumab in this cohort.
Eptinezumab received marketing approval in 2022 by the European Medicines Agency (EMA) and was introduced to the German market in September 2022 (24). Its safety and efficacy were assessed through several randomized controlled trials. The PROMISE 1 and PROMISE 2 trials focused on EM and CM respectively, evaluating different dosages of eptinezumab (36,37). These trials reported a reduction of 3.9 MMDs for eptinezumab 100 mg and 4.3 MMDs for 300 mg in EM, and 7.7 and 8.2 in CM during weeks 9–12 compared to baseline. Additionally, the DELIVER study, including EM and CM patients with two to four prior preventive treatments, demonstrated a reduction of 4.8 and 5.3 MMDs for eptinezumab 100 mg and 300 mg (6). However, it is important to note that no clinical trial assessed patients with more than four prior treatment failures including several mAb treatment attempts, which represent a significant proportion of patients treated with eptinezumab in the real world (6). The number of prior preventive treatments is known to be a negative predictor of response (38). Consistent with this understanding, in our cohort of highly refractory patients, the observed reductions were notably lower.
Other real-world data regarding eptinezumab treatment remains limited, yet it also suggests a potential correlation between the number of prior preventive treatments and the drug's efficacy as previously described for other CGRP-R mAbs (39). In a study conducted in the United Arab Emirates involving 53 patients over 24 weeks, the majority were relatively young (mean age 36.4 ± 10.6 years) with an average migraine duration of 7.0 ± 7.9 years. A significant portion of patients were treatment-naïve for preventive measures (30.2%). In this study, MMDs decreased from 12.2 (±5.0) to 3.6 (±4.2) over 24 weeks, with 58.5% experiencing a reduction of at least 75% in MMDs by weeks 21–24 (26). In Singapore, a cohort of 29 migraine patients with an average of 3.4 prior treatments was treated with eptinezumab 100 mg for six months with a decrease in MMDs relative to baseline by 4.3 at month 3 and 4.9 at month 6 (25).
The most recent data on eptinezumab are from a retrospective multicenter study in Germany, which is the most similar to our study. Scheffler et al. (27) analyzed the treatment response of 79 patients with episodic or CM who received eptinezumab 100 mg intravenously for three months. Due to national reimbursement policies, patients underwent all first-line preventive treatments before being eligible for eptinezumab, resulting in a minimum of four prior treatments for those with EM and five for those with CM. They observed a significant reduction in MHDs by 4 days, MMDs by 3 days, and AMD by 2 days during the third month of treatment. Importantly, 35 patients in this cohort had received two or more CGRP(-R) mAbs in addition to first line treatment prior to eptinezumab infusion. Patients with at least two prior CGRP(-R) treatments exhibited a 30% response rate for MMDs of 32.1%, and those with three prior treatments had a response rate of 35.3%. In our study, we observed a slightly lower response rate at three months, with 23.1%. This disparity in response rates could potentially be attributed to the higher initial migraine burden in our cohort, with an average of 16.1 MMDs and 85% of patients reporting a CM, compared to 12.0 MMDs and 76% with CM in the previous German study. Additionally, a significant proportion of patients in our cohort experienced daily headaches, a known negative predictor of treatment response, with a response rate of only 10% in our study (38). When excluding patients with daily headaches, the response rates at three months rose to 30%, mirroring the findings from the previous German study. Our study therefore confirms previous findings in highly treatment-resistant cohorts and expands them towards a longer treatment duration. The observation that 5 out of 11 responders at six months were non-responders at three months underscores the importance of extending treatment duration before assessing the overall effectiveness of eptinezumab in difficult-to-treat migraine patients. This delayed response suggests that some patients may need more time and a higher dose to achieve significant benefits, supporting the recommendation for a second treatment cycle with an increased dose of eptinezumab (300 mg) before deciding on continuation. This approach could help identify late responders who may benefit from sustained therapy, optimizing patient outcomes, especially in a patient group with limited treatment alternatives. Notably, among the patients who initially responded to treatment and still chose to increase their dose to 300 mg, three maintained their responder status at three months, while three others experienced a reduction in efficacy. The observed fluctuations in efficacy suggest that increasing the dose does not uniformly benefit all patients and highlights the importance of tailoring treatment decisions to the individual needs of each patient with migraine. In addition to assessing treatment efficacy, we also examined various patient reported outcomes (PROs), a facet not explored in the study by Scheffler et al. (27), that allows a more patient-centered evaluation of therapy success. Using the HIT-6 questionnaire to gauge the impact of headaches on individuals’ daily lives and functioning, we noted a clinically meaningful improvement in 29% of at least 2.3 points of treated patients at month 3, increasing to 39% by month 6. Similarly, reductions in patients’ MIDAS scores revealed a clinically meaningful improvement in migraine-associated disability in 61% of patients at month 3 and 52% at month 6. Furthermore, there was a decrease in patients’ levels of depression, anxiety, and especially stress as indicated by the DASS-21 subscales and a high overall satisfaction with the eptinezumab treatment, with 61.5% of patients reporting a subjective improvement after six months.
The favorable subjective response to eptinezumab therapy is underscored by the high treatment adherence rate, with 93% of patients continuing their treatment beyond 12 weeks. This remarkable adherence rate can be attributed partly to the alleviation of migraine symptoms, but also to the favorable side effect profile associated with eptinezumab. Notably, no adverse drug reactions were reported in our cohort during the observation period, aligning with data from the UAE where only one out of 53 patients reported adverse effects (26).
The main limitation of our study is its non-blinded retrospective design and the absence of a control group, which is likely to introduce biases and increase the likelihood of a placebo response. The potential influence of a placebo effect on our findings is particularly high given the more invasive route of administration compared to subcutaneous mAbs (40). However, it is important to note that this is a real-world study, and placebo effects are an inherently part of such settings. This reflects the practical outcomes of eptinezumab treatment in a real-world clinical environment, providing valuable insights into its effectiveness in a highly refractory patient population. Additionally, we encountered missing data on pain intensity and acute treatment use, and we did not collect information on attack duration, vegetative symptoms, migraine aura characteristics, or response rates to acute medication as potential variables for partial response. The missing data on acute medication use could have introduced selection bias, resulting in a relatively higher proportion of patients reporting MO, even though the absolute numbers remained unchanged. These gaps limit the comprehensiveness of our analysis. Another limitation of this study is the classification of poor tolerability as a failure of the first mAbs, which may be misleading when assessing ineffectiveness. However, with only four patients discontinuing due to adverse events, this aspect is not likely to impact the overall results. Furthermore, the limited number of patients reduced the study's power to differentiate treatment responses between eptinezumab 100 mg and 300 mg, as well as between episodic and CM. Our cohort comprises a challenging-to-treat, therapy-refractory population, with a significant proportion experiencing daily headaches, recruited from a tertiary headache center. Therefore, it is central to acknowledge that this patient group may not be representative of the broader migraine population.
Conclusion
Our retrospective analysis of 41 therapy-refractory migraine patients suggests that switching to the intravenous CGRP mAb eptinezumab may provide clinically relevant benefits for approximately one-third of patients who previously had an inadequate response to first-line preventive treatments, erenumab, galcanezumab, and/or fremanezumab. A treatment duration of at least six months and consideration of dose escalation appear to be reasonable strategies to maximize treatment potential. The response rates and overall outcomes in our study were significantly lower compared to studies involving treatment-naive or less treatment-experienced patients. In particular, patients with daily headaches showed a limited response to treatment. This disparity suggests that use of eptinezumab is likely to provide greater benefits when used earlier in the treatment algorithm. Nevertheless, our findings indicate that even in a highly refractory population, eptinezumab can still offer meaningful improvements in a subgroup of patients. Larger prospective studies are necessary to validate our results, assess predictors of a positive response, and refine the timing and sequencing of CGRP-targeted treatments to advance the personalization of migraine management.
Article highlights
30% of treatment-refractory patients responded with a reduction in monthly migraine days by 30% during weeks 21–24 of treatment with eptinezumab.
Clinically meaningful improvements were observed in 40% of patients based on HIT-6 scores and in 50% based on MIDAS scores after six months of treatment.
Eptinezumab treatment, up to 300 mg, was well tolerated by patients.
Evaluating treatment success after a minimum six-month treatment period appears reasonable.22% of patients who did not respond to the initial 100 mg eptinezumab treatment achieved a 30% reduction in MMDs after increasing the dose to 300 mg.
Supplemental Material
sj-docx-1-cep-10.1177_03331024241288875 - Supplemental material for Efficacy of eptinezumab in non-responders to subcutaneous monoclonal antibodies against CGRP and the CGRP receptor: A retrospective cohort study
Supplemental material, sj-docx-1-cep-10.1177_03331024241288875 for Efficacy of eptinezumab in non-responders to subcutaneous monoclonal antibodies against CGRP and the CGRP receptor: A retrospective cohort study by Paul Triller, Virginia N. Blessing, Lucas H. Overeem, Mira P. Fitzek, Ja Bin Hong, Kristin S. Lange, Uwe Reuter, and Bianca Raffaelli in Cephalalgia
Footnotes
Authors’ contributions
P.T: Investigation; Formal Analysis; Writing – Original Draft Preparation. V.N.B.: Conceptualization; Writing – Review & Editing. L.H.O.: Data curation; Writing – Review & Editing. M.P.F: Investigation; Writing – Review & Editing. J.B.H: Investigation; Writing – Review & Editing. K.S.L.: Investigation; Writing – Review & Editing. U.R.: Conceptualization; Supervision; Writing – Review & Editing. B.R.: Conceptualization; Investigation; Project Administration; Writing – Review & Editing.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P.T. reports personal fees from Abbvie. V.N.G. has nothing to disclose. L.H.O. has nothing to disclose. M.P.F. reports personal fees from Teva. J.B.H. has nothing to disclose. K.S.L. reports personal fees from Teva. U.R. reports personal fees from Amgen, Allergan, Abbvie, Lilly, Lundbeck, Novartis, electroCore, Medscape, StreaMedUp, and Teva, and research funding from Novartis. B.R. reports personal fees from Abbvie/Allergan, Eli Lilly, Lundbeck, Novartis, Perfood and Teva, and research funding from Lundbeck and Novartis.
Ethics approval
This study (EA1/015/24) was approved by local ethics committee (Charité Universitätsmedizin Berlin, Berlin, Germany) and adhered to the principles outlined in the Declaration of Helsinki. Written informed consent was not required for this retrospective analysis of chart data acquired during routine treatment according to national legislation and institutional requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Lundbeck Foundation, (grant number IIT-2023-005).
Supplementary Material
Supplementary material is available at Cephalalgia online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
